Cytokine Expression in Peripheral Blood Mononuclear Cell Cultures Obtained from Cattle with Different Stages of Natural Mycobacterium bovis Infection
Abstract
In bovine tuberculosis (bTB), cellular, humoral, or both types of immune responses have been observed. The purpose of this study was to examine the immune status of tuberculous cows based on the differential cytokine gene expression associated with Th1 (IFN-γ, IL-2), or Th2 (IL-4, IL-10) responses. Twenty-three (23) cows belonging to a dairy herd located in a rural region of the State of Hidalgo, México, were selected for the study. Single Intradermal Comparative Cervical Tuberculin (SICCT) Test, Interferon-Gamma (IFN-γ) Release Assay (BOVIGAM), and Enzyme-Linked Immunosorbent Assay (ELISA) were used for detection of cattle infected by M. bovis. Thirteen cows were positive to all the tests (Group 1); ten cows were positive only to ELISA (Group 2), and the remaining Group (Group 3, control) included cows negative to all the tests. Peripheral blood mononuclear cells (PBMC) from animals were in vitro stimulated by bovin purified protein derivative (PPD), avian PPD, and Concanavalin A (Con A) mitogen for 72h. Changes in the levels of expression of mRNA of the respective cytokines was measured by Reverse Transcription-Polymerase Chain Reaction (RT-PCR) using β-actin gene as internal control. In group 1, PPD bovis and Con A-stimulated cells exhibited high production of IFN-γ, IL-2 and IL-4, but not IL-10. In contrast, PPD avium-stimulated cells displayed a low production of cytokine transcripts. In group 2, cells showed a significant production of IL-10 in response to bovine PPD (P< 0.001). In the control group, a high production of IFN-γ and IL-2 was observed only in Con A-stimulated cells. Post-mortem examinations in animals of group 1 showed slight and medium lesions in lymph nodes, whereas in group 2, the lesions were more extensive. Results indicate differences on gene expression levels of cytokines considered to determine balance in Th1/Th2 response among the evaluated groups. In addition, high levels of antibodies against M. bovis and high IL-10 expression in PBMC together are indicators of progressive bTB when both tuberculin test and IFN-γ assay are negative in tuberculous anergic cattle. Inclusion of serology and IL-10 cytokine expression in in the diagnosis checklist improves detection of infected cattle to help control bovine tuberculosis.
Author Contributions
Academic Editor: Yanzhou Yang, Ningxia Medical University
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2021 Lugo-Arriaga, María Teresa, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Bovine Tuberculosis (bTB) is a contagious, chronic bacterial disease caused by Mycobacterium bovis (M. bovis), with worldwide distribution 1. The disease has a negative impact on the livestock industry and it represents a risk to human health 2. The progress or containment of the infection depends on the dynamic nature established between host immune response cells and the M. bovis bacterium. Cellular immunity, mediated by CD4+ T lymphocytes, involving cytokines that increase the microbicide activity of macrophages, play a significant role in the elimination of bacteria 3. Interferon gamma (IFN-γ), a pro-inflammatory cytokine seems to be the most critical effector molecule for the activation of the phagocytic cells, because it mediates its protective efficacy through the induction of reactive oxygen and nitrogen intermediates, necessary for the destruction of intracellular mycobacteria 4, 5. Hence, the measuring of IFN-γ level in whole-blood culture supernatant has been a diagnostic test widely used to identify tuberculous cattle from decades ago 6, 7, 8. Diverse studies have shown that a protective immune response is not only associated to IFN-γ but also to tumor necrosis factor-α (TNF-α) and interleukin (IL)-2 produced by memory T cells 9, 10, 11. Nevertheless, the production of IFN-γ and IL-2 is accompanied by synthesis of IL-4 and IL-10 after a M. bovis-challenge in BCG-vaccinated cattle 12. Some authors have shown that IL-10 regulates a strong pro-inflammatory immune response, which contributes to host-induced tissue harm 12,13. On the other hand, both tuberculin skin test and Interferon-Gamma (IFN-γ) Release Assay (BOVIGAM) are important to detect bTB-suspected cattle at onset and during the primary infection, but as the disease progresses, a decrease in the cell-mediated immune response take place concomitantly with an increase of antibodies to M. bovis3. Detection of serum antibodies to the pathogen is an alternative test that has been proposed to improve the sensitivity of bTB diagnosis in cattle 14. However, the scarcity of understanding concerning the immunological mechanisms in M. bovis infection in cattle has forced the search for biomarkers. In this sense, PPD bovis-stimulated peripheral blood mononuclear cells (PBMC) from M. bovis-infected animals with high pathology showed an increase in expression of transcripts to pro-inflammatory cytokines than did animals with low pathology 5. Therefore, this study aimed to examine the immunological status of tuberculous cows with different reactivity to immunodiagnostic tests of tuberculin skin test, IFN-γ assay (BOVIGAM), and ELISA to anti-M. bovis antibodies by measuring transcripts to cytokines IFN-γ, IL-2, IL-4, and IL-10 in PBMC culture, and their correlation with postmortem inspection.
Materials and Methods
Reagents
Bovine- and avian-PPD were purchased from PRONABIVE (Mexico City, Mexico). Ficoll-hypaque was acquired from Amersheim Biosciences (Uppasala, Sweden). The kit for the detection of bovine gamma interferon was from BOVIGAM™ (Applied Biosystems™ Thermo Fisher Scientific). RPMI-1640 culture medium, peroxidase-labeled protein G, L-glutamine, ortho-phenylendiamine, t-octylphenoxy polyeth oxyethanol (Triton X-100), polyoxyethylenesorbitan monolaurate (Tween 20), Canavalia ensiformis (Concanavalin A, Con A), ethidium bromide, and 2-mercaptoethanol were from Sigma Aldrich (St. Louis, MO, USA). TRIzol reagent, RNase inhibitor, dithiothreitol (DTT), recombinant DNase I (RNase-free), ampliTaq DNA polymerase, superScript III reverse transcriptase, and deoxyribonucleotides were acquired from Invitrogen Life Technologies (Carlsbad, CA, USA). Diethyl-pyrocarbonate (DEPC) was from Amresco Inc. (Solon, OH, USA). AmpliTaq DNA polymerase and oligo-d(T)16 were from Applied Biosystems Inc. (Hammonton NJ, USA). Chloroform and 2-propanol were acquired from Merck Chemicals Inc. (Gibbstown, NJ, USA). Sulfuric acid and salts were from JT. Baker Inc (Phillipsburg, NJ, USA).
Ethic Statement
The study design and sampling methodology were approved by Bioethics Committee for care and reasonable use of experimental animals in research projects of the Centro Nacional de Investigación Disciplinaria en Salud Animal e Inocuidad (CENID-SAI) belonging to the National Institute for Forestry, Agriculture and Livestock Research (INIFAP) in Mexico City, Mexico. Permission to perform the fieldwork was also obtained from CENID-SAI and INIFAP. Collection of blood samples and Single Intradermal Comparative Tuberculin (SICCT) test were performed by qualified veterinarians following official procedures from the Norma Oficial Mexicana (NOM-041-ZOO1995) of the National Campaign against tuberculosis in Animals 15.
Animals and Experimental Design
The study was conducted in a Holstein-Friesian dairy herd located in a rural region of the State of Hidalgo, México, bTB is endemic in the region. In a first field work session, the SICCT test was applied to all the animals of the herd. The same day blood samples were taken from all animals to carry out Enzyme-Linked Immunosorbent Assay (ELISA) to evaluate immune humoral response against M. bovis-antigens. The following week, after SICCT and ELISA tests were carried out and results obtained, heparinized blood samples were taken from reactor cows and cows with high antibody titers, as well as from non-reactor cows, but which showed high titers of antibodies to mycobacterial antigens, to perform the Interferon-Gamma (IFN-γ) Release Assay (BOVIGAM). Based on the results obtained in the three diagnostic tests, twenty-three cows were selected and grouped, as: Thirteen, positive to all the mentioned tests, formed Group 1; the other ten cows, that were only positive to ELISA, formed Group 2. A third Group included ten healthy cows, and tested negative by all the three diagnostic tests, served as control. The latter came from a bTB-free accredited herd. For the purpose of analysis of mRNA relative expression of IFN-γ, IL-2, IL-4, and IL-10 in PBMC culture of the animal groups, blood samples were collected into tubes containing sodium heparin anticoagulant (BD Vacutainer, Franklin Lakes NJ, USA). PBMC were isolated by Ficoll-Hypaque (GE Healthcare Ficoll-Paque™ PLUS Media (density 1.077 g/mL)) density gradient centrifugation for its subsequent in vitro stimulation with bovine PPD (20 μg/ml) and with the mitogen Concanavalina A.
Single Intradermal Comparative Cervical Tuberculin (SICCT) test.
Intradermal inoculation of bovine PPD and avian PPD in two sites 12 cm apart on the cow mid-neck was performed. The first site was injected 100 µl containing 3,250 IU/ml bovine PPD and the second site was injected with 100 µl containing 5,000 IU/ml avian PPD. The skin thickness was measured by a digital Vernier caliper before the intradermal injection and at 72 h later. The results were recorded as an increase in skin thickness at 72 h compared to the thickness pre-injection. Values of both, avian and bovine PPD were plotted. According to the official graphic of the Mexican Official Norm, the result was the value at the intersection of avian and bovine PPD values 15.
Interferon Gamma Release Assay
Heparinized whole blood samples were individually processed within 2–3 h after collection. They were distributed in 1.5 ml aliquots into four individual wells per animal in sterile flat-bottomed 24-well cell culture plate (Nunclon, Roskilde, Denmark). The first well of the different cultures was stimulated with 20 mg/ml bovine PPD, the second with 20 mg/ml avian PPD, the third with 10 mg/ml polyclonal mitogen Con A, and the fourth well was used as control without stimulation. Blood cultures were incubated at 37°C in a 5% CO2 humidified atmosphere for 24 h. After this time, the supernatant plasma was harvested and stored at -70°C until analyzed. The IFN-γ levels in the plasma supernatants were measured using a sandwich ELISA following the instructions from a commercial kit (Applied Biosystems™ Bovigam™ Tb Kit, Thermo Fisher Scientific) 16. Absorbance of standards and plasma samples were read at 450 nm using an ELISA plate reader (Benchmark-Plus Microplate Spectrophotometer Bio-Rad Laboratories, Hercules CA, USA). Results were evaluated considering the optical density at 450 nm (OD450) mean value of stimulated plasma samples. An animal was positive to the test if value to bovine PPD OD450 minus value to avian PPD OD450 were >0.1 OD, and a value for the OD450 with bovine PPD minus the OD450 nil antigen culture of >0.1.
Mycobacterium bovis Culture-Filtrate Protein Extract
Culture-filtrate protein extracts (CFPE) was prepared from M. bovis strain AN5 and M. avium strain D4. The bacterial were cultured individually at 37°C for over six weeks in modified Dorset-Henley liquid synthetic medium 17. At the completion of the incubation period, proteins from the cultures filtrates were precipitated by ammonium sulfate [(NH4)2 SO4] at a final saturation of 80%, at 4°C for 24h in constant agitation18. Then, the solution was centrifuged at 15,000 g for 60 min at 4°C, the pellet was suspended in 10 ml of phosphate buffered saline pH 7.2 (PBS) and dialyzed against PBS using dialysis bags with MW cutoff point from 10 kDa (Cellu.Sep H1, Membrane Filtration Products, Inc. Seguin TX, USA) at 4°C for 36h. Protein concentration in the CFPE was determined by the Bradford method 19, and it was adjusted to 3.5 mg/ml and divided into aliquots of 1 ml, which were stored at -70°C until used.
Comparative ELISA for Determination of Ig-G Class Antibodies to Mycobacterium bovis
A comparative ELISA was carried out using CFPE from M. bovis and M. avium antigens separately to assess IgG-class antibodies to M. bovis in serum. 20. Ninety-six-well flat-bottom ELISA plates (Nunclon, Denmark) were incubated with 5 mg/well of either CFPE from M. bovis or M. avium diluted in 0.1 M carbonate/bicarbonate buffer of pH 9.6 at 4°C for 24h. Antigen-coated ELISA plates were washed 3 times by 0.3% Triton X-100 in PBS (PBS-Triton), 3 times by 0.1% Tween-20 in PBS (PBS-Tween) and blocked in 200ml of a blocking buffer (3 % skim milk in PBS-Tween) with incubation for 1h at 37°C. After 6 washes in PBS-Tween, 100 µl blood serum (diluted 1:100 in blocking buffer) was added in each well and incubated for 1h at 37°C. The ELISA plates were washed again and 100 ml peroxidase-labeled protein G diluted 1:10,000 in blocking buffer was added per well, with incubation for 1h at 37°C. Another stage of washes was performed and subsequently 100 µl chromogenic substrate solution containing 0.04% O-phenylenediamine (Sigma P-3804) and 0.04% of hydrogen peroxide in citrate buffer pH 4.5 was added for detection of enzyme reaction. Reaction was stopped with 50 μl/well of 2 M sulfuric acid. Optical densities were obtained at 492 nm (OD492nm) using an automated ELISA plate reader (Benchmark-Plus Microplate Spectrophotometer). All assays were carried out in duplicate. Antibody response to M. bovis or M. avium CFPE was established previously by setting the cutoff value of the mean OD492 plus two standard deviations of sera from healthy animals.
Culture of Peripheral Blood Mononuclear Cells (PBMC)
PBMC were isolated from bovine blood sample collected using heparin as anticoagulant. For this, 6 ml of blood was diluted 1:2 in PBS, carefully placed over 2.5 ml of Ficoll-Hypaque gradient in a 15 ml centrifuge tube, and centrifuged at 1,600 g for 40 min at room temperature 21. The mononuclear cell layer was isolated and washed twice in RPMI-1640 culture medium by centrifugation at 440 g for 10 minutes. PBMC (5 x 106) were placed into each well of a 24-wells flat-bottom culture plate (Nunclon Denmark) and suspended in 1 ml RPMI-1640 culture medium supplemented with 10 mM HEPES buffer, 50 mM 2-mercaptoethanol, 2 mM L-glutamine, 100 U/ml penicillin, 0.1 mg/ml streptomycin, and 10% heat-inactivated fetal calf serum. Twenty mg/ml bovine PPD, 20 mg/ml avian PPD and 10 mg/ml Con A were added to the corresponding wells, and incubated for 72 h at 37°C in a 5% CO2 humidified atmosphere. Cells without antigen stimulation were used as negative control. At the end of the incubation period, the cells, were harvested and washed in RPMI-1640 medium by centrifugation at 440 g for 3 min at 4°C discarding supernatant before RNA extraction.
RNA Extraction
Total RNA from bovine PPD- or avian PPD- or Con A-stimulated PBMC and control without stimulus were extracted as described 22. After RPMI-1640 medium washing, cells (5 x 106) were incubated in 1 ml of TRIzol for 5 minutes at room temperature. A second incubation was performed in 200 ml chloroform for 3 min at 4°C. After centrifugation at 12,000 g for 15 minutes at 4°C, the aqueous phase was recovered and RNA was precipitated by incubation 10 minutes in 500 ml 2-propanol. The pellet was centrifuged at 12,000 g for 10 min at 4° C, next suspended in 1 ml 75% ethanol and centrifuged at 7,500 g/5 min/4°C. Subsequently, the RNA was suspended in 50 ml of DEPC water and heated for 10 minutes at 55°C. The extracted RNA was measured in a fluorometer (VersaFluorTMFluorometer System Bio-Rad, Richmond CA, USA). To eliminate any genomic DNA contamination, a recombinant DNase I, RNase-free was used to treat the extracted RNA.
Cytokine Transcripts
The reverse transcriptase-polymerase chain reaction (RT-PCR) technique was used to evaluate cytokine transcript expression by specific primers for amplification of each one of them [23-27 (Table 1). Beta-actin (β-actin) mRNA gene was used as internal control 27,28. The cDNA was synthesized from total RNA obtained from stimulated and unstimulated PBMC using the enzyme superScript III reverse transcriptase. The reaction mixture was: 5 ml of Oligo(dT)16 (50 mM); 10 ml of 5X first-strand buffer (250 mM); 2.5 ml of dNTP Mix (10mM); 5 ml of 0.1M DTT; 2.5 ml 40U RNase inhibitor; 5 ml 250 U enzyme superScript III reverse transcriptase and 250 ng total RNA in a total volume per reaction of 50 ml. The final reaction mixtures were incubated at room temperature for 10 min, followed by incubation at 42°C for 50 min and then by heat inactivation at 70°C for 15 min. Then, the cDNA was amplified by PCR, which was conducted in a final volume of 20 ml. The mixture contained 2 ml 10X buffer (200 mM Tris-HCl, pH 8.4, 500 mM KCl MgCl2); 2 ml of dNTP Mix (10mM); 1 ml of each primer (20 pmol) for the cytokines under study, 0.15 ml ampliTaq DNA polymerase (0.75U), 4 ml of each cDNA and DEPC water (up until 20 ml) 29. Polymerase chain reaction was conducted in a thermocycler iCicler IQTM (Bio-Rad. Hercules CA, USA) according to previously established conditions (Table 1). PCR product of each cytokine RNA was analyzed in an agarose gel dyed in ethidium bromide. Subsequently, it was compared with the β-actin mRNA level and denoted as relative intensity (cytokine intensity/β-actin intensity) using the software LabWorks 4.0.
Table 1. Sequence of the primers, size of the amplified fragment of the different citokines evaluated and programs used in reverse transcriptase-polymerase chain reaction (RT-PCR).| Cytokine and Reference | Oligonucleotide sequences | Amplicon length (base pairs) | Themocycler Program |
| IL-2Cerretti., et al (1986) 23 | 5’ AGATACAACTCT TGTCTTGC 3’5’ AGTCATTGTTGA GTAGATGC 3’ | 457 | 95º, 94º, 52º, 72º, 72ºC 4’ 1’ 1’ 1’:30’’ 7’ 33 CYCLES |
| IFN-γCerretti., et al (1986) 24 | 5’ TTCAGAGCCAAA TTGTCTCC 3’5’ CTGGATCTGCAG ATCATCCA 3’ | 184 | 95º, 94º, 51º, 72º, 72ºC 4’ 1’ 1’ 1’:30’’ 7’31 CYCLES |
| IL-4Heussler., et al (1992) 25 | 5’ CTATTAATGGGTCTCACCTACCA 3’3’ CTTGCCAAGCTG TTGAGATTC 5’ | 311 | 94º, 94º, 51º, 72º, 72ºC 5’ 30’’ 45’’ 1’ 5’33 CYCLES |
| IL-10Hash., et al (1994) 26 | 5’ GTTGCCTGGTCT TCCTGGCTG 3’5’ TATGTAGTTGAT GAAGATGTC 3’ | 471 | 94°, 94°, 53°, 72°, 72°C 4’ 1’ 1’ 1’:30’’ 7’30 CYCLES |
| β-actinDegen., et al (1983)27 | 5’ ACCAACTGGGAC GACATGGAG 3’5’ GCATTTGCCGTG GACAATGGA 5’ | 890 | 94º, 94º, 53º, 72º, 72ºC 5’ 30’’ 45’’ 1’ 5’28 CYCLES |
Post Mortem Examination
BTB-suspected animals were examined at post mortem in the abattoir for visible evidence of tuberculosis. The procedure involved visual inspection, palpation and incision of lungs, liver, kidneys and tracheobronchial, mediastinal and prescapular lymph nodes. Tissue samples were collected from cattle with suspected TB lesions during post mortem examination. The collected tissues were sliced at 0.5 to 1 cm intervals followed by evaluation to determining the severity of gross lesions. Pathology scoring method described by Vordermeier., et al. (2002) 30was used to evaluate the severity of the lesions. A score of 0, was assigned for absence of lesions; a score of 1, to a few mild lesions (1 to 2 mm in diameter); a score of 2, to a few medium lesions (1 to 5 mm in diameter) and necrotic tissue of a dimension of 5 by 5 mm; a score of 3 to multiple medium lesions presenting a size of greater than10 mm in diameter; and a score of 4 to severe multifocal lesions and large necrotic regions.
Statistical Analysis
Data were analyzed by parametric and non-parametric statistics using the Sigma Stat v 4.0 software (Jandel Scientific CA, USA). Statistical differences identified in the results were considered significant at P< 0.05 when compared with their respective controls.
Results
Tuberculin Skin Test
Thirteen from 23 tuberculosis-suspected cattle were extreme reactors to tuberculin test. These animals displayed a mean skin thickness value of >37 mm (± 9.7 mm) for bovine PPD, (Table 2). In contrast, the remaindaining animals in the test groups and the 10 clinically healthy animals in the control group did not show a significant reaction in skin after inoculation of M. bovis PPD or M. avium PPD. Group 1, was formed by 13 cows, which were positive to both Single Intradermal Comparative Cervical Tuberculin (SICCT) test and IFN-γ assay. Group 2, included 10 cows that were negative to tuberculin test and IFN-γ assay. Group 3, was constituted by 10 clinically healthy cows used as control group, which were negative to the different immunodiagnostic tests realized. Values denote mean ± standard deviation of optical density (OD492) obtained in serum samples from cattle.
Table 2. Results of a comparative ELISA that detects bovine serum antibodies to culture-filtrate protein extract from M. bovis or M. avium| Comparative ELISA | |||||
| Groups | n | SICCT test | IFN-γ (BOVIGAM) | M. bovis | M. avium |
| 1 | 13 | positive | positive | 0.489 ± 0.100 | 0.332 ± 0.130 |
| 2 | 10 | negative | negative | 0.771 ± 0.102 | 0.355 ± 0.123 |
| 3 | 10 | negative | negative | 0.197 ± 0.079 | 0.166 ± 0.048 |
IFN-γ Production
Plasma samples from 13 tuberculin-reactor animals showed an IFN-γ OD450 mean value upon bovine PPD-stimulation, which was 7.47-fold greater than non-stimulated samples (0.605 ± 0.313 vs 0.080 ± 0.033, respectively). Correspondingly, the IFN-γ level in response to stimulation by avian PPD was 0.393 ± 0.290, whereas that the upon Con A-stimulation the mean value was 1.026 ± 0.297. Thus, the group was considered positive (Table 2), according to the criteria of the BOVIGAMTM test. On the contrary, 10 bTB-suspected cattle (group 2, anergic) but negative to tuberculin test exhibited low IFN-γ levels after stimulation with either bovine or avian PPD (0.100 ± 0.026 and 0.124± 0.060, respectively). As a result, they were considered negative to this test (Table 2). Likewise, a low mean level production was obtained when stimulated by Con A (0.260 ± 0.173) in whole blood cultures of anergic animals. Therefore, IFN-γ level was 3.94-fold lower than that produced by tuberculin-positive animals.
The IFN-γ production in plasma samples from 10 tuberculin-negative healthy control animals was as follows: IFN-γ level in non-stimulated whole blood showed OD450 mean value of 0.069± 0.017, which did not increase after stimulation by either bovine or avian PPD (0.095 ± 0.015 and 0.117 ± 0.029, respectively). However, a high level of IFN-γ production was observed upon Con A stimulation, 2.923 ± 0.780; which was 2.85 fold higher than the reactor group, and 11.2 fold higher than the anergic group.
Antibodies to Mycobacterial Antigens Evaluated by ELISA
Serum antibodies in response to M. bovis antigen in tuberculin-positive cattle showed an OD492 mean value of 0.489 ± 0.100, which was 2.5 fold higher than the control (0.197 ± 0.079). While, bTB-suspected tuberculin-negative cattle with low IFN-γ release in response to Con A stimulation, showedmean value of 0.771 ± 0.102 (Table 2). The positive ELISA reaction detected in tuberculin-negative cattle showed a mean value of 1.57 fold higher than tuberculin-positive cattle (0.771 ± 0.102 vs 0.489± 0.100). With regard to ELISA reactions using M. avium antigen, the mean values of sera from tuberculin-positive cattle and bTB-suspected tuberculin-negative cattle were similar (0.332± 0.130 and 0.355 ± 0.123, respectively). However, the above results were 2.0 to 2.13-fold higher than the OD492 mean value(0.166 ± 0.048) when compared to the control group (Table 2).
Table 3. Macroscopic lung, tracheal, lymph node lesion scores in cows of the groups 1 and 2| HEAD-MANDIBULAR | NECK | THORAX | ABDOMEN | CARCASS | |||||||||||
| GROUPS | Animal | Lateral retropharyngeal | Medial retropharyngeal | Submandibular | Parotid | Deep cervical | Tracheal | Lung | Thoracic tracheal | Mediastinal | Hepatic | Mesenteric | Superficial Cervical | Inguinal | Internal iliac |
| GROUP 1 | 1 | 2 | 0 | 2 | 1 | 2 | |||||||||
| 2 | 2 | 0 | 2 | ||||||||||||
| 3 | 0 | 1 | 2 | 1 | 2 | 2 | 2 | ||||||||
| 4 | 2 | 1 | 1 | ||||||||||||
| 5 | 0 | 0 | |||||||||||||
| 6 | 2 | 0 | 2 | 1 | 1 | 1 | 1 | ||||||||
| 7 | 0 | 1 | 2 | 1 | 1 | ||||||||||
| 8 | 1 | 0 | 1 | 1 | 2 | 2 | 1 | 1 | |||||||
| 9 | 0 | 0 | |||||||||||||
| 10 | 2 | 1 | 1 | 2 | 1 | 1 | 1 | ||||||||
| 11 | 1 | 2 | 2 | 1 | 1 | 1 | |||||||||
| 12 | 0 | 0 | |||||||||||||
| 13 | 2 | 0 | 2 | 2 | 1 | 1 | 1 | ||||||||
| GROUP 2 | 1 | 0 | 3 | 3 | 1 | 1 | 3 | 2 | 1 | ||||||
| 2 | 0 | 4 | 1 | 2 | 4 | 1 | 4 | 1 | |||||||
| 3 | 0 | ||||||||||||||
| 4 | 0 | 1 | |||||||||||||
| 5 | 0 | 3 | 4 | 2 | 1 | 2 | 2 | 2 | 1 | ||||||
| 6 | 3 | 3 | 3 | 2 | 3 | 1 | |||||||||
| 7 | 0 | 4 | 2 | 4 | 2 | 2 | 2 | 4 | 2 | ||||||
| 8 | 0 | 4 | 4 | 3 | 3 | 2 | 3 | 3 | 3 | ||||||
| 9 | |||||||||||||||
| 10 | 3 | 3 | 4 | 3 | 3 | 2 | 1 | 1 | 1 | 1 | |||||
Study Groups for Cytokines
PBMC from cattle of each group were individually stimulated with bovine-PPD, avian-PPD, and Con A mitogen for 72h. Cytokine transcript responses were analyzed by semi-quantitative RT-PCR. The mRNA levels were calculated as the ratio between the band intensity of each cytokine and the corresponding β-actin (Figure 1). In group 1, the production of IFN-γ and IL-2 (108.2 ± 19 and 91.8 ± 9.8, respectively) in PBMC stimulated by bovine-PPD, was similar to Con A-stimulated cells, with the exception of IL-4 (70.2 ± 9.8). The mRNA levels upon stimulation by Con A were IFN-γ (118 ± 13), IL-2 (78.7 ± 9.5), and IL-4 (74.8 ± 9.8). In contrast, low transcript levels to IFN-γ (43.9 ± 6.6), IL-2 (19.3 ± 4.6), and IL-4 (5.9 ± 1.3) were detected in response to avian PPD. The expression of IL-10 was not detected in PBMC stimulated by bovine PPD, avian PPD nor Con-A (Figure 2A). On the contrary, in group 2, IL-10 had a significantly high production (85.7 ± 7.1) only upon stimulation by bovine PPD (Figure 2B). IL-10 was not detected in groups 1 and 3. Though there were transcripts to IL-10 upon PPD avium- or Con A-stimulation (31.4 ± 5.7 and 42.8 ± 4.6, respectively), the levels were similar to samples from non-stimulated cells (23.3 ± 3.8) (Figure 2 B). Bovine PPD also stimulated production of IL-4 (54 ± 7.7), which was 1.3 fold lower than group 1. The level of IL-4 transcript upon stimulation by PPD avium exhibited 3.7 fold more relative intensity (21.7 ± 2.9) than the bovines from group 1 (5.9 ± 1.3) (Figure 2. A and B). Similar production of IL-4 was also observed upon Con A-stimulation (20 ± 3.9). However, although cells produced IFN-γ and IL-2 upon stimulation by bovine PPD, avian PPD, or Con A, results were below the background level of the control group (Figure 2B).
Figure 1.Cytokine mRNA levels of bovine PPD stimulated Peripheral Blood Mononuclear Cells measured by semi-quantitative RT-PCR. A representative picture is shown. The expression levels of mRNA were calculated as the ratio between the band intensity of cytokine mRNA an that of corresponding β-actin mRNA in the linear range.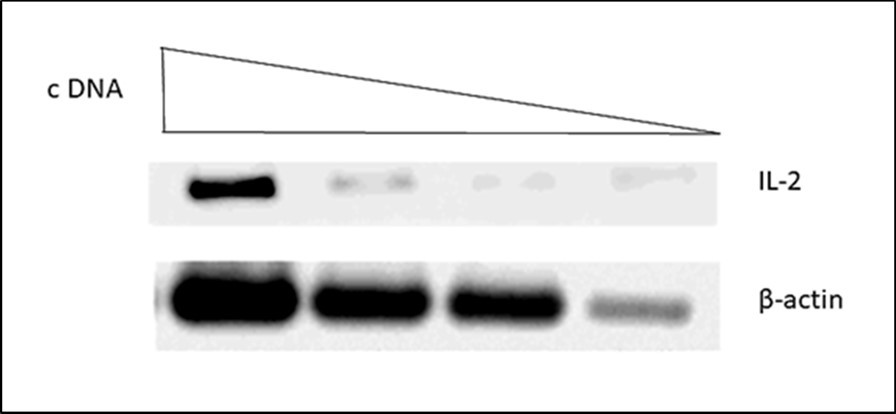
Figure 2.Semiquantitative RT-PCR analysis of cytokine mRNA expression in PBMC from naturally tuberculosis infected cattle after stimulation with bovine PPD (hatched bars), avian PPD (black dotted bars), Con-A (grey bars), or médium alone (White dotted bars) at 72 h of cell culture. (A) Group 1 (n=13), cows positive to all diagnosis tests (SICCT test, BOVIGAM and a serological comparative ELISA). (B) Group 2 (n=10), cows were only positive to comparative ELISA. (C) Control Group (n=10), cows negative to all disgnosis tests. Results are expressed as mean ± SD from relative intensity of the m RNAs (cytokine band/ β-actin band) measured by LabWorks 4.0 software.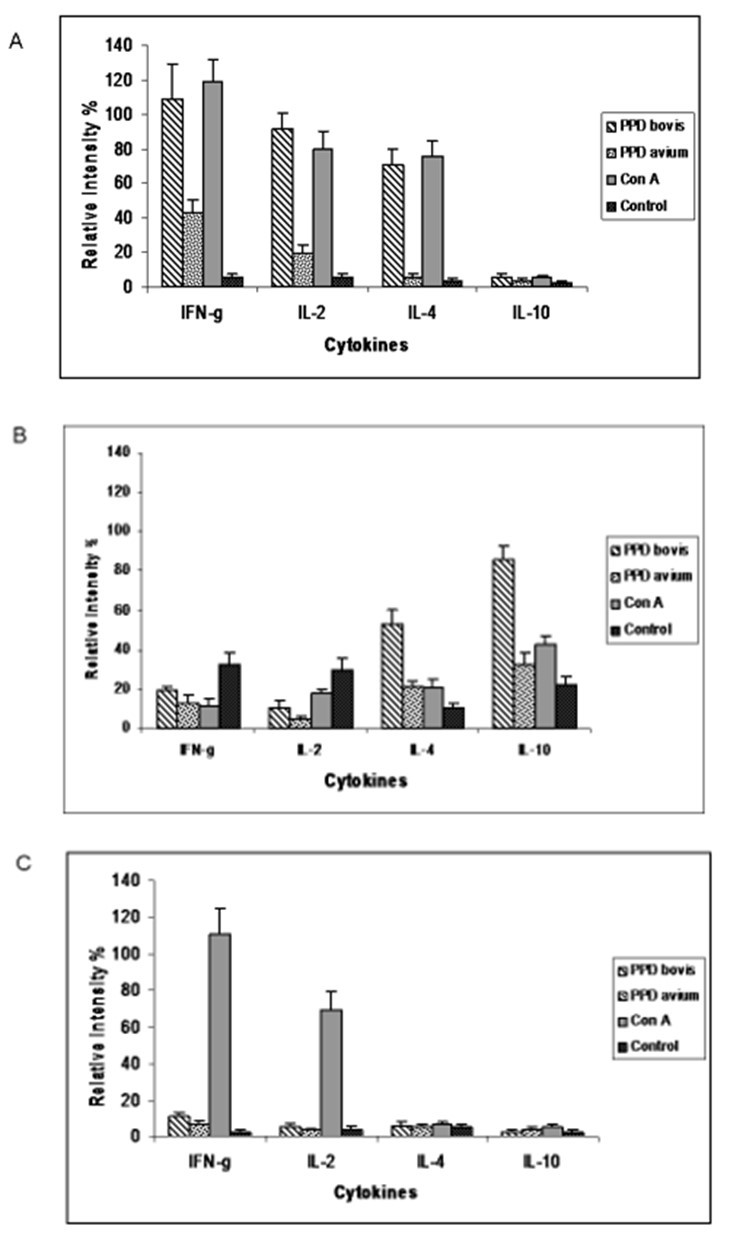
As expected, PBMC from healthy control group produced very low IFN-γ levels that were only detected after stimulation by bovine PPD (12 ± 3). In this group, IL-2, IL-4 and IL-10 transcripts were not observed upon stimulation, with neither bovine PPD nor avian PPD (Figure 2 C). Similarly, as expected, the control group produced IFN-γ (110 ± 13.6) and IL-2 (70.1 ± 10.7), but not IL-4 or IL-10 transcripts in response to stimulation by Con A.
Post-Mortem Examinations
Lymphatic tissue lesions were measured in parotid, submandibular, lateral and medial retropharyngeal, deep and superficial cervical, pulmonary, thoracic-tracheal, mediastinal, hepatic, inguinal, iliac, and mesenteric lymph nodes. In group 1, nine cows displayed mainly median lesions in deep cervical and lateral retropharyngeal lymph nodes. Twenty-three to 38% animals exhibited small lesions in examined lymph nodes, while 3 animals did not show any bTB-visible lesion in all the examined tissue. In group 2, 50-60% cows showed severe lesions in medial retropharyngeal, deep and superficial cervical lymph nodes, which were scored 3 and 4. Moreover, these animals showed severe pulmonary lesions that were considered as score 3. However, 2 cows did not show bTB-associated lesions in lymphatic tissues nor in lung lobes sliced into thin sections (Table 3).
Discussion
Ante mortem diagnosis to detect infected animals is critical for successful eradication and control of bovine tuberculosis. Tuberculin skin and interferon-gamma tests are both based on the detection of early cell-mediated immune response in tuberculosis infection are employed for eradication and control. However, at late disease stage, false negative results are observed due to waning cell-mediated immune response as opposed to a generally increasing humoral immune response. 16, 31, 32. After diagnosis, rapid removal of infected cattle is performed in order to reduce the risk of tuberculosis spread. On the other hand, in the presence of a virulent M. bovis strain, there are two types of animals, resistant and susceptible cattle to bovine tuberculosis. In susceptible animals, the infection is progressive possibly leading to anergy, while other M. bovis-infected animals are resistant to bTB. In this study, analyzed animals came from a herd where a comparative tuberculin skin test was applied for detection of cattle infected by M. bovis. At the time of the study, bovine tuberculosis prevalence determined by us was 25% using comparative tuberculin skin test. Therefore, blood sample was collected from all cows from the target herd for initial screening serologically by comparative ELISA, followed by subsequent studies on mostly animals with high levels of antibodies by BOVIGAM. Twenty-three bTB-suspected cattle were selected according to the results of the diagnostic tests used, 13 of them were positive to all the three tests (SICCT test, BOVIGAM and ELISA), while the remaining 10 were positive only to the serological test. The latter are considered probably anergic animals. Due to their negative test result to intradermal tuberculin test and the high levels of antibodies they had. In this sense, various authors have recommended alternative diagnostic tools based on the detection of serum M. bovis-specific antibodies 14,33. For this, mycobacterial antigens such as ESAT-6 and CFP-10 have been proposed to antigenic targets to improve the detection of M. bovis-infected animals 34. In addition, a large number of mycobacterial novel antigens recognized by serum antibodies from bTB-animals have showed being useful molecular candidates to the future development of a more sensitive serological assay 35. Our result showed that ELISA is an important serologic test to detect humoral response 36. So that, serological test should be considered as an ancillary test to be used in parallel with tuberculin skin test and IFN-γ assay. Gamma interferon release assay (BOVIGAM) has the ability to identify naturally infected animals at an early stage, when they have a negative intradermal tuberculin test result. Furthermore, BOVIGAM assay have greater sensitivity and specificity than intradermal tuberculin test showing greater correlation with pathology; however, it fails to detect anergic animals, a problem it shares with the tuberculin test 20,37.
On the other hand, other investigations on biomarkers such as cytokines from immune response have been conducted. The measurement of pro-inflammatory and anti-inflammatory cytokines could support to understand bovine immune response to develop diagnostic tools and design vaccines. In this way, PBMC from experimentally M. bovis-infected animals have been incubated by PPD bovis and after culture, cytokine mRNA production analyzed. In a study, transcripts to IFN-γ, TNF-α, iNOS and IL-4 were higher in bTB-cattle with high pathology than in those with low pathology 5. With regard to IL-10 cytokine, bTB-cattle with high pathology showed 2-fold less IL-10 mRNA than did animals with low pathology 5,38. This outcome has suggested that a strong immunological response is associated with increased pathology. A similar study showed that transcripts to IL-2, IL-17, and sometimes IL-10 could be potential predictors of disease progression in cattle exposed to M. bovis39. We also performed a PBMC culture to identify pro-inflammatory and anti-inflammatory cytokine transcripts by RT-PCR. We found that positive animals to all tests exhibited transcripts to IFN-γ, IL-2 and IL-4 by PBMC upon PPD bovis-specific or Con A-mitogen stimulation. Our results coincide with those from Thacker, et al (2007) 5 who found high IFN-γ and IL-4 levels in bTB-cattle with high pathology as cited. The presence of IL-4 in these animals is consistent with high serum anti-PPD bovis antibody titers, because IL-4 is an important factor to humoral immune response. IL-4 is also an anti-inflammatory cytokine that regulates the cell-mediated immune response.
In contrast, we found that bTB-suspected animals were negative to both tuberculin test and IFN-γ assay, and showed a high transcript levels for IL-10 upon stimulation by PPD bovis. This is consistent with the negative result to IFN-γ assay in these animals since IL-10 inhibits production of IFN-γ from PPD-stimulated whole blood cells. Some of the biological effects that are induced by IL-10 are deactivation of macrophages and decreased production of reactive nitrogen and oxygen species; consequently, in its absence, a stronger Th1 immune response is incited, while elevated levels of IL-10 are associated with increased susceptibility to mycobacterial infection, rendering the Th2-associated cytokine IL-10 a critical anti-inflammatory. Hence, the Th2-associated cytokine IL-10 is considered a critical anti-inflammatory mediator of innate and adaptive responses to pathogenic mycobacteria. It appears that mostly an inverse relationship exists between IL-10 and IFN-γ40. In this regard, Welsh., et al. (2005) 41 analyzed PBMC cytokine mRNA of experimentally infected cattle, and reported high IL-10 levels prior to infection, which gradually declined following infection as higher IFN-expression was detected. However, the authors registered a significant increase in the expression of IL-10 in cattle that showed the greatest severity of disease. In addition, levels higher in expression IL-10 correlated with decreasing CMI and increasing humoral responses.
Sheridan., et al (2017) 42 have demonstrated that IFN-γ levels increased when IL-10 was neutralized by addition of an anti-IL-10 antibody in bovine PPD-stimulated whole blood culture 36. We think animals from group 2 were found in an anergy status because cytokines decreased when PBMC were stimulated by Con A-mitogen. Moreover, positive animals to all tests showed small and medium lesions in lymph nodes during post-mortem examination. This result suggested that the animals were fighting the pathogen, or were tested at onset or during primary M. bovis-infection. Consistently, three animals from group 1 didn’t show lesions in tissues analyzed in the study. We concluded cows were infected with M. bovis, since stimulation of PBMC by M. avium showed a limited production of cytokine transcripts. On the contrary, animals from group 2 displayed the highest anti-M. bovis antibody levels in blood serum and a high IL-10 production only in M. bovis-stimulated cells. Likewise, these animals exhibit multifocal severe lesions in lymphatic tissues. Thus, the results suggested that animals from group 2 were experiencing progressive tuberculosis; possibly, they may be in an advanced stage. Hence, negative animals to both tuberculin skin test and IFN-γ assay, with suggestive signs of bTB should be subjected to ELISA test together with a bovine PPD stimulated-PBMC IL-10 cytokine expression. Therefore, the elucidation and understanding on the dynamics of Th1/Th2 cytokine profile changes at different stages of infection contributes to the identification and development of diagnostic biomarkers more efficient. In view of the above, if animals are not detected by tuberculin test or IFN-γ, we suggest the use of a serologic test to detect anti-M. bovis antigen antibodies together with IL-10 cytokine expression after cultured PBMC whole cells are stimulated by bovine PPD. Because, anergic animals constitute an important source of infection within herds, especially in those that are under disease control phase. The establishment of diagnostic methodologies based on the detection of specific antibodies against M. bovis antigens, together with tests that evaluate cellular immunity, will help improve disease control programs and reduce the risk of infection to humans and cattle, considering the different patterns of immune response that are observed as the disease progresses, as observed in the study conducted. Thus, the use of parallel testing that evaluate both types of immune response would allow herds sanitation to be achieved in less time, since the recurrence of reactors in herds declared free of tuberculosis can be prevented through planned follow-up.
Conclusion
The expression analysis of the main cytokines of the Th1/Th2 immunological profile carried out indicates differences among the evaluated groups, showing that high anti M. bovis antibody titers in blood serum and high IL-10 production in PBMC are indicators of progressive bTB when both tuberculin test and IFN-γ assay are negative in tuberculous anergic cattle. Consequently, the evaluation of these parameters or biomarkers in tandem with tests that evaluate cellular immunity will help to improve disease control programs.
Acknowledgements
Funding for this study came from the Instituto Nacional de Investigaciones Forestales, Agrícolas y Pecuarias (INIFAP) México, with a registry number in the Integral System of Institutional Management of 14294534013.
References
- 1.Good M, Bakker D, Duignan A, Collins D M. (2018) The history of in vivo tuberculin testing in bovines: Tuberculosis, a "one health" issue. Front Vet Sci. 5, 59.
- 2.Sorensen A, Beest F M van, R K Brook. (2014) Impacts of wildlife baiting and supplemental feeding on infectious disease transmission risk: a synthesis of knowledge. Prev Vet Med. 113, 356-63.
- 3.Welsh M D, Cunningham R T, Corbett D M, Girvin R M, McNair J et al. (2005) Influence of pathological progression on the balance between cellular and humoral immune responses in bovine tuberculosis. , Immunology 114, 101-111.
- 4.Walravens K, Wellemans V, Weynants V, Boelaert F, V de Bergeyck et al. (2002) Analysis of the antigen-specific IFN-gamma producing T-cell subsets in cattle experimentally infected with Mycobacterium bovis. , Vet Immunol Immunopathol 84, 29-41.
- 5.Thacker T C, Palmer M V, Waters W R. (2007) Associations between cytokine gene expression and pathology in Mycobacterium bovis infected cattle. Vet Immunol Immunopathol. 119, 204-213.
- 6.J S Rothel, S L Jones, L A Corner, J C Cox, P R Wood. (1992) The gamma-interferon assay for diagnosis of bovine tuberculosis in cattle: conditions affecting the production of gamma-interferon in whole blood culture. , Aust Vet J 69, 1-4.
- 7.Schiller I, Waters W R, H M Vordermeier, Nonnecke B, Welsh M et al. (2009) Optimization of a whole-blood gamma interferon assay for detection of Mycobacterium bovis-infected cattle. , Clin Vaccine Immunol 16, 1196-202.
- 8.Vordermeier M, S V Gordon, R G Hewinson. (2011) Mycobacterium bovis antigens for the differential diagnosis of vaccinated and infected cattle. , Vet Microbiol 151, 8-13.
- 9.Maggioli M F, M V Palmer, Thacker T C, H M Vordermeier, J L McGill et al. (2016) Increased TNF-α/ IFN-γ/IL-2 and decreased TNF-α/IFN-γ production by central memory T cells are associated with protective responses against bovine tuberculosis following BCG vaccination. , Front Immunol 7, 421.
- 10.Maggioli M F, Palmer M V, Thacker T C, Vordermeier H M, Waters W R. (2015) Characterization of effector and memory T cell subsets in the immune response to bovine tuberculosis in cattle. PLoS One. 10(4), 0122571.
- 11.A O Whelan, Villarreal-Ramos B, H M Vordermeier, P J Hogarth. (2011) Development of an antibody to bovine IL-2 reveals multifunctional CD4 T (EM) cells in cattle naturally infected with bovine tuberculosis. PLoS One. 6(12), 29194.
- 12.Widdison S, L J Schreuder, Villarreal-Ramos B, C J Howard, Watson M et al. (2006) Cytokine expression profiles of bovine lymph nodes: effects of Mycobacterium bovis infection and Bacille Calmette-Guérin vaccination. Clin Exp Immunol.144. 281-289.
- 13.Shu D, Heiser A, D N Wedlock, Luo D, Lisle G W de et al. (2014) Comparison of gene expression of immune mediators in lung and pulmonary lymph node granulomas from cattle experimentally with Mycobacterium bovis. Vet Immunol Immunopathol. 160, 81-89.
- 14.Casal C, Infantes J A, Risalde M A, Díez-Guerrier A, Domínguez M et al. (2017) Antibody detection tests improve the sensitivity of tuberculosis diagnosis in cattle. Res Vet Sci. 112, 214-221.
- 15. (1995) Norma Oficial Mexicana NOM-031-ZOO. Campaña Nacional Contra la Tuberculosis Bovina (Mycobacterium bovis). Secretaria de Agricultura, Ganadería y Desarrollo Rural.
- 16.Wood P R, Jones S L. (2001) BOVIGAM: an in vitro cellular diagnostic test for bovine tuberculosis. Tuberculosis (Edinb)81(1-2):. 147-55.
- 17.C O Thoen. (1990) The genus Mycobacterium. In:. Diagnostic Procedures in Veterinary Bacteriology and Mycology. 5th edition Carter GR, Cole Jr JR, editors , San Diego, CA 287-298.
- 18.Nagai S, Matsumoto J, Nagasuga T. (1981) Specific skin-reactive protein from culture filtrate of Mycobacterium bovis BCG. , Infect Immun 31, 1152-1160.
- 19.Bradford M M. (1976) A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding. , Anal Biochem 72, 248-254.
- 20.Díaz-Otero F, Banda-Ruíz V, Jaramillo-Meza L, Arriaga-Díaz C, González-Salazar D et al. (2003) Identification of Mycobacterium bovis infected cattle by immunological and molecular methods. , Vet. Méx 34, 13-26.
- 21.Boyum A. (1976) Isolation of lymphocytes, granulocytes and macrophages. , Scand J Immunol 5, 9-15.
- 22.Chomczynski P, Sacchi N. (1987) Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. , Anal Biochem 162, 156-159.
- 23.D P Cerreti, McKereghan K, Larsen A, M A Cantrell, Anderson D et al. (1986) Cloning, sequence, and expression of bovine interleukin 2. Proc Natl Acad Sci USA 83, 3223-3227.
- 24.D P Cerreti, McKereghan K, Larsen A, Cosman D, Gillis S et al. (1986) Cloning, sequence, and expression of bovine interferon-gamma. , J Immunol 136, 4561-4564.
- 25.Heussler V T, Eichhorn M, Dobbelaere D A. (1992) Cloning of a full-length cDNA encoding bovine interleukin 4 by the polymerase chain reaction. , Gene 114, 273-278.
- 26.S M Hash, W C Brown, A C Rice-Ficht. (1994) Characterization of a cDNA encoding bovine interleukin 10: kinetics of expression in bovine lymphocytes. , Gene 139, 257-61.
- 27.J L Degen, M G Newbauer, S J Degen, C E Seyfreid, D R Morris. (1983) Regulation of protein synthesis in mitogen-activated bovine lymphocytes. Analysis of actin-specific and total mRNA accumulation and utilization. , J Biol Chem 258, 12153-12162.
- 28.Covert J, Splitter G. (1995) Detection of cytokine transcriptional profiles from bovine peripheral blood mononuclear cells and CD4+ lymphocytes by reverse transcriptase polymerase chain reaction. , Vet Immunol Immunopathol 49, 39-50.
- 29.Beltan E, Horgen L, Rastogi N. (2000) Secretion of cytokines by human macrophages upon infection by pathogenic and non-pathogenic Mycobacterium. Microbial Pathogenesis. 28, 313-318.
- 30.H M Vordermeier, M A Chambers, P J Cockle, A O Whelan, Simmons J et al. (2002) Correlation of ESAT-6-specific gamma interferon production with pathology in cattle following Mycobacterium bovis BCG vaccination against experimental bovine tuberculosis. , Infect. Immun 70, 3026-3032.
- 31.T A Clegg, Good M, Doyle M, Duignan A, S J More et al. (2017) The performance of the interferon gamma assay when used as a diagnostic or quality assurance test in Mycobacterium bovis infected herds. Prev Vet Med. 140, 116-121.
- 32.De La Rua-Domenech, Goodchild R, A T Vordermeier, Hewinson H M, Christiansen R G et al. (2006) Ante mortem diagnosis of tuberculosis in cattle: a review of the tuberculin tests, gamma-interferon assay and other ancillary diagnostic techniques. Res Vet Sci. 81-190.
- 33.Casal C, Díez-Guerrier A, Álvarez J, Rodríguez-Campos S, Mateos A et al. (2014) Strategic use of serology for the diagnosis of bovine tuberculosis after intradermal skin testing. , Vet Microbiol 170, 342-351.
- 34.H M Vordermeier, S V Gordon, R G Hewinson. (2011) Mycobacterium bovis antigens for the differential diagnosis of vaccinated and infected cattle. , Vet Microbiol 151, 8-13.
- 35.K P Lyashchenko, Grandison A, Keskinen K, Sikar-Gang A, Lambotte P et al. (2017) Identification of novel antigens recognized by serum antibodies in bovine tuberculosis. Clin Vaccine Immunol. , H.M., Stabel, J.R., Thacker, T.C., Palmer, M.V., Waters, WR 24(12), 00259-17.
- 36.Quevillon E, Díaz-Otero F, Jaramillo-Meza L, Lascurain-Ledesma R, J A Gutiérrez-Pabello et al. (2013) Comparison of immune peripheral blood cells in tuberculin reactor cattle that are seropositive or seronegative for Mycobacterium bovis antigens. , Vet Immunol Immunopathol 153, 194-201.
- 37.S D Neill, Cassidy J, Hanna J, D P Mackie, J M Pollock et al. (1994) Detection of Mycobacterium bovis infection in skin test-negative cattle with an assay for bovine interferon-gamma. Vet Rec. 135, 134-135.
- 38.Cruz-Fierro M, Jaramillo-Meza L, C I Espitia-Pinzón, Pérez-González R, Manzo-Sandoval A et al. (2020) Evaluación experimental de vacuna BCG y extracto proteico en bovinos, vía expresión de citocinas. Spei Domus. 16, 1-27.
- 39.Smith K, Kleynhans L, R M Warren, W J Goosen, M A. (2021) Cell-mediated immunological biomarkers and their diagnostic application in livestock and wildlife infected with Mycobacterium bovis. , Front Immunol 4(12), 639605.
- 40.Welsh M D, Cunningham R T, Corbett D M, Girvin R M, McNair J et al. (2005) Influence of pathological progression on the balance between cellular and humoral immune responses in bovine tuberculosis. , Immunology 114, 101-11.
