The Effect of Medical Theraphy on Plasma Homocysteine Levels and Carotid Intima-Media Thickness in Parkinson's Disease
Abstract
Objective:
We aimed to investigate homocysteine levels and carotid intima-media thickness (CIMT) in Parkinson’s Disease (PD), to determine relationship of these parameters and as well as to determine whether CIMT in patients with PD was associated to age, disease duration, age of disease onset, stage, the Unified Parkinson Disease rating scale (UPDRS), the drugs used in therapy.
Methods:
The study population consisted of 55 PD patients (37 male) and 25 healty subjects. The severity of neurological impairment was assessed with UPDRS and the Hoehn-Yahr scale. CIMT and homocysteine levels were measured. Anti-parkinsonian treatments were recorded and the total daily dose of levodopa was calculated for each patient.
Results:
Homocysteine levels were significantly higher in the patient group compared to the control group (p=0.002). A positive correlation was found between CIMT and homocysteine (r=0.29 p=0.03), but no a relationship between CIMT and UPDRS scores, disease duration, age of disease onset, and stage. Mean levodopa dosage did not predict CIMT 0.6 mm (AUC: 0.546, 95%CI 0.372-0.720, p=0.59). Homocysteine 14 µmol/l predicted CIMT 0.6 mm with 64% sensitivity and 69% specificity (AUC: 0.654, 95%CI 0.488-0.819, p=0.07).
Dıscussion:
This study revealed that homocysteine levels in levodopa + dopa decarboxylase enzyme inhibitor (DDEI) group were increased which was correlated with a mild increasement of CMIT. This might indicate to the importance of clinical and radiological follow up of PD patients who are under treatment of levodopa + DDEI.
Conclusion:
Our Findings May Suggest The Role Of CIMT As A Meaningful Clinical Marker For Follow-Up Of Patients With PD
Author Contributions
Academic Editor: Biswarup Ghosh, Thomas Jefferson University, Dept. of Neuroscience, Philadelphia, US
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2016 Yasemin Kaya, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Idiopathic Parkinson’s disease (IPD) is the second most common neurodegenerative disorder after Alzheimer’s Disease 1. Various studies have reported that the treatment of IPD with levodopa is associated with the elevation of serum homocysteine (Hcy) 2. Levodopa is metabolized and changes O-methylation in the brain and peripheral tissues. This reaction is carried out by the means catechol O-methyltransferase (COMT) enzyme, and requires S-adenosyl methionine (SAM) as the methyl donor and produces the demethylated product S -adenyl homocysteine (SAH). Then, SAH is converted to homocysteine. Homocysteine is converted back to methionine via methylene tetra hydro folate reductase and betaine homocysteine methyltransferase. Methylene tetra hydrofolate reductase requires a folate cofactor. The catabolism of levodopa interferes with homocysteine metabolism at several steps 3.
Some studies have stated that the toxic effects of hyperhomocysteinemia (HHcy) have been suggested in cardiovascular and cerebrovascular diseases 4. HHcy is an independent risk factor for coronary artery disease 5. Elevated Hcy levels may be a risk factor for atherothrombotic vascular disease; this condition may worsen the course of IPD. Intima media thicknes (IMT) is one of the markers of early atherosclerosis and large peripheral arteries can be evaluated for early atherosclerosis 6, 7. Structural, functional, and hemodynamic alterations of the carotid artery are measured with ultrasound and is used as a marker of atherosclerosis 8.
The aim of this study was to determine Hcy levels in IPD, to investigate the relationship between Hcy levels and carotid intima media thicknes (CIMT), to determine whether CIMT in patients with IPD was associated with age, disease duration, age of disease onset, stage, Unified Parkinson’s Disease rating scale (UPDRS), and the drugs used in therapy, and Hcy levels.
Methods
Patients with IPD were recruited from the Movement Disorders Clinic of the School of Medicine of the Ordu University, Turkey. Fifty-five patients with IPD who registered at our movement disorders outpatient clinics between June 2013 and May 2014 were included in this study. The patients who did not want to participate in the study and were uncooperative were excluded from the study. The diagnosis of IPD was confirmed by movement disorders specialists in neurology, according to the UK Parkinson’s Disease Society Brain Bank Criteria 9. The severity of neurological impairment was assessed with the Unified Parkinson’s Disease rating scale (UPDRS scale, ranging from 0 to 147) and the Hoehn -Yahr scale (HY scale, ranging from 0 to 5) 10, 11. The healthy control was result of the study attempt by F.H. Beşir et al. that Turkish healthy individuals (withoutParkinson's disease and coronary artery disease) were examined by measurements made CIMT according to age groups 12. The study was approved by the institutional ethics committee of Ordu University.
Anti-parkinsonian treatments were recorded and the total daily dose of levodopa was calculated for each patient. For dopaminergic agonists, we calculated the equivalent levodopa dosage as follows: 4 mg ropinirole=1 mg pramipexole= 100 mg levodopa 13, 14.
Blood Tests
Blood samples were collected in gel tubes that did not include anticoagulant to measure total cholesterol, triglycerides (TG), high-density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C), and uric acid (UA); additionally, EDTA tubes were used to measure Hcy after 12 hours overnight. Blood samples were centrifuged at 1800*g for15 minutes and plasma and serum samples were obtained. Total cholesterol, TG, HDL-C,LDL-C, and UA levels were measured cholorometrically using an Abbott original reagent on Abbott Architect 8000 auto analyzer. Plasma Hcy was also determined by tandem mass spectrometry (LC-MS/MS).
Ultrasonographic Study
The ultrasound evaluation of the common carotid artery (CCA) was performed as described previously using an (Hitachi Preirus, Tokyo, Japan) that incorporated a13 -MHz probe equipped with a Doppler system. After the patients had rested in the supine position for at least 10 minutes with their neck in a slightly hyper extended position, optimal visualization of the right and left CCA was performed. The IMT was measured 1 cm proximal to the start of the carotid bulb dilatation on the CCA in the far wall. The thickened side was evaluated.
Statıstıcal Analysıs
Statistical analyses were performed using the Statistical Package for the Social Sciences (SPSS), Version 20. Descriptive statistics were used to summarize all measurements. All continuous data were shown as mean ± standard deviation. The patient and healthy control group were compared using the chi square test and student’s t-test. Pearson’s test, or if measurements were ordinal, Spearman’s correlation tests, were used for correlation analysis. The subgroups of IPD that had Hcy levels of >15 µmol/l and≤15 µmol/l were compared using the Mann-Whitney U-test and the chi square test. Based on the generated cross-tables, G-test (likelihood-ratio chi-square test) was applied in cases in which the expected frequency was below 5 and Fisher’s exact test was implemented for those with an excepted frequency below 1. The Kruskal-Wallis Test was used to compare drugs subgroups. Parameters that showed a significant difference in the Kruskal -Wallis Test were compared in groups of two, using the Mann-Whitney U-test. The cut-off values were calculated with the ROC curve analysis. The level of statistical significance was established at p < 0.05.
Results
Mean age of IPD was 69.11±8.77 years and mean Hcy level was 13.80±3.65 µmol/l. In the present study, mean CIMT according to the age groups was as follows: 50-59 years of age group 0.65±0.15 mm, 60-69 years of age group 0.72±0.09 mm, 70-79 years of age group 0.85 mm. Sociodemographics, IPD characteristics, mean levodopa dosage, lipids, uric acid, homocysteine, and CIMT levels are presented in Table 1.
Table 1. Demographics and IPD characteristic features, homocysteine, lipids, vitamin B12, folate levels, and CIMT of the patients| Parkinson’s Disease(PD)n= 55 | |
| Mean age years (min-max) | 69.11±8.77 (42-85) |
| Gender n (%) male | 37 (67.3%) |
| female | 18 (32.7%) |
| Age of onset of disease years (min-max) | 65.08±8.53 (44-84) |
| Duration of PD years(min- max) | 4.92±3.77 (1-15) |
| Mean levodopa dosage(mg/day) (min-max) | 503.86±270.81 (25-1100) |
| Stage of disease | |
| 1 (H.Y.S.) n (%) | 17 (31.5%) |
| 2 (H.Y.S.) n (%) | 26 (48.1%) |
| 3 (H.Y.S.) n (%) | 10 (18.5%) |
| 4 (H.Y.S.) n (%) | 1 (1.9%) |
| UPDRS (min-max) | 28.48±18.16 (8-99) |
| UPDRS total | 1.97±1.32 (0-5) |
| UPDRS cognitive | 18.02±11.03 (5-60) |
| UPDRS motor sections | 7.94±7.21 (1-36) |
| UPDRS activities of daily living | 0.48±1.23 (0-6) |
| UPDRS complication of treatment | 13.80±3.65 (7.3-26.7) |
| Homocysteine(µmol/l) | 202.40±40.75 (126-293) |
| Total cholesterol (mg/dl) | 145.11±87.11 (40-469) |
| Triglycerides (mg/dl) | 145.11±87.11 (40-469) |
| HDL-C (mg/dl) | 44.68±12.02 (24-68) |
| LDL-C (mg/dl) | 128.05±32.78 (74.4-206) |
| Uric acid (mg/dl) | 5.15±2.03 (2-8.8) |
| CIMT (mm) | 0.75±0.30 (0.4-2.3) |
In the Parkinson’s patients group, correlation analyses were applied between CIMT and clinical-demographic features, UPDRS scores, mean levodopa dosage, cholesterol, Hcy, and UA levels. Positive correlation was found between CIMT and disease stage and Hcy level. Although there was found no correlation between CIMT, and other parameters (i.e. UPDRS scores, disease duration, age of disease onset, mean levodopa dosage) (Table 2). We have revealed a positive correlation between the Hcy levels and the mean levodopa dosage(r=0.31 p=0.01). However it should be also noted that there was no significant correlation between the Hcy levels and UPDRS scores, disease duration, age of disease onset and stage.
Table 2. Correlation between CIMT and homocysteine, cholesterol, uric acid levels demographics, clinical features.| CIMT | r | P |
|---|---|---|
| Age | 0,24 | 0.06 |
| Duration of PD | -0.02 | 0.9 |
| Age of onset of dısease | 0.18 | 0.27 |
| Stage | 0.28 | 0.03 |
| Mean levodopa dosage | 0.06 | 0.64 |
| UPDRS total | 0.01 | 0.94 |
| UPDRS cognitive | -0.06 | 0.68 |
| UPDRS motor sections | 0.08 | 0.6 |
| UPDRS activities of daily living | -0.06 | 0.68 |
| UPDRS complication of treatment | -0.08 | 0.62 |
| Homocysteine | 0.29 | 0.03 |
| Total cholesterol | -0.17 | 0.31 |
| LDL-C | 0.04 | 0.79 |
| HDL-C | -0.04 | 0.78 |
| LDL-C | -0.18 | 0.3 |
| Uric acid | 0.28 | 0.31 |
The subgroups of drugs (levodopa+DDEI, levodopa+DDEI+entacapone, and other drugs) were compared each other and significant differences were found in terms of age, age of disease onset, Hcy, UPDRS total, UPDRS motor sections, UPDRS activities of daily living and mean levodopa dosage as given details below (Table 3). Post hoc analysis was performed to determine which group differences is (groups were compared in pairs)
Table 3. Comparison of levodopa+DDEI, levodopa+DDEI+ entacapone and other drugs of subgroups| Levodopa+ | Levodopa+ | other drugs | p | |
| DDEI+ | DDEI | n: 13 | ||
| Entacapone: | n: 30 | |||
| n: 12 | ||||
| Age (year) | 72.75±7.52 | 70.53±8.92 | 62.46±6.03 | 0.001b, c |
| Age of onset of the disease (year) | 66.44±11.90 | 67.33±5.91 | 57.63±6.47 | 0.01b, c |
| Duration of disease (year) | 5.56±4.24 | 5.10±3.17 | 3.75±4.86 | 0.2 |
| Homocysteine (µmol/l) | 12.23±2.47 | 14.85±4.06 | 12.8±2.90 | 0.05 a, c |
| UPDRS total | 34.25±15.81 | 32.60±19.9 | 14.22±5.49 | 0.002 b, c |
| UPDRS cognitive | 2.25±1.66 | 2.20±1.19 | 1.22±1.09 | 0.14 |
| UPDRS motor sections | 21.5±10.73 | 20.20±11.74 | 10.11±5.03 | 0.009 b, c |
| UPDRS activities of daily living | 9.62±5.92 | 9.55±8.33 | 2.88±0.92 | 0.002 b, c |
| Mean levodopa dosage (mg/day) | 593.75±258.51 | 566.66±257.46 | 275.96±185.36 | 0.001 b, c |
| Total cholesterol | 214.22±48.81 | 190.61±37.02 | 230.60±21.46 | 0.08 |
| Triglycerides | 193.0±138.8 | 120.54±52.24 | 157.10±52.03 | 0.37 |
| HDL-C | 44.66±13.98 | 42.97±11.68 | 53.7±5.50 | 0.173 |
| LDL-C | 132.92±43.47 | 124.63±30.88 | 133.96±24.68 | 0.74 |
| Uric acid | 4.27±2.33 | 5.2±2.11 | 5.9±1.74 | 0.37 |
| CIMT | 0.74±0.22 | 0.79±0.37 | 0.66±0.08 | 0.35 |
By comparing the levodopa+DDEI with the levodopa+DDEI+entacapone subgroup ,we have revealed higher Hcy levels in the LD+DDEI group than the levodopa+DDEI+entacapone subgroup (p=0.02).
Howewer when we compared the LD+DDEI subgroup with the other drugs subgroup, we have found that age, age at disease onset, UPDRS total, UPDRS motor, UPDRS daily life, mean levadopa dosage were significantly increased in the levodopa+DDEI subgroup (p=0.000, p= 0.001 p= 0.001 p=0.002 p=0.001 p=0.000, p=0.03 respectively).
Furthermore, although we have found that Hcy levels were not significantly different between the LD+DDEI+entacopone and the other drugs subgroups (p=0.38). The age, age at disease onset, UPDRS total, UPDRS motor, UPDRS daily life and mean levadopa dosage were sigificnatly higher in levodopa+DDEI+ENT subgroup (respectively p= 0.001 p=0.045 p= 0.001 p=0.009 p=0.000 p=0.001).
When we compared levodopa+DDEI subgroup with the levodopa+DDEI+entacapone subgroup we have no found any differences in terms of age, age at disease onset, UPDRS total, UPDRS motor, UPDRS daily life, mean levadopa dosage (respectively p=0.15 P=0.32 P= 0.30 P= 0.32 P=0.38 P=0.38).
In term of CIMT, no difference was detected among the drug subgroups.
The subgroups with Hcy level of > 15 and ≤ 15 µmol/l were compared and Hcy levels and mean levodopa dosage were significantly higher; HDL-C levels were significantly lower in the subgroup with a homocysteine level > 15. In terms of CIMT and UPDRS scores no significant difference was detected between the subgroups (Table 4).
Table 4. Comparison of patients with homocysteine levels ≤15 and >15 µmol/l of subgroups| Homocysteine | p | ||
| >15 µmol/l(N= 15) | ≤15 µmol/l (N=40) | ||
| Age (year) | 70.93±10.4 | 68.43±8.12 | 0.23 |
| Gender n (%) Male | 12 (%21.8) | 25 (%45.45) | 0.2 |
| female | 3 (%5.45) | 15 (27.03) | 0.2 |
| Age of onset of the disease (year) | 66.31±7.8 | 64.44±8.97 | 0.67 |
| Duration of disease (year) | 5.38±3.82 | 4.68±3.80 | 0.35 |
| B12(pg/ml) | 300.65±144.13 | 314.07±167.26 | 0.78 |
| Homocysteine(µmol/l) | 18.19±3.15 | 12.16±2.18 | 0 |
| Folic acid(ng/ml) | 7.65±3.69 | 8.98±5.3 | 0.57 |
| UPDRS total | 36.10±27.19 | 25.66±13.05 | 0.45 |
| UPDRS cognitive | 2.30±1.15 | 1.85±1.37 | 0.29 |
| UPDRS motor sections | 20.60±15.93 | 17.07±8.78 | 0.74 |
| UPDRS activities of daily living | 12±11.06 | 6.44±4.59 | 0.2 |
| UPDRS complication of treatment | 1±1.88 | 0.29±0.86 | 0.1 |
| Mean levodopa dosage (mg/day) | 656.66±318.85 | 446.56±229.56 | 0.01 |
| 1 (H.Y.S.) n (%) | 5 (% 9.25) | 12 (% 22.22) | 0.3 |
| 2 (H.Y.S.) n (%) | 5 (% 9.25) | 21 (%38.88) | 0.3 |
| 3 (H.Y.S.) n (%) | 3 (% 5.55) | 7 (%12.96) | 0.3 |
| 4 (H.Y.S.) n (%) | 1(% 1.89) | 0 | - |
| Total cholesterol (mg/dl) | 193.50±44.82 | 205.96±39.40 | 0.59 |
| Triglycerides (mg/dl) | 136.7±59.09 | 148.62±97.35 | 0.92 |
| HDL-C(mg/dl) | 37.90±11.0 | 47.51±11.47 | 0.04 |
| LDL-C(mg/dl) | 129.33±41.17 | 127.57±30.07 | 0.87 |
| Uric acid(mg/dl) | 6.06±1.90 | 4.7±2.02 | 0.24 |
| CIMT (mm) | 0.78±0.46 | 0.74±0.22 | 0.78 |
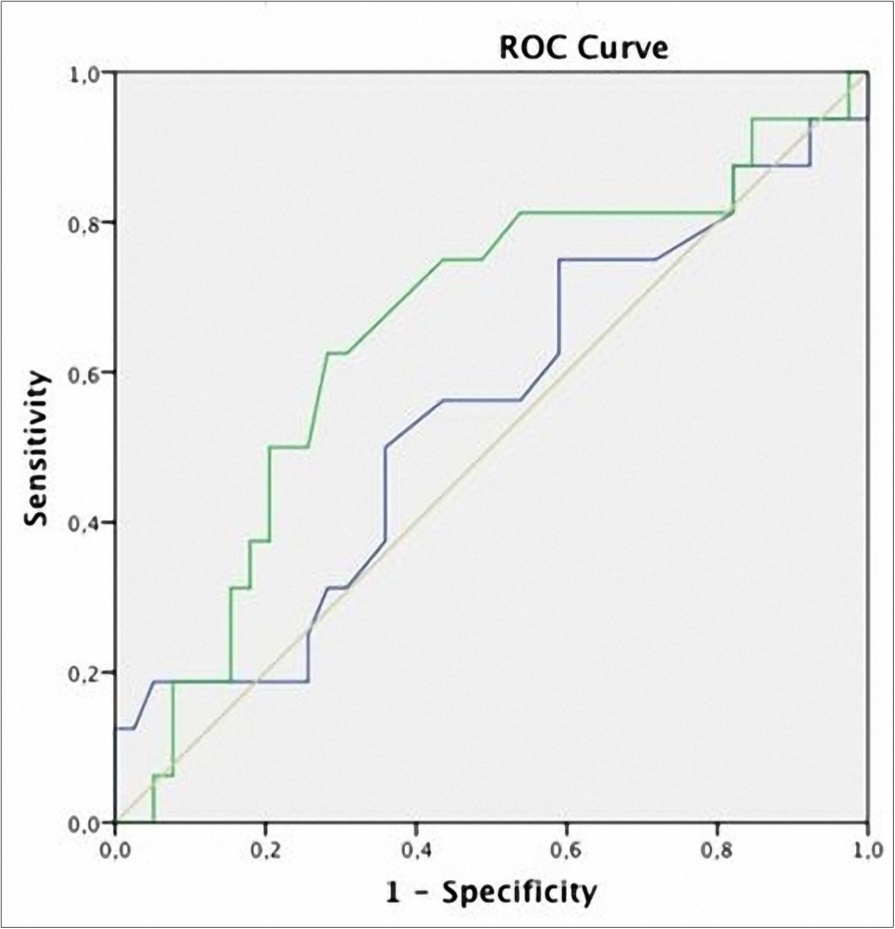
Mean levodopa dosage did not predict CIMT 0.6 mm 14 (AUC: 0.546, 95%CI 0.372-0.720, p=0.59). Homocysteine 14 µmol/l predicted CIMT 0.6 mm with 64% sensitivity and 69% specificity (AUC: 0.654, 95%CI 0.488-0.819, p=0.07)(Figure 1)
Discussion
In our study, homocysteine levels were significantly higher in the Parkinson’s patients group when compared to the healthy controls. There was a positive correlation between CIMT and Hcy. Also positive correlation found between mean levodopa dosage and Hcy levels (r=0.31 p= 0.018). A comparison of the subgroups with levodopa+DDEI, levodopa+DDEI+entacapone, and other drugs showed that the levodopa+DDEI subgroup had higher homocysteine levels than the levodopa+DDEI+entacapone and the other drug subgroups. Homocysteine levels were not significantly different between levodopa+DDEI+entacapone and the other drug subgroups.
F.H Beşir et al. have investigated CIMT levels in healthy Turkish people and found out that the mean CIMT according to the age groups was as follows: 50-59 years of age group 0.58±0 .09 mm, 60-69 years of age group 0.69±0.05mm 70-79 years of age group 0.73±0.06mm 14. In the present study, mean CIMT according to the age group was; 50-59 years of age group 0.65±0.15mm, 60-69 years of age group 0.72±0.09mm, 70-79 years of age group 0.85mm. These results establish that patients with Parkinson’s diseases have overall higher mean CIMT than healthy individuals.Normal thickness of carotid intima-media was 0.6 mm 12 . Homocysteine 14 µmol/l predicted CIMT 0.6 mm with 64% sensitivity and 69% specificity. It was also demonstrated that the mean levadopa dosage does not predict CIMT 0.6 mm.
There was a positive correlation between CIMT and stage. The positive correlation between CIMT and stage might be affected by various parameters including age, disease duration, and the increase noted in mean levodopa dosage by increasing the stage of the disease.
We have found that the mean age, age of onset of the disease as well as duration of disease in the dopamine agonist group were significantly lower in compared to the other drug groups. This can be related in our patients, dopamine agonists and MAO inhibitors were applied usually at early stages in younger patients. In this respect, we have revealed that UPDRS total, UPDRS motor sections and UPDRS activities of daily living of the patients using dopamine agonists were significantly lower than the other drug subgroups
The subgroups with levodopa+DDEI, levodopa+DDEI+entacapone and other drugs were compared each other according to Hcy levels. Hcy levels were significantly higher in the levodopa+DDEI subgroup, as expected. In terms of CIMT, no significant difference was detected between the subgroups of drugs. The levodopa+DDEI subgroup had statistically significantly higher Hcy levels than the levodopa+DDEI+entacapone and the other drug subgroups; however, the mean Hcy level in the levodopa+DDEI subgroup was 14.85±4.06, which is below 15. It is possible that the initiation of therapy with agonist drugs and/or MAO-B inhibitors and the subsequent addition of levodopa at low doses with gradual up-titration of the dosage (mean dosage 566.66 mg/day) might have beneficial effects on maintaining the Hcy levels with in normal limits.
Previous studies have demonstrated an increase plasma homocysteine (Hcy) levels in patients with IPH and associated this increase with the use of levodopa 3, 15, 16, 17. In a previous meta-analysis, plasma Hcy levels were significantly higher in levodopa-treated patients compared to healthy controls and untreated Parkinson’s patients. Homocysteine levels in untreated patients with IPD were not significantly higher compared to healthy controls. Patients with PD treated with levodopa plus catechol-O-methyl transferase inhibitor (COMT-I) showed lower plasma Hcy concentrations compared to levodopa-treated patients 18. Nevrly et al. found that the addition of entacapone to the treatment regimens of patients using levodopa resulted in lower homocysteine levels only when compared to the patients using levodopa 19.In the present study, we demonstrated higher homocysteine levels only in the group using levodopa+DDEI when compared to the group using levodopa+DDEI+entacapone.
Plasma Hcy levels usually range between 5 and 15 µmol/l in healthy adults. Hyperhomocysteinemia (HHcy) may be classified as moderate (15-30μmol/L), intermediate (31-100 µmol/l), and severe (>100 µmol/l) 20. In our study, Hcy levels were 13.80±3.65 in Parkinson’s patients, among which 12 patients were using levodopa+DDEI+entacapone and 13 patients using other drugs. Only half of our patients were using levodopa and dopamine decarboxylase enzyme inhibitors. Hcy elevations are expected to occur more frequently in the patients using this combination; therefore, the levels in these patients might have affected the mean level, which turned out to be low.
Although we have revealeted that Hcy levels were found to be decreased in all of our study groups in compared to the normal population, we have shown that levodopa+DDEI group had significantly increased Hcy levels compared to the other study groups (p<0.05) It can be hypothesized that the mediterian diet style which is common in the black sea region of the Turkey can be reasonable of the decresa value of Hcy.
Hyperhomocysteinemia has been related to an increased risk of cardiovascular disease in several studies 9, 21, 22. Homocysteine contributes to arterial disease through various mechanisms, such as endothelial dysfunction, increased permeability of lipid and lipoprotein oxidation, vascular inflammation, smooth muscle proliferation, and abnormalities in the clotting cascade 23, 24.
There have been a few studies and inconsistent results regarding the coincidence of Parkinson's disease (PD) and atherosclerotic diseases such as cerebrovascular disease. Carotid intima-media thickness is known as a marker for subclinical atherosclerosis 25. Various studies have produced different results for CIMK in Parkinson's disease. Alexa et al. reported that CIMT was higher in PD patients compared to the controls 26. Oncel et al. demonstrated that CIMT was thickened in patients using levodopa compared to those who were not 27.Lee et al. found that CIMT in PD patients was significantly lower than in the controls 25 .Okura et al. did not demonstrate a relationship between CIMK and Hcy in their study 9.
In our study, the comparison of CIMT between the healthy controls and patients with Parkinson’s did not reveal a significant difference, whereas Parkinson’s patients had higher CIMT levels compared to the patients in the control group. There was also a positive correlation between CIMT and Hcy. When we investigated the other parameters that may have possible effects on CIMT in Parkinson’s patients, we noted that there was no correlation between CIMT and age of onset of the disease, duration of disease, UPDRS, and equivalent dose. There was a positive correlation with stage only. Okura et al. found that the factor associated significantly with mean IMT was age 9.We also demonstrated a positive correlation between age and CIMT, although the level of significance was at the limit.
Conclusion:
We have found significantly increased Hcy levels in levodopa +DDEI subgroup which was associated with a nonsignificantly increasement of CMIT. This would impress the importance of clinical and radiological follow up of PD patients with the history of CIMT who are under treatment of levodopa +DDEI.
Finally, regarding the well known strong link between the increased CIMT and atherosclerosis, these above findings may suggest the role of CIMT as a meanful clinical marker for follow-up of patients with IPD. Thus, further studies with larger number of cases are required to explore the association between the homocysteine levels, the clinical course of IPD and the CIMT.
Disclosures:
The authors do not have an affiliation with or financial interest in any organization that might pose a conflict of interest.
Acknowledgments:
None
References
- 1.Ataie A, Ataee R, Mansoury Z. (2013) Homocysteine Intra Cerebroventricular Injection Induces Apoptosis İn The Substantia Nigra Cell Sand Parkinson’s Disease Like Behavior İn Rats. Int J Mol Cell Med Spring. 2(2), 80-5.
- 2.Szadejko K, Szabat K, Ludwichowska A. (2013) Homocysteine And Its Role In Pathogenesis Of Parkinson’s Disease And Other Neurodegenerative Disorders.Przegl Lek. 70(7), 443-7.
- 3.Ozer F, Meral H, Hanoğlu L. (2006) Macitkoldaş plasma homocysteine levels İn Patients treated with levodopa: Motor And cognitive associations. Neurological research. 28, 853-858.
- 4.Zheng Z, Wang J, Yi L. Hindawi Publishing Corporation. Biomed Research International (2014) Correlation Between Behavioural And Psychological Symptoms Of Alzheimer Type Dementia And Plasma Homocysteine Concentration. Doi: 10.1155/2014/383494
- 5.Zhou S, Zhang Z, Xu G. (2014) Notable epigenetic role of hyperhomocysteinemia in atherogenesis. Lipids In Health And Disease 21(13), 134.
- 6.Poredos Pavel. (2004) Intima-Media Thickness: Indicator Of Cardiovascular Risk And Measure Of The Extent Of Atherosclerosis. Vascular Medicine. 9, 46-54.
- 7.Xiang J, Zhang T, Yang Q W. (2013) Carotid artery atherosclerosis is correlated with cognitive impairment in an elderly urban Chinese non-stroke population. , J Clin Neurosci 20(11), 1571-5.
- 8.Okura T, Miyoshi K, Irita J. (2014) Hyperhomocysteinemia Is One Of The Risk factors Associated With Cerebrovascular stiffness In Hypertensive Patients, Especially Elderly Males. Sci Rep. 11(4), 5663.
- 9.Rc Rosen, Riley A, Wagner G. (1997) The International Index Of Erectile Dysfunction (IIEF): A Multidimensional Scale For Assessment Of Erectile Dysfunction. Urology. 49, 822-830.
- 10.Fahn S, Elton R L. (1987) Parkinson’s Disease Rating Scale.In:Fahn S,Marsden CD,Goldsteijn M,Calne DB,Editors.Recent Developments In Parkinson’s Disease.Florham Park, NJ:Macmillan Healthcare Information. P: 153-63.
- 11.Hoehn M M, Yahr M D. (1967) Parkinsonism: Onset, Progression And Mortality. , Neurology 17, 427-42.
- 12.Beşir F H, Yazgan S, Celbek G. (2012) Sağlıklı Erişkinlerde Karotis Intima-Media Kalınlığının Normal Değerleri Ve Etkileyen Parametreler. Anadolu Kardiyol Derg. 12, 427-33.
- 13.Ozer F, Meral H, Hanoğlu L. (2007) Cognitive Impairment Patterns In Parkinson’s Disease With Visual Hallucinations. Journal Of Clinical Neuroscience. 14, 742-746.
- 14.Grosset K, Needleman F, Macphee G, al Et. (2004) Switching from ergot to nonergot dopamine agonists in Parkinson’s disease: a clinical series and five-drug dose conversion table. , Mov Disord 19(11), 1370-4.
- 15.Yasui K, Kowa H, Nakaso K. (2000) . Plasma Homocysteine And MTHFR C677T Genotype In Levodopa-Treated Patients With PD. Neurology 55, 437-40.
- 16.Kuhn W, Roebroek R, Blom H. (1998) Elevated Plasma Levels Of Homocysteine In Parkinson’s Disease. Eur Neurol. 40, 225-7.
- 17.Shin J Y, Ahn Y H, Paik M J. (2012) Elevated Homocysteine By Levodopa Is Detrimental To Neurogenesis In. Parkinsonian Model. Doi: 10.1371. Plosone 7(11), 50496.
- 18.Hu X W, Qin S M, Li D. (2013) Elevated Homocysteine Levels İn Levodopa-Treated İdiopathic Parkinson's Disease: A Meta-Analysis. Acta Neurol Scand. 128(2), 73-82.
- 19.Nevrly M, Kanovsky P, Vranova H. (2010) Effect Of Entacapone On Plasmahomocysteinelevels İn Parkinson’s disease patients. Neurol Sci. 31(5), 565-9.
- 20.Kaul S, Zadeh A A, Shah P K. (2006) Homocysteine Hypothesis For Atherothrombotic Cardiovascular Disease: Not Validated. J Am Collcardiol. 48, 914-923.
- 21.Faria-Neto J R, Chagas A C, Bydlowski S P. (2006) Hyperhomocystinemia In Patients With Coronary Artery Disease. Braz J Med Biol Res. 39(4), 455-63.
- 22.Agata R, Pawel P J, Wojciech K. (2013) Homocysteine Level And Mechanisms Of Injury In. Parkinson’s Disease As Related To MTHFR, MTR, And MTHFD1 Genes Polymorphisms And Ldopa Treatment. Current Genomics 14, 534-542.
- 23.Domagala T B, Undas A, Libura M.et al.(1998). Pathogenesis Of Vascular Disease In Hyperhomocysteinaemia. , J Cardiovasc Risk 5, 239-247.
- 24.Faraci F M.Lentz SR.(2004) Hyperhomocysteinemia, Oxidative Stress, And Cerebral Vascular Dysfunction. , Stroke 35, 345-347.
- 25.Lee J M, Park K W, Seo W K. (2007) Carotid Intima-Media Thickness In Parkinson’s Disease.Movdisord. 22(16), 2446-9.
Cited by (2)
This article has been cited by 2 scholarly works according to:
Citing Articles:
M. S. Tai, Kuo Ghee Ong, T. Toh, Hafez Hussain, Abdul Rashid Mat Mahidin et al. - Inflammatory Heart Diseases (2019) Semantic Scholar
IntechOpen eBooks (2019) OpenAlex