Author Contributions
Academic Editor: Qiang Cheng, Biomedical Informatics Institute, and Computer Science Department
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2018 Pedram Azari, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Editorial Note
A scaffold is defined as a 3D porous material that features characteristics such as biocompatibility, and biodegradability to serve as a temporary platform for tissue regeneration. While hydrophilicity was not initially mentioned as one of the requirements for scaffolding materials, the success of bio-derived hydrophilic natural polymers e.g. collagen, gelatin, won great acclaims for hydrophilic scaffolds 1. However, more recent application of hydrophobic electrospun biopolyesters in tissue engineering provides an opportunity to look at this matter from a different perspective and to identify the suitability of hydrophobic scaffold materials as well.
For the last two decades, electrospun biopolyesters have been an important class of scaffolding materials offering the advantages of electrospinning and biocompatibility of polyesters in one package. Electrospinning is a direct and versatile method that has the flexibility to be applied for fiber fabrication from various biopolymers 2. It produces morphologies and dimensions similar to those of natural extracellular matrix (ECM) 3, an interwoven random fibrous structure of collagen and elastin with diameters ranging from tens to hundreds nanometers creating a matrix with considerable surface area for biomolecular interactions 4, 5. The morphological similarity of the electrospun scaffolds to natural ECMs facilitates cell seeding, adhesion, proliferation, and cell differentiation 4, 6. Two other common features of electrospun fibers is their high surface area to volume ratio and their porosity, which can lead to infiltration of cells into scaffolds resulting in stronger cell attachment 7. The large surface area of the scaffolds is favorable for loading bioactive materials within the matrix 8 while the porous nature of the electrospun scaffolds facilitates food absorption and waste release by the cells 9.
Componential aspects of the scaffold materials are also of great significance. An ideal scaffold mimics the natural morphological and componential properties of an ECM 4. Biopolyesters are attractive materials for scaffolding applications, mainly due to their relatively low cost of synthesis, ease of degradation, and the ability to tailor their degradation rate through changes in the structure. This class of polymers degrade through hydrolysis of the ester bonds, producing non-toxic degradation products 10.
Polycaprolactone (PCL), polylactic acid (PLA), poly3-hydroxybutyrate (PHB), and its copolymer with hydroxvalerate (PHBV) are the most popular commercially available biopolyesters that have been successfully electrospun and produced promising platforms for tissue engineering applications. All these materials are aliphatic linear polyesters produced from renewable resources. Their rheological and viscoelastic properties facilitate processing and fabrication of them into desired morphologies 3. Electrospun scaffolds based on these biopolyesters were successfully used for various types of cells including stem cells 11, osteoblasts 12, dermal fibroblasts 13, chondrocytes 14, keratocytes 15, vascular 16, and neural tissue 17. Figure 1 shows laser microscope representation of the surface, water contact angle, and field emission scanning electron microscopy (FESEM) of PHB electrospun fibers seeded with keratocytes as an example for biopolyesters. All these materials in the form of electrospun membranes exhibit hydrophobic behavior when analyzed by water contact angle (WCA) (Figure 1.b, θ ~120°). This behavior is mainly due to the chemical structure of the biopolyesters and the absence of hydrophilic functional groups as well as relative roughness of the electrospun surfaces, and the air trapped in the micropores of fibrous network 18. Electrospun materials, due to their fibrous nature, typically possess rough surfaces. In surface topography analysis, a rough surface has deviations and irregularities compared to a smooth surface 19. While surface roughness, in a general sense, improves the cellular response to the substrate for higher availability of the surface area 20, hydrophobicity has mainly been considered as a less favorable attribute for the scaffolding materials 21. Hydrophobic materials tend to exhibit poor wettability and cell attachment as well as uncontrolled biological interactions. Therefore, several methods have been considered to rectify these shortcomings and to introduce a more hydrophilic nature to the materials. These suggested modification techniques can be divided in four categories: plasma treatment, alkaline treatment, coating, and blending with other bioactive materials 3.
Figure 1.Electrospinning setup for fabrication of randomly deposited fibers (a); Water-in-air contact angle for untreated electrospun PHB fibers (b, θ =128°); 3D confocal laser microscopic image of PHB with average roughness of Sa = 11.02 µm (c); and Electrospun PHB fibers seeded with rabbit keratocytes showing a successful cell attachment (d).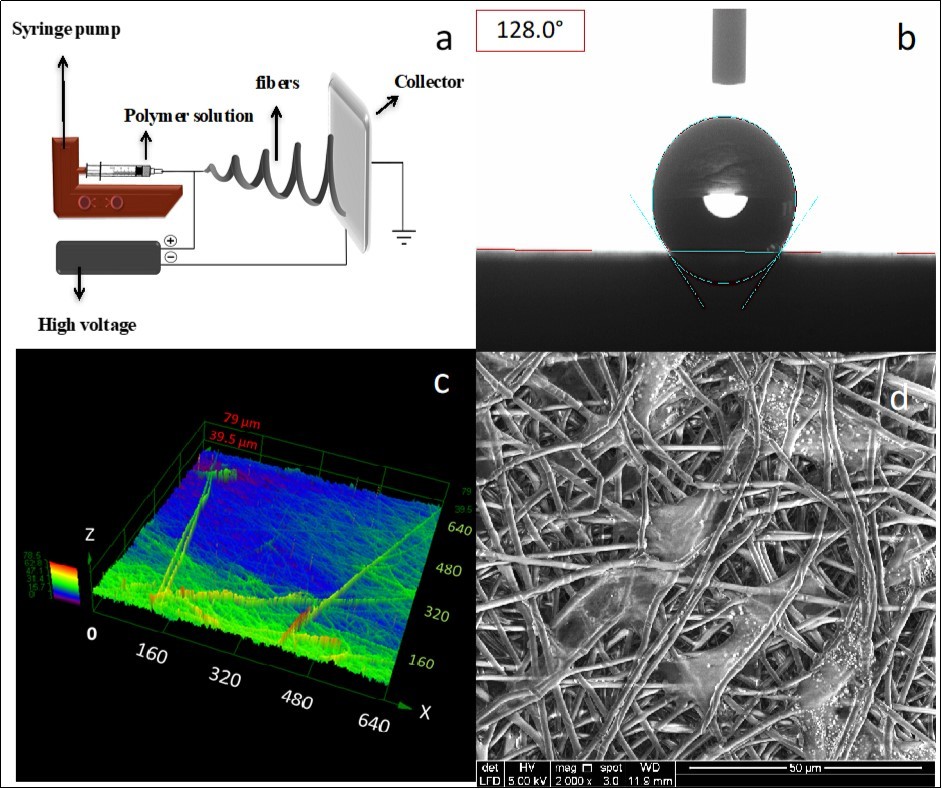
Despite being hydrophobic and contrary to the expectation, electrospun biopolyesters have proven to be capable of promoting cell growth with desirable proliferation rates 15. Volger et al. reported that hydrophilic surfaces with contact angle of θ < 65° that do not lead to high level of cell attachment efficiency 22. Similar reports suggested that fibroblast cells indicate maximum attachment at contact angles between 60° and 80° 23. These results imply that a certain level of hydrophobicity can encourage and facilitate cell attachment and growth. This could be explained through protein adsorption and transient hydrogen bonding mechanisms. Attachment of the cells to the scaffold happens through a sequence of physico-chemical reactions in which protein adsorption plays a key role as mediator for cell adhesion. Adsorbed proteins to the scaffold provide the signal to the cells through adhesion receptors for which a series of cytoplasmic, transmembranal, and extracellular proteins are required 23. It is well-known that moderate hydrophobic surfaces are capable of better absorption of proteins in comparison to the hydrophilic surfaces, which have more resisting forces at the surface 24, 25, 26, 27. The phenomenon could be due to unique physico-chemical properties of the water. As the main component in culture media, water has a unique self-association property. A water molecule has two hydrogen bond acceptors (unshared electron pairs of oxygen) and two hydrogen bond donors (protons) allowing the molecule to associate with maximum four neighboring molecules. Since these associations are relatively weak and almost equivalent to the energy level transferred from one molecule to another neighboring molecule, the hydrogen bonds possess a transient nature. The presence of Lewis acid/base sites on hydrophilic surfaces suppresses self-association of water molecules as they enter a competition through hydrogen bonding resulting in presence of repulsive forces at the interface while surfaces with θ ≥ 65° can establish a wider range of attractive forces 22.
Although the natural hydrophobicity of the electrospun biopolyesters is higher than the reported optimum numbers for scaffolding materials, it has been proven that relative hydrophobicity can offer rather more favorable platforms for such applications. As mentioned earlier, several surface modification methods are also available for treatment of super-hydrophobic biopolyester candidates to reduce the hydrophobicity 3. Furthermore, WCA ≈ 120° applies to unprocessed electrospun biopolyester. The general practice of scaffold preparation forcell culturing involves various wet processing steps including sterilization with ethanol, washing with phosphate buffered saline (PBS), and soaking in culturing medium 28. These wet processing steps can cause a significant reduction in WCA, providing promising cell-scaffold interactions even on the unmodified electrospun biopolyesters.
While not discrediting the previous understanding in regard to hydrophilic polymeric scaffolds, this short note suggests that surfaces with moderate hydrophilicity could be optimal platforms for tissue engineering applications. In conclusion, electrospun biopolyesters should be considered within the unique family of scaffolding materials that offers desirable features for tissue engineering with addressable shortcomings.
Acknowledgments
The authors would like to acknowledge the financial support of Tecnologico de Monterrey, Mexico, for the special grant (grant number: 002EICII01) awarded to the Nano Sensors and Devices Focus Group, School of Engineering and Sciences, Tecnologico de Monterrey, Monterrey, Mexico.
References
- 1.F J O'Brien. (2011) Biomaterials & scaffolds for tissue engineering. Materials Today. 14(3), 88-95.
- 2.Akram H. (2014) An in-vitro study on the proliferative potential of rat bone marrow stem cells on electrospun fibrous polycaprolactone scaffolds containing micro-hydroxyapatite particles. Materials Research Innovations. 18, 520-524.
- 3.Cipitria A. (2011) Design, fabrication and characterization of PCL electrospun scaffolds-a review. , Journal of Materials Chemistry 21(26), 9419-9453.
- 4.Wang X, Ding B, Li B. (2013) Biomimetic electrospun nanofibrous structures for tissue engineering. Materials Today. 16(6), 229-241.
- 5.B N Sathy. (2017) Designing Scaffolds for Bone Tissue Engineering. Translating Biomaterials for Bone Graft: Bench-top to Clinical Applications.
- 6.Liang D, B S Hsiao, Chu B. (2007) Functional electrospun nanofibrous scaffolds for biomedical applications. Adv Drug Deliv Rev. 59(14), 1392-412.
- 7.Wu J, Hong Y. (2016) Enhancing cell infiltration of electrospun fibrous scaffolds in tissue regeneration. Bioactive Materials. 1(1), 56-64.
- 8.Azari P. (2014) Improved processability of electrospun poly[(R)-3-hydroxybutyric acid] through blending with medium-chain length poly(3-hydroxyalkanoates) produced by Pseudomonas putida from oleic acid. Materials Research Innovations. 18(6), 345-349.
- 10.P A Gunatillake, Adhikari R. (2003) Biodegradable synthetic polymers for tissue engineering. Eur Cell Mater. 5(1), 1-16.
- 11.M C Phipps. (2011) Mesenchymal stem cell responses to bone-mimetic electrospun matrices composed of polycaprolactone, collagen I and nanoparticulate hydroxyapatite. PLoS One. 6(2), 16813.
- 12.Rozila I. (2016) Differential osteogenic potential of human adipose‐derived stem cells co‐cultured with human osteoblasts on polymeric microfiber scaffolds. , Journal of Biomedical Materials Research Part A 104(2), 377-387.
- 13.J L Lowery, Datta N, G C Rutledge. (2010) Effect of fiber diameter, pore size and seeding method on growth of human dermal fibroblasts in electrospun poly (ɛ-caprolactone) fibrous mats. Biomaterials. 31(3), 491-504.
- 14.W J Li. (2003) Biological response of chondrocytes cultured in three‐dimensional nanofibrous poly (ϵ‐caprolactone) scaffolds. , Journal of Biomedical Materials Research Part A 67(4), 1105-1114.
- 15.Azari P. (2015) Electrospun Biopolyesters as Drug Screening Platforms for Corneal Keratocytes. , International Journal of Polymeric Materials and Polymeric Biomaterials 64(15), 785-791.
- 16.Vaz C. (2005) Design of scaffolds for blood vessel tissue engineering using a multi-layering electrospinning technique. Acta biomaterialia. 1(5), 575-582.
- 17.Ghasemi-Mobarakeh L. (2008) Electrospun poly (ɛ-caprolactone)/gelatin nanofibrous scaffolds for nerve tissue engineering. Biomaterials. 29(34), 4532-4539.
- 18.Hosseini S. (2017) Polymethacrylate Coated Electrospun PHB Fibers as a Functionalized Platform for Bio-Diagnostics: Confirmation Analysis on the Presence of Immobilized IgG Antibodies against Dengue Virus. Sensors. 17(10), 2292.
- 19.Grasselli G, Wirth J, Egger P. (2002) Quantitative three-dimensional description of a rough surface and parameter evolution with shearing. International Journal of Rock Mechanics and Mining Sciences 39(6), 789-800.
- 20.Deligianni D D. (2000) Effect of surface roughness of hydroxyapatite on human bone marrow cell adhesion, proliferation, differentiation and detachment strength. Biomaterials. 22(1), 87-96.
- 21.E S Place. (2009) Synthetic polymer scaffolds for tissue engineering. Chemical Society Reviews. 38(4), 1139-1151.
- 22.E A Vogler. (1999) Water and the acute biological response to surfaces. , Journal of Biomaterials Science, Polymer Edition 10(10), 1015-1045.
- 23.Chang H-I, Wang Y. (2011) Cell responses to surface and architecture of tissue engineering scaffolds, in Regenerative medicine and tissue engineering-cells and biomaterials. , InTech
- 24.Xu L-C, C A Siedlecki. (2007) Effects of surface wettability and contact time on protein adhesion to biomaterial surfaces. Biomaterials. 28(22), 3273-3283.
- 25.Chen S. (2015) Biomimetic honeycomb-patterned surface as the tunable cell adhesion scaffold. Biomaterials science. 3(1), 85-93.
- 26.Fairman C. (2013) Protein resistance of surfaces modified with oligo (ethylene glycol) aryl diazonium derivatives. , ChemPhysChem 14(10), 2183-2189.
Cited by (2)
- 1.Amiri Zahra, Molavi Amir Mahdi, Amani Amir, Moqadam Kurosh Hamzanlui, Vatanchian Mehran, et al, 2024, Fabrication, characterization and wound-healing properties of core–shell SF@chitosan/ZnO/Astragalus arbusculinus gum nanofibers, Nanomedicine, 19(6), 499, 10.2217/nnm-2023-0311
- 2.Ng , Azari , Nam , Xu , Pingguan-Murphy , 2019, Electrospin-Coating of Paper: A Natural Extracellular Matrix Inspired Design of Scaffold, Polymers, 11(4), 650, 10.3390/polym11040650

