Auricular Vagus Nerve Stimulation Improves Chronic Pain and Pain-Related Cytokine Levels: A Clinical Study
Abstract
Periauricular Vagus Nerve Stimulation (pVNS) has been proven safe and effective in reducing chronic pain and related comorbidities in numerous clinical studies. This multicenter, interventional study used a non-randomized, interrupted time-series analysis to test the efficacy of an 8-week treatment protocol using the Stivax neurostimulator device. Subjects (n=33, 15 F, 18 M, age 40-77) were recruited at 3 clinic sites in California and Colorado. All subjects had long-term chronic pain and had failed other treatments. Subjects were treated with the Stivax device 3 times (2 weeks on, 1 week off). Subjective assessments of pain (Visual Analog Scale), disability (Oswestry Disability Index), depression (PHQ-9), and activity (IPAQ-E) were collected at baseline and weekly. Objective blood levels of pain-related cytokines collected at the end of weeks 2 and 8. Most subjects reported reduced pain, disability, and depression, with increased activity levels. At the end of week 8, subjects reported an average reduction in pain by 38.5% (3 subjects reported no pain), depression by 43.6% (2 subjects reported no depression), disability by 38.6% (2 subjects reported no disability), and an average 26.1% increase in activity level (5 subjects doubled their activity level). Levels of the pain-related cytokines IL-1ꞵ, IL-2, IL-3, IL-7, IL-10, IL-15, IL-17α, IL-21, TNF-α, IFN-γ, and FLT3-ligand showed improvement at week 8. pVNS is believed to “reset” central sensitization underlying chronic pain and other central sensitization syndromes, engaging the body’s pain modulation systems. Our results indicate that pVNS can clinically significantly improve chronic pain and associated morbidities without adverse effects.
Author Contributions
Academic Editor: Peter Awhen, Nigeria
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2023 James Anderson, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Author note
Co-author credential: Forest Tennant (MD).
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Chronic pain affects more than 20% of adults in the United States, with 8% suffering from chronic pain that interferes with daily life and work1. Chronic pain can significantly reduce patient quality of life and have broad psychosocial effects2. Much of the pathophysiology of chronic pain is different from acute pain and can be very difficult to treat3. Periauricular Vagus Nerve Stimulation (pVNS) is a safe and effective minimally invasive treatment to reduce chronic pain and its comorbidities4, 5, 6, 7, 8, 9, 10, 11. Studies of transcutaneous neurostimulation include a broad range of chronic diseases and pain conditions as well as a large variety of subjective and objective measures of symptoms and biomarkers. In this study, we tested the effect of pVNS on pain and pain-related comorbidities in a sample of patients with long-term chronic pain that did not respond to standard therapy. Our results showed improvement in subjective measures of pain, disability, depression, and activity levels, as well as improvement in circulating levels of pain-related cytokines. This report focuses on the effect of vagal stimulation on self-reported measures that affect quality of life and cytokines related to pain and inflammation.
METHODS
Subjects
The study population consisted of 33 subjects, aged 40-77, 15 female and 18 male recruited across three clinic sites in California and Colorado, USA. Informed consent was obtained from all subjects. Data were collected during clinical visits from October 2019 through February 2020. All subjects suffered from long-term chronic pain that failed to respond to other treatment modalities. Exclusion criteria for this study were pregnancy, hemophilia, prescription blood thinners, cardiac arrhythmias, pacemakers, bipolar disorder or similar psychological or psychiatric disability, drug or alcohol abuse within the past six months, and any ongoing infection or other serious medical issues. The subject dropout rate was 6.1% (2 of 33 subjects). One subject withdrew due to discomfort while sleeping with the device implanted, while the other withdrew due to transportation difficulties. Data from these subjects is excluded from analysis.
Study Design
This multicenter, interventional study was modeled on a non-randomized, interrupted time-series analysis design. During this eight-week treatment protocol, each subject was assessed during their weekly clinic visits. Subjective assessments and objective clinical measurements were recorded. The study design called for observing various outcome measures taken each week. The goal of these measures was to determine the correlation with the treatment intervention.
Clinical Protocol
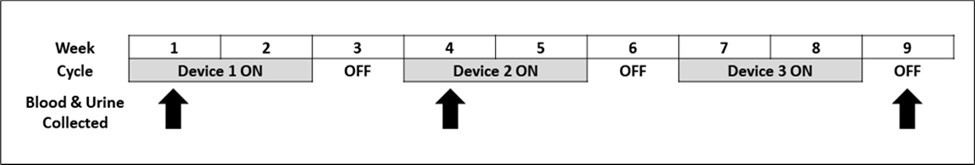
Subjects were examined during their first visit, and baseline assessments and clinical measurements were taken. The Stivax neurostimulator device was applied during the first visit. The Stivax neurostimulator electrodes were percutaneously implanted in the triangular fossa of either the left or right ear. This first device remained activated on the test subject for two weeks and was then removed. One week after the removal of the first device, a second device was applied for another two weeks, then removed. One week after the removal of the second device, the third and final device was applied for the final two-week activation period. See ( Figure 1 )for a diagram of the experimental design.
Neurostimulation Device
The Stivax device (Biegler GmbH, Austria) is an FDA-approved electro-acupuncture device. The device consists of two electrodes percutaneously implanted into the skin and retained in place with adhesive and an electrical stimulator connected via wire. The stimulator is a constant-current signal generator that produces a square monophasic wave, delivering 200 μs pulses at 1 Hz with an adjustable current of 0.065-1.12 mA. The device intensity (mA) is manually set based on the subject’s comfort level. The stimulator is activated continuously during the two weeks it is worn, alternating between 40 minutes on and 20 minutes off of continuous pulses and 20 minutes of no pulses.
Outcome Measures
During each of the nine weekly clinic visits, subjects were given subjective assessments covering four dimensions of chronic pain: pain levels, disability level, depression, and physical activity level. Blood and urine samples were collected during the first, third, and final clinic visits.
Subjective Assessments
Weekly assessments consisted of four self-reported measures. Pain levels over the previous week were assessed using the Visual Analog Scale (ranging from 0-10), including both the lowest (Weekly Low) and highest (Weekly High) levels of chronic pain. Disability level was assessed using the Oswestry Disability Index (OWI). Depression was assessed using the Patient Health Questionnaire (PHQ-9). Physical activity levels were assessed using the International Physical Activity Questionnaire (IPAQ-E).
Objective Assessments
Blood and urine were collected at the first, third, and last visits (Figure 1). Blood serum was tested for levels of pain-related hormones, neurotransmitters, and endogenous opiate peptides as well as pain-related cytokines Interleukin 1ꞵ (IL-1ꞵ), Interleukin 2 (IL-2), Interleukin 3 (IL-3), Interleukin 7 (IL-7), Interleukin 10 (IL-10), Interleukin 15 (IL-15), Interleukin 17 alpha (IL-17α), Interleukin 2 (IL-21), Tumor Necrosis Factor-alpha (TNF-α), Interferon-gamma (IFN-γ), and FMS-like Tyrosine Kinase Receptor 3 ligand (FLT3-ligand). Urine samples were also collected for toxicology screening.
Statistics
Results are reported as the mean change (Δ) from baseline levels measured at the initial clinic visit. The mean Δ percentages were calculated by averaging Δ in units after week eight of the treatment program, thus applying appropriate weighting of incremental changes versus larger deltas in units. The standard error of the mean (SEM) was calculated by dividing the standard deviation of the sample by the square root of the sample size (n). The highlights of this study were chosen by ranking the test results by their mean Δ percentages and selecting those tests with SEMs of < 3 (lower standard errors).
Results
Subjective Measures
Most subjects reported improvement across all four subjective measures, including lower pain, disability, and depression, and increased activity levels. Regression analysis showed a steady improvement for all four subjective measures from baseline to week eight. At week eight, the average reduction in pain was 38.5%, the average reduction in depression was 43.6%, the average reduction in disability was 38.6%, and the average increase in activity level was 26.1%.
Pain
Chronic pain levels improved in the majority of subjects ( Figure 2 ). Low Pain, the minimum level of chronic pain, was reduced by 16.7%-100% in 71.0% of subjects. High Pain, the maximum level of chronic pain, was reduced by 10.0%-100% in 77.4% of subjects. Average Pain, the average of High Pain and Low Pain, was reduced by 11.1%-100% in 83.9% of all subjects. Weekly Low Pain was reduced by 44.0%, weekly High Pain levels were reduced by 29.8%, and weekly Average Pain was reduced by 38.5%. Average Pain was reduced by 50%-100% in 36.4% of subjects. While most subjects reported at least a modest improvement in pain levels, some reported a 100% reduction in pain. Ten subjects reported zero low-level pain, two subjects reported zero high-level pain, and three subjects had zero Average Pain at week eight.
Disability
Disability levels were reduced in almost all subjects ( Figure 3 ). The level of disability was reduced by 10.0%-100% in 93.5% of subjects. Disability was reduced by 50%-100% in 36.4% of subjects. The average disability reduction was 38.6%. The overwhelming majority of subjects reported improved disability, with two reporting a 100% reduction in disability.
Depression
Most subjects reported a reduction in depression (Figure 4 ) Reduced depression was reported in 74.2% of subjects with a range of 10.0%-100% reduction. Depression was reduced by 50%-100% in 48.5% of subjects. The average reduction in reported depression was 43.6%, five points on the PHQ-9.
Activity
Physical activity increased in more than half of the subjects ( Figure 5 ). Activity levels increased in 56.3% of patients, with an average increase of 26.1%. Activity levels were doubled in five subjects.
Figure 5.International Physical Activity Questionnaire (IPAQ-E)
Opioid Medication Use
Average opiate/opioid medication use was reduced by 42.9%, with three subjects discontinuing all opioid medications.
Side-Effects and Dropouts
Only one subject reported an adverse side effect during the study: discomfort during sleep due to the implanted electrode. This subject was one of two who dropped out of the study (6.1% dropout rate); the other subject dropped out due to weather-related difficulties traveling to the clinic. Both subjects dropped out after week four, but both had reported improvement in pain, disability, and depression. One subject reported an improvement in activity levels, while the other reported a decrease in activity.
Objective Measures
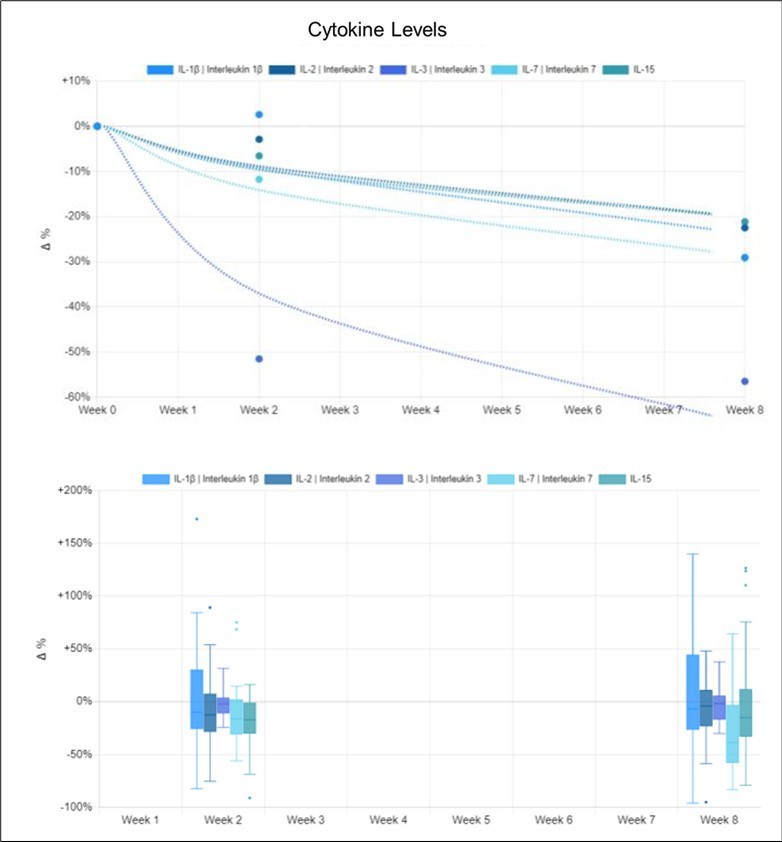
Sixteen pain-related cytokines showed improvement in response to therapy (Figure 6, Figure 7, Figure 8). Assessments of pain-related hormones and other analytes showed high variability between individuals and did not correlate with the treatment protocol; data are not reported here.
For all reported cytokines except IL-10, reduced levels indicate improvement in inflammation. IL-10 levels increased by an average of 144.5% above baseline at week eight. The decreases seen in all other average cytokine levels were: IL-1β 21.1%, IL-2 22.5%, IL-3 56.5%, IL-7 29.0%, IL-15 21.2%, IL-17α 21.7%, IL-21 14.2%, TNF-α 11.4%, IFN-γ 27.5%, and FLT-3 Ligand 37.8%.
Figure 7.Interleukin 10 (IL-10) Levels
Other Findings
Most subjects reported a reduction in their need for opiate pain medications, with three subjects discontinuing opiate pain medications entirely. None of the subjects reported any major negative adverse events related to the treatment. The subject dropout rate was 6.1% (2 of 33 subjects). One subject withdrew due to discomfort while sleeping with the device implanted, while the other withdrew due to transportation difficulties.
Discussion
Chronic Pain Involves Autonomic Dysfunction
The picture painted by researchers to explain the mechanisms and pathways involved in chronic pain is complex. Chronic pain can be summarized as a dysfunctional interaction between inflammatory and immune mechanisms, and autonomic dysfunction leading to maladaptive neuroplasticity. Vagus nerve stimulation has proven to be a valuable tool for treating pain and other pathologies that involve autonomic dysfunction, including heart disease and metabolic syndrome12, 13, 14. The vagus nerve is a desirable target for therapeutic intervention. Cervical and auricular branches of the vagus nerve are easy to non-invasively access, allowing external, transcutaneous electrical stimulation of both afferent and efferent nerve fibers. Additionally, the vagus nerve is involved in multiple bodily mechanisms and influences the hippocampal-pituitary-adrenal (HPA) axis, brain-gut axis, and other pathways4, 13.
Pathophysiology of Chronic Pain
Inflammation, Immunity, and Pain
Research has also shown a complex interplay between inflammation and the immune system in pain, including syndromes with chronic pain16, 17, 23, 24, 26, 28. Elevated levels of pro-inflammatorycytokines14, 21, 23, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35,hormones13, 36, 37, 38, and altered neurotransmitter levels9, 39are associated with a wide range of chronic pain conditions6, 6, 8, 9, 11, 12, 17, 18, 25, 26, 28, 29, 30, 31, 34, 40, 41, 42 These cytokines and other peptides can serve as both biomarkers of inflammation and chronic pain15, 18, 25, 31, 34, 40 as well as therapeutic targets28, 35, 43, 44, 45. Neuroinflammation is one important aspect of chronic pain conditions associated with depression, a significant comorbidity of these conditions46. Neuroinflammation also influences maladaptive neuroplasticity, rewiring the brain to be more sensitive to pain3, 19, due in part to a shift in the function of GABA-ergic neurons from inhibitory to excitatory activity19, 39, 46.
Central Sensitization
Central sensitization is a relatively new unifying concept to describe the pathological “amplification” of otherwise non-noxious stimuli19, 47in chronic pain and disorders such as fibromyalgia, myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS), and others3, 19, 20, 22. The sympathetic nervous system plays an important role in modulating the communication between peripheral sensory nerves and the central nervous system and has proven to be a good target for therapy to address conditions with chronic pain44,48. The vagus nerve (cranial nerve 10) is the primary parasympathetic innervation of the body, affecting everything from heart rate to gastrointestinal motility. Auricular branches of the vagus nerve travel through the outer ear, making it a useful location to “access” the vagus non-invasively. Transcutaneous vagal nerve stimulation is known to induce the release of neurotransmitters and endogenous opioid peptides, such as endorphins9,48, and improve inflammatory cytokine levels49 in patients with chronic inflammation.
The results of this study demonstrate that pVNS can decrease chronic pain, disability, and depression while improving pain-related limited activity in persons with chronic pain that has not responded satisfactorily to other treatments. Additionally, pVNS can lower elevated pro-inflammatory cytokines and raise levels of the anti-inflammatory cytokine IL-10. This adds further credence to other studies that have shown similar types of symptomatic and physiological improvement in study populations with various chronic conditions4, 56, 8, 9, 10, 45, 48, 49, 50, 51. Notably, patients in this study showed symptomatic improvement within a relatively short time frame of eight weeks, with several subjects reporting complete remission of pain, disability, or depression at the end of the study. Symptomatic improvement was seen in the majority of subjects after only one week of neurostimulation. For pain-related cytokines, results were mixed at the end of week two, with some showing improvement and others worsening. However, at the end of week eight, there was a clear improvement in the average levels of pain-related cytokines.
While lacking statistical significance, this study's improvement in subjective and objective pain-related measures was clinically significant. A definitive majority of subjects saw at least modest improvement in one or more measured domains, with several reporting a 100% reduction in pain, depression, or disability. These results are encouraging, especially for a minimally invasive treatment with no adverse events reported in any subjects aside from one subject who quit the study due to ear discomfort from the implanted electrodes during sleep.
Improved Cytokine Levels and Symptom Severity
A correlation between improved symptoms and decreased biomarkers of inflammation is expected, given what is known about the effects of cytokines in the body. Only two blood samples were collected from subjects during treatment. While this minimizes the risk of adverse events, it may limit the analysis of potentially illuminating alterations to the cytokine profile throughout treatment. However, a much larger sample size would be needed to detect statistically significant changes in pain-related biomarkers since cytokine levels and subjective assessments showed high variability within the study population. The results of this study both support and are supported by the results seen by numerous other researchers4, 5, 8, 9, 10, 49.
Clinical Significance
The most important outcome of this study is the impact of symptomatic improvement on the quality of life. With only 33 subjects in the study, two patients experienced complete remission of their chronic pain and 10 patients in total experienced reduced low-level pain to zero throughout the study. All subjects experienced at least a low level of chronic pain at baseline, meaning they had no pain-free days in the week prior to treatment. Such improvement is remarkable and potentially life-changing, especially in individuals who have suffered from chronic pain for years without finding relief. The reduction in opioid medication use is also important. There was an average 42% reduction in opioid use, with three subjects discontinuing opioid pain medication entirely. Any reduction in chronic opioid use is potentially significant on both an individual and societal level, given the high potential for dependence, abuse, and hyperalgesia, as well as their dubious effectiveness for treating chronic pain52, 53, 54, 55, 56, 57. Wider use of pVNS therapy can potentially improve the lives of people with chronic pain and other conditions ranging from metabolic syndrome to inflammatory arthritis, as well as unusual and difficult-to-treat conditions like fibromyalgia and ME/CFS.
Study Limitations
The primary limitation of this study is the small sample size (n=33). For all reported measures variability within the sample group was high, resulting in a high standard error. Given the small sample size and high SEM, it is unclear how well the study population represents a broader population with chronic pain. However, the exclusion criteria for this study were intentionally somewhat broad to diminish any confounding effects from serious medical conditions which can potentially affect the subjective and objective measures used. Many complex medical conditions are associated with chronic pain, but they can have vastly different overall pathologies.
Further studies with larger population samples are needed to achieve statistical significance with these measures. Additionally, longer term surveillance of patient outcomes may prove useful to reveal how long these symptomatic and physiologic changes persist after treatment. A larger sample could also facilitate the analysis of multiple factors that may influence treatment response, such as age, sex, site of pain, and underlying injuries or illnesses. A large-scale study with a large number of participants with various central sensitivity syndromes (including fibromyalgia, ME/CFS, and other conditions) could prove to be highly effective at uncovering how pVNS specifically affects central sensitivity as it relates to chronic pain.
Summary
Non-invasive Periauricular vagus nerve stimulation using a Stivax or similar device can be an effective treatment for chronic pain, even in difficult-to-treat patients. pVNS is safe, effective, and cost-effective compared to long-term pharmacotherapy or surgical interventions for chronic pain. It carries much less risk of adverse side effects than those therapies. pVNS should be considered as an early intervention for chronic pain in eligible patients to treat not only pain but also underlying autonomic dysfunction.
References
- 1.Dahlhamer J, Lucas J, Zelaya C, Nahin R, Mackey S. (2018) . Prevalence of Chronic Pain and High-Impact Chronic Pain Among Adults—United States, 2016.Morbidity and Mortality Weekly Report,67(36) 1001-1006.
- 2.M A Hadi, G A McHugh, S J Closs. (2019) . Impact of Chronic Pain on Patients’ Quality of Life: A Comparative Mixed-Methods Study.Journal of Patient Experience,6(2) 133-141.
- 3.Curatolo M, Arendt-Nielsen L, Petersen-Felix S. (2006) . Central Hypersensitivity in Chronic Pain: Mechanisms and Clinical Implications.Physical Medicine and Rehabilitation Clinics of North America,17(2) 287-302.
- 4.C R Brown. (2015) Peri-Auricular Percutaneous Neurovascular Stimulation as a Non-Narcotic Treatment Alternative for Acute and. Chronic Pain.Clinical Medicine and Diagnostics,5(4) 70-79.
- 5.Chakravarthy K, Chaudhry H, Williams K, P J Christo. (2015) . Review of the Uses of Vagal Nerve Stimulation in Chronic Pain Management.Current Pain and Headache Reports,19(12) 54, 10-1007.
- 6.D E Chou, G J, C H, M S Yugrakh. (2017) External Trigeminal Nerve Stimulation for the Acute Treatment of Migraine:. Open-Label Trial on Safety and Efficacy.Neuromodulation: Technology at the Neural Interface,20(7) 678-683.
- 7.J L Hawkins, L E Cornelison, B A, P L Durham. (2017) Vagus Nerve Stimulation Inhibits Trigeminal Nociception in a Rodent Model of Episodic Migraine.Pain Reports,2(6). 628-10.
- 8.P B Lee, Horazeck C, F S Nahm, B K Huh. (2015) Peripheral Nerve Stimulation for the Treatment of Chronic Intractable Headaches: Long-term Efficacy and Safety Study.Pain Physician,18(5). 505-516.
- 9.Li S, Sun C, Rong P, Zhai X, Zhang J. (2018) . Auricular Vagus Nerve Stimulation Enhances Central Serotonergic Function and Inhibits Diabetic Neuropathy Development in Zucker Fatty Rats.Molecular Pain,14 1744806918787368-10.
- 10.R L, S P Cohen, C A Gilmore, J M North, Kapural L. (2014) . , Treatment of Post-Amputation Pain with Peripheral Nerve Stimulation.Neuromodulation: Journal of the International Neuromodulation 17(2), 10-1111.
- 11.Yuan H, S D. (2017) Vagus Nerve Stimulation and Headache.Headache:. The Journal of Head and Face Pain,57(S1) 29-33.
- 12.A M, Veldhuijzen van Zanten, S J J C, Lip G Y H, Paton J F R et al. (2017) . Cardiovascular Autonomic Regulation, Inflammation and Pain in Rheumatoid Arthritis.Autonomic Neuroscience,208 137-145.
- 13.Hughes D. (2013) . 6).Hormone Levels Can Validate Presence of Pain and Affect Treatment. MPR. https://www.empr.com/home/mpr-first-report/painweek-2013/hormone-levels-can-validate-presence-of-pain-and-affect-treatment/ .
- 14.Zhang J-M, An J. (2007) . Cytokines, Inflammation, and Pain.International Anesthesiology Clinics,45(2) 27-37.
- 15.Bäckryd E, Ghafouri B, Larsson B, Gerdle B. (2016) Plasma Pro-Inflammatory Markers in Chronic Neuropathic Pain: A Multivariate. , Comparative, Cross-Sectional Pilot Study.Scandinavian Journal of Pain,10 1, 10-1016.
- 16.A D Cook, A D Christensen, Tewari D, S B McMahon, J A Hamilton. (2018) . Immune Cytokines and Their Receptors in Inflammatory Pain.Trends in Immunology,39(3) 240-255.
- 17.J D Clark, V L Tawfik, Tajerian M, W S Kingery. (2018) . Autoinflammatory and Autoimmune Contributions to Complex Regional Pain Syndrome.Molecular Pain,14 1744806918799127-10.
- 18.A N Khan, H E Jacobsen, Khan J, C G Filippi, Levine M. (2017) Inflammatory Biomarkers of Low Back Pain and Disc Degeneration: A Review.Annals of the New York Academy of Sciences,1410(1). 68-84.
- 19.Latremoliere A, C J Woolf. (2009) Central Sensitization: A Generator of Pain Hypersensitivity by. , Central Neural Plasticity.The Journal ofPain Journal of the American Pain 10(9), 895-926.
- 20.Meeus M, Nijs J. (2007) Central Sensitization: A Biopsychosocial Explanation for Chronic Widespread Pain. in Patients with Fibromyalgia and Chronic Fatigue Syndrome.Clinical Rheumatology,26(4) 465-473.
- 21.Oliveira C M B de, R K Sakata, A M Issy, L R Gerola, Salomão R. (2011) . , Cytokines and Pain.Brazilian Journal of Anesthesiology,61(2) 255, 10-1016.
- 22.Phillips K, D J Clauw. (2011) Central Pain Mechanisms. in Chronic Pain States – Maybe It Is All in Their Head.Best Practice & Research. Clinical Rheumatology,25(2) 141-154.
- 23.Ramesh G, A G MacLean, M T Philipp. (2013) . Cytokines and Chemokines at the Crossroads of Neuroinflammation, Neurodegeneration, and Neuropathic Pain.Mediators of Inflammation,2013 480739-10.
- 24.Ren K, Dubner R. (2010) . Interactions Between the Immune and Nervous Systems in Pain.Nature Medicine,16(11) 1267-1276.
- 25.Sturgill J, McGee E, Menzies V. (2014) . Unique Cytokine Signature in the Plasma of Patients with Fibromyalgia.Journal of Immunology Research,2014 938576-10.
- 26.Zhang C, Ward J, J R Dauch, R E Tanzi, H T Cheng. (2018) . Cytokine-Mediated Inflammation Mediates Painful Neuropathy from Metabolic Syndrome.PLOS ONE,13(2) 0192333-10.
- 27.Zhou Y-Q, Liu Z, Liu Z-H, Chen S-P, Li M. (2016) . , Interleukin-6: An Emerging Regulator of Pathological Pain.Journal of Neuroinflammation,13(1) 141, 10-1186.
- 28.Salaffi F, Giacobazzi G, M Di Carlo. (2018) . Chronic Pain in Inflammatory Arthritis: Mechanisms, Metrology, and Emerging Targets-A Focus on the JAK-STAT Pathway.Pain Research & Management,2018 8564215-10.
- 29.Capossela S, Pavlicek D, Bertolo A, Landmann G, J V Stoyanov. (2018) Unexpectedly Decreased Plasma Cytokines. in Patients with Chronic Back Pain.Journal of Pain Research,11 1191-1198.
- 30.R E Miller, R J Miller, Malfait A-M. (2014) . Osteoarthritis Joint Pain: The Cytokine Connection.Cytokine,70(2) 185-193.
- 31.Perini F, D’Andrea G, Galloni E, Pignatelli F, Billo G. (2005) . Plasma Cytokine Levels in Migraineurs and Controls.Headache,45(7) 926-931.
- 32.Sun J-M, Sun L-Z, Liu J, Su B, Shi L. (2013) . Serum Interleukin-15 Levels Are Associated with Severity of Pain in Patients with Knee Osteoarthritis.Disease Markers,35(3) 203-206.
- 33.Sun C, Zhang J, Chen L, Liu T, Xu G. (2017) . IL-17 Contributed to the Neuropathic Pain Following Peripheral Nerve Injury by Promoting Astrocyte Proliferation and Secretion of Proinflammatory Cytokines.Molecular Medicine Reports,15(1) 89-96.
- 34.Tsilioni I, I J Russell, J M Stewart, R M Gleason, T C. (2016) . Neuropeptides CRH, SP, HK-1, and Inflammatory Cytokines IL-6 and TNF Are Increased in Serum of Patients with Fibromyalgia Syndrome, Implicating Mast Cells.The Journal of Pharmacology and Experimental Therapeutics,356(3) 664-672.
- 35.Yuan F-L, Hu W, Lu W-G, Li X, Li J-P. (2011) . Targeting Interleukin-21 in Rheumatoid Arthritis.Molecular Biology Reports,38(3) 1717, 10-1007.
- 36.Herrera K. (2012) 4).Chronic Pain and Its Link to Hormones. Women’s International Pharmacy. https://www.womensinternational.com/blog/chronic-pain-link-to-hormones/
- 37.Tennant F. (2016) Testing and Replacement in Pain Patients Made Simple. https://www.practicalpainmanagement.com/treatments/hormone-therapy/hormone-testing-replacement-pain-patients-made-simple
- 38.Vincent K, C J Stagg, C E Warnaby, Moore J, Kennedy S et al. (2018) Luteal Analgesia”: Progesterone Dissociates Pain Intensity and Unpleasantness by Influencing Emotion Regulation Networks.Frontiers in Endocrinology,9. https://www.frontiersin.org/articles/10.3389/fendo.2018.00413
- 39.Dellwo A.. (2022).Role of GABA and Glutamate in Fibromyalgia and ME/CFS. Verywell Health. https://www.verywellhealth.com/gaba-glutamate-fibromyalgia-chronic-fatigue-716010 .
- 40.Burska A, Boissinot M, Ponchel F. (2014) . Cytokines as Biomarkers in Rheumatoid Arthritis.Mediators of Inflammation,2014 545493-10.
- 41.Chamessian A, Ven T Van de, Buchheit T, Hsia H-L, McDuffie M. (2017) . Differential Expression of Systemic Inflammatory Mediators in Amputees with Chronic Residual Limb Pain.PAIN,158(1) 68-74.
- 42.Ren G, Lutz I, Railton P, J P Wiley, McAllister J. (2018) . Serum and Synovial Fluid Cytokine Profiling in Hip Osteoarthritis: Distinct from Knee Osteoarthritis and Correlated with Pain.BMC Musculoskeletal Disorders,19(1) 39, 10-1186.
- 43.Ernberg M, Christidis N, Ghafouri B, Bileviciute-Ljungar I, Löfgren M. (2018) . Plasma Cytokine Levels in Fibromyalgia and Their Response to 15 Weeks of Progressive Resistance Exercise or Relaxation Therapy.Mediators of Inflammation,2018 3985154-10.
- 44.Ji R-R, Xu Z-Z, Gao Y-J. (2014) . Emerging Targets in Neuroinflammation-Driven Chronic Pain.Nature Reviews Drug Discovery,13(7), Article 7. https://doi.org/10.1038/nrd4334 .
- 45.Rivat C, Sar C, Mechaly I, Leyris J-P, Diouloufet L. (2018) . Inhibition of Neuronal FLT3 Receptor Tyrosine Kinase Alleviates Peripheral Neuropathic Pain in Mice.Nature Communications,9(1), Article 1, 10-1038.
- 46.A K Walker, Kavelaars A, C J Heijnen, Dantzer R. (2014) . Neuroinflammation and Comorbidity of Pain and Depression.Pharmacological Reviews,66(1) 80-101.
- 47.M de Goeij, Eijk L T van, Vanelderen P, O H Wilder-Smith, K C Vissers. (2013) Systemic Inflammation Decreases Pain Threshold in Humans. in vivo.PloSOne,8(12), e84159. https://doi.org/10.1371/journal.pone.0084159
- 48.Lerman I, Davis B, Huang M, Huang C, Sorkin L. (2019) Noninvasive Vagus Nerve Stimulation Alters Neural Response and Physiological Autonomic Tone to Noxious Thermal Challenge.PLOS ONE,14(2). 0201212-10.
- 49.do Carmo Almeida, C T, dos Santos Figueiredo, W F, Filho Barbosa et al. (2018) . Effects of Transcutaneous Electrical Nerve Stimulation on Proinflammatory Cytokines: Systematic Review and Meta-Analysis.Mediators of Inflammation,2018 1094352-10.
- 50.Cao J, Lu K-H, T L Powley, Liu Z. (2017) . Vagal Nerve Stimulation Triggers Widespread Responses and Alters Large-Scale Functional Connectivity in the Rat Brain.PLOS ONE,12(12) 0189518-10.
- 51.Kaniusas E, Kampusch S, Tittgemeyer M, Panetsos F, R F Gines. (2019) . Current Directions in the Auricular Vagus Nerve Stimulation I – A Physiological Perspective.Frontiers in Neuroscience,13. https://www.frontiersin.org/articles/10.3389/fnins.2019.00854 .
- 52.Baldini A, M Von Korff, Lin E H B. (2012) A Review of Potential Adverse Effects of Long-Term Opioid Therapy: A Practitioner’s Guide.The Primary Care Companion to CNS Disorders,14(3). 11-01326.
- 53.J C Ballantyne, K S LaForge. (2007) Opioid Dependence and Addiction During Opioid Treatment of Chronic Pain.PAIN,129(3). 235-255.
- 54.J C Ballantyne, N S Shin. (2008) Efficacy of Opioids for Chronic Pain:. , A Review of the Evidence.The Clinical Journal of 24(6), 469-478.
- 55.Chou R, J A Turner, E B Devine, R N Hansen, S D. (2015) The Effectiveness and Risks of Long-Term Opioid Therapy for Chronic Pain: A Systematic Review for a National Institutes of Health Pathways to Prevention Workshop.Annals of Internal Medicine,162(4). 276-286.
Cited by (2)
This article has been cited by 2 scholarly works according to:
Citing Articles:
Expert Review of Medical Devices (2025) Crossref
Expert Review of Medical Devices (2025) OpenAlex
John J. Finneran IV, B. Ilfeld - Expert Review of Medical Devices (2025) Semantic Scholar