Abstract
Beer is typically brewed from four basic ingredients (water, a starch source, brewer’s yeast and flavoring agent such as hops). In Ethiopian case hops are coasty ingredient of beer which is imported with hard currency of the country. But this flavoring agent (hops) in beer production can also be substituted by locally available flavouring agent called gesho “Rhamuns Prinoide” leaf. In this study the effect of drying temperature and time on brewing components of gesho “Rhamuns Prinoide” leaf was examined using oven dryer. Brewing components of gesho “Rhamuns Prinoide” leaf were compared by taking the commercial hop brewing component as a standard. Drying temperature and time have a significant effect (P< 0.05) on major beer brewing components (Resin, Hop oil, Polyphenol, Mineral and Protein) of gesho leaf. Not only drying temperature and time but also the interaction effect has significant effect on these major brewing components of the leaf. The optimal drying temperature and time of gesho leaf was also determined to commercialize the gesho leaf powder or pellet as hop substitute in commercial beer brewers as flavouring
Author Contributions
Academic Editor: Chang Li, Department of Food Science, Nanchang University
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2017 Ayalew De, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Beer is not the only widely consumed alcoholic beverage in the world but also the third most popular drink after water and tea. Beer is mainly brewed from four basic ingredients which are water, a starch source, brewer’s yeast, and a flavoring agent such as hops. Different types of beer are available in the market where this typicality comes from these ingredients, the additives used and the brewing processes. Beer is composed of a complex mixture of phenolic compounds extracted from the starch source and hop. Now a day’s special trend from micro breweries in the U.S.A. is the production of several range of extremely bitter beer created by the addition of extra hop during the brewing process.
The hop derived xanthohumol and the iso-alpha acid are primarily responsible for the perceived bitterness of beer. Most of the secondary metabolites are not only important for health benefits; it is also needed for beer flavor as well as stability. But some secondary metabolites of beer will make it quality loss during storage by the creation of haze (Piendl et al, 1989).
Beer contains nutrients such as carbohydrates, amino acids, minerals, and vitamins. In addition to nutrients beer also contains many of non-nutrient components including phenolic compounds.
The type and quality of beer brewed depends the following factors, the variety of barley, the malting process, temperature and pH of mashing, sparging, wort boiling, the variety of hops added during wort boiling, and fermentation process. It has been estimated that about 70–80% of beer polyphenols are malt-derived. Barley polyphenols are changed in the malting and brewing process. The phenols found in malt are less characterized when compared to the phenol derived from hops. The phenols extracted from malt during mashing and sparging known as polyphenol or tannins and it also react with proteins in the wort to form chill haze or permanent haze. In order to control these problem reduce sparge temperature below 77oc; reduce sparge time and the alkalinity of brewing liquor (De Keukeleire, 2000).
The hop is a perennial climbing plant; the aerial part dies off in the autumn but the rootstock stays in the soil, sometimes for many years. The plant needs a support up which to grow. In the wild, hops are found in hedgerows but for cultivation they are trained up strings attached to permanent wirework. Hops are used in beer production in small amount to impart its characteristic aroma and bitterness. Hops also contribute to the beer its antifungal and antibiotic properties so that the produced beer remains safe during storage (Verzele et al, 1991). The composition of the hops is extremely important for the quality of the beer produced from it and on dry weight basis the bitter substances (Resin) (18.5%), hop oil (0.5%), polyphenols (3.5%), protein (20.0%) and minerals (8.0%).The rest consists of cellulose and other materials which are unimportant for beer production. The most important components of hop for the production of beer are the bitter substances (resins) and essential oils or the hop oils (Kunze, 1996). During drying of hop the air speed and temperature should be carefully controlled so that volatile compounds which are heat sensitive will not be destroyed with increasing air temperature for prolonged time. But if the air speed is too low, the air will become saturated with moisture in the lower layers of the bed where lastly deposit moisture on the hops in the upper layer which finally causes discoloration of the hops (Verzele et al, 1991).
Gesho is a plant which grows up to six meters and it is cultivated in Ethiopia for its importance in the production of domestically fermented beverages like tella and teji. In Ethiopia the leaves and steams of gesho are crucial components in the production of traditional fermented beverages (Ashenafi M., 2006). Although gesheo leaf had been used as flavouring as well as antibiotic agent in traditional beverages, it had yet tried to commercialize it as hop substitute in commercial beer brewing industries. In doing so drying is the critical process in gesho leaf powder or pellet production. Because major brewing components of gesho leaf powder are volatile compounds which will be destroyed during drying by high temperature and moisture. Traditionally gesho leaf is dried simply using sunlight by spreading the leaf on the ground that had been already prepared for drying purpose where it negatively affect on its taste and flavor. In this study the optimal drying temperature and time had been investigated and hence it can potentially substitute the commercial hop imported as a flavouring agent in commercial beer brewing industries. So gesho leaf powder or pellet dried at the optimal drying condition had similar brewing components as that of the commercial hop imported by Dashen brewery Share Company.
The general objective of this study is: Determination of optimum drying conditions of gesho leaf powder as commercial hop substitute in modern beer brewing industries
Specific objectives:
To determine the optimum drying temperature of gesho leaf as hop substitute in commercial beer brewing industries
To determine the optimum drying time of gesho leaf as hop substitute in commercial beer brewing industries
To characterize the composition of dried gesho leaf for its major brewing components as hop substitute in commercial beer brewing industries
Material and Method
Sample collection and preparation
Gesho leaf which was physiologically matured collected from Amhara Agriculture ResearchInstitute (ARARI) at Adet Agriculture Research Center. The collected gesho leaf sample was cleaned manually to remove seed and other extraneous materials such as stalks and dusts before drying process conducted.
Gesho leaf powder preparation
Drying of gesho leaf was conducted by drying oven to investigate the effect of drying factors (drying temperature and time) on the quality of gesho leaf powder as commercial hop substitute in commercial beer brewing industries. During conducting the experiment, the sample was dried at different temperature and time with five levels (40oc, 45oc, 50oc, 55oc and 60oc) and (1hr, 1.5hr, 2hr, 2.5hr and 3hr) to investigate the optimum drying parameters of drying process. Then dried gesho leaf was milled using laboratory grain miller at Amhara Agricultural Research Institute grain laboratory and packed with polyethylene using bag seller. Finally products were characterized for its important brewing components (polyphenol, bittering substance, hop oil, mineral and protein content).
Characterization of the gesho leaf powder
Moisture content
The crucibles were washed and dried using drying oven for 1hr at 100oC, placed in the desiccators (with granular silica gel), cooled for 30 minute and weighed. 5.00g of the prepared samples were weighed and transferred into the dried and weighed crucibles. The crucible containing samples were dried in the oven at 110oc with one hour interval until constant weight of the crucible containing sample were weighed and recorded. At each interval of the drying step the sample were cooled in the desiccators to room temperature before weighing the sample. The amount of water present in a sample is considered to be equal to the loss of weight after drying the sample to constant weight at a temperature about the boiling point of water.
equation (1)
Where:
Mintial = Weight of sample before drying in the oven
Mfinal = Weight of the sample after drying in the oven
Total crude protein content
Protein was determined by the Kjeldahl method. All nitrogen is converted to ammonia by digestion with a mixture of concentrated sulphuric acid and concentrated orthophosphoric acid containing potassium sulphate as a boiling point raising agent and selenium as a catalyst. The ammonia released after alkalinization with sodium hydroxide is steam distilled into boric acid and titrated with sulphuric acid.
Digestion: 0.5gram of samples were taken in a tecator tube and 6ml of acid mixture (5parts of concentrated ortho-phosphric acid and 100 parts of concentrated sulfuric acid) was added, mixed- thoroughly and a 3.5ml of 30% hydrogen peroxide was added step by step. As soon as the violent reaction had ceased, the tubes were shaken for a few minutes and placed back into the rack. A 3.00g of the catalyst mixture (ground 0.5g of selenium metal with 100g of potassium sulfate) was added into each tube, and allowed to stand for about 10min before digestion. When the temperature of the digester reached 370oC, the tubes were lowered into the digester. The digestion was continued until a clear solution was obtained, about 1h.The tubes in the rack was transferred into the fume hood for cooling, a 15ml of demonized water was added, and shaken to avoid precipitation of sulfate in the solution.
Distillation: A 250ml conical flask containing 25ml of the boric acid-indictor solution was placed under the condenser of the distiller with its tips immersed into the solution. The digested and diluted solution was transferred into the sample compartment of the distiller. The tubes were rinsed with two portions of about 5ml de-ionized water and the rinses were added into the solution. A 25ml of 40% sodium hydroxide solution was added into the compartment and washed down with a small amount of water, stoppered and the steam switched on. A 100ml solution of the sample was distilled, and then the receiver was lowered so that the tip of the condenser is above the surface of the distiller. The distillation was continued until a total volume of 150ml is collected. The tip was rinsed with a few milliliter of water before the receiver was removed.
Titration: The distilled solution was titrated with 0.1N sulfuric acid to a reddish colour.
mg nitrogen in the sample = V*N*14---------------------equation (2)
g nitrogen sample/100g sample = mg of nitrogen*100/mg of the sample
equation (3)
Crude protein (%) = Total nitrogen (%) * F---------------equation (4)
Where:
V=Volume of sulfuric acid consumed to neutralize the test material (ml)
Vb=Volume of the acid consumed to neutralize the blank
F=Conversion factor of total nitrogen to crude protein (6.25)
14=Equivalent weight nitrogen
N= Normality of standard sulfuric acid
Total ash content
Ash was determined by incineration of known weights of the samples in a muffle furnace at 550oc until a white ash was obtained. Organic matter was burned off and the inorganic material remaining is cooled and weighed. Heating was carried out in stages, first to derive the water, then to char the product thoroughly and finally to ash at 550oc in a muffle furnace. The ashing dishes (made of porcelain) were placed into a muffle furnace for 30 min at 550oC. The dishes were removed and cooled in desiccators (with granular silica gel) for about 30 min at room temperature; each dish was weighed. 2.5g of powder sample was added into each dish. The dishes were placed on a hot plate under a fume-hood and the temperature was slowly increased until smoking ceases and the samples become thoroughly charred. The dishes were placed inside the muffle furnace at 550oC for 6 hr, and removed from the muffle and then placed in desiccators for 1hr to cool. The ash was clean and which in appearance. When cooled to room temperature, each dish plus ash was re-weighed. Weight of total ash was calculated by difference as:
equation (5)
Where:
Mash = Refers to weight of ash
Mwet= Refers to original weight of the sample
Polyphenol content
Polyphenol were determined by the modified vanillin assay method (Butter et al., 1982). 200 mg samples were weighed and then extracted with 10 ml absolute methanol for 20 minutes rotating screw cap culture tubes (130*100mm). The mixture then is centrifuged for 10minutes at 3000*G and the supernatant were used in the analysis. About 0.0 – 1.0 ml aliquot of catechin standard is dispended into two sets of culture tubes and each sample was brought to 1.0 ml by the addition of absolute methanol. Incubate the tubes in water bath. 5 ml of the working vanillin reagent was added at 1 min interval to one set of standards, and 5ml of the 4% HCl solution is added at 1 min interval to the second set of standards. After 20 min, the absorbance of sample solution and the standard solution were measured at 500 nm by using spectrophotometer. The absorbance of the blank is subtracted from the absorbance of the corresponding vanillin-contain sample. A standard curve has been constructed (Absorbance vs Catechin) and the linear portion of the curve will be extrapolated to produce the standard curve.
Finally, the polyphenol contents will be calculated. Values of tannins will be expressed in milligram of D-catechin equivalent per gram of sample.
Polyphenol in mg/100g = (absorbance density*weight of sample *100) -----equation (6)
equation (6.1)
Where:
Pw-Weight of polyphenol in gm
Ws-Weight of sample in gm (100gm)
Bitterness substance (Resin) content
Total resin content
For resin analysis 20g of the sample was dissolved in 100 ml of cold methanol in a conical bottom flask and the mixture was vigorously agitated by swirling the flask. Thereafter, the solution was filtered. The filtrate containing the resin was then dried and the total resin was calculated as a percentage of the original sample weight.
equation (7)
Where: Mdf and Mintial are weight of extract after dry and initial weight of the sample respectively
Soft and hard resin content
With regard to resin determination, 20 grams of each sample was dissolved in 20 ml of n-hexane thoroughly stirred and filtered using filter paper. Filtrate was dried to a constant weight at 50oc. The soft resin was calculated as the percentage of the original weight of sample dissolved in the n-hexane. Hard resin was determined by subtracting soft resin from total resin.
equation (8)
Where:
Mfinal = weight of the final dried sample
Mintial=Weight of the sample dissolved in the n-hexane
Hop oil content
In hop oil content analysis, 10 gram of the sample was taken in 1000 ml round bottom flask and then 750 ml of distilled water was added. The mixture was agitated by swirling the flask. A Clevenger apparatus was mounted and fitted on to the round bottom flask and then connected to a tap water source. The set up was held tight with a retort stand and the mixture was placed on a suitable electric heating mantle. When the water boils, the steam rises through the stockings, thus extracting the essential oil. The delivery from the condenser was connected to the separating funnel to receive the mixture of steam and oil on condensation. After 120 min, the set up was switched-off and allowed to cool. The water-oil mixture was decanted to separate the oil from the water at the water oil interface. After drying in an electric oven the mass was recorded and calculated as a percentage from the original sample.
equation (9)
Where:
Mdf = weight of the sample after drying phenomenon
Mintial = Initial weight of the sample
Data analysis
Chemical and physical measurements were stastically analyzed by design expert-7 software. The comparison between sample treatments and the indices were done by using analysis of variance with a probability (P < 0.05). In this experimental work all physical and chemical measurements were performed in triplicates
Result and discussion
As it is observed from the optimization table-one below, drying of gesho leaf at a temperature of 55oc for 2 hours has high desirability value (0.948) than the rest of the products processed at a given levels of factors (drying temperature and time). High desirability value which approaches to one indicates the best operating drying condition of the product to be processed in relation to the target components of the product in this case the brewing components of gesho leaf. On the contrary gesho leaf powder with low desirability value indicates inferior quality. So in gesho leaf drying, the best optimal drying temperature and time were 55oc for two hours. At optimal drying condition the brewing component of gesho leaf had equivalent values on dry wet basis with that of the commercial hop as it is stated in (Kunze, 1996).
Effect of drying temperature and time on resin content
From Figure 1 below (drying temperature and time interaction effect graph) the concentration of resin is high in a drying temperature range from 40oc to 50oc within the time interval of 1 to 2hours. Although the concentration of resin was high at those drying operating conditions, it couldn’t be recommend as point of optimal processing condition because the moisture content of the product was also high which leads to the deterioration of resin by the action of oxygen as it had been stated in (Kunze,1996). Then the product would have inferior quality which finally makes it to be less demanded in the market for commercial beer brewing industries as hop substitute flavouring agent. On the other side drying temperature greater than 55oc and time more than 2hrs indicate low amount of resin as a result of high temperature break down of α-acids which also makes inferior quality. At optimal drying temperature and time (55oc for 2hrs) the product had 18.015 percent which meets the standard of commercial hop set by American trade association.
Figure 1.Effect of temperature and time interaction on resin content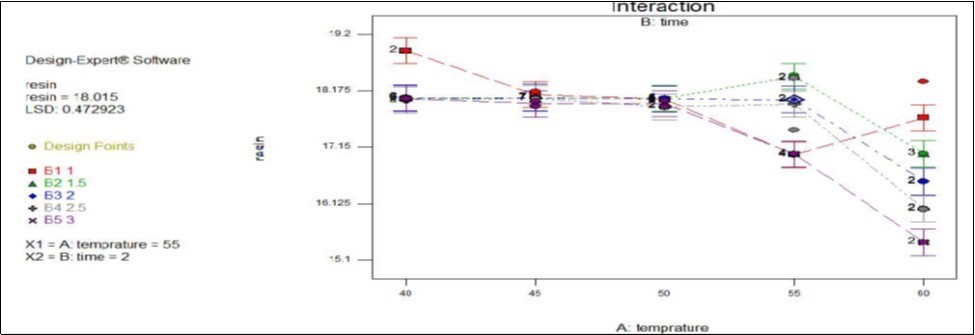
| No_ | Tern-'c | Tim-hr | moisture | protein | Polyph. | mineral | resin | hopoil | desirability |
| 1 | 55 | 2 | 10.125 | 18.055 | 3.53 | 8.055 | 18.015 | 0.965 | 0.948 |
| 2 | 50 | 2 | 14.34 | 18.015 | 3.565 | 7.985 | 18.02 | 1 | 0.94 |
| 3 | 50 | 2.5 | 12.06 | 18 | 3.455 | 7.98 | 17.88 | 0.965 | 0.932 |
| 4 | 50 | 3 | 10.34 | 17.98 | 3.42 | 7.975 | 17.94 | 0.8 | 0.923 |
| 5 | 40 | 1 | 17.435 | 18.085 | 3.82 | 8.02 | 18.03 | 0.985 | 0.909 |
| 6 | 60 | 3 | 18.64 | 18.06 | 3.885 | 8.06 | 17.685 | 0.525 | 0.862 |
| 7 | 45 | 2.5 | 16.34 | 17.885 | 3.665 | 8.115 | 18.035 | 1.305 | 0.852 |
| 8 | 60 | 1.5 | 12.055 | 18.05 | 3.86 | 8.06 | 17.03 | 0.485 | 0.843 |
| 9 | 50 | 1.5 | 20.34 | 18.025 | 3.97 | 7.96 | 18.03 | 0.87 | 0.836 |
| 10 | 55 | 1.5 | 13.36 | 17.885 | 3.565 | 8.025 | 18.43 | 1.005 | 0.827 |
| 11 | 40 | 2.5 | 19.57 | 18.08 | 4.9775 | 8.065 | 18.005 | 1.21 | 0.826 |
| 12 | 55 | 2.5 | 9.46 | 18.32 | 3.23 | 8.06 | 17.93 | 0.89 | 0.776 |
| 13 | 40 | 2 | 23.455 | 18.53 | 3.98 | 8.01 | 18.045 | 1.2 | 0.755 |
| 14 | 60 | 2 | 9.34 | 18.03 | 3.23 | 7.975 | 16.525 | 0.425 | 0.696 |
| 15 | 50 | 1 | 38.45 | 18.085 | 4.11 | 8.12 | 18.02 | 0.93 | 0.644 |
| 16 | 55 | 3 | 8.98 | 17.865 | 2.975 | 8.06 | 17.025 | 0.465 | 0.612 |
| 17 | 45 | 2 | 18.4 | 17.89 | 3.68 | 8.32 | 18.03 | 1.195 | 0.426 |
| 18 | 45 | 1.5 | 32.485 | 17.87 | 5.02 | 8.31 | 18.05 | 1.12 | 0.366 |
| 19 | 45 | 1 | 40.965 | 17.885 | 4.01 | 8.31 | 18.105 | 1.12 | 0.36 |
| 20 | 60 | 2.5 | 7.445 | 18.415 | 2.88 | 7.895 | 16.025 | 0.4 | 0.314 |
| 21 | 40 | 1.5 | 38.075 | 18.56 | 4.015 | 8.325 | 18.045 | 1.225 | 0.265 |
Effect of drying temperature and time on hop oil content
In Figure 2 below the hop oil content in gesho leaf powder decreases at drying temperature greater than 55oc and time greater than 2hrs. At these temperatures range the decrement of hop oil was due the action of high temperature destruction. But the hop oil content was high when the gesho leaf was dried at temperature range from 40oc to 55oc for a given time interval where the moisture content is above the recommended. At the optimal drying temperature and time (55oc for 2hrs), the product would have hop oil content of 0.98 percent on dry weight basis where the product meets the desired amount to substitute commercial hop in commercial beer brewing industries in relation to essential oil content.
Figure 2.Effect of temperature and time interaction on hop oil content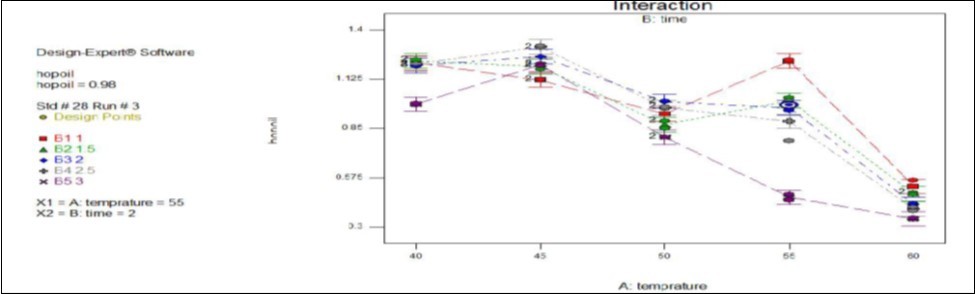
Effect of drying temperature and time on protein content
Considering Figure 3 below for a drying temperature greater than 55oc the protein content of gesho leaf powder decreases, the phenomenon was expected to be may due to denature small molecular weight proteins. For the drying process of gesho leaf in a temperature range 40 to 50oc the protein content of the product shows high variation, this should be more probably due to the impact of other ingredients change with drying time and temperature such as resin, hop oil and polyphenol content which finally bring change on proximate composition of the product. The protein content of the gesho leaf became nearly constant at optimal drying condition (drying temperature of 55oc and time duration of 2 hour) of the product and also at the optimal operating condition of the protein content was found 18.055 percent which was in agreement with range set by American trade association for commercial hop.
Figure 3.Effect of temperature and time interaction on protein content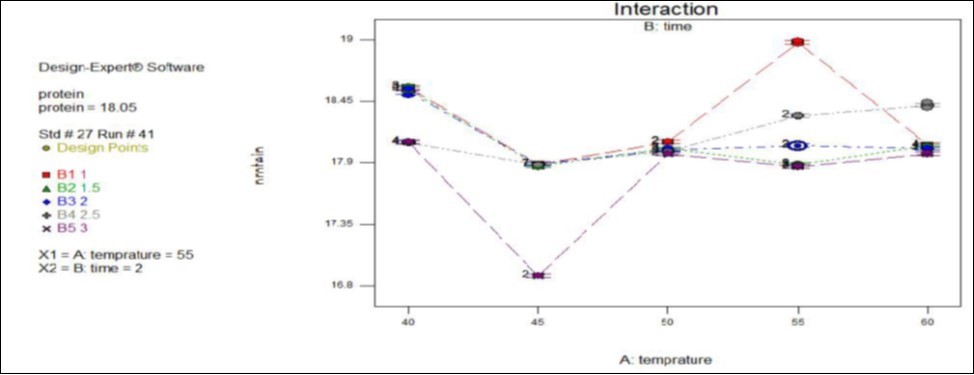
Effect of drying temperature and time on polyphenol content
As it is shown in the Figure 4 below the polyphenol content of the gesho leaf powder was found higher at a drying temperature range 40 to 50oc for nearly all drying time ranges (from 1 to 3hr) but for a temperature range greater than 55oc the polyphenol content decreases. The cause of polyphenol content to decrease in a drying temperature greater than 55oc was due the action of high heat on these semi-volatile components of gesho leaf. At the optimal drying condition (55oc for 2hrs) gesho leaf had polyphenol content 3.53% which is equal as that of commercial hop stated in (Kunze,1996).
Figure 4.Effect of temperature and time interaction on polyphenol content Temperature in oc,Time in hr and polphenol (%) in dry weight basis
Effect of drying temperature and time on mineral content
The amount of minerals in beer production shall be within the desired level if not it will totally disturb the overall brewing process through various interactions in the course of the process (kunze, 1996). In the Figure 5 below the mineral content of gesho leaf became higher when it was dried at a temperature range of 40 to 50oc for all drying time while the mineral content reduces for drying temperature greater than 55oc nearly for all temperature ranges. The reason for the fluctuation of mineral content was more probably due to the variation of other components which in turn disturbs the proximate composition of the product. At the optimal drying temperature and time (55oc for 2hrs) the mineral content of the product was 8.055 percent which was in agreement as stated in (Kunze, 1996).
Figure 5.Effect of temperature and time interaction on mineral content Temperature in oc,Time in hr and mineral (%) in dry weight basis
Effect of drying temperature and time on moisture content
High moisture content negatively affects the quality of gesho leaf powder where the α-acids break dawn by the action oxygen as a result the bitterness substance will be lost which indicates inferior quality. It was clear that in Figure 6 below shows the moisture content of gesho leaf powder decreases as drying temperature and time increases. During drying the moisture content reduced at the beginning dramatically and then it slowly reduces as drying time reaches around three hours. At optimal the drying condition (temperature 55oc and time 2hrs) the moisture content was 10.125 percent. At this moisture content the brewing components of gesho leaf would stay for long period of time without the loss of critical brewing components.
Figure 6.Effect of temperature and time interaction on moisture content
Conclusion
The optimal drying condition (drying temperature and time) for the production of gesho leaf powder or pellet as hop substitute in commercial beer brewing industry was found to be 55oc for two hours. At the optima drying condition the bitterness substances, the hop oil and polyphenol content of gesho leaf which are highly heat sensitive were found 18.025, 0.96% and 3.53% respectively while protein and mineral content which are less heat sensitive were 18.055% and 8.055% respectively. As a conclusion at the optimal drying condition gesho leaf can certainly substitute the imported commercial hop for commercial beer brewing industries in terms of beer brewing components. The finding also helps in saving foreign currency by import substitution of the commercial hop. Commercializing this flavouring agent as hop substitute improved the livelihood of farmer’s especially rural women where the cultivation was mainly held by women in Ethiopia. The finding also would reduce the coast of commercial beer brewing process so that it will increase the profitability of beer brewers.
Recommendation
Evaluation of optimally dried gesho leaf powder need further in commercial beer production by substituting the commercial need further study. The impact of soil type and growing environment on composition of brewing component of gesho leaf shall also need further investigation.
References
- 1.K, A, S I Dika. (2011) Bitter leaf as local substitute for hops in the Nigerian brewing industry. , Archive of Applied Sciences Research 3, 388-397.
- 2.P E üAjebesone, J O Aina. (2004) Potential African substitute for hops in tropical beer brewing. , Journal of food technology in Africa 9(1), 13-16.
- 3.üAshenafi M. (2006) A review on the microbiology of indigenous fermented foods and beverages of Ethiopia. Ethiopian journal biological sciences. 5(2), 189-245.
- 4.üBerhanu A 2013.Microbial profile of Tella and the role of gesho (Rhamnus prinoides) as bittering and antimicrobial agent in traditional Tella (Beer) production. , Journal internation food research 21(1), 23-35.
