Profile of Similarity of Electron Withdrawing Structure Towards Analgesic-Anti-Inflammatory Activity of The Novel Isatin Analogue: Design and Implementation of Phase I Drug Discovery
Abstract
Isatin (1H-indole-2,3-dione ) and derivatives demonstrate a diverse array of biological activities. Isatin and 5-halo derivatives has reacted to form the schiff’s bases , mannich bases and friedal craft alkylation’s to form C-C, C-N, C=N bonds. From the spectral studies, isatin has undergoes reaction at C-3 and N-1 position and synthesized lead in present schme and seen the similarity of structure and analgesic-anti-inflammatory activity.
Author Contributions
Academic Editor: Nasim Habibzadeh, Teesside university
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2018 Rahul Hajare. et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Isatin is a versatile compound isolated in 1988 and reported to possess a wide range of biological activity in mammals. Isatin also is a synthetically versatile substrate that can be used to prepare a large variety of heterocyclic compounds, such as indoles and quinolines, and as a raw material for drug synthesis. Schiff bases and mannich bases of isatin has known to possess a wide range of pharmacological properties including anticonvulsant, antibacterial, antiprotozoal, antifungal, antiviral, anti-HIV, tuberculostatic, analgesic, anticancer activities. In the view of this fact, we have planned the synthesis of some novel isatin analogue and screening them for analgesic-anti-inflammatory activity. Isatin, an extract from Strobilanthes cusia. The roots and the leaves of the plant, Strobilanthes cusia of the Acanthaceae family that is widely distributed in northern and central China, have been used in traditional Chinese medicine to treat a variety of ailments caused by microorganisms and virus.
Experimental:
Material and Methods
All reagents have obtained from Sigma Aldrich Bangalore, and Loba Chemie, Mumbai. All the solvents used in these studies has dried and distilled before use. Melting points have determined by open capillary tube method and are uncorrected. The purity and homogeneity of the synthesized compound has ascertained by T.L.C. on glass plate using silica gel G as adsorbent and solvent system benzene: ethanol (9:1). The spot has visualized by iodine vapor. The 1H-NMR spectra has recorded on CDCl3 (BRUCKRS). All spectra have obtained from Indian Institute of Technology Pawai Mumbai.
Methodology
Lab Scale R & D
Methods of Preparation of 5-substituted N-benzyl isatin derivatives: 6
Equimolecular quantity of isatin and benzyl chloride has taken in RBF. To this mixture 20 ml of dimethyl formamide and potassium carbonate has added. After gently shaking the mixture refluxed for 2hrs. The mixture has cooled and pours into the 100 ml of ice cold water. The resultant orange colour precipitate has collected and washed with water. After drying it has recrystallized from ethanol. M.P. is1200C-1300C 1, 2.
Methods of Preparation of Schiff’s bases of 5-substituted N-benzyl isatin derivatives:
Equimolecular quantity of N-benzyl derivatives and amines has added into 20ml of absolute ethanol in 250 ml of round bottom flask. In this mixture few drops of glacial acetic acid has added. The reaction mixture has refluxed for 2-3 hour extended and checked for completion through visual procedure. After completion of reaction the mixture has placed for 24 hour, filtered and recrystalized from ethanol 3, 4. (Table 1)
Table 1. List of compounds| CODE | COMPOUND | TEXTURE |
|---|---|---|
| P-01 |

|
Yellow crystalline |
| P-02 |
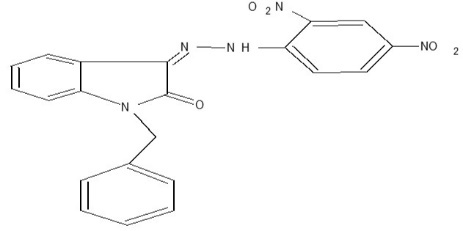
|
Orange crystalline |
| P-03 |
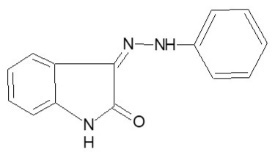
|
Yellow shiny crystalline |
| P-04 |
|
Orange crystalline |
| P-05 |

|
Red-orange crystaline |
| P-06 |

|
Brown crystaline |
| P-07 |
|
Red orange crystal |
| P-08 |

|
Brown |
| P-09 |

|
Yellow crystalline |
| P-10 |

|
Yellow crystalline |
| P-11 |

|
Yellow crystalline |
Preliminary Pharmacological Screening
Acute Toxicity Studies, Cut-off LD50
LD50 of test compounds has performed at Anuradha College of Pharmacy Chikhali as per the O.E.C.D. guideline. Test compounds have suspended in 1% CMC solution. The compounds have administered orally at a dose level of 300,500 and 1000 mg/kg body weight, to groups of 6 animals. After administration of test compounds the rats have observed for gross behavioral neurological autonomic and toxic effects. The toxicological effects have observed in terms of mortality. No death occurred within 24 h of dose of 300 and 500mg/kg but at a dose of 1000mg/kg 50% mortality has observed. As dose has increased further up to 4000mg/kg, all the animals have died. Hence 1000 mg/kg dose has considered as LD50. 1/10th of the LD50 has considered as an effective dose i.e.100 mg/kg.
Evaluation of Analgesic Activity. (Writhing test)
The method has followed described by International standards. The total number of writhing following intraperitoneal administration of acetic acid solution (1%, 10 ml/kg) has recorded over a period of 30 min, starting 5 min after acetic acid injection. The mice have treated with test (100 mg/kg) suspended in 1% CMC or standard drug (Aspirin, 300 mg/kg), 30 min before administration of acetic acid 5.
The number of writings and stretching has recorded and permitted to express the percentage of protection using the ratio = (Control mean-treated mean) ×100/control mean
Carrageenan-Induced Hind Paw Edema in Rats.
The acute hind paw edema has produced by injecting 0.1 ml of Carrageenan (prepared as 1% suspension in 1% CMC) locally into the plantar aponeurosis of the right hind paw of rats, to groups of six ,and standard drug, acetylsalicylic acid (ASA, 300 mg/kg), respectively. Test (drug 100 mg/kg) and ASA has administered 1 h prior to the injection of Carrageenan. The rat pedal volume up to the ankle joint has measured using plethysmometer at 0 h (just before) and 1h and 3 h after the injection of Carrageenan. Increase in the paw edema volume has considered as the difference. Percent inhibition of edema volume between treated and a control group has calculated as follows:
Percent inhibion = (Vc − Vt)/Vc × 100, Where Vt = mean relative change in a paw volume in test group and Vc = mean relative change in paw volume in control group.
Result and Discussion
The all synthesized compound has analyzed by 1H-NMR spectra on CDCl3.The reproducibility of the developed method has ascertained by considering the physical properties of the synthesized compound.
N-benzyl 3-(phenylimino)-1,3-dihydro-indole-2one (P-01)
Yield 70%, m.p.2600C, Rf value 0.6, 1H-NMR (1H) =N-NH d 12.82, (4-H) d 7.6, (2 Ar-H) N-CH2 d 6.6.
N-benzyl 3-[(2, 4 -dinitrophenylimino)-hydrazono]-1, 3-dihydro-indole-2-one (P-02)
Yield 70%, m.p. >3000C Rf value 0.53, 1H-NMR (1H) =N-NH d 12.82, (4-H ) d 7.6, (2 Ar-H) N-CH2 d 6.6.
3-(phenylimino)-1,3- dihydro-indole-2-one (P-03)
Yield 76%,m.p.>2200C,Rf value 0.46. 1H-NMR (1H) =N-NH d 12.7, (4-H) d 7.8, (4 Ar-H) d 7.3.
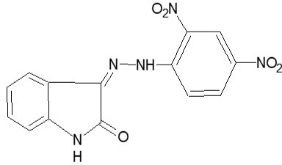
3-[(2, 4 -dinitro-phenylimino)-hydrazono]-1,3- dihydro-indole-2-one (P-04)
Yield 80%,m.p. >2900C, Rf value 0.5. 1H-NMR (1H) =N-NH d 12.15, (4-H) d 8.5, (2 Ar-H) d 7.4.
N-benzyl 3-(phenylimino)-5-Chloro-1,3-dihydro-indole-2-one (P-05)
Yield 40%, m.p. >3000C Rf value 0.7. 1H-NMR (1H) =N-NH d 12.8, (3-H) d 7.6 , (4 Ar-H) d 7.3, (2H) N-CH2 d 6.6.
3-(phenylimino)-5chloro-1,3-dihydro-indole-2-one (P-06)
Yield 70%, m.p. >3000C Rf value 0.53. 1H-NMR (1H) =N-NH d 11.14, (3-H) d 7.5 , (4 Ar-H) d 7.3.
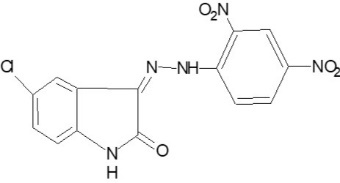
3-[(2, 4 -dinitro-phenylimino)-hydrazono]-5Chloro-1,3-dihydro-indole-2-one (P-07)
Yield 80%, m.p. >3500C Rf value 0.52. 1H-NMR (1H) =N-NH d 9.4, (3-H) d 8.3, (3 Ar-H) d 7.3.
3-(phenylimino)-5 Fluro-1,3-dihydro-indole-2-one(P-08)
Yield 45%, m.p. 2700C Rf value 0.9. 1H-NMR (1H) =N-NH d 12.7, (3-H) d 7.6, (4Ar-H) d 7.3.
3-[(2, 4 -dinitro-phenylimino)-hydrazono]-5 Fluro-1,3-dihydro-indole-2one (P-09)
Yield 50%, m.p. >3500C Rf value 0.51. 1H-NMR (1H) =N-NH d 12.7, (3-H) d 7.6, (2Ar-H) d 7.3.
3-(phenylimino)-5 Nitro-1,3-dihydro-indole-2-one (P-10)
Yield 72%, m.p. 3000C Rf value 0.9. 1H-NMR (1H) =N-NH d 12.7, (3-H) d 7.6, (2 Ar-H) d 7.3.
3-[(2, 4 -dinitro-phenylimino)-hydrazono]-5 Nitro-1,3-dihydro-indole-2-one (P-11)
Yield 80%, m.p. >3500C Rf value 0.5. 1H-NMR (1H) =N-NH d 9.4, (3-H) d 8.5, (3 ArH) d 7.5.
In the present study, new Schiff’s bases of 5-substituted N-benzyl isatin derivatives have synthesized and their anti-inflammatory and analgesic activities has evaluated against standard drug. The anti-inflammatory activity of the compounds have examined by using carrageenan induced rat paw oedema method and analgesic activity by Writhing test. The test compound P-01, P-02, P-06 and P-08 exhibit statistically significant at 3 hrs. Except P-03 .However the effect has comparable to aspirin at 3 hrs. (Table 2)
Table 2. Carrageenan Induced Rat Paw Oedema Method| Group | Test Material (dose) | Mean increase in paw volume + Standard deviation and % inhibition | ||
| 0 hr. | 1 hr. | 3 hr. | ||
| 1. | Control | 1.1 + 0.17 | 1.40 + 0.12 | 1.70 + 0.14 |
| 2. | Standard (Aspirin 300mg/kg) | 1.1 + 0.12 | 1.3 +0.12 (33%) | 1.4 + 0.14 (50 %) |
| 3. | P-01 (100mg/kg) | 1.2 + 0.12 | 1.4 + 0.14 (33%) | 1.5 + 0.14 (50%) |
| 4. | P-02 (100mg/kg) | 1.00+ 0.16 | 1.3+ 0.16 (00%) | 1.5 + 0.14 (16 %) |
| 5. | P-03 (100mg/kg) | 1.2 + 0.14 | 1.4 + 0.14 (33%) | 1.4 + 0.14 (33%) |
| 6. | P-06 (100mg/kg) | 1.1 + 0.10 | 1.3 + 0.12 (33%) | 1.3 + 0.4 (33%) |
| 7. | P-08 (100mg/kg) | 1.00 + 0.14 | 1.2 + 0.14 (33%) | 1.3 + 0.17 (50%) |
The results are expressed as MEAN (+ S.D.) from six observations. One way ANOVA is followed by Dunnet’s t- test. Group II ndto VIIth is compared with group I st (control). (P< 0.01); * = not significant (for 3 hrs.). No-one compound is significant at 1 hrs (P= 0.O74). (Figure 1) (Table 3)
Figure 1.Carageenan Induced rat paw edema
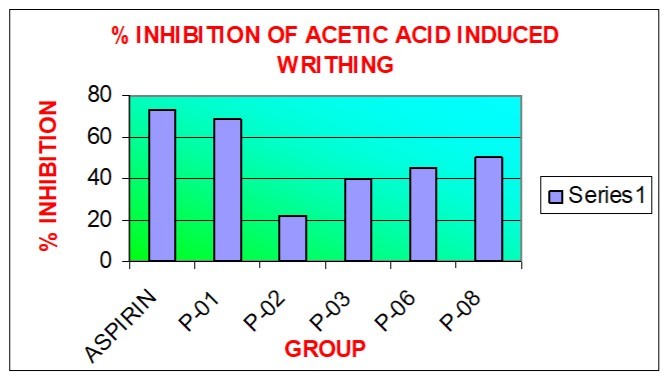
| S.N. | Group (Dose) | Response time ( + s.d. ) | No. of Writhing ( + s.d. ) | % Inhibition in Writhing |
| 1. | Control | 2.37 (0.33) | 59.83 (2.31) | ---------- |
| 2. | Standard (Aspirin 300mg/kg) | 5.54(0.41) | 15.85(2.63) | 73 |
| 3. | P-01 (100mg/kg) | 7.76(0.94) | 18.50(1.87) | 69 |
| 4. | P-02 (100mg/kg) | 3.13(0.33 | 46.66(1.60) | 22 |
| 5. | P-03 (100mg/kg) | 4.35(0.37) | 35.83(1.16) | 40 |
| 6. | P-06 (100mg/kg) | 4.85(0.27) | 32.83(1.16) | 45 |
| 7. | P-08 (100mg/kg) | 5.42(0.60 | 29.33(2.42) | 50 |
The results are expressed as mean (+ S.D.) from six observations. One way ANOVA is followed by Dunnet’s t- test. Group II ndto VIIth is compared with group I st (control).(P< 0.01); and are significant (Figure 2)
Conclusion:
The experiments reported here from results of biological activity of the compounds there has clear cut relation of compounds with each other due to structural similarity. A compound with N-benzyl-3-substituted isatin has synthesized using appropriate synthetic route and screened for analgesic and anti-inflammatory activity.The test compounds showed significant anti-inflammatory and analgesic activity compared with the standard drug Aspirin. On the study of the structure-activity relationship reveals that the presence of the electron withdrawing group does not affect anti-inflammatory activity and increase in electro-negativity increases the analgesic activity. (Table 4)
Table 4. Comparative study of the activity| S.N. | Compound | R | R I | Activity | |
|---|---|---|---|---|---|
| Analgesic | Anti-Inflammatory | ||||
| 1 | P-01 | C6H5 | H | 69 | 50 |
| 2 | P-02 | C6H4N202 | H | 22 | 16 |
| 3 | P-03 | H | H | 40 | 33 |
| 4 | P-06 | H | Cl | 45 | 33 |
| 5 | P-08 | H | F | 50 | 50 |
This article belongs to the Public Health. The experimental data and analysis part is very sufficient, and the conclusion is very reasonable. It is recommended to publish the article in your esteemed journal. I hope this helps.
Acknowledgments
This research work is in completed under the supervision and guidance Renowned Laboratory Scientist, Respected Dr. Ramesh S. Paranjape, Retired Director & Scientist ‘G’ National AIDS Research Institute, India. I express my sincere gratitude towards Respected Sir for motivation and being great knowledge source for this work.
Disclosures
Animal ethical approval
Funding
None
Abbreviations
References
- 1.Hajare Rahul, Paranjape Ramesh, Kulkarni Smita.Tailored microwave technology for Synthesis N-[(3Z)-5-chloro-1-(morpholin-4-ylmethyl)-2-oxo-1,2-dihydro-3H-indol-3 ylidene. pyridine-4 carbohydrazide as HIV-1 inhibitors.(2016) Drug Formulation & Bioavailability Congress” September 05-07 , Beijing, China .
- 2.Hajare Rahul, Paranjape Ramesh, Kulkarni Smita. (2016) Technology development and design of novel 1, 3, 5-tri substituted-1H-indole-2, 3-dione: HIV-1 Inhibitors with displays strategic nanomolar cytotoxicity. 251stACS National Meeting & Exposition held on March 13-17,2016 in , San Diego, CA, USA .
- 3.Hajare Rahul, Kulkarni Smita, Thakar Madhuri, Paranjape Ramesh. (2016) . Technology Development and Design of Novel 1, 3, 5-tri Substituted-1H-Indole-2, 3-Dione HIV-1 Inhibitors With Displays Strategic Nanomolar Cytotoxicity , World J Pharm Pharm Sci.; 5(6), 391.
Cited by (5)
This article has been cited by 5 scholarly works according to:
Citing Articles:
Noor Waleed, Ibrahim, M. Mahdi, Ayad M. R. Raauf - Iraqi Journal of Pharmaceutical Sciences (2025) Semantic Scholar
N. Ibrahim, M. Mahdi, Ayad M. R. Raauf - Materials Today: Proceedings (2021) Semantic Scholar
Materials Today: Proceedings (2021) Crossref
Materials Today Proceedings (2021) OpenAlex
Rahul Hajare - Organic & Medicinal Chemistry International Journal (2018) Semantic Scholar
International Physiology Journal (2018) OpenAlex