Liver Stiffness by ARFI does not Correlate with Decompensation and Portal Hypertension in Patients with Cirrhosis
Abstract
Background and Aims:
Establishing the degree of fibrosis is important in determining the prognosis of patients with chronic liver disease. Acoustic Radiation Force Impulse Imaging (ARFI) has been validated as a reliable method to estimate liver fibrosis. It remains unclear if ARFI readings may be a useful way to stage patients with established cirrhosis and predict the development of complications. The aim of this study was to determine if ARFI liver stiffness measurements correlate with the severity of liver disease in patients with cirrhosis, and predict the development of complications and decompensation.
Methods:
All patients attending our institution who had a prior clinical diagnosis of cirrhosis and an ARFI liver stiffness measurement (LSM) over 26 months were included. Area under the receiver operating characteristic (AUROC) curves were calculated for ARFI detection of any complication, any varices, medium or large varices, moderate or severe ascites, encephalopathy, Child Pugh Grade B or C and MELD ≥15.
Results:
ARFI LSM did not correlate with complications: any complication (AUROC 0.672), any varices (0.631), medium or large varices (0.610), moderate or severe ascites (0.681), Child Pugh B/C (0.691) or MELD ≥15 (0.711). Hepatic encephalopathy did correlate with higher LSM (0.854), but only in a small number of cases.
Conclusion:
ARFI in patients with cirrhosis does not correlate with the presence of portal hypertension or decompensated liver disease.
Author Contributions
Academic Editor: Divey Manocha, Upstate Medical University
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2017 Hoie Kidd Leong, et al
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
The natural history of chronic liver disease of any aetiology is progressive fibrosis that evolves into cirrhosis. 1 Once cirrhosis develops there is a risk of complications including oesophageal varices, ascites, hepatic encephalopathy and hepatocellular carcinoma (HCC). Establishing the degree of fibrosis is important in determining the prognosis of patients with chronic liver disease, and has historically been evaluated by liver biopsy, which reliably predicts the natural history of liver disease and remains the accepted gold standard for the assessment of fibrosis in the liver2. However, liver biopsy is invasive and painful, and carries a risk of morbidity. The accuracy of liver biopsy can also suffer from intra-observer variability, inter-observer variability and sampling error3.
Ultrasound elastography is a non-invasive method of estimating liver fibrosis by measuring liver stiffness. Two commonly used methods are Fibroscan® and Acoustic Radiation Force Impulse (ARFI;Siemens, Berlin, Germany). ARFI measures liver stiffness by delivering narrow high energy ultrasound pulses that result in slight tissue displacements, which in turn generate low amplitude shear waves that travel in a lateral direction away from the line of the ultrasound beam. Tracking ultrasound pulses can then measure the speed of these shear waves, the speed of which correlates with liver stiffness.
ARFI has been validated as a reliable method to estimate liver fibrosis4, 5, 6 and has similar accuracy to Fibroscan®7. The advantages of ARFI over Fibroscan® are that ARFI is integrated into a routine ultrasound machine, which allows the screening of liver lesions, more precise selection of sampling site and the measurement can be adapted according to the distance between the skin and liver capsule. Factors that adversely affect the accuracy of ARFI include obesity8, skin liver distance9 and ratio between the interquartile range (IQR) of measurements and median velocity of measurements (IQR:median velocity) of greater than 0.3010. While ARFI has been accepted to have good diagnostic accuracy in determining fibrosis, there is conflicting evidence in the literature whether ARFI has the same accuracy in predicting complications of cirrhosis. Most studies that have investigated the correlation between ARFI and complications of cirrhosis have focused on oesophageal varices11, 12. Some studies have shown high predictive ability of ARFI for the development of varices13, while others show no correlation14. Similar inconsistency is seen with regards to Fibroscan® and varices14, 15.
Given the lack of clarity in the literature with regards to ARFI and patients with cirrhosis, the aim of this study was to determine if ARFI liver stiffness measurements (LSM) correlate with the severity of liver disease in patients with an established clinical diagnosis of cirrhosis, and correlate with the development of complications and decompensation in a large cohort of cirrhotic patients managed at a tertiary centre.
Methods
All patients that visited a tertiary institution who had a prior clinical diagnosis of cirrhosis, based on histology, ultrasound, biochemistry and/or clinical signs, and an ARFI LSM between August 2012 and November 2014 were included in this study. For each patient, medical records were searched to find demographic data, chronic liver disease aetiology, biochemical and haematological results, the presence of cirrhosis and presence of complications. Patients with an IQR:median velocity ratio of >0.30 were excluded from the study (n=82). The patient cohort is shown diagrammatically in Figure 1.
Figure 1.Diagrammatical representation of the patient cohort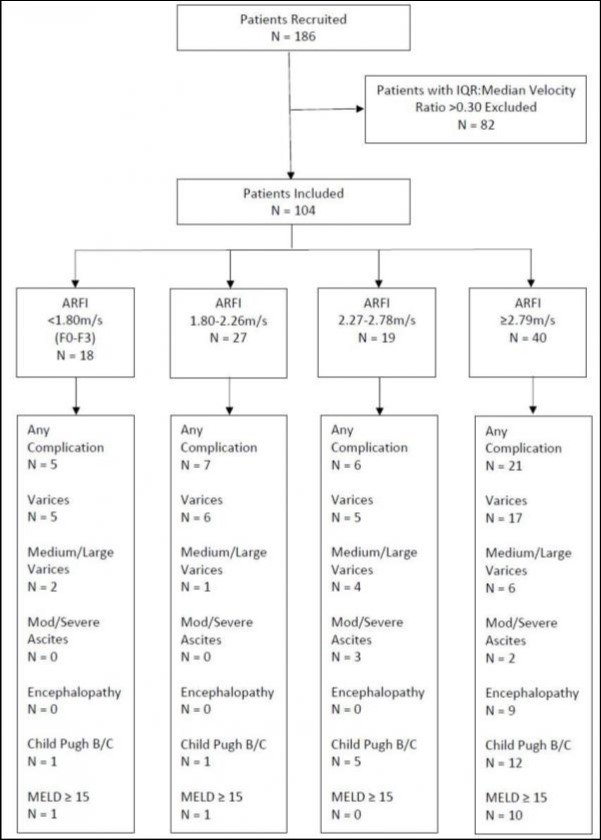
Demographic data included gender, aetiology of liver disease, body mass index (BMI) and age at time of ARFI. The BMI calculation done closest to the time of ARFI was recorded; however, from March 2014, all patients had their height and weight recorded at the time of ARFI for BMI calculation. Blood test results included full blood examinations, liver function tests and prothrombin time international normalised ratio (INR) closest to the time of the ARFI measurements. A patient was defined as having cirrhosis if they had either biopsy proven cirrhosis or cirrhosis diagnosed based on the results of clinical, laboratory and imaging studies found in the medical records16. The diagnosis of varices was determined by upper gastrointestinal endoscopy within one year of ARFI measurement. The diagnosis of ascites was based on clinical examination or ultrasound. The diagnosis of hepatic encephalopathy was based on clinical assessment by a consultant hepatologist. If it was unclear from the medical records whether a patient experienced a particular complication, this was treated as missing data. Child Pugh grade and MELD scores were calculated for all patients who had data available for calculation.
Interpretation of ARFI values as estimates of liver fibrosis were taken from previous validation studies such that <1.35m/s = F0-F1, 1.35-1.54m/s = F2, 1.55-1.80m/s = F3 and >1.80m/s = F44, where F0 is normal liver, F1 is mild fibrosis, F2 is moderate fibrosis, F3 is severe fibrosis (pre-cirrhotic), and F4 is cirrhosis as determined on histology by Scheuer or Metavir scores.
ARFI Procedure
LSM were obtained with ARFI using a Siemens Acuson S2000 ultrasound system by at least two different ultrasonographers who were blinded to the results of each other. Patients were fasted for at least six hours and measurements were taken with the patient in the supine position. A region of interest was set in the right hepatic lobe through an intercostal approach two to four centimetres below the liver capsule. The region of interest was placed in an area of parenchyma that was free of blood vessels. Measurements were taken by each sonographer until ten successful LSM were obtained and were expressed in meters per second (m/s). The overall median of all the LSM by both sonographers were used as the ARFI measurement for this study. The mean, standard deviations, IQR and the number of failed measurements were also recorded. The IQR is calculated using all the successful LSM measurements obtained by each sonographer. This value divided by the average of the median LSM from both sonographers gives the IQR:median velocity ratio.
Statistical
Statistical analyses were performed using Stata 13. The accuracy of ARFI LSM in estimating the liver disease complications was assessed using area under the receiver operating characteristic (AUROC) curves that graph sensitivity plotted against 1-specificity for all possible cut-off values. In addition to the AUROC curves, 95% confidence intervals and p values were calculated.
Ethics and Consent
This study was approved by The Melbourne Health, Office for Research, Human Ethics committee as a quality assurance project. Verbal consent was obtained by the doctor booking the ultrasound. According to our ethics committee, no written consent was required as this was deemed a low risk project.
Results
There were 186 patients with clinically diagnosed cirrhosis who had an ARFI during the study period. After excluding patients with an IQR:median velocity of >0.30, 104 patients were included in the study, with the baseline characteristics presented in Table 1.
Table 1. Baseline Characteristics| Characteristic | Value |
|---|---|
| Total Number of Patients | 104 |
| Male: Female | 55(52%):49(47%) |
| Age – Median ± IQR (Mean, Standard Deviation) | 56 ± 17 (55.95, 13.52) |
| BMI – Median ± IQR (Mean, Standard Deviation) | 26.57 ± 10.53 (27.40, 7.65) |
| ARFI Median Velocity – Median ± IQR (Mean, Standard Deviation) | 2.44 ± 1.02 (2.50, 0.73) |
| ARFI IQR:Median Velocity Ratio – Median ± IQR (Mean, Standard Deviation) | 0.19 ± 0.10 (0.20, 0.06) |
| Aetiology of Liver Disease | |
| Hepatitis C | 39 (38%) |
| ·NAFLD | 24 (23%) |
| Alcohol | 17 (16%) |
| Hepatits B | 12 (12%) |
| Other Aetiology | 17 (16%) |
| Varices: No Varices: Missing Data | 65 (63%): 33 (32%): 6 (6%) |
| Medium/Large Varices: Small/No Varices: Missing Data | 13 (13%): 85 (82%): 6 (6%) |
| Moderate/Severe Ascites: Mild/None Ascites: Missing Data | 5 (5%): 93 (89%): 6 (6%) |
| Encephalopathy: No Encephalopathy: Missing Data | 9 (9%): 89 (86%): 6(6%) |
| Child Pugh A: B: C: Missing Data | 65 (63%): 19 (18%): 2 (2%): 18 (17%) |
| MELD <15: ≥15: Missing Data | 76 (73%): 12 (12%): 16 (15%) |
| F0-F1 on ARFI (<1.35) | 4 (4%) |
| F2 on ARFI (1.35-1.54) | 9 (9%) |
| F3 on ARFI (1.55-1.80) | 6 (6%) |
| F4 on ARFI (>1.80) | 85 (82%) |
There was a slight male predominance (52%), with a median and IQR for age of 56 years ± 17. Hepatitis C was the most common aetiology (38%), followed by non-alcoholic fatty liver disease (NAFLD) (23%). The majority of patients (85/104, 82%) had an ARFI LSM consistent with cirrhosis (F4), the remaining 19 patients with clinical cirrhosis had an ARFI between 0.97 and 1.72 m/s. Most patients had well compensated cirrhosis with 63% having Child Pugh A and 73% having MELD scores <15. Sixty three percent of patients (n=65) had varices, with 13 (13%) shown to have medium or large varices, 5 patients (5%) had moderate or severe ascites and 9 (9%) had overt hepatic encephalopathy. Data was unavailable for 6 patients with regards to varices, ascites and hepatic encephalopathy.
The association of a higher ARFI LSM with the presence of varices; medium or large varices; moderate to severe ascites; hepatic encephalopathy; a higher Child Pugh grade or MELD score ≥ 15 was determined using the AUROC curve Table 2 The higher ARFI LSM correlated with the presence of encephalopathy in cirrhotic patients with an AUROC of 0.854 (Figure 2); however, this was only in a small number of patients. The presence of other complications only had a very weak correlation with ARFI LSM, with optimal cut-offs for each complication presented in Table 3. The negative predictive value for moderate to severe ascites and for hepatic encephalopathy was 100% with cut-offs of 2.27m/s and 2.79m/s respectively. Similarly, the negative predictive value for Child Pugh B or C, or MELD >15 were 92% and 96% for 2.27m/s and 2.79m/s respectively. However, the positive predictive value was poor for all complications of cirrhosis.
Figure 2.AUROC for ARFI prediction of hepatic encephalopathy in cirrhotic patients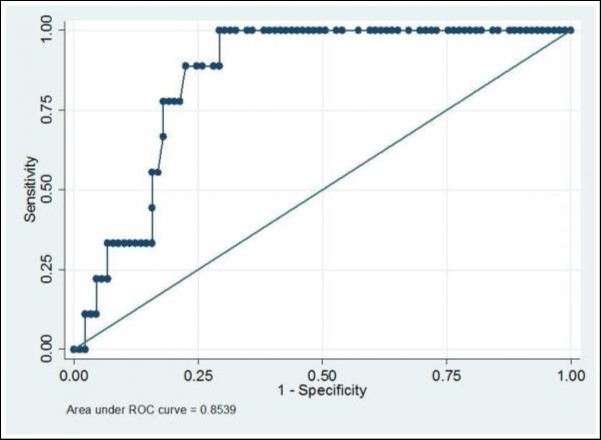
| Event | AUROC (95% Confidence Interval) |
| Any Complication | 0.672 (0.561 – 0.784) |
| Any Varices | 0.631 (0.511 – 0.751) |
| Medium or Large Varices | 0.61 (0.443 – 0.777) |
| Moderate or Severe Ascites | 0.681 (0.492 – 0.869) |
| Encephalopathy | 0.854 (0.772 – 0.936) |
| Child Pugh B/C | 0.691 (0.572 – 0.810) |
| MELD≥15 | 0.711 (0.557 – 0.864) |
| Event | Optimal ARFI LSM Cut-off | Sensitivity (%) | Specificity (%) | Positive Predictive Value (%) | Negative Predictive Value (%) |
| Exclusion of IQR:Median Velocity Ratio >0.30 | |||||
| Any Complication | 2.67 | 59 | 75 | 61 | 73 |
| Any Varices | 2.67 | 58 | 71 | 50 | 77 |
| Medium or Large Varices | 2.34 | 77 | 52 | 20 | 94 |
| Moderate or Severe Ascites | 2.27 | 100 | 46 | 9 | 100 |
| Encephalopathy | 2.79 | 100 | 71 | 26 | 100 |
| CPS B/C | 2.27 | 86 | 52 | 37 | 92 |
| MELD ≥15 | 2.79 | 83 | 66 | 28 | 96 |
Discussion
In this study, we show that the ARFI results do not reliably translate to risk of cirrhotic complications. One possibility to explain this finding is that the ARFI measurement itself was inaccurate. Every effort was made during our study to ensure the accuracy and the reproducibility of the ARFI LSM. Firstly, there is no consensus in the literature on the ideal region of interest when performing ARFI LSM. It has been suggested that ARFI LSM on the right lobe are more accurate than the left lobe17 and the ideal depth placement from the liver capsule is 3-4cm with a linear probe and 5-6cm with a curved probe9. The region of interest in our study was selected based on these guidelines. To further improve accuracy, we have excluded patients with an IQR:median velocity ratio of >0.3010. Obesity is another factor that can impact on the accuracy of ARFI8. Our study did have a reasonably high average BMI for cirrhotic patients and this may have impacted on the results. However, we decided not to have a BMI exclusion criteria as we felt this may impact on the generalisability of the findings.
Another possible explanation may be that liver stiffness does not continue to increase as patient’s progress from compensated to decompensated cirrhosis, and therefore assessing complication risks by ultrasound elastography may be suboptimal. Progression of disease in a cirrhotic liver is complicated, and changes in the vasculature of the sinusoids and the presence of microthrombi reduce the flow through the liver to contribute to portal hypertension and its associated clinical sequelae 18. The development of varices also reduces liver blood flow through the sinusoids, as some portal blood flow is diverted around the liver. This reduced liver blood flow may result in some reduction in liver stiffness, despite an increase in fibrosis. The cirrhotic liver also progressively loses steatosis and inflammation, both of which are known to increase liver stiffness by ultrasound elastography19, 20.
Liver histology has long been the gold standard of assessing both the aetiology and the stage of liver disease. The Laennec classification is a histological sub-classification based on the severity of fibrosis in the cirrhotic liver, and has found that stratifying patients in this way may predict the likelihood of complications21, 22 . Based on this information it would be expected that liver stiffness as assessed by ARFI, would continue to rise with progression of disease; however, the results of our study show that progression of liver disease may not simply be a reflection of increasing fibrosis.
Previous studies that have attempted to correlate LSM by ARFI with the development of complications of cirrhosis have been conflicting, which attests to the complexity of changes that occur with liver disease progression. Many studies have tried to use LSM to determine who to screen for oesophageal varices; and high accuracy has been reported by some authors (AUROC 0.959) 23, where others have shown a very poor predictive value (AUROC 0.58) 14. Ascites was not well correlated with liver stiffness in our study or by other investigators (AUROC 0.62) 24. The assessment of portal hypertension might be better served by ARFI of the spleen, which has shown reasonable correlation with the presence of ascites and varices (AUROC 0.80) 24.
Our results show a statistically significant correlation between ARFI LSM with the presence of hepatic encephalopathy (AUROC 0.854). This may have been influenced by the limited number of patients who experienced clinical encephalopathy, although it is feasible that encephalopathy could be predicted by liver stiffness. This increased stiffness may reflect the intrahepatic shunting of blood and reduced hepatocyte mass, which are both important for the development of encephalopathy. As the pathophysiology of hepatic encephalopathy is complex, it is likely that the LSM will be variable and not correlate well with all patient populations 25. This may explain why other authors, also with similarly low numbers of patients with clinical encephalopathy, have shown that liver stiffness and encephalopathy did not correlate well in their cohort (AUROC 0.70) 14. Future studies with larger numbers of more advanced liver disease will be required to examine this in depth.
The MELD score is used to assess patients for transplantation as it is a guide to 3 month survival from cirrhosis, whereas the Child Pugh score is an established indicator of 1 and 2 year mortality from advanced liver disease. As discussed above, progression of liver disease is associated with increasing fibrosis, although other changes such as loss of inflammation and steatosis along with increased intrahepatic shunting and thrombosis 26, 27 may coexistently reduce liver stiffness. Therefore, it is not surprising that our study found that LSM by ARFI is only moderately successful in distinguishing Child Pugh A from Child Pugh B or C (AUROC 0.69); or MELD < 15 from MELD >15 (AUROC of 0.71).
A limitation of this study is that the presence of complications was determined retrospectively. In addition, our study also only focused on ARFI liver stiffness, and there has been some suggestion that spleen stiffness may have better predictive ability with complications such as oesophageal varices and may warrant further investigation 23, 28.
Our study has shown that ARFI LSM, in patients with cirrhosis, offers poor correlation with the presence of liver disease complications, with the possible exception of hepatic encephalopathy. It is likely that other factors are significantly involved with decompensation and the presence of clinical complications, rather than just hepatic fibrosis. However, it is also possible that the co-existing reduction in inflammation and steatosis that may occur with worsening liver failure could be responsible for a reduced liver stiffness that would preclude further increases in ARFI scan results. This may limit the clinical utility of ARFI in detecting the risk of hepatic complications or decompensated liver failure, although larger studies would be needed to better define to what extent this may occur.
Acknowledgments:
The authors would like to sincerely thank Dr Andrew Clouston for helpful discussions and providing valuable references for this manuscript
References
- 1.H B LEFTON, ROSA A, COHEN M. (2009) Diagnosis and epidemiology of cirrhosis. , The Medical clinics of North America; vii 93(4), 787-99.
- 2.J L DIENSTAG. (2002) The role of liver biopsy in chronic hepatitis C. Hepatology. , (Baltimore, Md), 36(5 Suppl 1, 152-60.
- 3.BRAVO A A, SHETH S G, CHOPRA S. (2001) Liver biopsy. The New England journal of medicine. 344(7), 495-500.
- 4.FRIEDRICH-RUST M, NIERHOFF J, LUPSOR M. (2012) Performance of Acoustic Radiation Force Impulse imaging for the staging of liver fibrosis: a pooled meta-analysis. Journal of viral hepatitis. 19(2), 212-9.
- 5.BOTA S, HERKNER H, SPOREA I. (2013) Meta-analysis: ARFI elastography versus transient elastography for the evaluation of liver fibrosis. Liver international : official journal of the International Association for the Study of the Liver 33(8), 1138-47.
- 6.FIERBINTEANU BRATICEVICI C, SPOREA I, PANAITESCU E, TRIBUS L. (2013) Value of acoustic radiation force impulse imaging elastography for non-invasive evaluation of patients with nonalcoholic fatty liver disease. Ultrasound in medicine & biology 39(11), 1942-50.
- 7.FRIEDRICH-RUST M, M F ONG, MARTENS S. (2008) Performance of transient elastography for the staging of liver fibrosis: a meta-analysis. Gastroenterology. 134(4), 960-74.
- 8.PETTA S, V DI MARCO, CAMMA C, BUTER A G, CABIBI D et al. (2011) Reliability of liver stiffness measurement in non-alcoholic fatty liver disease: the effects of body mass index. Alimentary pharmacology & therapeutics. 33(12), 1350-60.
- 9.POTTHOFF A, ATTIA D, PISCHKE S. (2013) Influence of different frequencies and insertion depths on the diagnostic accuracy of liver elastography by acoustic radiation force impulse imaging (ARFI). European journal of radiology. 82(8), 1207-12.
- 10.BOTA S, SPOREA I, SIRLI R, POPESCU A, JURCHIS A. (2013) Factors which influence the accuracy of acoustic radiation force impulse (ARFI) elastography for the diagnosis of liver fibrosis in patients with chronic hepatitis C. Ultrasound in medicine & biology 39(3), 407-12.
- 11.SALZL P, REIBERGER T, FERLITSCH M. (1980) Evaluation of portal hypertension and varices by acoustic radiation force impulse imaging of the liver compared to transient elastography and AST to platelet ratio index. Ultraschall in der Medizin , Stuttgart, Germany : 35(6), 528-33.
- 12.PARK Y, S U KIM, S Y PARK.(2015)A novel model to predict esophageal varices in patients with compensated cirrhosis using acoustic radiation force impulse elastography,PLoS One,10(3):. 0121009.
- 13.MORISHITA N, HIRAMATSU N, OZE T.Liver stiffness measurement by acoustic radiation force impulse is useful in predicting the presence of esophageal varices or high-risk esophageal varices among patients with HCV-related cirrhosis. , Journal of gastroenterology 2014; 49(7), 1175-82.
- 14.VERMEHREN J, POLTA A, ZIMMERMANN O. (2012) Comparison of acoustic radiation force impulse imaging with transient elastography for the detection of complications in patients with cirrhosis. Liver international : official journal of the International Association for the Study of the Liver 32(5), 852-8.
- 15.SPOREA I, RATIU I, BOTA S, SIRLI R, JURCHIS A. (2013) Are different cut-off values of liver stiffness assessed by transient elastography according to the etiology of liver cirrhosis for predicting significant esophageal varices? Medical ultrasonography. 15(2), 111-5.
- 16.BELLENTANI S, TIRIBELLI C, SACCOCCIO G. (1994) Prevalence of chronic liver disease in the general population of northern Italy: the Dionysos Study. Hepatology. , (Baltimore, Md) 20(6), 1442-9.
- 17.TOSHIMA T, SHIRABE K, TAKEISHI K.New method for assessing liver fibrosis based on acoustic radiation force impulse: a special reference to the difference between right and left liver. , Journal of gastroenterology 2011; 46(5), 705-11.
- 18.CERINI F, VILASECA M, LAFOZ E.Enoxaparin reduces hepatic vascular resistance and portal pressure in cirrhotic rats. , Journal of hepatology 2016; 64(4), 834-42.
- 19.POWELL E E, W G COOKSLEY, HANSON R, SEARLE J, J W HALLIDAY et al. (1990) The natural history of nonalcoholic steatohepatitis: a follow-up study of forty-two patients for up to 21 years. Hepatology. , (Baltimore, Md) 11(1), 74-80.
- 20.E M BRUNT. (2016) Nonalcoholic Fatty Liver Disease: Pros and Cons of Histologic Systems of Evaluation. International journal of molecular sciences. 17-1.
- 21.S U KIM, OH H J, I R WANLESS, LEE S, K H HAN et al.The Laennec staging system for histological sub-classification of cirrhosis is useful for stratification of prognosis in patients with liver cirrhosis. , Journal of hepatology 2012; 57(3), 556-63.
- 22.B K KIM, H J O, J Y PARK. (2013) Early on-treatment change in liver stiffness predicts development of liver-related events in chronic hepatitis B patients receiving antiviral therapy. Liver International. 33(2), 180-89.
- 23.RIZZO L, ATTANASIO M, M R PINZONE.A new sampling method for spleen stiffness measurement based on quantitative acoustic radiation force impulse elastography for noninvasive assessment of esophageal varices in newly diagnosed HCV-related cirrhosis. BioMed research international 2014;2014:. 365982.
- 24.MORI K, ARAI H, ABE T.. Spleen Stiffness Correlates with the Presence of Ascites but Not Esophageal Varices in Chronic Hepatitis C Patients. BioMed research international 2013; 2013: 7.
- 25.FREDERICK R T. (2011) . Current Concepts in the Pathophysiology and Management of Hepatic Encephalopathy. Gastroenterology & Hepatology 7(4), 222-33.
- 26.BOSCH J, PIZCUETA P, FEU F, FERNANDEZ M, J C GARCIA-PAGAN. (1992) Pathophysiology of portal hypertension. Gastroenterology clinics of North America. 21(1), 1-14.
Cited by (4)
This article has been cited by 4 scholarly works according to:
Citing Articles:
BMJ Open Gastroenterology (2022) Crossref
BMJ Open Gastroenterology (2022) OpenAlex
Amjad Alhyari, C. Görg, C. Dietrich, Corrina Trenker, Lena Strauch et al. - BMJ Open Gastroenterology (2022) Semantic Scholar
Gastroenterology Hepatology and Endoscopy (2018) OpenAlex
H. K. Leong, R. Gibson, S. Sood, D. Nadebaum, A. Gorelik et al. - (2018) Semantic Scholar
