Phytoplankton Community of Taylor Creek in the Niger Delta Using Diversity Indices
Abstract
This study evaluated the phytoplankton community of Taylor creek from Polaku to Agbia between November 2013 and July 2014. Sampling was carried out in 12 locations along the stretch of the creek following standard protocol. The phytoplankton enumeration was done and identified accordingly. In all the 12 locations, 100 species of phytoplankton belonging to 14 taxonomic groups including Bacillariophyta (31 species), Chlorophyta (25 species), Pyrrophyta (4 species), Cyanophyta (17 species), Spermatophyta (9 species), Euglenophyta (3 species), Phaeophyta (1 species), Heterokontophyta (2 species), Myxophyta (1 species), Xanthophyta (1 species), Chrysophyta (2 species), Lycopodiophyta (1 species), Cryptophyta (1 species), Dinophyta (2 species). Bacillariophyta, Chlorophyta, Cyanophyta and Spermatophyta with occurrence rate of 36%, 34%, 10% and 10%, respectively were the predominant phytoplankton in the study area occurring in all the locations across the period of the study. The various species have some distinct environmental condition that enable them thrive in their niche, and this determines the structure of the phytoplankton in the study area. The diversity indices provided information about the distribution and health condition of the creek. The study showed significant relationships between Shannon-Wiener, Menhinick, Margalef, equitability and Fisher-alpha indices. The ecosystem showed moderate to heavy pollution based on Shannon Wiener index. There is the need for improved method of waste disposal and other anthropogenic activities being carried in and/ or within the creek.
Author Contributions
Academic Editor: Narcisa Vrinceanu, Faculty of Engineering, "Lucian Blaga†University of Sibiu / 4 Emil Cioran Street, 550025 Sibiu, Romania.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2018 Emmanuel Nwabueze Ogamba, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Plankton are aquatic organisms that drift with water movement and possesses no locomotive organs 1. Phytoplankton rely on sunlight for photosynthesis to produce their own food. Phytoplankton are among the major organisms that make up the aquatic food chains, and play essential role in the aquatic food web. Phytoplankton are food to smaller organisms such as fish in the aquatic ecosystem. These phytoplankton consist of microscopic algae, such as diatoms, and dinoflagellates. As such they are among the primary producers in the aquatic ecosystem 2.
Phytoplankton are among the organisms that are used for the assessment of pollution in aquatic ecosystem 3. The distribution, abundance and composition of the phytoplankton is essential in surface water quality assessment 4. Phytoplankton also provide vital information about the nutrient level in an aquatic ecosystem. Basically in the aquatic ecosystem, pollution could influence green pigment of many algae.
Specific taxa of phytoplankton have been reported in literature, and some of these phytoplankton could also cause diseases. This is because some species of phytoplankton produce powerful biotoxins that could be toxic to aquatic life forms such as fishes and even humans that consume fishes contaminated by this harmful phytoplankton.
The main types of algae are the Diatoms (Bacillariaceae) with yellow or brown pigment in the chromatophores which are so abundant that they mask the green colour of chlorophyll; Green algae (Chlorophyta) having both floating and attached forms; Blue green algae (Cyanophyta) having diffused chlorophyll in the algal cells which is masked by blue green pigments; and Filamentous green algae or pond scum 5. Phytoplankton such as Navicula, Fragilaria, Asterionella and Nitzschia (Bacillariaceae) are good water quality indicators 5, while Spirogyra, Oedogonium(Filamentous green algae) are also used as pollution indicator. Cyanophyta such as Anabaena, Gloetrichia, Oscillatoria etc are good phytoplankton species that fix nitrogen.
Phytoplankton is used to assess the health of aquatic ecosystem, so as to enhance the effectiveness of surface water management 6. As such environmental law include the assessment of plankton communities in surface water close to industrial area. Specifically, European Water Framework Directive expects member states to monitor phytoplankton abundance and composition in an aquatic ecosystem 7, 8.
In many coastal regions of the Niger Delta, Nigeria pollution do occur due to human activities in the water ways. In some regions, municipal wastes are discharged into the water bodies with little or no obstruction. Runoff is also contributing to surface water contamination. As such there is need to assess pollution level in the aquatic ecosystem. To this regard several diversity indices have been widely used to assess environmental contamination, spatial and temporal distribution of biological organisms in surface water. This present study assesses the phytoplankton communities of Taylor creek in the Niger Delta using diversity indices.
Materials and Methods
Study Area
Taylor Creek and its tributaries is an offshoot of Orashi River and empties into River Nun. The area of study lies between Polaku community to Agbia community in Yenagoa Local Government Area of Bayelsa State, Nigeria. The water is a major source of livelihood of indigenous people of the area because of fishing activities. The creek also receiving domestic wastes including sewage from the residents of the area. Artisanal dredging is also carried out along the creek. The creek has tributaries at Oku-Oba, Imbiyai-Oba, Kala-oba, Opu-oba, Pini-oba, Etele-bou (Kemie) in the study area. The climatic condition of the area is similar to other parts of the Niger Delta which are characterized by 5 months’ of dry season (November to March of the following year) and 7 months’ wet season (April to October).
Sampling Process
The phytoplankton was collected from 12 locations viz: A (Izewaribi) (006o 16’ 48.5” E and 05o 01’ 50.1” N), B (Oku-oba) (006º 17’ 02.9” E and 05º 01’ 56.7” N), C (Amase-pou) (006º 17’ 48.8” E and 05º 02’ 18.5” N), D (Imbiyai-oba) (006º 17’ 59.3” E and 05º 02’ 05.2” N), E (Kala-oba) (006º 18’ 30.2” E and 05º 02’ 01.0” N), F (Obunagha) (006º 18’ 38.2” E and 05º 02’ 02.5” N), G (Opu-oba) (006º 18’ 56.2” E and 05º 02’ 05.7” N), H (Pini-oba) (006º 19’ 18.5” E and 05º 02’ 18.8” N), I (Court Kiri) (006º 19’ 27.4” E and 05º 02’ 51.8” N), J (Ogboloma) (006º 20’ 11.4” E and 05º 03’ 00.0” N), K Etelebou (Kemie) (006º 20’ 38.1” E and 05º 02’ 57.4” N) and L (Unka) (006º 21’ 29.6” E and 05º 03’ 29.6” N) with aid of plankton net with a mesh size of 30-70nm with a 0.5m diameter by 2m long conical plankton net with 30cm3 container. The samples were carefully dispensed into 1litre wide mouthed plastic containers and immediately fixed with 4% formalin, stored in a cool box for identification and enumeration 9. In the laboratory samples were allowed to stand for a minimum of 24 hours before the supernatant was carefully pipetted off until a 50ml concentrated sample was achieved 10.
Counting and Identification
In the laboratory, plankton samples were allowed to settle by gravity for 24 hours before decanting carefully the supernatant 10to achieve 50 ml volume. From the stock sample, 1 ml sub-sample was taken with the help of a Pasteur pipette and transferred into a Sedgwick Rafter counting chamber, and then allowed to settle for approximately 5-10 minutes. A DC2 camera (Lieder Model; MC 332) was attached to a computer and used for the identification processes. Identification guides of Botes 11, Vashishta et al. 12 were used for plankton identification.
Statistical Analysis
The diversity obtained was analysed using Paleontological statistics software package by Hammer et al. 13. Pearson correlation of the diversity indices carried out using SPSS version 20. The charts presented in this study were plotted using Microsoft excel. The Renkonen’s Number was calculated based on the method described by Ogbeibu 14.
Results and Discussion
A total of one hundred (100) species of phytoplankton belonging to fourteen (14) taxonomic groups were recorded from Taylor Creek and its tributaries. Bacillariophyta was represented by 31 species, Chlorophyta (25 species), Pyrrophyta (4 species), Cyanophyta (17 species), Spermatophyta (9 species), Euglenophyta (3 species), Phaeophyta (1 species), Heterokontophyta (2 species), Myxophyta (1 species), Xanthophyta (1 species), Chrysophyta (2 species), Lycopodiophyta (1 species), Cryptophyta (1 species), Dinophyta (2 species) (Figure 1). The occurrence distribution of the taxa is presented in Table 2. Bacillariophyta, Chlorophyta, Cyanophyta and Spermatophyta with occurrence rate of 36%, 34%, 10% and 10%, respectively were the predominant phytoplankton in the study area occurring in all the locations across the period of the study. While Phaeophyta, Myxophyta, Xanthophyta Lycopodiophyta and Cryptophyta has the least number of species. (Figure 2)
Figure 1.Mean of taxonomic group-based phytoplankton population density at the sampling locations in Taylor creek between November 2013 to July 2014
Figure 2.Distribution occurrence of each taxonomic group among the population of phytoplankton between November 2013 to July 2014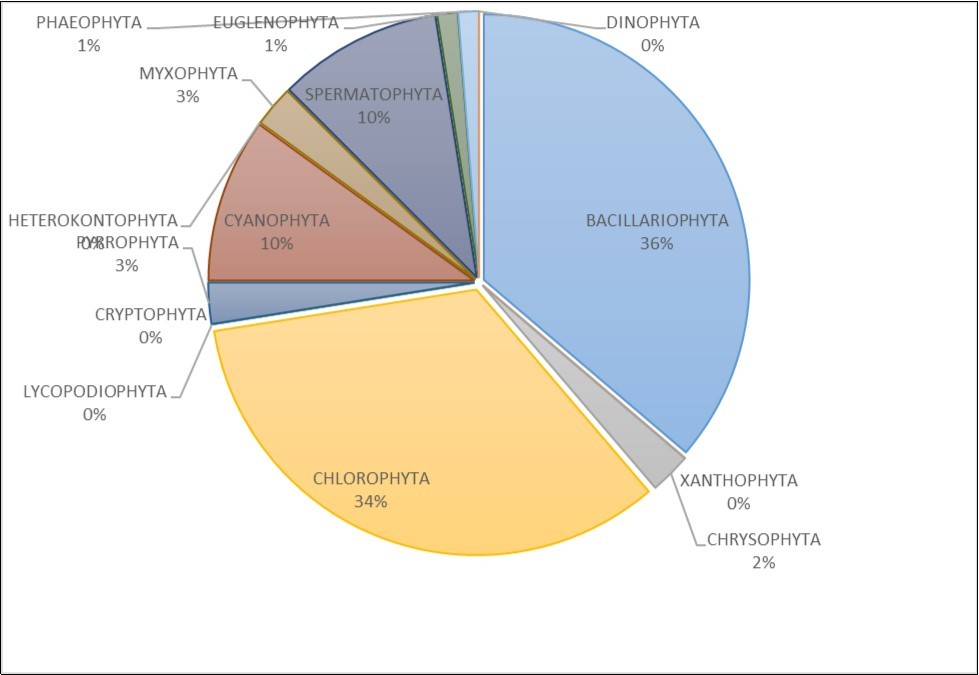
Location A (Polaku, Izewaribi): The phytoplankton species counted from this location are Bacillariophyta (31 species), Chlorophyta (27 species, Pyrrophyta (2 species), Cyanophyta (8 species), Spermatophyte (8 species), Euglenophyta (1 species), Phaeophyta (1 species), Myxophyta (2 species), Chrysophyta (2 species) and Cryptophyta (1 species), making a total of 81 species. Ecological indices at this Location was high with Margalef index (1.826), Shannon-Wiener index (1.581), fairly high with Evenness index (0.540) and low with Dominance (0.268) (Table 1).
Location B (Polaku, Oku-Oba: A total of 73 species of phytoplankton belonging to Bacillariophyta (37 species), Chlorophyta (16 species), Pyrrophyta (1 species), Cyanophyta (12 species), Spermatophyta (5 species), Euglenophyta (1 species), Myxophyta (1 species) were recorded in this study location. Ecological indices show a very minimal variation within this Location over the period. Some of the diversity indices assessed were Dominance, Shannon Wiener index, Evenness index and Margalef index with 0.337, 1.334, 0.542 and 1.398, respectively (Table 1).
Location C (Koroama, Amase-pon): This Location had Bacillariophyta, Chlorophyta, Cyanophyta, Spermatophyta, Phaeophyta and Dinophyta with 14 species, 17 species, 4 species, 2 species, 3 species and 1 species, respectively. The abundance was fairly distributed due to human activities and disturbances. Some of the indices studied were Dominance (0.306), Evenness (0.668), Margalef index (1.346) and Shannon-Wiener index (1.388) (Table 1).
Location D (Koroama, Imbiyai-Oba): The encountered taxa of phytoplankton were Bacillariophyta (24 species), Chlorophyta (11 species), Pyrrophyta (2 species), Cyanophyta (5 species), Spermatophyta (2 species), Heterokontophyta (1 species). A total of 45 species were recorded within the Location in the taxonomic group. Diversity indices values Dominance (0.361), Evenness index (0.603), Shannon-Wiener index (1.285) and Margalef index (1.313) (Table 1).
Location E (Obunagha, Kala-Oba): Phytoplankton species recorded in this Location were 69 species including Bacillariophyta (36 species), Chlorophyta (21 species), Cyanophyta (4 species), Spermatophyta (5 species), Phaeophyta (2 species), Chrysophyta (1 species). Diversity indices were Dominance (0.375), Evenness index (0.565), Margalef index (1.181) and Shannon-Wiener index (1.221).
Location F (Obunagha): Phytoplankton recorded in this Location were 5 taxonomic groups including Bacillariophyta (27 species), Chlorophyta (19 species), Cyanophyta (3 species), and Spermatophyta (3 species). Hence, a total of 52 species were recorded within the Location. Diversity indices showed that Dominance (0.410) and Shannon-Wiener index (1.037), Evenness index (0.705) were high and Margalef Index (0.759) were low.
Location G (Okolobiri, Opu-Oba): This Location recorded 6 taxas of phytoplankton with a total number of 69 species belonging to Bacillariophyta (33 species), Chlorophyta (22 species), Cyanophyta (8 species), Spermatophyta (3 species), Euglenophyta (2 species), and Chrysophyta (1 species). Diversity indices showed that Dominance, Evenness, Margalef index and Shannon-Wiener index were 0.347, 0.592, 1.181 and 1.267, respectively.
Location H (Okolobiri, Pini-Oba): The taxonomic groups recorded in this Location were Bacillariophyta (13 species), Chlorophyta (8 species), Cyanophyta (6 species), Spermatophyta (3 species), and Phaeophyta (1 species) making a total number of 31 species. Diversity indices such as Dominance, Evenness index, Margalef index and Shannon-Wiener index had a value of 0.290, 0.786, 1.165, and 1.369, respectively (Table 1).
Location I (Okolobiri, Court-Kiri): This Location possessed 5 taxonomic groups comprising of 36 species. The taxonomic groups are distributed into Bacillariophyta (13 species), Chlorophyta (16 species), Cyanophyta (3 species), Spermatophyta (3 species), and Chrysophyta (1 species). Diversity indices were Dominance (0.343), Shannon-Wiener index (1.242), Evenness index (0.693) and Margalef index (1.116) (Table 1).
Location J (Ogboloma): A total of 6 taxonomic groups and 51 species were recorded in this location. The taxas were Bacillariophyta (19 species), Chlorophyta (15 species), Cyanophyhta (9 species), Spermatophyta (7 species), and Heterokontophyta (1 species) of the total. Diversity indices were Dominance (0.276), Shannon-Wiener index (1.384), Evenness index (0.798) and Margalef index (1.017) (Table 1).
Location K (Ogboloma (Etelebou) Kemie): The phytoplankton taxas encountered in this Location were Bacillariophyta (with 15 species), Chlorophyta (with 25 species), Cyanophyta (with 18 species), Spermatophyta (with 4 species), Heterokontophyta (with 1 species) and Xanthophyta (with 1 species). Diversity indices were Dominance (0.291), Shannon-Wiener index (1.3679), Evenness index (0.654) and Margalef index (1.202) (Table 1).
Location L (Agbia, Unka): This Location recorded a total of 57 phytoplankton species. The taxonomic group found in this location include Bacillariophyta (15 species), Chlorophyta (29 species) Cyanophyta (5 species), Spermatophyta (4 species), Phaeophyta (1 species), Lycopodiophyta (1species), Cryptophyta (1species), and Dinophyta (1 species). Diversity indices values of dominance, Margalef index, Shannon-Wiener index and Evenness index were 0.342, 1.731, 1.379 and 0.496, respectively (Table 1). with "The species present across the Locations were in increasing order of A > B > E > G > K > L > F > J > D > C > I > H with total species of 81, 73, 69, 69, 64, 57, 52, 51, 45, 41, 36 and 31, respectively (Figure 3).
Table 1. Overall distribution of plankton community in the study area using diversity indices| Indices | Locations | |||||||||||
| A | B | C | D | E | F | G | H | I | J | K | L | |
| Dominance | 0.268 | 0.337 | 0.306 | 0.361 | 0.375 | 0.410 | 0.347 | 0.290 | 0.343 | 0.276 | 0.291 | 0.342 |
| Simpson | 0.733 | 0.663 | 0.694 | 0.639 | 0.626 | 0.590 | 0.653 | 0.710 | 0.657 | 0.724 | 0.709 | 0.658 |
| Shannon | 1.581 | 1.334 | 1.388 | 1.285 | 1.221 | 1.037 | 1.267 | 1.369 | 1.242 | 1.384 | 1.367 | 1.379 |
| Evenness | 0.540 | 0.542 | 0.668 | 0.603 | 0.565 | 0.705 | 0.592 | 0.786 | 0.693 | 0.798 | 0.654 | 0.496 |
| Menhinick | 1.006 | 0.819 | 0.937 | 0.894 | 0.722 | 0.555 | 0.722 | 0.898 | 0.833 | 0.700 | 0.750 | 1.060 |
| Margalef | 1.826 | 1.398 | 1.346 | 1.313 | 1.181 | 0.759 | 1.181 | 1.165 | 1.116 | 1.017 | 1.202 | 1.731 |
| Equitability | 0.720 | 0.686 | 0.775 | 0.717 | 0.681 | 0.748 | 0.707 | 0.850 | 0.772 | 0.860 | 0.763 | 0.663 |
| Fisher_alpha | 2.603 | 1.907 | 1.936 | 1.859 | 1.579 | 1.010 | 1.579 | 1.687 | 1.577 | 1.373 | 1.621 | 2.534 |
| Berger-Parker | 0.363 | 0.507 | 0.415 | 0.533 | 0.522 | 0.519 | 0.478 | 0.419 | 0.444 | 0.373 | 0.391 | 0.509 |
Figure 3.Distribution of total phytoplankton population density at the sampling locations in Taylor creek between November 2013 to July 2014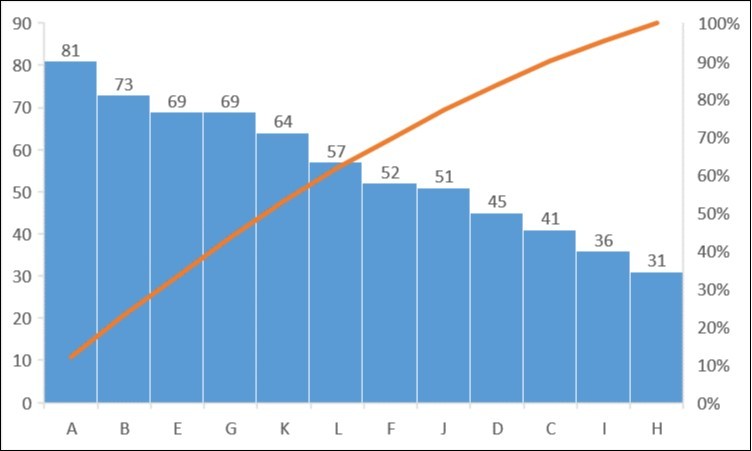
The variations in the diversity indices among the various study locations may be associated to anthropogenic activities in and within the creek that is causing an alteration in the water quality especially with regard to phosphate, Sulphate and Nitrite which encourage algal bloom and eutrophication. Also, surface runoff resulting from several activities and sewage could also be contributing to differences in the diversity indices in the various locations
During the study, Bacillariophyta, Chlorophyta, Cyanophyta and Spermatophyta were the predominant phytoplankton occurring in all the locations, hence dominance, Shannon Wiener, Simpson, evenness, Menhinick, Margalef, equitability, Fisher_alpha and Berger-Parker indices were used to show the relationship between the various locations (Table 2). The phytoplankton of Taylor creek during the period of study showed that dominance was highest in location C (0.500) and lowest in location E (0.088). These values suggest slight variation in dominance of few species in the various locations. Shannon-Wiener index ranged from 0.00 to 2.414. Basically this index range from 0 – 5, where <1 (heavy pollution), 1 to 2 (moderate pollution) and above 3 (indicates stable conditions) 15, 16, 17. Based on these criteria, the Taylor creek is moderate to heavily polluted. Evenness index ranged from 0.577 to 1.000. Simpson index ranged from 0.000 – 0.912. In most of the locations, high Simpson index is an indication of mature and stable community, while areas with low values may suggest the effect of stress. According to Whittaker 18, Shah and Pandit 15, locations with low species always have higher Simpson value, and when dominance is shared by large number of species. As such, in this study, anthropogenic activities could be impacting on some of the phytoplankton community of the creek. In most cases, an increase in Simpson index makes evenness to go in opposite direction 15. Simpson showed negative significant relationship with evenness (r = -0.375, P< 0.01) (Table 3). This trend has been previously reported by Shah and Pandit 15.
Table 2. Diversity indices of the study area of the four predominant phytoplankton from November 2013 to July 2014 at Taylor creek, Bayelsa state, Nigeria| Diversity indices | Bacillariophyta | Chlorophyta | Cyanophyta | Spermatophyta |
| Location A | ||||
| Dominance | 0.115 | 0.111 | 0.188 | 0.406 |
| Simpson | 0.885 | 0.889 | 0.813 | 0.594 |
| Shannon-Wiener index | 2.414 | 2.377 | 1.733 | 0.974 |
| Evenness | 0.745 | 0.829 | 0.943 | 0.883 |
| Menhinick | 2.785 | 2.502 | 2.121 | 1.061 |
| Margalef | 4.158 | 3.641 | 2.404 | 0.962 |
| Equitability | 0.892 | 0.927 | 0.967 | 0.887 |
| Fisher_alpha | 12.500 | 9.857 | 10.910 | 1.743 |
| Berger-Parker | 0.207 | 0.222 | 0.250 | 0.500 |
| Location B | ||||
| Dominance | 0.118 | 0.141 | 0.153 | 0.360 |
| Simpson | 0.882 | 0.859 | 0.847 | 0.640 |
| Shannon-Wiener index | 2.363 | 2.133 | 1.979 | 1.055 |
| Evenness | 0.759 | 0.844 | 0.905 | 0.957 |
| Menhinick | 2.302 | 2.500 | 2.309 | 1.342 |
| Margalef | 3.600 | 3.246 | 2.817 | 1.243 |
| Equitability | 0.895 | 0.927 | 0.952 | 0.960 |
| Fisher_alpha | 8.203 | 11.410 | 10.490 | 3.167 |
| Berger-Parker | 0.216 | 0.250 | 0.250 | 0.400 |
| Location C | ||||
| Dominance | 0.174 | 0.260 | 0.250 | 0.500 |
| Simpson | 0.827 | 0.741 | 0.750 | 0.500 |
| Shannon-Wiener index | 1.834 | 1.773 | 1.386 | 0.693 |
| Evenness | 0.894 | 0.654 | 1.000 | 1.000 |
| Menhinick | 1.871 | 2.183 | 2.000 | 1.414 |
| Margalef | 2.274 | 2.824 | 2.164 | 1.443 |
| Equitability | 0.942 | 0.807 | 1.000 | 1.000 |
| Fisher_alpha | 5.571 | 7.753 | 0.000 | 0.000 |
| Berger-Parker | 0.214 | 0.471 | 0.250 | 0.500 |
| Location D | ||||
| Dominance | 0.142 | 0.124 | 0.280 | 1.000 |
| Simpson | 0.858 | 0.876 | 0.720 | 0.000 |
| Shannon-Wiener index | 2.175 | 2.146 | 1.332 | 0.000 |
| Evenness | 0.800 | 0.950 | 0.947 | 1.000 |
| Menhinick | 2.200 | 2.714 | 1.789 | 0.707 |
| Margalef | 3.107 | 3.336 | 1.864 | 0.000 |
| Equitability | 0.907 | 0.977 | 0.961 | 0.000 |
| Fisher_alpha | 7.504 | 23.150 | 9.284 | 0.796 |
| Berger-Parker | 0.280 | 0.182 | 0.400 | 1.000 |
| Location E | ||||
| Dominance | 0.088 | 0.134 | 0.375 | 0.680 |
| Simpson | 0.912 | 0.866 | 0.625 | 0.320 |
| Shannon-Wiener index | 2.546 | 2.199 | 1.040 | 0.500 |
| Evenness | 0.851 | 0.820 | 0.943 | 0.825 |
| Menhinick | 2.500 | 2.400 | 1.500 | 0.894 |
| Margalef | 3.907 | 3.285 | 1.443 | 0.621 |
| Equitability | 0.940 | 0.917 | 0.946 | 0.722 |
| Fisher_alpha | 9.655 | 9.332 | 5.453 | 1.235 |
| Berger-Parker | 0.139 | 0.238 | 0.500 | 0.800 |
| Location F | ||||
| Dominance | 0.127 | 0.163 | 0.333 | 0.556 |
| Simpson | 0.873 | 0.837 | 0.667 | 0.444 |
| Shannon-Wiener index | 2.255 | 2.059 | 1.099 | 0.637 |
| Evenness | 0.795 | 0.784 | 1.000 | 0.945 |
| Menhinick | 2.228 | 2.294 | 1.732 | 1.155 |
| Margalef | 3.267 | 3.057 | 1.820 | 0.910 |
| Equitability | 0.908 | 0.894 | 1.000 | 0.918 |
| Fisher_alpha | 7.669 | 8.541 | 0.000 | 2.622 |
| Berger-Parker | 0.207 | 0.316 | 0.333 | 0.667 |
| Location G | ||||
| Dominance | 0.147 | 0.124 | 0.281 | 0.333 |
| Simpson | 0.854 | 0.876 | 0.719 | 0.667 |
| Shannon-Wiener index | 2.117 | 2.350 | 1.321 | 1.099 |
| Evenness | 0.755 | 0.807 | 0.937 | 1.000 |
| Menhinick | 1.945 | 2.772 | 1.414 | 1.732 |
| Margalef | 2.885 | 3.882 | 1.443 | 1.820 |
| Equitability | 0.883 | 0.916 | 0.953 | 1.000 |
| Fisher_alpha | 5.926 | 13.350 | 3.184 | 0.000 |
| Berger-Parker | 0.250 | 0.273 | 0.375 | 0.333 |
| Location H | ||||
| Dominance | 0.208 | 0.188 | 0.278 | 0.333 |
| Simpson | 0.792 | 0.813 | 0.722 | 0.667 |
| Shannon-Wiener index | 1.748 | 1.733 | 1.330 | 1.099 |
| Evenness | 0.821 | 0.943 | 0.945 | 1.000 |
| Menhinick | 2.021 | 2.121 | 1.633 | 1.732 |
| Margalef | 2.415 | 2.404 | 1.674 | 1.820 |
| Equitability | 0.898 | 0.967 | 0.959 | 1.000 |
| Fisher_alpha | 7.028 | 10.910 | 5.245 | 0.000 |
| Berger-Parker | 0.333 | 0.250 | 0.333 | 0.333 |
| Location I | ||||
| Dominance | 0.207 | 0.250 | 0.333 | 1.000 |
| Simpson | 0.793 | 0.750 | 0.667 | 0.000 |
| Shannon-Wiener index | 1.672 | 1.661 | 1.099 | 0.000 |
| Evenness | 0.887 | 0.752 | 1.000 | 1.000 |
| Menhinick | 1.664 | 1.750 | 1.732 | 0.577 |
| Margalef | 1.949 | 2.164 | 1.820 | 0.000 |
| Equitability | 0.933 | 0.854 | 1.000 | 0.000 |
| Fisher_alpha | 4.322 | 4.745 | 0.000 | 0.525 |
| Berger-Parker | 0.308 | 0.438 | 0.333 | 1.000 |
| Location J | ||||
| Dominance | 0.114 | 0.173 | 0.161 | 0.265 |
| Simpson | 0.886 | 0.827 | 0.840 | 0.735 |
| Shannon-Wiener index | 2.333 | 1.987 | 1.889 | 1.475 |
| Evenness | 0.859 | 0.810 | 0.945 | 0.874 |
| Menhinick | 2.753 | 2.324 | 2.333 | 1.890 |
| Margalef | 3.736 | 2.954 | 2.731 | 2.056 |
| Equitability | 0.939 | 0.904 | 0.971 | 0.917 |
| Fisher_alpha | 13.980 | 9.500 | 14.490 | 7.824 |
| Berger-Parker | 0.211 | 0.333 | 0.222 | 0.429 |
| Location K | ||||
| Dominance | 0.153 | 0.206 | 0.414 | 0.375 |
| Simpson | 0.847 | 0.794 | 0.586 | 0.625 |
| Shannon-Wiener index | 1.970 | 1.842 | 1.242 | 1.040 |
| Evenness | 0.897 | 0.701 | 0.577 | 0.943 |
| Menhinick | 2.138 | 1.800 | 1.414 | 1.500 |
| Margalef | 2.652 | 2.485 | 1.730 | 1.443 |
| Equitability | 0.948 | 0.838 | 0.693 | 0.946 |
| Fisher_alpha | 7.757 | 5.043 | 3.152 | 5.453 |
| Berger-Parker | 0.214 | 0.320 | 0.611 | 0.500 |
| Location L | ||||
| Dominance | 0.236 | 0.130 | 0.280 | 0.375 |
| Simpson | 0.764 | 0.870 | 0.720 | 0.625 |
| Shannon-Wiener index | 1.582 | 2.249 | 1.332 | 1.040 |
| Evenness | 0.811 | 0.790 | 0.947 | 0.943 |
| Menhinick | 1.549 | 2.228 | 1.789 | 1.500 |
| Margalef | 1.846 | 3.267 | 1.864 | 1.443 |
| Equitability | 0.883 | 0.905 | 0.961 | 0.946 |
| Fisher_alpha | 3.706 | 7.669 | 9.284 | 5.453 |
| Berger-Parker | 0.333 | 0.241 | 0.400 | 0.500 |
Margalef index usually takes into consideration the species richness and has no limit for comparison. According to Shah and Pandit 15, the benefit of Margalef index is used to compare species richness across the study area and it yielded a value above 1, being greater than Simpson values. In this study, Margalef index ranged from 0.000 to 4.158. The Menhinick index (0.577 – 2.785), made effort to estimate the species richness but was constrained by the sample size. Like Shannon-Wiener and Margalef indices, the lower values of Menhinick index could be due to environmental pollution in the creek and/ or lower number of species. In addition, climatic condition such as temperature and nutrients in the water could also lead to variation in the phytoplankton community in the creek. Berger-Parker index (ranging from 0.179 – 1.000) is the inverse of individuals in the community that belong to the most common single species. The Berger-Parker index ranged from 0.000 to 14.490. This index provides information about the spatial and temporal distribution pattern of a species 19. It has low sensitivity towards the sample size 20, 21 and independent of sample size. According to Shah and Pandit 15, Fisher’s alpha provides useful information about species richness. Fisher’s alpha showed positive significant correlation with Simpson (r=0.613), Berger parker (r=0.960), Margalef (r=0.868), Menhinick (r=0.871) and Shannon-Wiener index (r=0.922) at p<0.01 (Table 3).
Table 3. Pearson correlation matrix between the various diversity indices of the four predominant phytoplankton of Taylor creek, Bayelsa state, Nigeria| Parameters | Dominance | Simpson | Shannon-Wiener index | Evenness | Menhinick | Margalef | Equitability | Fisher_alpha | Berger_parker |
| Dominance | 1 | ||||||||
| Simpson | -1.000** | 1 | |||||||
| Shannon-Wiener index | -.922** | .922** | 1 | ||||||
| Evenness | .375** | -.375** | -.533** | 1 | |||||
| Menhinick | -.871** | .871** | .928** | -.341* | 1 | ||||
| Margalef | -.868** | .868** | .970** | -.491** | .970** | 1 | |||
| Equitability | -.769** | .769** | .494** | .012 | .520** | .464** | 1 | ||
| Fisher_alpha | -.612** | .613** | .717** | -.249 | .785** | .721** | .261 | 1 | |
| Berger_parker | .960** | -.960** | -.865** | .174 | -.846** | -.828** | -.773** | -.543** | 1 |
The equitability index ranged from 0.000 – 1.000. Basically, equitability index values range from 0 and 1. Higher and lower values is an indication of low and high diversity respectively. While equitability index of 1 suggest that all groups have same frequency 22. In other words, equitability index provided information about the spread of the species.
presents the renkonen’s Number for Phytoplankton Community of the study area. Renkonen’s number between Locations indicates significant difference at critical level of 50% in Locations E & A, G & A, L & A, J & E, J & H and J & I. Again these showed that varying level of anthropogenic activities is influencing the phytoplankton community of the creek. (Table 4).
Table 4. Renkonen’s Number for Phytoplankton Community of the study area| PHYTOPLANKTON | A | B | C | D | E | F | G | H | I | J | K | L |
| A | 100 | 48.5 | 33.9 | 43.2 | 50.2* | 45.1 | 52.6* | 41.8 | 38.5 | 48.6 | 44.3 | 52.3* |
| B | 100 | 29.6 | 46.6 | 43.3 | 48.2 | 45.2 | 32.4 | 44.5 | 49.7 | 38.1 | 41.3 | |
| C | 100 | 31.7 | 36.4 | 32 | 39.1 | 41 | 47.1 | 38.1 | 34.3 | 47.6 | ||
| D | 100 | 44.2 | 41.5 | 44.9 | 38.9 | 37.6 | 40.4 | 31.4 | 41.2 | |||
| E | 100 | 46.2 | 31.6 | 42.2 | 48.5 | 56.3* | 40.3 | 43.4 | ||||
| F | 100 | 43.8 | 40.4 | 36.3 | 40.5 | 44.4 | 32.5 | |||||
| G | 100 | 43.7 | 48.3 | 47.4 | 43.3 | 47.2 | ||||||
| H | 100 | 41.6 | 50.2* | 32.2 | 37.8 | |||||||
| I | 100 | 54.8* | 34.7 | 46.8 | ||||||||
| J | 100 | 44 | 36.6 | |||||||||
| K | 100 | 49.5 | ||||||||||
| L | 100 |
Conclusions
This study on the phytoplankton dynamics in Taylor creek in Bayelsa state, Nigeria found that the abundance and diversity of phytoplankton in this study differs based on the locations which could be attributed to varying human activities in the creek. The Bacillariophyta and Cyanophyta are the two major taxonomic groups found in the area. In this study, it was found that location A had the highest phytoplankton abundance amongst the (12) sampling stations, and location H had the lowest phytoplankton abundance. The phytoplankton taxa found during the study were Bacillariophyta, Chlorophyta, Cyanophyta, Chrysophyta, Spermatophyta, Phaeophyta, Pyrrophyta, Englenophyta, Heterokontophyta, Myxophyta, Dinophyta, Xanthophyta, Lycopodiaphyta and Crytophyta. The diversity indices provided useful information about the habitat quality, species richness and evenness and the association with the biotic and abiotic composition of the environment. The ecosystem was found to be moderately to heavily polluted based on Shannon-Wiener index
References
- 2.NOAA. (2014) What are Phytoplankton?. In National Ocean Service. Retrieved fromhttp://oceanservice.noaa.gov/facts/phyto.html .
- 3.Davies O A.Tawari CC Abowei JFN (2009). Zooplankton of Elechi Creek. , Niger Delta, Nigeria.Environ. Ecol.,26: 2441-2346.
- 4.Townsend C R, Herpen J L.Begon M (2000).Essentials of ecology. 3rdedition. Lack well science publishers. , London
- 5.Arora S.(2008).Fundamentals of Environmental Biology,3rdEdition,KalyaniPublishers. , New Delhi, India, Pp.655
- 6.Bill B, Lawrence C, Sian D. () K (2007).Guidance on the quantitative analysis of phytoplankton in Freshwater samples.
- 7.CEN (2004).Water quality- guidance Standard for the Routine Analysis of Phytoplankton Abundance and Composition using Inverted Microscopy.(Utermohl technique),CEN.TC.230/WG2/TG3/N83.
- 8.Wetzel R G. (2001) . Limnology: Lake and River Ecosystems (3rd ed.) , San Diego, CA:AcademicPress .
- 9.Public American.Health Association(APHA) (1998).Standard Method for examination of water and waste water. 20thedition New York:American Public Health Association.
- 10.Robert J L, Akshithala K P, Xufeng N, Sean E M. (2012) . Effect of Ammonia in Pulp Mill Effluents on Estuarine Phytoplankton Assemblages: Field Descriptive and Experimental Results,Aquatic Botany,74: 343-367.
- 11.Botes L.(2003).Phytoplankton Identification Catalogue. GloBallast Monograph Series No.7. , Saldanha Bay, South Africa
- 12.Vashishta B R, Sinha A K, Singh V P. and Company Pvt. Ltd.,Ram Nagar (2014) Botany for degree students. , New Delhi. Pp612, Algae. S. Chand
- 13.Hammer Ø, Harper D A T, P D Ryan. (2001) PAST: Paleontological statistics software package for education and data analysis.PalaeontologiaElectronica. 4(1), 9.
- 14.A E Ogbeibu. (2005) A practical approach to research and data handling.Mindex Publishing Company Limited. , Benin-City Nigeria
- 15.Shah J A, Pandit A K.Application of diversity indices to crustacean community of Wular Lake,Kashmir Himalaya.InternationalJournalof BiodiversityandConservation,5(6):. 311-316.
- 16.Stub R, Appling J W, Hatstetter A M, Hass I J. (1970) The effect of industrial waste of Memphis and Shelby country on primary planktonic producers.Bioscience. 20, 905-912.
- 19.Rosenzweig M L. (1995) Species Diversity in Space and Time. , Cambridge UniversityPress,Cambridge,UK
Cited by (27)
This article has been cited by 27 scholarly works according to:
Citing Articles:
Microorganisms (2025) Crossref
Microorganisms (2025) OpenAlex
Jiao Yang, Subha Bhassu, Ghazanfer Ali, Thenmoli Govindasamy, Muhamad Afiq Aziz et al. - Microorganisms (2025) Semantic Scholar
The handbook of environmental chemistry (2024) OpenAlex
(2024) Crossref
Asian Journal of Biological Sciences (2024) Crossref
Asian Journal of Biological Sciences (2024) OpenAlex
T. Auta, Benjamin Felix Sani, M. A. Sadauki - Asian Journal of Biological Sciences (2024) Semantic Scholar
Indonesian Journal of Agricultural Research (2023) OpenAlex
A. Iyiola, Sylvester Chibueze Izah, Sonia Morya, Ademola Akinsorotan, Matthew Ogwu - Indonesian Journal of Agricultural Research (2023) Semantic Scholar
(2023) Crossref
Sustainable development and biodiversity (2023) OpenAlex
Glory Richard, Sylvester Chibueze Izah, Olalekan Raimi Morufu, Iyingiala Austin-asomeji - Bio-Research (2023) Semantic Scholar
Elsevier eBooks (2023) OpenAlex
Elsevier eBooks (2023) OpenAlex
Journal of Fisheries and Aquatic Science (2022) Crossref
Journal of Fisheries and Aquatic Science (2022) OpenAlex
Resham Rajput, N. Wast - Journal of Fisheries and Aquatic Sciences (2022) Semantic Scholar
International journal of research studies in biosciences (2019) OpenAlex
F. O. Youkparigha, D. E. Patani - International Journal of Research Studies in Biosciences (2019) Semantic Scholar
