Disability and prognostic factors of MS severity: An Algerian cohort study
Abstract
Background
The evolution of multiple sclerosis (MS) is highly variable. Predicting this evolution at the beginning of the disease will help in the therapeutic management. The objective of this study was to describe the clinical characteristics and to identify early predictors of long-term disability among MS patients in Algeria.
Methods
We performed a descriptive and retrospective study of 400 MS patients followed over a 4-years period from July 2012 to July 2016 in the neurology department of Mustapha Bacha hospital in Algiers. The following parameters were systematically assessed for each patient: Age at onset, gender, relapses, initial demyelinating event, interval between first and second attack, residual deficit after first attack, onset of the progressive course, time of assignment of the successive scores of irreversible disability and type of disease modifying therapies. Univariate and multivariate Cox models were used to determine factors influencing time to Expanded Disability Status Scale (EDSS) 4 and 6.
Results
During the follow-up of this cohort of MS patients, a total of 144 (36%) and 83 (20%) patients reached EDSS scores 4 and 6 respectively. The median times from the onset of MS to assignment of a score 4 and 6 was 10 years and 19 years, respectively. Using the MSSS severity score, and after univariate analysis we identified several risk factors predictive of disease severity. These included male sex (P= 0.002), age of onset ≥ 40 years (P= 0.001), pyramidal (P= 0.0001), cerebellar (P= 0.002), and poly-symptomatic (P=0.0001) onset of the disease, incomplete recovery after the first attack (P= 0.0001), a high number of attacks during the first 2 years (P= 0.04) and the progressive form (P=0.001), and all these factors were correlated with a high MSSS score. However, prognostic factors in the multivariate binomial logistic regression analysis were limited to pyramidal onset, incomplete recovery after the first attack, short interval between the first and second attack, and progressive onset.
Conclusion
On the whole, the early predictive factors of disability in MS among Algerian patients were age of onset over 40 years, pyramidal onset, incomplete recovery after the first attack, short interval between the first and second attack and progressive form at onset. To increase the generalizability of findings, a national MS registry is strongly recommended, as well as long-term follow-up epidemiological studies.
Author Contributions
Academic Editor: Mourad Tayebi, The University of Surrey
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2023 N.HECHAM.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Multiple sclerosis (MS) is a demyelinating inflammatory and degenerative disease that is the most frequent cause of disability in young adults 1. Genetic predisposition, unknown environmental factors, or a combination of both may explain the variable prevalence of MS in the world 2, 3. Also, ethnicity may affect the frequency of MS among people living in the same area 4. Migration from a low to a high-risk area before the age of fifteen increases the MS risk 5, 6.
MS in North Africa (NA) has seen epidemiological and phenotypic variation over the past few decades. Based on the Kurtzke classification, Algeria is located in a low-risk zone for MS 7. Over the past decade, MS is relatively common but scarcely documented in Algeria, but our experience in the practice suggests that the occurrence of MS is increasing. This notion was raised in a recent study that estimates up to 2.8 million people are living with MS worldwide (35.9 per 100,000 population) and that MS prevalence has increased in every region of the world since 2013,
A particularly severe profile of MS has been demonstrated in several studies in NA countries (Morocco, Tunisia and Algeria) 9, 10, 11, 12, 13. Some of these studies have been carried out on multicenter cohorts. Predicting this evolution at the onset of the disease will help in therapeutic management. Most of the studies evaluating the predictive factors of disability have been performed in Western populations and they are identical in all these populations 14, 15, 16. A late onset disease, male gender, incomplete recovery from first episode, short interval between first and second episodes, high number of attacks during the first two years, and motor, cerebellar or sphincter symptoms at onset were associated with poor prognosis. However, these predictors of disability have not been studied in Arab populations, with the exception of the Yamout study which found that the predictors of disability in MS among the Lebanese population were not very different from predictors in western countries 17. The purpose of this study was to describe the clinical characteristics and to identify early predictors of long-term disability among MS patients in Algeria.
Methods
We performed a descriptive and retrospective study of MS patients seen at our MS clinic, followed over a 4-year period from July 2012 to July 2016 in the neurology department of University hospital Mustapha Bacha in Algiers. All patients gave informed consent for their data to be used and the study was approved by the local research ethics committee.
We systematically reviewed medical records of each subject. Subjects whose records were incomplete, did not confirm a diagnosis of clinically definite MS were excluded. Only the cases with definite MS according to Mc Donald’s classification were included in the cohort (dissemination in space and time were required: clinical presentation or additional data needed for MS diagnosis). These patients were classified according to international accepted disease course classification 18: relapsing-remitting (RR), secondary-progressive (SP) and primary progressive (PP).
A series of clinical variables were systematically assessed for each patient. They included: gender, medical history, key episodes in the course of Ms including relapses, onset of the progressive course, expanded EDSS, time of assignment of the successive scores of irreversible disability and type of disease modifying therapies, The initial demyelinating event was categorized by site (optic nerve, spinal cord, brain stem or cerebellum) and polysymptomatic signs. Recovery from the first relapse was classified as incomplete (persistence of neurologic signs) or complete (absence of neurologic signs). To determine the extent of neurologic disability, the EDSS score (range 0 to 10) was recorded during each visit of the patient to the neurologist. Disability was defined as irreversible when a given score persisted at least 6 months, excluding transient worsening of disability related to relapses. The Multiple Sclerosis Global Severity Score MSSS) was originally developed to compare the relative severity of disease across all levels of EDSS for a duration of disease, using a single clinical assessment at one point in time 19.
MRI of the brain was carried out in all patients, T1, T2 and flair sequences were performed. The cerebrospinal fluid (CSF) was examined to look for oligoclonal bands (OCB) to support the diagnostic in certain subjects.
Statistical analysis was conducted using SPSS for windows version 13.0, Comparison of categorical data was made according to the chi-squared test. The quantitative variables were presented as number of patients (%), the value of demographic data (gender, age, age at onset of MS), clinical data (course at onset of MS and initial symptoms, time to diagnosis, time from onset of MS to the second neurologic episode, extent of recovery from the first relapse, number of relapses during the first 2 years of the disease) were analyzed. The ANOVA test was used for the comparison of several means. The Kaplan-Meier technique was used for estimating the time to the assignment of an irreversible score of EDSS 4 and EDSS6. Survival curves were compared using the log-rank test.
Results
Characteristics and initial course of the patients:
The demographics and clinical characteristics of the 400 patients (299 females, 101 males) registered in the data base are presented in Table 1 and Table 2. Overall, the sex ratio female: male was 2.9, the mean patients age was 39,86 (95%CI= (30.16 – 37.36)) years. The mean age at MS clinical onset was 31.2 (IC 95% = (30.27- 32.14)) years, and the mean duration of follow-up from multiple sclerosis clinical onset to the last visit was 33.76 (IC 95%= (30.16 – 37.36)) months. The initial course of the disease was relapsing-remitting in 362 patients (90.5%), whereas it was progressive in 38 (9.5%) patients. In the first group, 309 patients (77.2%) had residual deficit after the first relapse, and 197 patients (49.2%) had two relapses or more within the first 2 years after clinical onset (mean number of relapses during the first 2 years of MS was: 1.5 ± 0.8). The main results for relapsing-remitting and progressive initial courses of MS are compared in table 2. The two groups differ in several respects: the first group was characterized by a greater female preponderance (p=0.001), an earlier onset of MS (p< 0.0001). Review of the patient’s initial symptoms found that the primary progressive group experienced motor weakness, cerebellar and polysymptomati symptoms more frequently (95%- 47.4%- 50%) than those with relapsing-remitting MS (52%- 33.4% - 28%) (P= 0.001). OCB were analyzed in 396 patients. OCB in the CSF were found in 78% of patients. Initial cerebral MRI was studied in all patients and spinal MRI in 144 patients. Three to four Barkhof criteria were fulfilled in 93.5% of patients and spinal lesions were found in 84.7%.
Table 1. Demographic and clinical analysis of MS dataset| Characteristics | (Value n=400) |
| Gender, n (%) | |
| Male | 101 (25) |
| Female | 299 (75) |
| Age at onset of MS : No (%), years | |
| < 20 | 48 (12) |
| 20 to 39 | 267 (66,7) |
| 40 to 49 | 74 (18.5) |
| ≥ 50 | 11 (3) |
| Mean ± SD | 31,2 ± 9,4 |
| Mean ± SD disease duration ( years) | 8,6 ± 6,1 |
| Range | (1-30) |
| Initial symptoms : no of patients (%) | |
| Visual disturbance | 100 (25) |
| Motor weakness | 224 (56) |
| Sensory disturbance | 140 (35) |
| Cerebellar | 139 (34,7) |
| Brainstem | 82 (21) |
| Sphincter disturbance | 32 (8) |
| Combined symptoms | 119 (30) |
| Disease course: no of patients (%) | |
| Relapsing-remitting | 299 (74,7) |
| Secondary progressive | 63 (15,7) |
| Primary progressive | 38 (9,5) |
| Proportion of incomplete recorvery from the first relapse, % | 112 (28) |
| Time from MS onset to diagnosis, month, mean ± SD | 33,7 ± 3.6 |
| Median (25 and 75 quartile estimate of median) | 14,5 (1 - 160) |
| Median time to EDSS score of 4, year (25 and 75 quartile estimate of median) | 10 (8,6 – 11,4) |
| Median time to EDSS score of 6, year (25 and 75 quartile estimate of median) | 19 (14,2 – 23,7) |
| MSSS, median (IQR) | 4,3 (3,9- 4,5) |
| Progression index, mean (IQR) | 0.49 (0,44- 0,54) |
| Positive OCB, n (%) | 309 (78) |
| Positive Barkhof criteria in initial MRI, n (%) | 373 (93,5) |
| All MS | RRMS | PPMS | P Value a | |
| Number | 400 | 362 | 38 | |
| Number of patients who had reached | 0,0001 | |||
| EDSS 4 | 144 | 110 | 34 | |
| Censored, n (%) | 256 (64) | 252 (69,6) | 4 (10,5) | |
| EDSS 6 | 83 | 57 | 26 | |
| Censored, n (%) | 317(80) | 305(84,3) | 12(32) | |
| Median time (years)b from disease onset to | ||||
| EDSS 4 | 10 (8,6 – 11,4) | 12 (9,9 – 14,1) | 4 (3,4 – 4,5) | 0,0001 |
| EDSS 6 | 19 (14,2 – 23,7) | 20 (14,2-23,7) | 9 (7,4-10,6) | 0,0001 |
| MSSS ≤ 5, n (%) | 232 (58) | 229(99) | 3(1) | 0,0001 |
Progression and assessment of disability:
During the follow-up of the 400 patients, a total of 144 (36%) and 83 (20%) patients reached EDSS scores 4 and 6 respectively. The median times from the onset of MS to assignment of a score 4 and 6 was 10 (95% confidence interval (CI) = 8.6- 11.4) years and 19 (95% confidence interval (CI)= 14.2- 23.7) years, respectively. The median interval from onset of disease to the assignment of each of these scores was significantly shorter in the group of patients with a progressive initial course of disease than among those who had relapsing–remitting disease at onset ( Figure 1. a, b, Table 2). It was 12 vs 4 years and 20 vs 9 years P<0, 0001, respectively. 57 patients with relapsing onset (15,7%) had converted to secondary progressive form. The mean time to the SP form was 22 years ((95% CI = (19.9 - 24.1)).
Considering EDSS and disease duration, we calculated 2 scores which are the progression index (EDSS to disease duration ratio) and the MSSS score (algorithm also considering EDSS and disease duration). The median progression index in our cohort was 0.33. The progression index was significantly shorter in patients having a progressive form than in patients with a relapsing–remitting onset (p = 0.001). The median MSSS score was 4.26. 42% of patients had rapid disease progression with a MSSS score > 5. The onset of the disease with pyramidal, cerebellar, and poly-symptomatic involvement was correlated with a MSSS score >5 (P<0.05).
Figure 1.a) Kaplan–Meier estimates of the time from the onset of multiple sclerosis to the assignment of a score of 4 and 6 on the EDSS. b) Kaplan–Meier estimates of the time from the onset of multiple sclerosis to the assignment of a score of 6 on the EDSS according to the initial course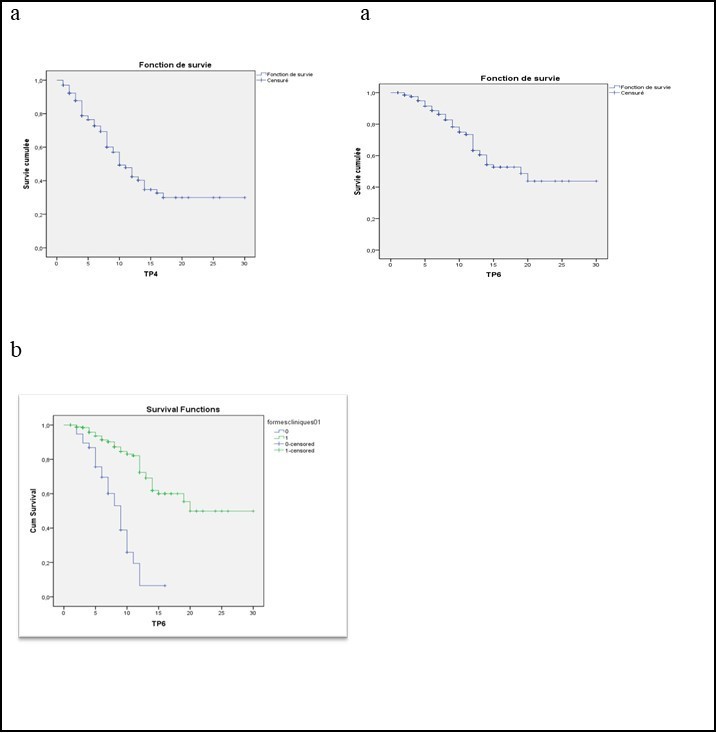
Factors influencing disability progression
The variables included in the multivariate Cox models to estimate the most predictive risk factor(s) for disability (EDSS 4 and EDSS 6) were: age of onset, sex, pyramidal involvement, cerebellar involvement, poly-symptomatic involvement, clinical form, number of relapses in the first two years, interval between the first and second years (P1-P2), and incomplete recovery or sequelae after the first relapse. Combinations of all variables were performed for at least two predictors. Variables included in the logistic regression were significant at P<0.05. This analysis showed that 04 factors are predictive of disability (Table 3): age of onset over 40 years, pyramidal onset, incomplete recovery after the first attack, progressive form at onset.
Using the MSSS severity score, and after univariate analysis we identified several risk factors predictive of disease severity. These included male sex (P= 0.002), age of onset ≥ 40 years (P= 0.001), pyramidal (P= 0.0001), cerebellar (P= 0.002), and poly-symptomatic (P=0.0001) onset of the disease, incomplete recovery after the first attack (P= 0.0001), a high number of attacks during the first 2 years (P= 0.04) and the progressive form (P=0.001), and all these factors were correlated with a high MSSS score. However, prognostic factors in the multivariate binomial logistic regression analysis were limited to pyramidal onset, incomplete recovery after the first attack, short interval between the first and second attack, and progressive onset.
Treatment data.
A total of 330 (82.5%) received disease modifying drugs for at least 6 months. Treatment was started 25.1 ± 13.4 months after diagnosis of multiple sclerosis on average, and consisted of beta-interferon (63.5%), Natalizumab (5.7%), cyclophosphamid (10.5%) and azathioprine (2.7%). The mean duration of interferon treatment was 3.16 years (95 CI %= 2.90 - 3.41).
Table 3. Cox proportional hazard models for time to Expanded Disability Status Scale 6| Univariate analysisHazard ratio 95% CI p Value | Multivariate analysis Hazard ratio 95% CI p Value | |||||
| Age at onset > 40 years | 1.81 | 1.19 – 2.76 | 0,05 | 1.71 | 1.12 – 1.61 | 0.012 |
| Female sex | 0.82 | 0.56 – 1.20 | 0.315 | |||
| Pyramidal initial symptoms | 1.62 | 0.19 – 0.61 | 0.031 | 1.68 | 1.10 – 2.58 | 0.016 |
| cerebellar initial symptoms | 1.21 | 0.69 – 2.13 | 0.488 | |||
| Poly symptomatic initial symptoms | 1.15 | 0.64 – 2.06 | 0.622 | |||
| Progressive form at onset of MS | 2.10 | 1.35 – 3.27 | 0.001 | 2.10 | 1.35 – 3.27 | 0.001 |
| Incomplete recovery from the first relapse | 3.15 | 2.15 – 4.62 | 0.0001 | 3.15 | 2.15 – 4.62 | 0.0001 |
| No. of relapses during the first 2 years of the disease, | 1.49 | 0.86 – 2.58 | 0.149 | |||
| interval between the first 2 relapses | 1.13 | 0.93 – 1.36 | 0.191 | |||
Discussion
Our study has shown that Algerian patients have similar characteristics to European and North Africans patients: predominantly female, average age of onset 30 years, relapsing–remitting onset in almost 90% of our cohort 20, 21, 22. However, some specific features were found: Isolated motor dysfunction was the most frequent onset symptom, exceeding visual and sensory signs, an initial poly-symptomatic involvement was relatively as frequent (30%), and recovery from the initial neurological episode was found in about one-third of the patients. These phenotypic characteristics underline the particular severity of North African MS. Indeed, originating from North Africa has been described in some publications as a factor of severity for MS 9, 10, 11, 23. The course of MS was more severe compared to European patients such that the EDSS 4 and 6 scores were readily reached presaging a more severe disease outcome. Thus, the study of Sidhom 12 revealed that the disability progression profile was more rapid in North African patients than in European patients. It was shown after statistical study by the Cox method that ethnic origin was a risk factor for reaching EDSS 6. The severity of the disease was more common in patients born in France and whose parents were of North African origin, reflecting genetic and environmental influences on the progression of the disease. For disability measures, the overall MSSS distribution in our cohort is similar to European and US data 19, 24. In a Tunisian and European study 12, the MSSS was 4.8 and 5.1. The same result is observed for the progression index which is similar to our cohort, it was 0.51 and 0.41. In contrast, the Moroccan study found a higher MSSS score and progression index (6.39 and 0.97) 10. The median time to reach EDSS 4 was similar to that reported in the North African studies and was shorter than in the European studies 10, 11, 22, 24. The median time to EDSS 6 (19 years (95% CI = (14.2 - 23.7)) was relatively short compared with European cohorts. It was 27.9 years in Canada 26, 24 years in the United States 27 and between 20 and 24 years in France 28, 23. The evolution of MS is therefore very variable, ranging from a severe disease that is rapidly disabling to a mild form that causes minimal long-term disability. Predicting this evolution at the beginning of the disease will help in the therapeutic management. Most of the studies evaluating the predictive factors of disability have been performed in Western populations and they are identical in all these populations 27, 29. However, these predictive factors of long-term disability have not been studied in Arab populations, with the exception of the Yamout study in Lebanon 17.
Late age at onset is considered predictive of early progression to disability in most studies 29, 30, 31. The presence of pyramidal and cerebellar signs has been described in several studies as a fact associated with a poor long-term prognosis 31, 21, 25. Incomplete recovery after the first attack is also predictive of poor long-term disability and secondary progression 28, 32. We also confirmed that the median time to irreversible disability (EDSS 4 or 6) was shorter in patients with a progressive form than in those with a remittent form. This result is in agreement with previous studies 33. A short interval between the first and second attacks (P1-P2) was also considered to be a poor prognosis. Scalfari et al. found a statistically significant association between P1-P2 interval and disability outcome 34. However, these authors described this result as a less predictive factor than the number of relapses in the first two years. Other studies 25, 28 reported similar results, showing that a longer P1-P2 interval was correlated with a longer time to disability and passage to secondary progressive form.
It has been described that MSSS-based analysis identifies more risk factors for severity than when the EDSS is used alone. For example, in the two American studies using MSSS, the male gender was correlated with the severity of the disease 35, 36. While the studies that used the EDSS alone did not identify gender as a risk factor for severity 37. These observations support the suggestion that the MSSS was a more nuanced indicator of disease severity than the EDSS, since it incorporates two factors that are not accounted for by raw EDSS scores: duration of the disease and change in EDSS score over time. This hypothesis was confirmed by the study of Pachner et al, which showed that disease severity as measured by the MSSS was practically stable over several years of follow-up, thus predicting individual-level disease severity over time 38. Benign MS is defined by an EDSS ≤ 2 or ˂3 after 10 years or more of disease progression 39, 40. In a recent study the authors propose an alternative definition (EDSS ≤1.0, absence from any disability, and the ability to work after 15 years of disease duration) which might truly reflect 41. In our series we found 31 patients (8%) meeting this definition. Although the definition of benign forms remains debated, it is generally admitted that they are characterized by the absence of significant ambulatory impairment 10 to 20 years after the onset of the disease. However, the practical impact of this concept remains limited for two reasons: on the one side, it is a progressive modality that can only be determined a posteriori, and on the other side, epidemiological studies with sufficient hindsight tend to show that a significant proportion of initially "benign" patients nevertheless end up worsening with time, following the usual evolution of other MS patients 41. The suggestion that MS is progressing more slowly than previously thought may represent a change in the type of patient with MS being seen in MS clinics today, perhaps driven by an increased recognition of MS, new diagnostic techniques, availability of immunomodulatory drugs to treat MS and increased survival (related to better health care and management of chronic diseases). All these factors might contribute to the differences between older and newer natural history studies. In fact, the specific component of these factors remains to be determined, in particular the impact of immunomodulatory drugs on long term disability progression 42. There were some limitations to our study that require discussion: we did not study the impact of disease-modifying drugs therapy. Moreover, considering variability in the duration and type of treatment in our population, it may be very hard to include this component in our evaluation. The accessibility of medical care in some centres, a less awareness of MS in the general public and the low socio-economic among some patients may have influenced this time of diagnosis, the time of treatment and the severity of the disease but those data which were not assessed in this study. Nevertheless, this is a detailed study of MS patients in Algeria, and it should provide baseline characteristics of MS patients in this region, since environmental and genetic factors are thought to have significant roles in MS 6.
We wish that in the future other epidemiological studies from other centers in the country will appear to clarify the characteristics and evolution of the disease in our region where multiple sclerosis is little studied. A national registry is strongly recommended, as well as long-term follow-up epidemiological and genetic studies.
References
- 1.Frischer J M, Bramow S, Dal-Bianco A, Lucchinetti C F, Rauschka H et al.The relation between inflammation and neurodegeneration in multiple sclerosis. Brain 2009;132(pt 5) : 1175-89.
- 2.Cabre P, Signate A, Olindo S. (2005) Role of return migration in the emergence of MS in French West Indies. , Brain 128-2899.
- 3.Gouider R, Mrabet S, Sidhom Y, Kacem I, Lubetzki C. (2020) Specific characteristics of multiple sclerosis in North Africa: Role of genetic and environmental factors. Bull Acad Natl Med. 215-1.
- 4.Ascherio A, Mungrt K. (2004) Multiple sclerosis. In:. Neuroepidemiology: From Principles to Practice Nelson LM, Tanner CM, Van Den Eeden SK, McGuire VM, editors , New York: Oxford 188-222.
- 5.Delasnerie-Laupretre N, Alperovitch A. (1992) Migration and age at onset of multiple sclerosis: some pitfalls of migrant studies. , Acta Neurol Scand 85, 408-411.
- 6.Kurtzke J F, Delasnerie-Laupretre N, Wallin M T. (1998) Multiple sclerosis in North African migrants to France. Acta Neurol Scand. 98, 302-309.
- 7.Kurtzke J F. (1980) Geographic distribution of multiple sclerosis: an update with special reference to Europe and the Mediterranean region. Acta Neurol Scand. 62, 65-80.
- 8.Walton C, King R, Rechtman L, Kaye W, Leray E. (2020) Rising prevalence of multiple sclerosis worldwide: Insights from the Atlas of MS, third edition. Mult Scler. 26(14), 1816-1821.
- 9.Hecham N, Nouioua S, Sifi Y, Toubal N, Aissa L A. (2014) Multiple sclerosis: progression rate and severity in a multicenter cohort from Algeria. Mult Scler. 1923-4.
- 10.Araqi-Houssaini A, Lahlou I, Benkadmir Y, Elotmani H, Hajjaj I. (2014) Multiple sclerosis severity score in a cohort of Moroccan patients. Mult Scler. 764-5.
- 11.Sidhom Y, Damak M, Riahi A, Hizem Y, Mrissa R. (2014) Clinical features and disability progression in multiple sclerosis in Tunisia: do we really have a more aggressive disease course? J Neurol Sci. 15, 343-1.
- 12.Sidhom Y, Maillart E, S Tezenas du Montcel, Kacem I, Lubetzki C. (2017) Fast multiple sclerosis progression in North Africans: Both genetics and environment. 88(13), 1218-1225.
- 13.Hentati E, Ben Sassi S, Nabli F, Mabrouk T, Zouari M et al. (2018) Disability progression in multiple sclerosis: a Tunisian prospective cohort study. Neurol Sci. 39(5), 879-884.
- 14.Binquet C, Quantin C, G Le Teuff, Pagliano J F, Abrahamowicz M et al. (2006) The prognostic value of initial relapses on the evolution of disability in patients with relapsing-remitting multiple sclerosis. Neuroepidemiology. 27(1), 45-54.
- 15.Mandrioli J, Sola P, Bedin R, Gambini M, Merelli E. (2008) A multifactorial prognostic index in multiple sclerosis. Cerebrospinal fluid IgM oligoclonal bands and clinical features to predict the evolution of the disease. J Neurol. 255(7), 1023-31.
- 16.Weinshenker B G, Rice G P, Noseworthy J H, Carriere W, Baskerville J et al. (1991) The natural history of multiple sclerosis: a geographically based study. 3. Multivariate analysis of predictive factors and models of outcome. , Brain 114, 1045-56.
- 17.Yamout B, Itani S, Arabi A, Hamzeh D, Yaghi S. (2010) Prognostic factors of multiple sclerosis in Lebanon. Int. 120(3), 206-10.
- 18.Lublin F D, Reingold S C. (1996) Defining the clinical course of multiple sclerosis: result of an international survey. National multiple sclerosis society (USA) Advisory Committee on clinical trials of new agents in MS. , Neurology 46, 907-911.
- 19.Roxburgh R H, Seaman S R, Masterman T, Hensiek A E, Sawcer S J. (2005) Multiple Sclerosis Severity Score: using disability and disease duration to rate disease severity. Neurology. 64(7), 1144-51.
- 20.Weinshenker B G, Rice G P, Noseworthy J H, Carriere W, Baskerville J et al. (1991) The natural history of multiple sclerosis: a geographically based study. 3. Multivariate analysis of predictive factors and models of outcome. , Brain 114, 1045-56.
- 21.Debouverie M, Pittion-Vouyovitch S, Louis S, Guillemin F. (2008) Natural history of multiple sclerosis in a population-based cohort. , Eur J Neurol 15-916.
- 22.HTS Benamer, ESM Ahmed, Al Din AS.Frequency and clinical patterns of multiple sclerosis in Arab countries: A systematic review.J Neurol Sci2009;. 278-1.
- 23.Debouverie C, Lebrun S, Jeannin S. (2007) More severe disability of North Africans vs. Europeans with MS in France. Neurology 29-32.
- 24.Kister I, Chamot E, Bacon J H, Niewczyk P M, De Guzman RA. (2010) Rapid disease course in African Americans with multiple sclerosis. Neurology. 75(3), 217-23.
- 25.Damasceno A, F Von Glehn, Brandão C O, Damasceno B P, Cendes F. (2013) Prognostic indicators for long-term disability in multiple sclerosis patients. J Neurol Sci. 15, 324-1.
- 26.Tremlett H, Paty D, Devonshire V. (2006) Disability progression in multiple sclerosis is slower than previously reported. Neurology. 66(2), 172-7.
- 27.Pittock S J, Mayr W T, McClelland R L, Jorgensen N W, Weigand S D. (2004) Disability profile of MS did not change over 10 years in a population-based prevalence cohort. Neurology. 62(4), 601-6.
- 28.Confavreux C, Vukusic S, Adeleine P. (2003) Early clinical predictors and progression of irreversible disability in multiple sclerosis: an amnesic process. , Brain 126, 770-82.
- 29.FBS Briggs, Thompson N R, Conway D S. (2019) Prognostic factors of disability in relapsing remitting multiple sclerosis. Mult Scler Relat Disord. 30, 9-16.
- 30.Alsaedi E Q, Alsaedi M Q, Mansuri F A. (2022) . Clinical Profile and the Extent of Disability in Multiple Sclerosis Patients in Madinah, Saudi Arabia. Cureus 14(6), 25851.
- 31.Scalfari A, Neuhaus A, Degenhardt A, Rice G P, Muraro P A et al. (2010) The natural history of multiple sclerosis: a geographically based study 10: relapses and long-term disability.Brain. 133, 1914-29.
- 32.Vasconcelos C C, Aurenção J C, Thuler L C, Camargo S, Alvarenga M P et al. (2016) Prognostic factors associated with long-term disability and secondary progression in patients with Multiple Sclerosis. Mult Scler Relat Disord. 8, 27-34.
- 33.Confavreux C, Vukusic S. (2006) Natural history of multiple sclerosis: a unifying concept. Brain. 129-606.
- 34.Scalfari A, Neuhaus A, Daumer M, Deluca G C, Muraro P A et al. (2013) Early relapses, onset of progression, and late outcome in multiple sclerosis. JAMA Neurol. 70(2), 214-22.
- 35.Kister I, Chamot E, Bacon J H, Niewczyk P M, De Guzman RA. (2010) Rapid disease course in African Americans with multiple sclerosis. Neurology. 75(3), 217-23.
- 36.Kister I, Chamot E, Bacon J H, Cutter G, Herbert J. (2011) New York State Multiple Sclerosis Consortium. Trend for decreasing Multiple Sclerosis Severity Scores (MSSS) with increasing calendar year of enrollment into the New York State Multiple Sclerosis Consortium. Mult Scler. 17(6), 725-33.
- 37.Langer-Gould A, Popat R A, Huang S M, Cobb K, Fontoura P et al. (2006) Clinical and demographic predictors of long-term disability in patients with relapsing-remitting multiple sclerosis: a systematic review. Arch Neurol. 63(12), 1686-91.
- 38.Pachner A R, Steiner I. (2009) The multiple sclerosis severity score (MSSS) predicts disease severity over time. J Neurol Sci. 15, 278-1.
- 39.Glad S B, Aarseth J H, Nyland H, Riise T, Myhr K M. (2010) Benign multiple sclerosis: a need for a consensus. Acta Neurol Scand Suppl. 44-50.
- 40.Sartori A, Abdoli M, Freedman M S.Can we predict benign multiple sclerosis? Results of a 20-year long-term follow-up study. , J Neurol 264(6), 1068-1075.
