Severe Ischemic Stroke due to Progression of Cervical Carotid Artery Dissection
Abstract
Spontaneous dissection of the cervical segment of the internal carotid artery (CS-ICA) is uncommon but not rare. A 43-year-old man suddenly developed complete right hemiparesis with motor aphasia. 3 weeks after the initial symptom onset. Magnetic resonance imaging showed enlargement of an intramural hematoma at the cervical carotid artery; it severely compressed and completely occluded the arterial lumen. Diffuse ischemia was observed in the area of the left middle cerebral artery (MCA) immediately after onset and he underwent emergency left superficial temporal artery-MCA double bypass within 6 hours. There was no fatal deterioration and his neurological deficit was resolved. We discuss our treatment of spontaneous dissection of the CS-ICA to prevent ischemic damage.
Author Contributions
Academic Editor: ESRA DOGRU HUZMELI, MUSTAFA KEMAL UNIVERSITY SCHOOL OF PHYSICAL THERAPY AND REHABILITATION
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2017 Nobuhiro Inoue, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Earlier population-based studies on the incidence cervical internal carotid artery (ICA) dissection revealed an annual incidence rate of 2.6 - 2.9 per 100,000 individuals. Cervical ICA dissection is among the most common contributors to stroke in young and middle-aged adults (under 45 years) without vascular risk factors. They are less likely to have cerebrovascular atherosclerosis; approximately 2% of overall ischemic stroke is seen in that population.1, 2, 3, 4, 5 Vertebral artery dissection (VAD) tends to be less common than cervical ICA dissection in Europe and America, however, reliable epidemiologic data are not available.6, 7, 8 According to a Japanese study, the most common site of dissection in the Japanese was the intracranial vertebro-basilar artery;9 dissection of the cervical segment of the internal carotid artery (CS-ICA) is rarely encountered. In more than 85% of the dissected vessels, ICA stenosis resolved or improved substantially3, 10, 11 and 92% of patients had a good treatment outcome.12 As most patients tend to do well regardless of the type of treatment,8, 10, 13 identification of the optimal treatment in patients with ICA dissection is difficult.6, 14, 15 We encountered a 43-year-old Japanese male with sudden-onset severe ischemic stroke attributable to the progression of spontaneous dissection of the CS-ICA that had been diagnosed 3 weeks earlier.
Case Report
Prior written consent for the publication of our findings was obtained from the patient.
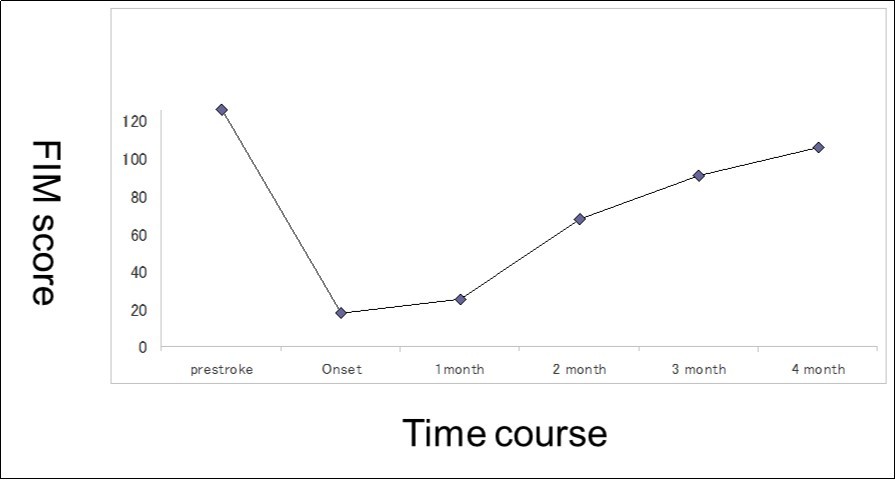
This 43-year-old Japanese male presented with right hand dysesthesia. He has not recognized his neck pain and Horner’s syndrome( oculosympathetic palsy). Magnetic resonance imaging(MRI) performed on a 3T TX scanner (Philips Achieva, The Netherlands) demonstrated dissection of the left cervical carotid artery (CCA; Figure 1A,1B ); no ischemic area was detected in the left cerebral brain. Axial section views of the CCA segment revealed a typical crescent hematoma; a bright, hyper-intense circle representing the narrowed arterial lumen was visualized. A T1-volumetric isotropic turbo spin echo acquisition (VISTA) sequence resulted in the diagnosis of dissection of the CS-ICA (Figure 1C). He had no conventional stroke risk factors such as hypertension, diabetes, hyperlipidemia, a history of cardiac valvular disease, arterial fibrillation, or features of inherited connective tissue disorder. Also absent was a history of arterial trauma and we were unable to identify any etiological factor(s). One week later he developed Gerstman symptoms; border zone ischemia was observed in the left cerebrum (Figure 2-Ic). He was admitted and the administration of antiplatelet agents was started. The left middle cerebral artery (MCA) was supplied via antegrade circulation (Figure 2-Ib) and an increase in the flow-void intensity was noted (compare Figure 1C with Figure 2-Ia). MRI studies acquired one week later revealed spreading of the ischemic area to beyond the watershed zone between the left anterior cerebral artery (ACA) and the MCA, and the MCA and the posterior cerebral artery (PCA) (Figure 2-IIc). The area of dissection had spread from the ICA bifurcation to the top of the ICA. The blood supply to the left MCA territory derived from the circle of Willis (Figure 2-IIb). The flow-void intensity had increased further (compare Figure 2-Ia with Figure 2-IIa). Two weeks after his admission he suddenly experienced complete right-sided hemiparesis with motor aphasia. MRI performed within 45 min demonstrated diffuse ischemia in the left MCA territory (Figure 2-IIIc), the left A1 segment could not be visualized. The left MCA and its A1 segment were completely occluded (Figure 2- IIIb). The flow-void intensity indicated complete occlusion of the true lumen (Figure 2-IIIa). He underwent emergency left superficial temporal artery (STA)-MCA bypass surgery within 6 hours; double anastomosis was successful. External decompression was performed to prevent neurological deterioration due to severe brain swelling. An intensive rehabilitation has been continued for him after operation. His functional independence measure (FIM) score recorded over the course of 4 months showed remarkable improvement ( Figure 3) as did the standard language test for aphasia.
Figure 1.First hospitalization. A. Maximum intensity projection ( MIP )image of arteries in the cervical segment. B. Magnetic resonance angiography of the left carotid artery. C. T1 VISTA sequence. Note the flow void reflective of the narrowed lumen next to intramural hematoma (hyperintense crescent)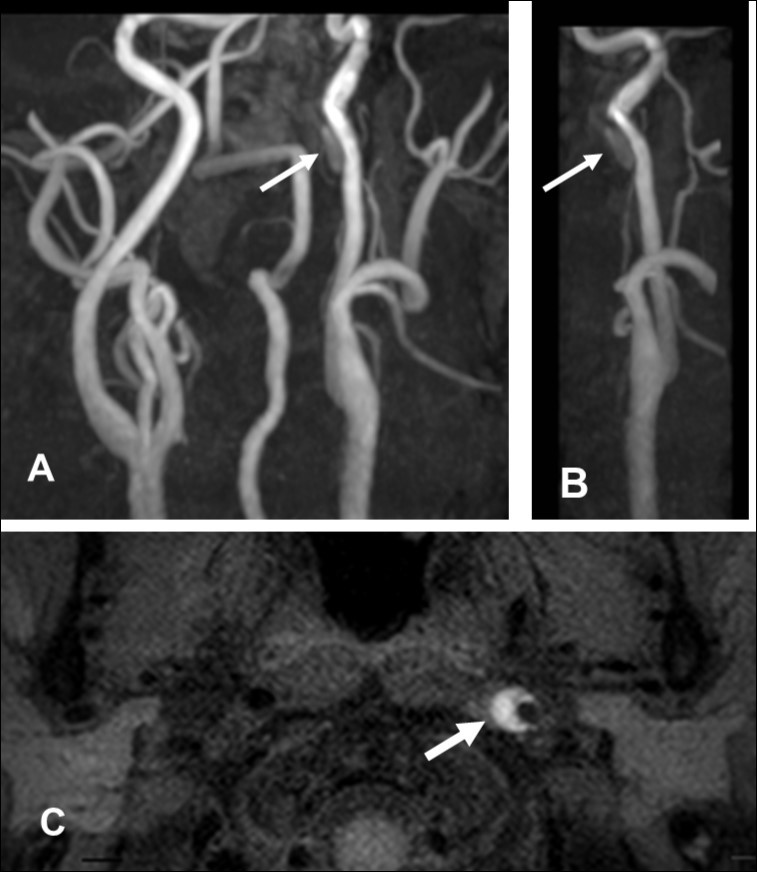
Figure 2.Time course Columns I, II, and III are images obtained 1, 2, and 3 weeks, respectively, after the start of dissection of the cervical segment of the internal carotid artery. Ia, IIa, IIIa.T1 VISTA images of the same segment shown in Fig. 1C. The dark area reflective of the low void progresses to hyperintensity by week 3. IIIa shows complete occlusion of the true lumen by the intramural hematoma (arrow ). Ib, IIb, IIIb. TOF-MRA images of the intracranial arteries. (a) The left MCA is supplied via antegrade flow from the left carotid artery (arrow). (b) Flow from the left carotid artery disappeared from the ICA bifurcation to the top of the ICA. Blood supply to the left MCA derives from the circle of Willis via the anterior- or the posterior communicating artery (arrows). By week 3, flow to the left MCA, even via the left A1 segment of the anterior cerebral artery had disappeared completely. Ic, IIc, IIIc. Diffusion-weighted images at the basal ganglia level. Week 1: a few hyperintense areas are visualized in the border zone of the left cerebrum (arrow). Week 2: Note the hyperintensity area in the watershed zone between the left ACA and the MCA, and between he MCA and the PCA (arrow). The image acquired on week 3 shows a diffuse hyperintensity area in the left MCA territory (arrow).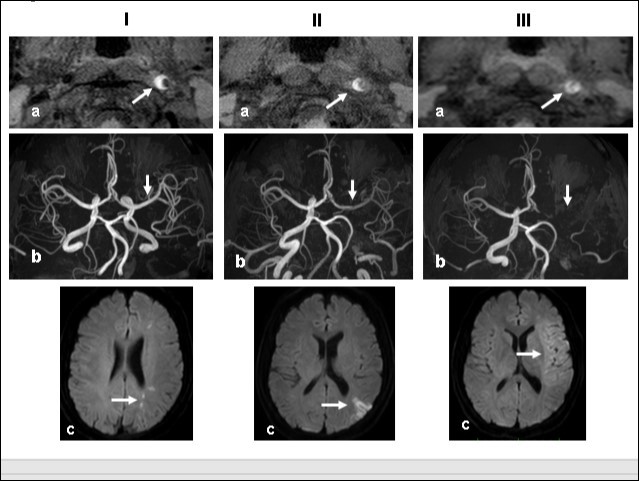
Discussion
This is a first report which was able to detect the progression of the dissection of cervical carotid artery chronologically within few weeks beyond to the severe ischemic stroke. In regard with the treatment of the cervical carotid artery dissection has been addressed by the administration of anticoagulants or antiplatelet agents, and by surgical procedures such as removal of the intramural hematoma, arterial resection with vein graft replacement, endovascular treatment consisting of percutaneous ballon angioplasty and stent placement, by occlusion of the distal extracranial carotid artery, and by superficial temporal artery-MCA bypass.1, 6, 10, 16, 17 The usefulness of antithrombic therapy has not been validated7, 13, 14, 18 and no evidence-based consensusguidelines for surgical intervention are available.14, 15, 17. In fact, the dissection of CS-ICA progressed despite of the antiplatelet agent administration in our case. Stent placement procedures may result in embolization from displaced intraluminal or intramural thrombi16, 17and surgical outcomes still need to be carefully evaluated.12 Although fewer than 5% of patients with dissection of the CS-ICA died due to massive cerebral infarct or edema,3, 6, 7, 14 4 - 5% were at risk for stroke recurrence in the first 2 weeks after CS-ICA dissection.13, 19 According to Morris et al.,20 in the absence of ischemia at the time of diagnosis, the risk of stroke after cervical artery dissection is limited to the first 2 weeks post-dissection, suggesting that stroke-prevention therapy should be started as early as possible after the onset of the first symptoms.21
In our patient, the left MCA was fed by scantblood flow through the circle of Willis until 2 weeks after the start of arterial dissection. Further one week later, the carotid artery was completely occluded and this led to severe ischemic stroke in the territory of the left MCA unexpectedly. The mechanism(s) underlying cerebral ischemic symptoms is thought to be hemodynamic at first as a result of significant stenosis or occlusion of the carotid artery involved by dissection extending to the A1 segment.6, 22. In our case, immediate STA-MCA anastomosis was completed within the therapeutic time window23, 24, 25 and narrowly escaped fatal deficits developed. There has not been established authentic strategies for the spontaneous dissection of the CS ICA to prevent complete stroke yet. In conclusion, patients with spontaneous dissection of the CS ICA is needed to treat optimally within few weeks after initiation of the dissection.
References
- 1.Ehrenfeld W K, Wylie E J. (1976) Spontaneous dissection of the internal carotid artery. , Arch Surg 111, 1294-130.
- 2.Schievink W I, Mokri B, Whisnant J P. (1993) Internal Carotid artery dissection in acommunity Rochester. , Minnesota,1987-1992 Stroke 24, 1678-1680.
- 3.Schievink W I, Mokri B, O’ Fallon WM. (1994) Recurrent spontaneous cervical artery dissection. , N Engl J Med 330, 397-397.
- 4.Giroud M, Fayolle H, Andre N, Dumas R, Becker F et al. (1994) Incidence of internal carotid artery dissection in the community of Dijon. , J Neurol Neurosurg Psychiatry 57, 1443.
- 5.Zhang F-L, Guo Z N, Liu Y, Luo Y, Yang Y.Dissection extending from extra-to intracranial arteries. A case report of progressive ischemic stroke. , Medicine 96(21), 1-4.
- 6.Mokri B. (1997) Spontaneous dissections of internal carotid arteries. , The Neurologist 3, 104-119.
- 7.Schievink W I. (2001) Spontaneous dissection of the carotid and vertebral arteries. , N Engl J Med 344, 898-906.
- 8.Lee V H, Brown R D, Mandrekar J N, Mokri B. (2006) Incidence and outcome of cervical artery dissection. A population-based study. , Neurology 67, 1809-1812.
- 9.Takagi Makoto. (2005) Cerebral arterial dissections. (in Japanese with English Abstract) , Clin Neurol 45, 846-848.
- 10.Mokri B, Sundt T M, Houser W, Piepgras D G. (1986) Spontaneous dissection of the cervical internal carotid artery. Ann Neurol. 19, 126-13.
- 11.Bassetti C, Carruzzo A, Sturzenegger M, Tuncdogan E. (1996) Recurrence of cervical artery dissection. A prospective study of 81 patients. , Stroke 27, 1804-1807.
- 12.Hart R G, Easton J D. (1983) Dissections of cervical and cerebral arteries. , Neurol Clin 1, 155-182.
- 13.Arauz A, Hoyos L, Espinoza C, Cantu C, Roman G Barinagarrementeria. (2006) Dissection of cervical arteries: long -term follow-up study of 130 consecutive cases. Cerebrovasc Dis. 22, 150-154.
- 14.Saver J L, Easton J D, Hart R G. (1992) Dissections and trauma of cervicocerebral arteries In:. Barnett HJM et sl editors Stroke, pathophysiology, diagnosis, and management. 2nd ed , London: Churchill Livingstone 671-688.
- 15.Mokri B. (1994) Dissections of cervical and cephalic arteries. Sundt’s occlusive cerebrovascular disease. 2nd ed. Philadelphia:WB Sander. 45-70.
- 16.Marks M P, Dake M D, Steinberg G K, Norbash A M, Lane B. (1994) Stent placement for arterial and venous cerebrovascular disease: Preliminary experience. , Radiology 191, 441-446.
- 17.Beletsky V, Nadareishvili Z, Lynch J, Shuaib A, Woolfenden A.Norris JW for the Canadian Stroke Consortium. Cervical arterial dissection. Time for a therapeutic trial?. , Stroke2003; 34, 2856-2860.
- 18.CADISS nonrandomized arm and meta-analysis. Antiplatelets vs anticoagulation for dissection. , Neurology2012; 79, 686-689.
- 19.Weimar C, Kraywinkel K, Hagemeister C, Haaβ A, Katsarava Z et al.Schmid E, Diener H-C, on behalf of the German Stroke Study Collaboration. Recurrent stroke after cervical artery dissection. , J Neurol Neurosurg Psychiatry2010; 81, 869-873.
- 20.Morris N A, Merkler A E, Gialdini G, Kamel H. (2017) Timing of incident stroke risk after cervical artery dissection presenting without ischemia. , Stroke 48, 551-555.
- 21.Biousse V, D’Anglejan- Chatillon J, Touboul P-J, Amarenco P, Bousser M-G. (1995) Time course of symptoms in extracranial carotid artery dissections. A series of 80 patients. , Stroke 26, 235-239.
- 22.Lucas C, Moulin T, Deplanque D, Tatu L, Chavot D.and the DONALD Investigators. Stroke patterns of internal carotid artery dissection in 40 patients. , Stroke1998; 29, 2646-2648.
- 23.Jones T H, Morawetz R B, Crowell R M, Marcoux F W, FitzGibbon S J et al. (1981) Thresholds of focal cerebral ischemia in awake monkeys. , J Neurosurg 54, 773-782.
Cited by (1)
This article has been cited by 1 scholarly work according to:
Citing Articles:
Journal of Neurological Research And Therapy (2017) OpenAlex