Abstract
Background
In natural conditions Cyclops are the first intermediate hosts in the life cycle of Spirometra species. In this paper we describe simple method of culturing Copepod of the genus Cyclops for use the first intermediate host in experimental life cycle of Spirometra species.
Methods
Paramecium was first cultured to be used as food for Cyclops. Sample of water was collected from a pond within Sokoine University. About 100 ml of water and pre-boiled wheat grains were transferred in a Petri dish and kept under laboratory conditions for 7 days, a swarm of Paramecium was formed. An adult female egg sacked Cyclops from a natural water pond in Tarangire National Park, Tanzania was added in a new Petri dish containing tap water, 0.3 ml of Paramecium suspension and 4 pre-boiled wheat grains. The mixture was kept under laboratory conditions temperature 26-29⁰C and observed daily.
Results
Eggs from the single Cyclops hatched to nauplius. The average time of developing to nauplius I was 1.2 days, nauplius I to copepodite I was 6.9 days, and copepodite I to adult female Cyclops was 26.3 days. The average measurements of naupliusI were 120.2µm length and 80.0µm width while the adult female was 846.3µm length and 284.6µm width. The adult female produced 1 to 8 broods (mean 4.3). The life span of Cyclops averaged 43.1 days.
Conclusion
The Cyclops cultured in the laboratory were fed Paramecium and used as first intermediate host in the experimental life cycle of Tanzanian Spirometra species.
Author Contributions
Academic Editor: Tewodros Alemneh, Researcher, Faculty of Veterinary Medicine, University of Gondar, Ethiopia.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2021 Nicholas Jairo
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Cyclops are free living forms found everywhere there is water, in damp leaf litter on the ground, ponds, ditches of stagnant water, streams and rivers. They feed on plankton and other small aquatic organisms. The life cycle is adapted to their natural habitat in ponds and other accumulations of stagnant water. Fresh water copepods may act as a biological control for malaria by consuming mosquito larvae.2 They also serve as intermediate hosts of many animal parasites and parasites of humans. Copepods are worldwide distributed. In Africa, freshwater copepods have been described in Mali by 14. In the life cycle of Spirometra species copepod of the genus Cyclops is the first intermediate host. For experimental work in the laboratory, Cyclops can be cultured by using different methods. 3 cultured copepod Eucyclopsserrulatus using Chilomonas paramecium, wheat infusion and pre-boiled wheat grain as food. The adult female egg sacked Cyclops was used. In order Cyclops to survive under laboratory conditions the adult Cyclops and nauplius have to be fed Paramecium 1970; 5, 6, 11. In Tanzania there is no records showing a study on culture of Cyclops. In the present study, culture of Cyclops started with an adult female single egg sac bearing which was fed Paramecium as food. Therefore, this paper reports how Cyclops were cultured in the Tanzanian laboratory from an adult female egg sacked Cyclops fed with live Paramecium and stock of Cyclops used in the experiment of life cycle of Spirometra species.
Materials and Methods
Culture of Paramecium
Sample of water was collected from a pond located within Sokoine University campus. About 100 ml was transferred in a Petri dish, 12 cm in diameter and 2.5 cm high, added 4 pre-boiled wheat grains. He Petri dish with contents was kept under laboratory conditions, temperature 26-29⁰C and observed daily. On day 7, there was a swarm of Paramecium around wheat grains. About 0.3 ml of the swarm was taken with a Pasteur pipette and transferred to another Petri dish of the same size containing tap water and 4 pre-boiled wheat grains. In this container Paramecium formed a swarm. Again same amount of swarm suspension was transferred to another Petri dish. In order to obtain pure culture of Paramecium, the procedure was repeated four times.
Culture of Cyclops
An adult female egg sacked Cyclops collected from natural pond in Tarangire National Park, Tanzania was cultured in the laboratory of Sokoine University. The egg sacked female Cyclops (Figure 1) was picked with a Pasteur pipette, transferred in a Petri dish 6 cm diameter and 2 cm high, containing tap water 50 ml, cultured Paramecium suspension 0.5 ml, 2 grains of pre-boiled wheat grains and trace amount of calcium carbonate powder. On day 3, the eggs hatched to nauplius. A total of six Petri dishes were used for culture, in each six Cyclops were transferred. Cyclops were maintained in the laboratory at temperature 26-29⁰C and observed daily under microscope until developed to adult stage. The new adult females developed egg sacs. They were observed until hatching, the time of developing egg sacs to hatching nauplii was recorded. The nauplii were separated into groups each with six, placed in Petri dishes with food contents and observed daily. The time from hatching to death was recorded as longevity. Number of days taken to reach each stage was recorded, and the cluster size was determined. Some gravid females were transferred to new Petri dishes and anaesthetized with carbonated water. They were picked and placed on a slide. Egg sac was teased with a needle to release eggs. The eggs were counted under microscope. Tea strainer sieve was used to separate the adult female Cyclops from hatched nauplii according to 12. Nauplii from 3 Petri dishes were killed by using 5% formalin and counted under microscope (Figure 2). Nauplii in the remaining 3 Petri dishes were allowed to develop to adult stage. The adult female egg sacked Cyclops appeared on day 12. The culture was transferred to another Petri dishes where multiplied and released nauplii. The procedure was repeated four times, the final culture of Cyclops was used for the experiment (Figure 3). Part of the Cyclops culture was maintained by feeding Paramecium for 1 year.
Figure 1.Egg sacked Cyclops species used in this study.
Figure 2.Nauplius hatched from egg sacked Cyclops.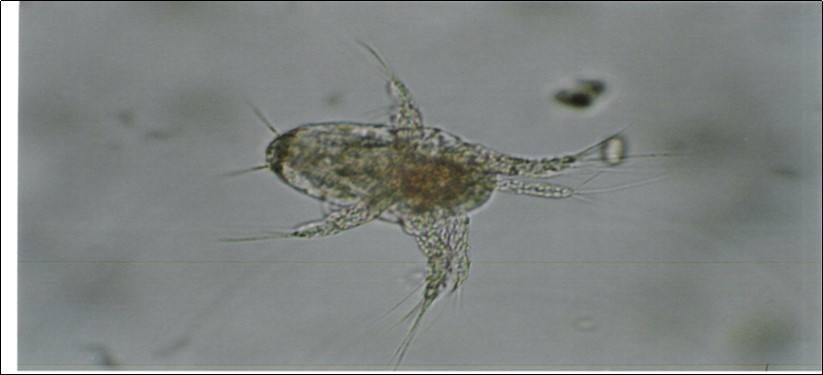
Results
After 7 days of culture there was a swarm of Paramecium around pre-boiled wheat grains in Petri dish.
Growth of Cyclops cultured in Petri dishes had an average time of, egg sac to nauplius I was 1.2 days, while from nauplius I to copepodite I was 6.9 days. The time taken from nauplius I to adult female egg sac bearing was 26.3 days. The average measurements of nauplius I were 120.2 µm in nlength and 80.0 µm in width while the adult female measurements were 846.3 µm in length and 284.6 µm in width.The number of broods produced by an adult female was 1 to 8 (mean, 4.3) and the life span of Cyclops in this experiment was 43.1 days.
Discussion
In the present study, the results shows that Cyclops were successfully cultured in the laboratory using Paramecium as food. Paramecium alone. Paramecium supported the whole life cycle of Cyclops. In previous studies copepods were maintained in the laboratory by using different methods. 7 cultured copepods in the laboratory which were used in the experiment of life-cycle of Spirometramansonoides. Cyclops were fed microorganisms (molds, bacteria, euglenoid and small ciliates) that developed in hay infusion. 9 cultured copepod by feeding Chlorella to nauplii and early copepodite stages, zooplankton containing rotifers and paramecia were fed to copepodite and adults. 4 cultured Cyclops abyssorum by feeding Euglena gracilis and Artemia.10 cultured Cyclops visinus by feeding pure algal diet (Chlamydomonas reinhardti or Cryptomonas sp.). 1 reported that Cyclops require not only green algae but also large organisms such as ciliates and rotifers as food to complete the whole life cycle. The present study agrees with previous studies that Paramecium acan be used as a source of food for Cyclops. In this study, the optimal temperature of the culture of the Cyclops was 26° to 29ºC. Many studies have been done in temperate regions where the temperature range was 10° to 20°C. The difference in temperature has not affected the development of Cyclops. In the present study, the time for development was shorter than the time in lower temperatures. This agrees with the study done by 5 who reported that the time for development of Cyclops at lower temperature is longer than at higher temperature. 8, 13 reported the average period from egg to egg bearing adult female was 29.4 and 30.7 days respectively. These results could be due to the effect of temperature.
Conclusion
Laboratory maintenance of Cyclops can be achieved by using Paramecium as the source of food. The method is simple because the food organism Paramecium proliferates in the Cyclops culture and supports a rapid growth and reproduction of the Cyclops. Cyclops is maintained with minimum care because a stable and equilibrium state of micro-ecosystem is established in the Petri dish. This culture method can be used in the study of Spirometra life-cycle in which Cyclops serve as the first intermediate host. However, another study is needed for the identification of Cyclops which exist in Tanzania.
Acknowledgements
The author wish to acknowledge Dr. E. Ernest of Tanzania Wildlife Research Institute, Arusha, Dr. B. Kissui of Tarangire Lion Research Project, for their help and co-operation during sample collection. Also Prof. A.A.Kassuku (late) of Sokoine University of Agriculture for his guidance. The study was supported by the Mzumbe University Post Graduate Studies Funds. The results presented here also formed part of the graduate study (PhD) programme of the author at the Sokoine University of Agriculture, Morogoro, Tanzania.
References
- 1.Adrian R, T M Frost. (1993) Omnivory in cylopoid copepods: comparisons of algae and invertebrates as food for three, differently sized species.Journal of Plankton Research15:. 643-658.
- 2.Hopp U, Maier G, Bleher R. (1997) Reproduction and adult longevity of five species of planktonic cyclopoid copepods reared on different diets; a comperative study.Freshwater Biology38:. 289-300.
- 3.Kumazawa H. (2000) Laboratory maintenance ofEucyclopsserrulatus(Copepoda:. , Cyclopoida).Journal of Parasitology International49 3, 189-193.
- 4.B G Lewis, Luff S, Whitehouse W. (1971) . Laboratory culture of Cyclops abyssorum Sars, 1863 (Copepoda, Cyclopoida). Crustaceana 176-182.
- 5.Maier G. (1990) The effect of temperature on the development, reproduction and longevity of two common cyclopoid copepods-Eucyclopsserrulatus(Fischer) andCyclops strenuous(Fischer).Hydrobiologia203:. 165-175.
- 6.Maier G. (1992) The reproductive biology ofCyclopsvivinus.Journal of Plankton Research14:. 127-135.
- 7.J F Mueller. (1959) The laboratory propagation ofSpirometramansonoides(Muller, 1935) as an experimental tool, II. Culture and infection of the copepod host, and harvesting of the procercoid.Transactions of American Microscopic Society78:. 245-255.
- 8.S H Park, C Y, S. (2005) A new culture system for in situ Observation of the growth and development ofEucyclopsserrulatus(Copepoda:. , Cyclopoida).Korean Journal of Parasitology43 4, 141-147.
- 9.Riviere F, B H Kay, Klein J M and Secham, Y. (1987) andBacillus thuringiensis varIsraelensisfor the biological control ofAedesandCulexvectors (Diptera: Culicidae) breeding in crab holes, tree hole and artificial containers.Journal of Medical Entomology24:. 425-430.
- 10.Santer B, Bosch F van den. (1994) Herbivorous nutrition ofCyclopsvicinus: the effect of a pure algal diet on feeding, development, reproduction and life cycle.Journalof Plankton Research16:. 171-195.
- 11.Smyly W J P. (1970) Observations on rate of development, longevity and fecundity ofAcanthocyclopsviridis(Jurine) (Copepoda, Cyclopoida) in relation to type of. Prey.Crustaceana18: 21-26.
- 12.Vijerberg J. (1989) Culture techniques for studies on the growth, development and reproduction of copepods and cladocerans under laboratory andin situconditions a review.Freshwater Biology21:. 317-373.

