Mucormycosis: A Surge in Mucorales Fungal Infection in Post – Covid Patients in Indian States and Insight into Known and Unknown Factors
Abstract
An overview of mucormycosis surge among post‑COVID patients in India. It reviews risk factors, clinical features, diagnostics, and urgent surgical and antifungal management, alongside gaps in evidence.
Author Contributions
Academic Editor: ANUBHA BAJAJ, Consultant Histopathology, A.B. Diagnostics, New Delhi, India.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2021 S.G.Borkar
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Mucormycosis (also known as phycomycosis or zygomycosis) is a rare opportunistic and invasive fungal infection in human caused by fungi belonging to the fungal order Mucorales and the family Mucoraceae 1, 2. It was first described by Paultauf in 1885 3. It represents the third most common angioinvasive fungal infection followed by candidiasis and aspergillosis. The genus most commonly associated with human infections causing mucormycosis are Rhizopus followed by Mucor, Cunninghamella, Actinomucor, Apophysomyces, Cokeromyces, Lichtheimia, Mycotypha, Rhizomucor, Saksenaea, Syncephalastrum, and Thamnostylum.4.
It usually affects the immunocompromised individuals and is rarely seen in apparently healthy individuals 5. In the compromised host, mucormycosis infection results from altered immunity in which rapid proliferation and invasion of fungal organisms ensue in deeper tissues. The mucormycosis can be acute and chronic. The classical triad is characterized by paranasal sinusitis, ophthalmoplegia with blindness, and unilateral proptosis with cellulitis. Facial or eye pain and necrotic wound of the palate of the nasal mucosa may occur. Black necrotic eschar in the nasal cornets is a characteristic sign 6.
This infection occurs in approximately 50% of the cases in individuals with Diabetes mellitus due to the greater availability of glucose to the pathogen that causes mucormycosis, the decrease in serum inhibitory activity against the Rhizopus in lower pH, and the increased expression of some host receptors that mediate the invasion and damage to human epithelial cells by Rhizopus or black fungus. Mucormycosis is also a rare presentation for patients in the emergency department (ED), primarily affecting immunocompromised patients.
The mucormycosis fungus is an allergenic form of mold (fungi) that usually grows fast and in thick patches. It is often white or greyish in color. It most oftenly grows near air conditioning, HVAC systems and ducting, due to moisture from condensation. Old damp carpets can also harbour mucor spores for invasion. These molds live throughout the environment particularly in soil and in association with decaying organic matter, such as leaves, compost piles, and animal dung. They are more common in soil than in air, and in summer and fall than in winter or spring.
Infection of mucormycosis is caused by asexual spore of the mucorales fungus. The tiny spores become airborne and settle on the oral and nasal mucosa of humans. In majority of immunologically competent hosts, these spores will be limited by a phagocytic response. If this response fails, germination will follow and hyphae will develop. As polymorphonuclear leukocytes are less effective in removing hyphae, in immunocompromised individuals, the infection becomes established in these cases. It further progresses as the hyphae begin to invade arteries, wherein they propagate within the vessel walls and lumens causing thrombosis, ischemia, and infarction with dry gangrene of the affected tissues. Hematogenous spread to other organs (lung, brain, and so on) can occur and results in sepsis 7 . Mucormycosis of the oral cavity can be of two different origins. One is from disseminated infection where the gateway of entry is inhalation (through the nose) and the other is through direct wound contamination with dissemination to other viscera as a common complication. When it arises from nose and PNS, the infection may cause palatal ulceration leading to necrosis and the affected area appears black in preponderance of the cases. When the infection spreads from direct wound contamination, the clinical findings may appear anywhere in the oral cavity, including the mandible. A significant difference between infection involving the maxilla and mandible is cavernous sinus thrombosis, a serious complication of maxillary infections.8
Incidence and Frequencies of Mucormycosis
Until recently, the frequencies of mucormycosis was very less. Population based incidence estimates for mucormycosisin the San Francisco Bay Area during 1992–1993 suggested a yearly rate of 1.7 cases per 1 million population. Bellazreg et.al 9 report five cases of confirmed mucormycosis in patients hospitalized in the Infectious Diseases department of Sousse, Tunisia, between 2000 and 2013. They reported the locations of mucormycosis in the patients as rhinocerebral, rhino-orbital, auricular, pulmonary and cutaneous.
The Mucorales isolated were Rhizopus arrhizus in 3 cases and Lichteimia corymbifera in 2 cases. All patients were treated with amphotericin B and 2 patients had, in addition, surgical debridement. Two patients died and 2 kept peripheral facial paralysis.
Mucormycosis has poor prognosis with a mortality rate of 17–51%. In Tunisian studies, the mortality rate was 65% https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4484545/ (9). Mortality is higher in case of diagnostic delay of more than five days and monocytopenia in patients with active malignant blood diseases. Surgical treatment associated with antifungals improves prognosis.
A surge in the cases of mucormycosis in post covid patients were observed in the second wave of covid infection in India where the mucormycosis cases increased to 150 % in 3 weeks (May 25th to June 20th 2021) upto 31,216 patients with 2109 deaths ( i.e 6.7% of deaths). The fungal disease was commonly being observed in patients who were given steroids for a long time, who have been hospitalised for a long time, were on oxygen support or ventilator, faced poor hospital hygiene or those who have been taking medication for other illnesses such as diabetes. The Covid medications can leave the body weak and low on immunity. These can also escalate the blood sugar levels in both diabetics and non-diabetic Covid-19 patients and this high blood sugar favors the invasion of the mucormycosis fungus.
The most common mucormycosis fungal genera are Rhizopus (47%) and Mucor (18%). Lichteimia is responsible for 5% of the cases https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4484545/ (4). In a previous Tunisian study, Rhizopus had been isolated in 70% of patient while Lichteimia was isolated in 60 % patients 9.
Updates on the Taxonomy of Mucorales with an Emphasis on Clinically Important Taxa
According to the revised taxonomy 10 the fungi causing mucormycosis are classified in the (new) phylum Glomeromycota (earlier zygomycota), class Glomeromycetes (earlier Zygomycetes), subphylum Mucoromycotina, order Mucorales. The order Mucorales has now been assigned to the phylum Mucoromycota and is comprised of 261 species in 55 genera. Of these accepted species, 38 have been reported to cause infections in humans, as a clinical entity known as mucormycosis. The Mucorales which cause the mucormycosis in human belong to the genus Rhzipus, Mucor, Lichtheimia formerly Absidia), Cunninghamella, Rhizomucor, Apophysomyces, Saksenaea, Actinomucor, Cokeromyces, Mycotypha,, Syncephalastrum. andThamnostylun (Table 1).
Table 1. Genera of the Mucorales, their assignment to families according Hoffmann et al. 11, the number of species included in these genera, and their clinical importance.| Genus | Family (According to Hoffmann et.al,2013) | Accepted species | Clinically relavent species |
| Actinomucor | Mucoraceae | 1 | 1 |
| Apophysomyces | Saksenaeaceae | 6 | 4 |
| Cokeromyces | Mucoraceae | 1 | 1 |
| Cunninghamella | Cunninghamellaceae | 14 | 4 |
| Lichtheimia | Lichtheimiaceae | 6 | 3 |
| Mucor | Mucoraceae | 76 | 13 |
| Mycotypha | Mycotyphaceae | 3 | 1 |
| Rhizomucor | Lichtheimiaceae | 2 | 2 |
| Rhizopus | Rhizopodaceae | 10 | 4 |
| Saksenaea | Saksenaeaceae | 5 | 4 |
| Syncephalastrum | Syncephalastraceae | 2 | 1 |
| Thamnostylum | Lichtheimiaceae | 4 | 1 |
| Mucorales total | 130 | 39 |
Out of 260 species of different Genera of Mucorales, 39 species from 12 Genera are clinically relevant to cause Mucoromycosis. The sequences for these genus are available 12. Approximately one-third cases of mucormycosis are caused by Mucor spp. Rhizopus spp , Apophysomyces spp, Cunninghamella spp, and Saksenaea spp occupy the second position while Lichtheimia spp. are the third most common mucoralean fungi shown to be responsible for development of mucormycosis. Other less common genera includes Rhizomucor, Mycotypha, Syncephalastrum, Thamnostylum and others, including their species which are being isolated and responsible for mucormycosis infection (table 2).
Table 2. Clinically relevant species of the Mucorales| Species | Previous Names/ Important Synonym | Reference | |
| 1 | Actinomucor elegans | Mahmud et al. 2011 13 | |
| 2 | Apophysomyces elegans * | Alvarez et al. 2010 14 | |
| Apophysomyces mexicanus | Bonifaz et al. 2014 15 | ||
| Apophysomyces ossiformis | Álvarez et al. 2010 14 | ||
| Apophysomyces trapeziformis | Álvarez et al. 2010 14 | ||
| Apophysomyces variabilis | Álvarez et al. 2010 14 | ||
| 3 | Cokeromyces recurvatus | Ryan et al. 2011 16 | |
| 4 | Cunninghamella bertholletiae | Navanukroh et al. 2014 17 | |
| Cunninghamella blakesleeana | García Rodríguez et al. 2012 18 | ||
| Cunninghamella echinulata | Álvarez et al. 2009 19 | ||
| Cunninghamella elegans | Yu et al. 2015 20 | ||
| 5 | Lichtheimia corymbifera | Absidia corymbifera, Mycocladus corymbifer | Alastruey-Izquierdo et al. 2010 21 |
| Lichtheimia ornata | Absidia ornate | Alastruey-Izquierdo et al. 2010 21 | |
| Lichtheimia ramose | Absidia ramosa ,Mycocladus ramosus | Alastruey-Izquierdo et al. 2010 21 | |
| 6 | Mucor amphibiorum | Walther et al. 2013 12 | |
| Mucor ardhlaengiktus | Mucor ellipsoideus | Álvarez et al. 2011 22 | |
| Mucor circinelloides | Mucor circinelloides f. circinelloides,Rhizomucor regularior, Rhizomucor variabilis var. regularior | Wagner et al. 2019 23 | |
| Mucor griseocyanus | Mucor circinelloides f. griseocyanus | Wagner et al. 2019 23 | |
| Mucor indicus | de Repentigny et al. 2008 24 | ||
| Mucor irregularis | Rhizomucor variabilis | Lu et al. 2013 25 | |
| Mucor janssenii | Mucor circinelloides f. janssenii | Walther et al. 2013 12 | |
| Mucor lusitanicus | Mucor circinelloides f. lusitanicus | Álvarez et al. 2011 22 | |
| Mucor plumbeus | Walther et al. 2013 12 | ||
| Mucor racemosus | Walther et al. 2013 12 | ||
| Mucor ramosissimus | Hesseltine & Ellis 1964 26 | ||
| Mucor variicolumellatus | Álvarez et al. 2011 (as M. fragilis) 22 | ||
| Mucor velutinosus | Álvarez et al. 2011 22 | ||
| 7 | Mycotypha microspora | Trachuk et.al.2018 27 | |
| 8 | Rhizomucor miehei | Walther et al. 2013 12 | |
| Rhizomucor pusillus | Iwen et al. 2005 28 | ||
| 9 | Rhizopus arrhizus (incl. var. delemar) | Rhizopus oryzae | Dolatabadi et al. 2014 29 |
| Rhizopus homothallicus | Chakrabarti et al. 2010 30 | ||
| Rhizopus microspores | Rhizopus microsporus var. azygosporus,var. chinensis, var. oligosporus, var. rhizopodiformis, var. tuberosus | Dolatabadi et al. 2013 31 | |
| Rhizopus schipperae | Weizmann et al. 1996 32 | ||
| 10 | Saksenaea erythrospora | Weizmann et al. 1996 32 | |
| Saksenaea erythrospora | Álvarez et al. 2010 33 | ||
| Saksenaea loutrophoriformis | Crous et al. 2017 34 | ||
| Saksenaea trapezispora | Álvarez et al. 2010 33 | ||
| Saksenaea vasiformis | Álvarez et al. 2010 33 | ||
| 11 | Syncephalastrum racemosum | Schlebusch et al. 2005 35 | |
| 12 | Thamnostylum lucknowense | Xess et al. 2012 36 |
The fungi classified in the order Mucorales are mainly saprophytic, rapidly growing, and able to grow at temperatures higher than 37°C (except for Mucor spp.). These molds are supposedly ubiquitous in nature and widely found on organic substrates, including bread, decaying fruits, vegetable matter, crop debris, soil between growing seasons, compost piles, and animal excreta. These colonize all kinds of wet, organic materials and represent a permanent part of the human environment, as pioneers on all kinds of wet organic materials and causative agents of rot and spoilage.
Interestingly, some of the Mucoralean fungi like Rhizopus oryzae have also been used for centuries to ferment traditional Asian and African food and are utilized for the production of several varieties of European cheese 37 and soy products. As producers of a broad spectrum of enzymes, Mucorales are used in biotechnology for the biotransformation of several medically and pharmaceutically important compounds 38 such as steroids and terpenoids.
However, it is not clear whether species e.g. Rhizopus oryzae which are used in chees product or enzyme production are the same which cause mucormycosis in human or there are subspecies/variant of the species which are pathogenic to human.
The order includes numerous thermotolerant or thermophilic species that are able to grow at human body temperature and can cause life-threatening infections of mucormycosis, mostly in patients with impaired immunity.
Medically important species that have been affected by recent changes include Lichtheimia corymbifera, Mucor circinelloides, and Rhizopus microsporus. The species concept of Rhizopus arrhizus (syn. R. oryzae) is still a matter of debate. Currently, species identification of the Mucorales is best performed by sequencing of the internal transcribed spacer (ITS) region. Due to molecular phylogenetic studies, the taxonomy of the order has changed widely during the last years. Characteristics such as homothallism, the shape of the suspensors, or the formation of sporangiola are shown to be not taxonomically relevant.
Our understanding of Mucorales biology has severely suffered from a largely unresolved taxonomy. Even in the medical setting, these fungi are often not identified to the species level. However, significant progress in understanding Mucorales taxonomy has been made in recent years.
The mucorales have coenocytic or non- septed hyphae. Septae are usually formed to delimit reproductive structures or swollen parts. In tissue infection, they may produce septae but not at regular distances as in ascomycetous fungi. Mucorales produce their uni-celled asexual spores (sporangiospores) endogenously, i.e., inside specialized cells known as sporangia or sporangiola. The spore forming specialized cells are named according to their shape and the number of spores they contain. Sporangia are globose cells containing a high (uncountable) number of sporangiospores. Sporangiola are globose cells containing one to a countable number of sporangiospores while merosporangia are elongated cells containing one to a countable number of sporangiospores. The main characterizing feature of the order Mucorales used to be the columella—a sterile central vesicle inside the sporangium. The members of the Mucorales reproduce sexually by formation of zygospores. These are thick-walled, pigmented, and often ornamented zygotes that are formed by the fusion of two differentiated hyphal ends, including fusion of the cytoplasm and nuclei.
Mucormycoses are generally angioinvasive, have an acute course, and affect predominantly immunocompromised individuals, whereas entomophthoromycoses are usually subcutaneous, show a chronic course, and mostly affect immunocompetent individuals. In histopathologic sections, mucormycoses and entomophthoromycoses are very similar, showing broad, belt-like, non- or rarely septated hyphae. However, it has been suggested that both entities can be differentiated in hematoxylin–eosin stained sections, where only the hyphae of the Entomophthorales are surrounded by eosinophilic sleeves 39.
The first molecular phylogenies based on two phylogenetic markers (fragment of coding or non-coding DNA used in phylogenetic reconstructions) 40 resulted in the revision of the family concepts of the Mucorales due to polyphyly (descent from more than one ancestral group) of the sporangiola forming taxa previously united in the Thamnidiaceae. By using an extended set of species and three loci, Hoffmann et al. 11 further enhanced the family concept and recognized 13 families exclusive of the Umbelopsidaceae that now belong to a separate order 41. Molecular Taxonomy and Re-Evaluated Criteria for Genus and Species Delimitation Members of the Mucorales seem to have a dynamic organisation of their genomes. The genome of Rhizopus arrhizus (syn. R. oryzae), and also most likely those of Mucor circinelloides and Phycomyces blakesleeanus have undergone whole genome duplication 42, 43, while in Lichtheimia corymbifera only single genes were duplicated 44. several markers that are traditionally used in the phylogeny of higher fungi cannot be applied to Mucorales due to the presence of paralogous genes 45, 46. As a result, the number of studies based on at least two, unlinked markers that meet the criteria of phylogenetic species recognition by genealogical concordance is comparatively low 47. Appropriate markers for the opportunistic genera Apophysomyces, Lichtheimia, Mucor, and Rhizopus are limited. In Lichtheimia, all the established phylogenetic markers, such as the partial genes of actin, beta-tubulin, the first (rpbq1) and the second largest subunit (rpb2) of RNA polymerase II, and the translation elongation factor-1 alpha (tef) possess paralogs 21. In Mucor, the genes of actin, tef, beta-tubulin, and calmodulin are also not single-copy genes 48. In Rhizopus arrhizus, tef has paralogs, but the polymorphisms are restricted to the third position of the triplet codon so that tef could be used as marker if these positions were excluded from the analyses 29. Due to this lack of additional markers, most descriptions of new species are still based on a single locus, usually ITS sequence alone, or combined with LSU sequences 49 for which genealogical concordance phylogenetic species recognition cannot be applied. This practice can be acceptable in taxa with well-defined molecular taxonomy and species variabilities. However, in unrevised genera lacking molecular species concepts (e.g., Absidia), the description of new species needs to be based on a comprehensive molecular and phenotypic study of the sibling species as well. Some of the recent descriptions of new species and new genera 50 are only based on morphological features. Considering the morphological plasticity of the Mucorales shown by numerous reclassifications of species based on molecular data, it is recommended that new species descriptions should include molecular data. This is especially important because a subsequent molecular evaluation of species is not always possible, as several strain collections do not provide strains outside their country or overseas. When molecular phylogenies were available, it became apparent that phenotypic characteristics traditionally used in mucoralean taxonomy were not always taxonomically informative. In the past, the formation of sporangiola or a combination of sporangiola and sporangia has been used to define new genera 51. However, according to molecular phylogenies 12, sporangiola-forming species appear intermittantly among Mucor species. Likewise, in the Mucor circinelloides complex 51, only two out of 14 species form sporangiola. Thus, the formation of sporangiola could either be a plesiomorphic character in the Mucorales that is genetically fixed, but not revealed in all species, or one that has evolved several times in surprisingly short phylogenetic distances, and often in connection to dung as habitat. Distinctly curved (circinate) sporangiophores, combined with persistent walls of the sporangia, occur not only in the genus Circinella, but also in two different lineages of the genus Mucor 51.
In addition to morphological characters, the formation of zygospores in crosses of two strains has been traditionally used to define biological species boundaries 52. Crosses between different but closely related species were assumed to result in the formation of azygospores 53, or lead to the production of zygospores that differ in size, colour, ornamentation, or number from intraspecific zygospores 54. Azygospores resemble zygospores. They can be either formed on a single suspensor or on two suspensors, where the septum between the differentiated hyphal tips (gametangia) remains and no plasmogamy takes place 53. However, Wagner et al. 51 showed for the Mucor circinelloides relationship that the differences between intra and interspecific zygospores can be inconspicuous and only related to the size of the zygospores and the height of their ornamentation. Consequently, the differentiation between intra and interspecific zygospores might require elaborate studies, including numerous crosses. The simple presence of zygospores between two strains cannot be used as criterion of specificity.
The important and most predominant genus causing mucormycosis are (Figure 1)
Figure 1.Important genera of Mucorales causing Mucormycosis in human.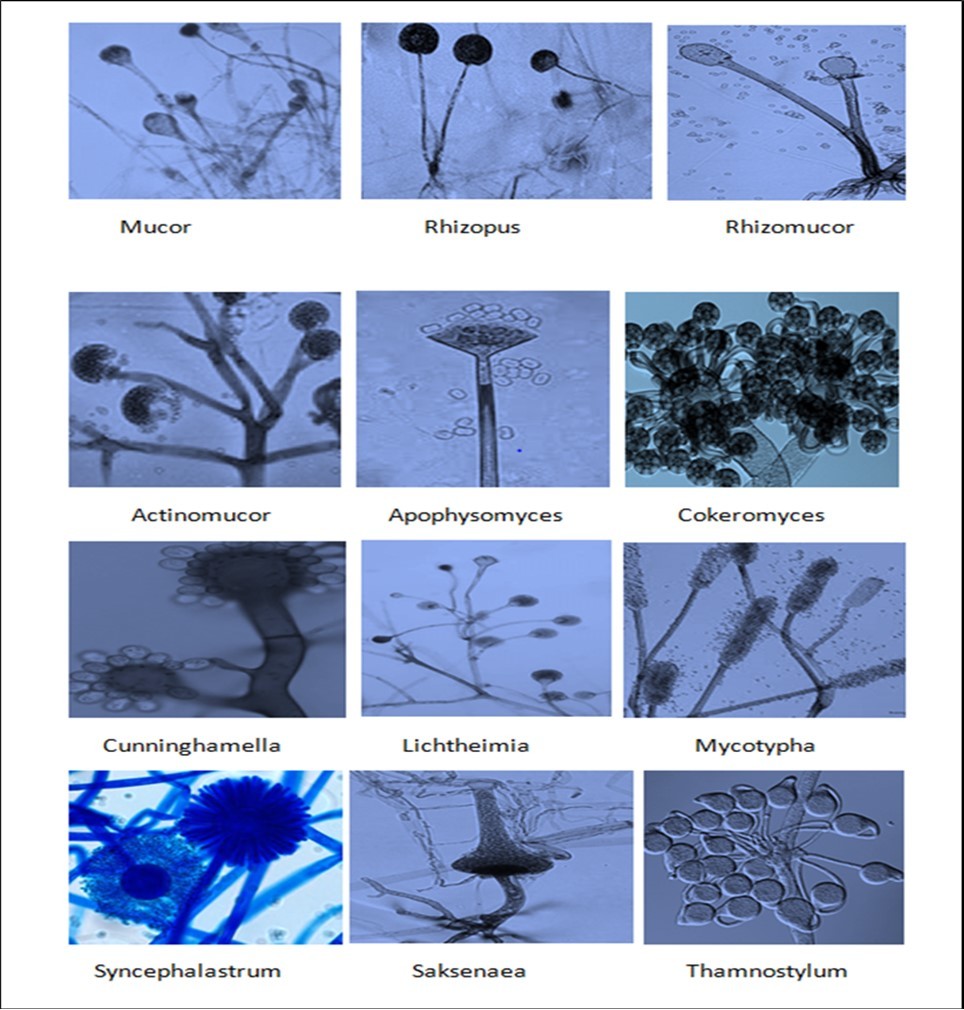
Mucor
Mucor belongs to the main genera causing mucormycoses 55. To date, 12 species of Mucor are known to be involved in infections 23. Members of the genus predominantly cause cutaneous infections but also disseminated and gastrointestinal manifestations have been reported 23. However, the clinical importance of the genus is still not clear because the causative agents of infections are only morphologically identified to the genus level in most cases 55 and might in fact represent the morphologically similar Rhizomucor or Lichtheimia species.
The genus Mucor is currently made up of 76 accepted species and is by far the largest genus in the Mucorales. Several molecular studies revealed the polyphyly of Mucor 12. Mucor was previously characterized by the formation of sporangia and equally shaped suspensors, as well as the absence of apophysis, rhizoids (root-like hyphae), and sporangiola. Recently, it was shown that Mucor species are able to form rhizoids 12. This explains the misclassification of several Mucor species in the genus Rhizomucor due to the formation of rhizoids in culture. Sequence analyses also revealed that taxa with an apophysis, such as Mucor durus (syn. Circinella rigida), and with sporangiola, such as Mucor ctenidius (syn. Backusella ctenidia), belong to the genus Mucor 12 and all Zygorhynchus species were transferred to Mucor 12.
On the basis of morphology and mating experiments, the most clinically relevant Mucor species, M. circinelloides, used to be divided in four formae: f. circinelloides, f. griseocyanus, f. janssenii, and f. lusitanicus 56. Through the use of multi-locus analyses of five different markers, morphological traits and mating experiments, a recent study showed that the former formae represent, in fact, one or two separate species 21. Mucor velutinosus 33 was supported as a sibling species of M. janssenii and five new species were described. Strains that were, in a previous study, misleadingly assigned to M. fragilis, are now assigned to M. variicolumellatus 12 because M. fragilis is related to Mucor hiemalis, according to the original description and illustration 23. Mucor ellipsoideus 22 was synonymised with M. ardhlaengiktus due to their similar ITS sequences 12.
Based on case reports or strain source information 12, the Mucor species potentially involved in human infections are M. amphibiorum, M. ardhlaengiktus (syn. M. ellipsoideus), M. circinelloides, M. griseocyanus, M. indicus, M. irregularis (syn. Rhizomucor variabilis), M. janssenii, M. lusitanicus, M. racemosus, M. ramosissimus, M. variicolumellatus, and M. velutinosus. Mucor amphibiorum is known as a pathogen of amphibians, but a single human isolate has been reported 12. The involvement of M. racemosus in invasive human infections is doubtful because it is morphologically similar to M. circinelloides, it does not grow at temperatures above 34 ◦C and there has been no DNA-based report of the species from invasive infections 21. The only molecularly-verified reports of this species refer to superficial infections 21. Mucor griseocyanus has not been described from invasive human infections, but was isolated from nails 21. Reports on M. hiemalis likely refer to its sibling species, M. irregularis (syn. Rhizomucor variabilis) 57 as M. hiemalis is not able to grow at temperatures higher than 30 ◦C 58. All case reports of this species are based solely on morphology and were published before Mucor irregularis was described 22. In agreement with the typical clinical picture of M. irregularis infections, the majority of these reports describe chronic cutaneous infections of healthy individuals 57.
Genus: Rhizopus
Rhizopus can be encountered in several different niches from the warm and moist Southern Asia to the colder Northern Europe. The most described member of the Rhizopus genus is Rhizopus oryzae. This species has not only raised significant research interest, but also is used in full-scale industrial applications. One long-time use of these fungi is in tempe, a dish from soybeans fermented by R. oryzae or the related Rhizopus microsporus, which has been indigenous to Southeast Asia since 500 years ago, and used as a common meal by millions of people. In addition, several other foodstuffs and beverages also use Rhizopus species in their processing. Besides this the Rhizopus is used in the production of organic acids, mainly l-lactic acid and fumaric acid and the enzymes amylases, pectinases, cellulases, proteases, and phytases . Rhizopus has also been investigated for the treatment of industrial wastewater from organic sources and production of animal feed 59.
The Rhizopus genus is not known only for its positive characteristics. It is also a known cause of food spoilage, particularly of crops, which causes huge economic losses during storage and transportation. Rhizopus stolonifer is a prime example behind Rhizopus-soft rot disease. This species is even able to spoil food after preventive treatment, since its enzymes are remarkably heat stable and are active even after 40 min at 100 °C.
The genus Rhizopus is characterized by the formation of unbranched, pigmented sporangiophores that arise singly or in whorls and that bear sporangia with an apophysis. Rhizoids are formed opposed to the sporangiophores 47. Based on morphological and molecular data 61, the following Rhizopus species: R. americanus, R. caespitosus, R. homothallicus, R. lyococcus, R. microsporus, R. schipperae, R. koreanus, R. arrhizus (with the varieties arrhizus (syn. R. oryzae) and var. delemar), R. sexualis, and R. stolonifer have been accepted. The recently described R. koreanus is a sibling species of R. stolonifer that differs from the latter only slightly by the size of the columellae 47. The species was already detected as a cryptic sibling species of R. stolonifer based on ITS sequences 12 and probably also by amplified polymorphic DNA (RAPD) patterns 62.
The genus Rhizopus is by far the most important causative agent of mucormycoses worldwide and the main cause of rhino–orbital–cerebral infections. Pulmonary, cutaneous, and disseminated disease manifestations are also frequently reported. The majority of cases are caused by two species: most frequently by R. arrhizus (syn. R. oryaze), followed by R. microsporus 55. Rhizopus Arrhizus (Syn. R. Oryzae)
R. arrhizus was described by Fischer 63 three years before R. oryzae 64. In contrast to the description of R. oryzae, the description of R. arrhizus was short, without figures, and no type material was mentioned. As a consequence, the name R. oryzae was preferred by most authors. However, Ellis 65 designated an ex-neotype strain for R. arrhizus, thus legitimizing the name R. arrhizus. Consequently, the name R. arrhizus has priority. Because the ex-type strain of R. oryzae clusters with the ex-neotype strain of R. arrhizus, R. oryzae should be treated as synonym of R. arrhizus 29.
The variety arrhizus produces and accumulates lactic acid in the medium because it possess two slightly differing genes for lactate dehydrogenase (ldhA and ldhB), while the fumaric and malic acid producing var. delemar possess only ldhB 66. Based on morphology, Zheng et al. 67 recognized three varieties: var. arrhizus, var. delemar, and var. tonkinensis. However, var. tonkinensis was not supported by molecular phylogenetic analyses 29. According to Zheng et al. 67, the morphological differences between var. arrhizus and var. delemar were small and quantitative, including the predominant position of swellings in the sporangiophore, the main origin of the sporangiophores (aerial hyphae or stolons = runner hyphae, horizontally growing unbranched hyphae), slight differences in the shape of the columellae, and a larger maximum spore size of var. delemar. However, these morphological features are not fully consistent. Strain CBS 395.34 was morphologically identified as var. arrhizus 67, but molecular identification related it to var. delemar 29. Additionally, no differences in ecology, distribution, and pathogenicity could be detected between the two varieties. In multi-locus studies, var. arrhizus and var. delemar were well-recognized without evidence of recombination 68. Although zygospore formation is a rare event in both varieties, zygospores have been observed in crosses between the variety arrhizus and variety delemar 60.
Genome sequencing of R. arrhizus var. delemar revealed a dynamic organization of the genome and indicated an ancestral, whole-genome duplication 42. On the genome scale, 76 orthologous proteins could not resolve var. arrhizus and var. delemar 69. A whole-genome phylogeny based on 1,620,389 single-nucleotide polymorphisms (SNPs) from 13 isolates revealed three clades: one clade containing only var. arrhizus, one clade containing only var. delemar strains, and the third clade containing strains with mixed ancestry from both var. arrhizus and var. delemar. No differences in virulence were found between strains of these clades 69. Gryganskyi et al. 70 analysed 192 orthologous proteins from the genomes of 21 Rhizopus strains, including R. microsporus, R. arrhizus var. arrhizus and var. delemar, and R. stolonifer. They found two well-supported clades for both varieties and a phylogeny based on morphology and physiology that was congruent with the molecular phylogeny.
The morphological characters used for their non-molecular phylogeny are in conflict with previous studies particularly (1) the diameter of the sporangia: Gryganskyi et al. 70 found them to be 160–240 µm in var. arrhizus, and 250–300 µm in var. delemar, while Schipper 60 and Zheng et al. 67 described them as up to 176 µm in diameter for both varieties; (2) the shape of the columella: the collumellae of var. arrhizus as ellipsoidal and those of var. delemar as subglobose or conical, while Zheng et al. 67 found the columellae of R. arrhizus to be subglobose, hemiglobose, or roundish conical; and (3) the Gryganskyi study found the sporangiospores of var. arrhizus to be ridged but not striate, but Zheng et al. 67 found them to be striate as well.
Rhizopus Microspores
Three species related to R. microsporus that were distinguished only by the shape, the size, and the ornamentation of the sporangiospores were reduced in rank due to positive mating tests 60 and subsequently treated as varieties of R. microsporus, namely var. chinensis, var. oligosporus, and var. rhizopodiformis. Later, additional varieties, var. azygosporus 67 and var. tuberosus 71 were described. Typically, domesticated foodborne strains with reduced and irregular sporulation were assigned to the variety oligosporus, while clinical strains were mostly attributed to var. rhizopodiformis.
However, in sequence based approaches 61 these varieties were not detected. Phylogenetic analyses of three loci (its, act, tef) revealed recombination between two of the three subgroups within R. microsporus, with both groups containing all the morphologically-defined varieties 31. There was no correlation between the phylogenetically-defined subgroups and the varieties. Based on these results, the varieties of Rhizopus microsporus were reduced to synonyms 31. On the genome level, this species shows a large degree of molecular diversity 70.
Genus: Rhizomucor
Rhizomucor species cause about 5% of the mucormycoses worldwide 55; the percentage is reduced by the cases of Rhizomucor variabililis because this is now transferred to Mucor irregularis.
Rhizomucor species mainly cause pulmonary infections followed by disseminated, cuteanous, and rhino–orbital–cerebral manifestations 55. All mesophilic species of Rhizomucor (Rm), including Rm. chlamydosporus, Rm. endophyticus, Rm. regularior (syn. Rm. variabilis var. regularior), and Rm. variabilis were transferred to Mucor based on molecular data 12. In its current classification, Rhizomucor only harbours thermophilic species with maximum growth temperatures above 50◦C and minimum growth temperatures below 20◦C. These species, including Rm. miehei, Rm. pusillus, Rm. nainitalensis, and Rm. pakistanicus, possess subglobose to short ellipsoidal sporangiospores. There are no strains, sequences, or additional reports available for the latter two species and it cannot be excluded that they represent synonyms of Rm. pusillus or Rm. miehei.
Genus: Actinomucor
Actinomucor elegans is the representative species of the Actinomucor genus. This class of fungi is characterized by having rhizoids, stolons and short branches of sporophores under the apex sporangia.While morphologically similar to the Mucor family, the Actinomucor differs by having (1) stolons, and (2) rhizoids and sporophores deriving from the rhizoids. The Actinomucor share its similarity with the Rhizopus and Absidia of the same family by having stolons, but differ in the forms and types of columella and sporophores.
Only a few cases of Actinomucor infections causing mucormycosis have been reported 72. The genus Actinomucor consists only of a single species, Actinomucor elegans, for which three varieties are described: var. elegans, var. meitauzae, and var. kuwaitiensis. These varieties differ in shape, size, and ornamentation of the sporangiospores 73 and their growth on Czapek’s agar 74. However, the varieties are not detected in ITS-based phylogenetic analysis, suggesting that these differences might not be phylogenetically relevant 20.
The Actinomucor can be grown in between 18℃- 40℃, with the optimal temperature being 30℃. Growth is inhibited by strong light, and it is best cultured in the dark. Optimal pH range is between pH3.5 to 9, but it grows best at pH 7.0. It grows best in 72.6-98% relative humidity. Cultures best on shakers than stationary incubators.
Genus: Apophysomyces
Apophysomyces is widely distributed in soil and decaying vegetation as a common environmental contaminant. Itis found most commonly in tropical to subtropical regions 75.
The genus Apophysomyces currently contains a single species, Apophysomyces elegans. Apophysomyces elegans is a rare cause of human mucormycosis. The infection is usually acquired via traumatic implantations, such as accidental injuries, insect bites, surgery, and contamination of burn wounds. Invasive soft tissue infections develop on burns or wounds contaminated by soil. Unlike other mucormycosis, the affected host is usually otherwise immunocompetent. Apophysomyces elegans infections present most commonly as necrotizing fasciitis, osteomyelitis, and angioinvasion. Systemic and secondary renal and bladder infections have also been reported.
Apophysomyces elegans colonies are fluffy and cottony. The inoculated plate covers with profuse woolly mycelium in 2 to 3 days. Surface of the colony is white initially and turns to brownish gray or yellowish cream as the culture gets older. Reverse is white to pale yellow. Apophysomyces elegans is a thermotolerant fungus. It grows rapidly at 42°C. Apophysomyces elegans has broad (4-8 µm in diameter), branching hyphae. Sparse septations may occasionally be observed. Rhizoids arise either between the points where the sporangiophores originate or opposite to the sporangiophores. Sporangiophores are long (max. 530 µm), unbranched, and straight or curved. They arise from a foot cell-like hyphal segment and taper slightly towards the apex. There exists a subapical thicker portion which is darker and brownish in color. The apex of the sporangiophore widens to form a typical funnel or bell-shaped apophysis. The columella of Apophysomyces elegans looks like a half circle and sporangia are pyriform (pear-shaped). The sporangia are hyaline initially and become colored by maturation. Upon dissolution of the sporangium, a small collar remains at the base of the columella. Sporangiospores of Apophysomyces elegans are smooth-walled and oblong (3-4×5-8 µm in size). They may be pale brown in color. Zygospores are formed in existence of compatible mating strains.
Sporulation of Apophysomyces elegans is poor on routine cultivation media. It produces typical “cocktail glass” shaped sporangia on nutrient deficient media. Yeast extract water culture and incubation of the culture media at 35°C enhance sporulation very effectively. Apophysomyces elegans mostly resembles Absidia. However, its bell-shaped (but not conical) apophyses, existence of the foot-cell, rhizoids produced opposite to the sporangiophores upon cultivation on plain agar, the darker and thicker subapical segment, and inability to sporulate on routine culture media help in differentiation of Apophysomyceselegans .
In vitro susceptibility data reported so far are very limited 14, 15. MIC breakpoints for interpretation of in vitro susceptibility results have not been defined. Some of the isolates may yield relatively low amphotericin B MICs. Similar to the other members of the class Zygomycetes, amphotericin B appears as the sole antifungal drug which may be active against Apophysomyces elegans. Extensive surgical debridement and amphotericin B therapy may be efficacious in vivo.
Genus: Cokeromyces
The fungal colonies are Light gray on PDA plates. On YPD plates yeastlike colonies grew at 37°C. Microscopic observations of the cellufluor mounts prepared from portions of the fungal colony reveals branching, broad, generally aseptate hyphae, some of which terminate in spherical vesicles. Sporangia-like structures appears at the ends of long, curved stalks attached to each vesicle. The PDA slide cultures shows Hyaline, smooth-walled, broad sporangiophores, each terminating in a large, spherical vesicle in 10 days old culture incubated at 30°C. Arising from the vesicles are elongate, recurved stalks or pedicles, each bearing globose sporangiola, 10 to 12 um in diameter. In addition numerous spherical, dark brown-black zygospores, borne on suspensors from opposite hyphae, are evident in the slide cultures. Subcultures of the fungus did not grow on MYC, even after 14 days of incubation. At 37°C, yeastlike colonies in texture composed primarily of spherical, thick-walled, budding cells appears. Based on colony morphology and microscopic appearance, the fungus can be identified as Cokeromyces recurvatus 76.
Genus: Cunninghamella
Although infections of Cunninghamella are rare, Cunninghamella betholletiae is emerging as an opportunistic human pathogen, predominantly in immunocompromised people, leukemia patients, and people with uncontrolled diabetes. Cunninghamella bertholletiae infections are often highly invasive, and can be more difficult to treat with antifungal drugs than infections with other species of the Mucorales, making prompt and accurate recognition and diagnosis of mycoses caused by this fungus is an important medical concern.
C. bertholletiae is found globally, with increased prevalence in Mediterranean and subtropical climates than in temperate zones and can grow at higher temperatures. It typically grows as a saprotroph and is found in a wide variety of substrates, including soil, fruits, vegetables, nuts, crops, and human and animal waste. C. bertholletiae can also cause significant infections in agricultural crops. Hosts include plants in the genera Daucus, Gossypium and Tetragonia.
C. bertholletiae hyphae appear hyaline, but masses of fungi are darker in colour. Colonies initially appear white, and become grey and powdery when they sporulate. C. bertholletiae displays very rapid growth on Sabouraud's agar (up to 20mm per day), which differentiates it from members of the Ascomycota and Basidiomycota. However, culturing clinical materials infected by this species has been known to yield false negative results. This species has very wide (10-20 μm), aseptate or partially septate hyphae, which contributes to a high capacity for cytoplasmic streaming which allows rapid diffusion of nutrients from a local nutrient source, which causes high growth rates and rapid nutrient depletion in culture or on limited substrates. Like other members of the order Mucorales, C. bertholletiae is thermotolerant, with a maximum growth temperature of 45-50˚C.
Hyphae branch at right angles and may appear twisted. When growing in animal tissue, hyphae spread in all dimensions. C. bertholletiae produces spores in globose sporangia atop sporangiophores that are typically tall enough to be visible without a microscope. Sporangiophores vary in length, and branch laterally to form concentric circles of shorter branches. They lack the columella and apophysis present in sporangiophores of many other species of the Mucorales. Unlike other members of the Mucorales, Cunninghamella species produce only one spore in each sporangium. Sporangia form a halo around a central, round vesicle at the apex of a sporangiophore. Spores are round to oval in shape and rough, with small spines or wart-like bumps. The hyphae of C. bertholletiae may or may not produce rhizoids at the base of the sporangiophores. The sexual reproduction in C. bertholletiae is through the formation of zygospores. Specifically, in the case of C. bertholletiae, heterothallic mating occurs when hyphae of opposite mating types are stimulated by mutually-secreted pheromones to grow toward each other and differentiate into gametangia. When they meet, these gametangia fuse (plasmogamy) and form a multinucleate, dikaryotic zygosporangium flanked by suspensor cells derived from the contributing hyphae. Each zygosporangium produces one zygospore, which, after a dormant period of weeks to months, undergoes nuclear fusion (karyogamy) to produce a diploid nucleus. The diploid nucleus then undergoes meiosis and chromosomes recombine to produce recombinant progeny genomes. A germosporangium forms, containing haploid spores, which are released into the environment to initiate the growth of a new mycelium.
It can be transmitted between ecological niches via water and air. In majority of cases, human infection is through airborne spores, although infections of deep wounds and medical devices can also occur through water contamination. The risk factors for infection are similar for other mucormycoses, including diabetic ketoacidosis, and immunosuppression from chemotherapy, organ transplantation, and malnutrition. Leukemia is a particularly high risk factor. HIV-associated cases have been reported, but serious cases are more often seen in leukemia patients. Disseminated infections have also been noted in renal and hepatic transplant patients. Infection often occurs through traumatic introductions into the body (i.e. through a wound). Cunninghamella bertholletiae can infect a wide variety of human tissue types, exhibits hyphal growth in the body and is angioinvasive. Like other Mucorales, under appropriate host conditions, it can grow very aggressively and destroy tissue structure. Typically, initial pathology is from thrombosis and infarction.
Common classes of mucormycoses include pulmonary, rhinocerebral (particularly when invasion into the vasculature of the brain is involved), multi-organ, cutaneous, and gastrointestinal (primarily in premature babies and malnourished children). Rhinocerebral infection and gastrointestinal cases are most immediately life-threatening. Pulmonary infections, as well as disseminated infections with pulmonary origins, are most common for C. bertholletiae, which has been identified in 7% of mucormycosis cases globally, and 3.2% of cases in the United States.
Although C. bertholletiae is only responsible for a small percentage of mucormycoses, it is cited as having the worst prognosis of the Mucorales. There are few identified cases per year, but C. bertholletiae infections and other mucormycoses are increasing in prevalence in North America, possibly due to growing populations of aging and immunosuppressed people.
Vascular invasion and tissue necrosis, often with black discharge, are good indicators of infection with Mucorales. C. bertholletiae can also grow at higher temperatures. The difficulty of culturing C. bertholletiae and other species within Mucorales from tissue samples makes laboratory analysis necessary to determine the causative organism of a mucormycosis. Polymerase chain reaction-based sequencing of fungal isolates is preferred as a reliable diagnostic tool due to possible difficulty of isolating C. bertholletiae from patients in culture. However, preliminary antifungal treatment should never be delayed if C. bertholletiae infection is suspected, as infections can often cause rapid and invasive tissue damage.
Genetic differences within the species C. bertholletiae can also be important determinants of pathogenicity and virulence. Recently, DNA barcoding of the internal transcribed spacer (ITS) region of C. bertholletiae ribosomal DNA was performed to improve upon current diagnostic techniques, providing more accurate and detailed between and within species discrimination compared to traditional analysis of colony colour and morphology, maximum growth temperature, and reproductive characteristics.
Because of its fast growth and invasiveness, treatment for C. bertholletiae infection can be expected to often require surgery in addition to antifungal treatment. Immediate surgery is especially important in case of rhinocerebral infection, in order to avoid dissemination into the vasculature of the brain and to avoid permanent optic nerve damage. Surgical debridement is a common treatment. Antifungal drugs that are used successfully against C. bertholletiae infection include amphotericin B, itraconazole, voriconazole and posaconazole. However, compared to other Mucorales species, C. bertholletiae has decreased responsiveness to some antifungals that are commonly prescribed to treat mucormycoses, and samples should be tested for individual antibiotic susceptibility if possible. Lipid formulations of amphotericin B are preferred for treatment of C. berthollettiae, because the high dosage required to treat infection can have significant toxic effects when administered in traditional formulations. Relapse after antifungal treatment and surgery is rare if a patient's clinical course initially improves during therapy.
Genus: Lichtheimia
Originally the genus Absidia united the species with pyriform sporangia with distinct apophysis (a dilatation of the sporangiophore underneath the sporangium) and hyaline, branched sporangiophores into the genus Lichtheimia. Phylogenetic and physiological studies showed that Absidia-like fungi represent three separate lineages 93: (1) the mesophilic genus Absidia sensu stricto that forms zygospores protected by long appendages of the suspensors; (2) the mycoparasitic genus Lentamyces that does not grow at temperatures above 30 ◦C; and (3) the thermotolerant and opportunistic genus Lichtheimia that produces zygospores with equatorial rings and suspensors without appendages.
Garcia-Hermoso et al. 94 showed that the clinical isolates in their study actually belonged to two species, Lichtheimia corymbifera and Lichtheimia ramosa, which were treated as synonyms previously. Alastruey-Izquierdo et al. 95 revised the whole genus and recognized five species in Lichtheimia: L. corymbifera, L. hyalospora, L. ornata, L. ramosa, and L. sphaerocystis. Later a sixth species, L. brasiliensis, was described 96. Only L. corymbifera, L. ornata, and L. ramosa are reported to cause human infections 31.
The clinical importance of Lichtheimia spp. depends on the geographical region. In Europe and Africa Lichtheimia species are the second most frequently reported aetiological agents of mucormycoses behind Rhizopus spp. while in America the number of cases is rather low. Most cases caused by Lichtheimia spp. show a cutaneous or pulmonary manifestation but also rhino-orbital-cerebral and disseminated infections occur 14.
Lichtheimia corymbifera is described to form subglobose to broadly ellipsoidal spores, while L. ramosa is thought to develop ellipsoidal to cylindrical spores only. When Nottebrock et al. 97 crossed strains of the two species identified by the shape of their spores, zygospores were produced, Subsequently, the two species were treated as synonyms until Garcia-Hermoso et al. 94 showed them to be distinct species. Alastruey-Izquierdo et al. 31 found strains of both species with an intermediary spore shape, such that misidentified strains could be responsible for the positive mating results of Nottebrock et al. 97.
The clinically relevant Lichtheimia species can be distinguished phenotypically. Lichtheimia ramosa has a higher growth rate at 43 ◦C than L. corymbifera and L. ornata. Lichtheimia ornata can be distinguished from L. corymbifera by its densely packed giant cells (large, irregularly shaped cells) formed on yeast extract agar.
Important for the differentiation of Absidia and Lichtheimia are the different maximum growth temperatures and the formation of a septum directly underneath the sporangium (subsporangial septum) in Absidia but (with rare exceptions) not in Lichtheimia 31
Genus: Mycotypha
Mycotypha rarely causes infections in humans, but recently Mycotypha microspora, has been found to involve in the clinical manifestation of the mucormycosis as a life-threatening disease.
Mycotypha microspora (also known as Microtypha microspora) was isolated from a Citrus aurantium peel in 1932 by E. Aline Fenner 82, in the Netherlands, where it was found to be pathogenic. Additionally, it has reportedly been found in decaying wood and a hospital washroom in Germany. One specific case noted its presence in stool samples from a child with leukaemia. Mycotypha africana, which is another species in the genus Mycotypha, is closely related to M. microspora. The fungus has subsequently been isolated from both outdoor and indoor settings around the world, and is typically found in soil, dung and faeces. Other species present in the genus Mycotypha include M. indica. These species are distributed worldwide and have been geographically collected from countries including Japan, India, Finland, Zimbabwe, and certain states in the U.S. such as Arizona, Washington D.C., Kansas, California, and Iowa.
Mycotypha microspora genus name is derived from the cattail-like appearance of its fructifications and tiny spores. It has a dense granular protoplasm and is composed of several hyphae and vacuoles. The structure is highly branched, with mycelium of varying diameters. It consists of two kinds of unispored sporangia: an inner layer containing globose spores and an outer layer with obovoid or cylindrical spores. During the growth period, the fungal body is coenocytic. After the fungus gradually matures, septation occurs at approximately the same time as sporulation. Mycotypha microspora colonies grow rapidly and abundantly on nutrient-rich media, such as carrot agar and potato dextrose. However, no growth occurs on low pH media. M. microspora is mesophilic, with optimal growth of cultures occurring at a temperature of 35 °C, with a threshold of 10 °C under which growth is inhibited. The fructifications typically form at night and thus respond unfavourably to light.
Only a few reported cases exist where the species has been found to cause an infection in humans. M. microspora has recently been implicated as a causative factor in the pathogenesis of gastrointestinal mucormycosis in humans. The disease develops due to the binding of spore coating (CotH) proteins from the fungus to glucose regulator protein 78 (GRP78) host receptors in endothelial cells. Tissue necrosis blocks the entry of antifungals to infected sites, therefore preventing clearance and promoting circulation of the disease. Mucormycosis is highly invasive in immunocompromised patients, and can mainly infect the body at pulmonary, rhinocerebral, cutaneous, and gastrointestinal sites. Factors that put an individual at risk for manifestation of the disease include corticosteroid use, diabetes, and ongoing neutropenia. CotH proteins are found in fungi of the order Mucorales, and blocking their function weakens their ability to invade endothelial cells, and reduces mucormycosis presentation in mice.
Given that the disease is rare, there is a lack of experimental findings assessing the efficacy of specific treatment regimens for mucormycosis. The most reliable antifungal agent against mucormycosis is amphotericin, however the use of this in combination with voriconazole led to acute kidney injury upon admission of a 41-year-old patient who was dually infected by Aspergillus fumigatus and M. microspora. In order to prevent permanent kidney damage, therapy was switched to administering the broad spectrum antifungal isavuconazole for 15 days, however this also led to complications in the patient. Ultimately, this gastrointestinal Mycotypha infection was treated with a combination of posaconazole and micafungin, which proved to be more effective than monotherapy, and the patient was eventually cured of the disease by surgically removing a part of the stomach in order to manage the gastrointestinal bleeding 22. This rare case provides some insight into potential treatment protocols for M. microspora and A. fumigatus infections in humans, however, further research that focuses on infections caused solely by M. microspora is essential in formulating a specific treatment regimen against this species.
Genus: Saksenaea
Saksenaea is the important pathogenic genera reported from India and Europe (83, 84]. Microscopic examination of fungal isolate using Giemsa stains shows hyaline and non-septate hyphae with right-angle branching. Culture of tissue samples on SDA (Sabouraud–glucose agar; Britania) grow as a white aerial mycelium after 5 days at 300C and 37 oC. Colonies on SDA are fast-growing and white with no pigment on the reverse. The fungus grow as sterile mycelia in malt extract agar, potato dextrose agar, SDA, Czapek–Dox agar (Becton Dickinson), and in an agar block with sterile distilled water and salt water (0.85 %NaCl).
Sporulation is achieved by floating the agar block containing fungal culture in a nutritionally deficient medium solution for 7 days at 37 OC 83. A few typical flask-shaped sporangia arise singly from erect sporangiophores. Sporangiophores are hyaline at first but soon became light brown with dichotomously branched and darkly pigmented rhizoids at their base. Sporangiospores are mostly ellipsoid but biconcave in the lateral view. A mucilaginous plug is observed on the apex of immature sporangia. These characteristics described identify the isolate as a member of the genus Saksenaea.Molecular identification is based on PCR amplification and sequencing of ITS.
Genus: Syncephalastrum
Syncephalastrum is a very rare causative agent of human mucormycosis 85, 86. It has so far been isolated in cases of cutaneous infection and otomycosis.
The genus Syncephalastrum is characterised by the formation of cylindrical merosporangia on a terminal swelling of the sporangiophore. Sporangiospores are arranged in a single row within the merosporangia. Syncephalastrum racemosum is the type species of the genus and a potential human pathogen; however, well-documented cases are lacking. It is found mainly from soil and dung in tropical and subtropical regions. It can also be a laboratory aerial contaminant. The sporangiophore and merosporangia of Syncephalastrum species may also be mistaken for an Aspergillus species, if the isolate is not examined carefully.
The fungal colonies of Syncephalastrum grow very rapidly and fill the petri dish or culture tube with cottony to fluffy, white to light grey, becoming dark grey with the development of sporangia. Maximum growth temperature is 40°C. From the front, the color is white initially and turns to dark gray to black in time. Reverse is pale or yellowish-brown.
Sporangiophores are erect, stolon-like, often producing adventitious rhizoids, and show sympodial branching (racemose branching) producing curved lateral branches. The main stalk and branches form terminal, globose to ovoid vesicles which bear finger-like merosporangia directly over their entire surface. At maturity, merosporangia are thin-walled, evanescent and contain five to ten (up to 18) globose to ovoid, smooth-walled sporangiospores (merospores).
Microscopic examination reveals broad (4-8 µm in diameter), nonseptate or sparsely septate hyphae, sporangiophores, merosporangia (finger-shaped, tubular sporangia), merosporangiospores (merospores), and rhizoids. Septation of the hyphae is mostly observed as the culture gets old. Sporangiophores are frequently branched and rather short. They end up in a vesicle (80 µm in diameter). Around this vesicle are the merosporangia (4-6 x 9-60 µm), which are filled with linear series (chains) of sporangiospores. Each merosporangium contains a single row of 3-18 merosporangiospores. Merosporangiospores (3-7 µm, may rarely reach 10 µm in diameter) are one-celled and spherical to cylindrical in shape. Zygospores, when produced, are black, spherical, and 50-90 µm in diameter. They have conical projections.
Genus: Thamnostylum
The colonies are fast-growing, initially white and cottony, reaching a diameter of approximately 10 cm after 7 days incubation on malt extract agar (MEA; Difco, Detroit, MI) and potato dextrose agar (PDA; Difco, Detroit, MI) at 25°C in the dark,. Colonies gradually turn pale yellow to olive brown after 7 days at 25°C on MEA and PDA. Aerial sporangiophores are produced from the substrate mycelium and stolons; these usually produce a terminal sporangium and a subterminal cluster of obpyriform sporangiola.
Dehisced sporangia mounted in 2% KOH–phloxine in distilled water bore a distinct columella and a collar. Whorls of obpyriform, recurved pedicillate sporangiola are typically formed on subterminal vesicles on the sporangiophores can be observed. Sporangiola mostly produce 8 to 16 sporangiospores that are oval, smooth, hyaline and measures 3 - 7 x 2 - 3 μm. These characteristic are descriptive of the culture of Thamnostylumlucknowense, a species that was originally isolated from soil in Lucknow, India, and designated as Helicostylumlucknowense was subsequently transferred by Upadhyay 87 to Thamnostylum based on the shared production of stolons and rhizoids, apophysate sporangia, and pyriforme sporangiola borne on circinate stalks.
General Signs of Mucormycosis Infection
The most common signs of mucormycosis infection includes
Excruciating Headaches
When a patient inhales the fungal spores which invades the sinus cavities and nerves, in turn causes a person to experience symptoms like persistent pain and headaches.
Vision Impairment
Changes to the eyes or vision distortion could also be warning signs of the spreading infection. As the fungi grows and spreads, vision can be distorted as well. Some people may also experience a sort of swelling in one eye, have hazy or poor vision, or develop bloodshot eyes.
Swelling in the Cheeks, Eyes or Parts of the Face
Swelling, local pain on the cheekbone, or experiencing one-sided facial pain or sort of numbness could also be primary sign of the infection. Apart from swelling, the fungal infection could also affect skin health and give rise to multiple lesions, necrosis like symptoms.
Altered Mental State, Confusion
Since the fungal infection is known to make its way to the brain, critical symptoms like delirium, memory loss, neurological impairment, the altered mental state could be signs of the infection.
Blackish Discolouration Around the Bridge of the Nose
Facial distortion is the primary feature of the infection. In the most severe cases, the infection can result in the growth of black patches around the eyes and nose. In some cases, sporadic growth of the fungal infection can lead a person to lose his or her teeth or jaws.
The symptoms of mucormycosis depend on the site of invasion of the fungus in human body Ribes et.al [2; Spellberg et.al, 88; Petrikkos et.al, 89; Soare et.al 90 and are referred as:
Rhinocerebral (Sinus and Brain) Mucormycosis
Rhinocerebral (sinus and brain) mucormycosis is an infection in the sinuses that can spread to the brain. This form of mucormycosis is most common in people with uncontrolled diabetes and in people who have had a kidney transplant.
The symptoms of Rhinocerebral mucormycosis include: One-sided facial swelling, Headache, Nasal or sinus congestion, Black lesions on nasal bridge or upper inside of mouth that quickly become more severe and fever.
Pulmonary (Lung) Mucormycosis
The symptomsinclude: Fever, Cough, Chest pain and Shortness of breath
Cutaneous (Skin) Mucormycosis
The symptoms include: Blisters or ulcers on the skin, and the infected area may turn black. Other symptoms include pain, warmth, excessive redness, or swelling around a wound.
Gastrointestinal Mucormycosis
The symptoms include: Abdominal pain, Nausea and vomiting and Gastro-intestinal bleeding
Disseminated Mucormycosis
Disseminated mucormycosis typically occurs in people who are already sick from other medical conditions, so it can be difficult to know which symptoms are related to mucormycosis. Patients with disseminated infection in the brain can develops mental status changes or coma.
Frequent clinical presentations include rhinocerebral, pulmonary, and cutaneous forms (superficial) and less frequently, gastrointestinal, disseminated, and miscellaneous forms. The rhinocerebral (rhinomaxillary) form is the most common form of infection commonly seen in patients with uncontrolled diabetes mellitus. The disease usually initiates in the nasal mucosa or palate and extends to the paranasal sinuses spreading through the surrounding vessels such as angular, lacrimal, and ethmoidal vessels. In addition, mucormycosis can also involve the retro-orbital region by direct extension. Once fungal hyphae enter into the bloodstream, they can spread to other organs such as cerebrum or lungs which can be fatal for the patient.
Diagnosis and testing for Mucormycosis
Healthcare provider may perform a tissue biopsy, in which a small sample of affected tissue is analyzed in a laboratory for evidence of mucormycosis under a microscope or in a fungal culture.
Mucormycosis Types and the Fungal Species Responsible
The mucorales species responsible for different types of mucormycosis are given in table 3.
Table 3. Types of mucormycosis and mucorale species involved.| Mucormycosis | Fungal sp responsible | |
| 1 | Rhinocerebral | Mucor, Rhizopus, Rhizomucor, Lichtheimia (Absidia) cunninghamella, and Apophysomyces. |
| 2 | Pulmonary | Absidia, Cunninghamella, and Syncephalastrum |
| 3 | Cutaneous | Rhizopus oryzae, Lichtheimia corymbifera, Apophysomyces elegans, Mucor, Saksensea,Cunninghamella, and Rhizomucor |
| 4 | Desseminated | Rhizomucor pusillu |
Scenario of Mucormycosis as Post Covid Infection in India
As cases of mucormycosis have been escalating in the country amid the second wave of the Covid-19 pandemic, several states have declared this rare fungal infection caused by a group of molds as a notifiable disease under the Epidemic Diseases Act, 1897. Many Covid-19 patients in India, of late, found to have contracted this potentially fatal infection. Recently, the Union health ministry urged the states and Union territories to make mucormycosis a notifiable disease under the Epidemic Diseases Act, 1897, stating that the infection is leading to prolonged morbidity and mortality among Covid-19 patients. A notifiable disease is required by law to be reported to the government authorities. The collection of information allows the authorities to monitor the disease and provides early warning of possible outbreaks.
Mucormycosis, also known as ‘black fungus’ in India, is more common among people whose immunity has lowered due to Covid-19, diabetes, kidney disease, liver or cardiac disorders, age-related issues, or those on medication for auto-immune diseases like rheumatoid arthritis. The disease begins to manifest as a skin infection in the air pockets located behind the forehead, nose, cheek-bones and in between the eyes and teeth. It then spreads to the eyes, lungs and can even spread to the brain. It leads to blackening or discoloration over the nose, blurred or double vision, chest pain, breathing difficulties and coughing of blood.
In India, the infection of mucormycosis has increasingly been seen in recovering Covid patients, with a 31,216 total cases with 2,109 deaths (i.e 6.7% of deaths) in a period of 3 week (May-June 2021) (table 4) during secod wave of covid-19, that means it has grown over 150 per cent over the last three weeks, putting additional stress on an already embattled healthcare system fighting off the deadly second surge of Covid wave.
Table 4. Surge in Mucormycosis cases in different states of India during May-june 2021.| Name of state | 25th May | 18th June | |||
| No.of Patient | No.of Patient | (%) increase in patient | No of Deaths | (%) mortality | |
| Gujrat | 2859 | 5418 | 54.52 | 323 | 5.96 |
| Maharashtra | 2770 | 7359 | 165.66 | 653 | 8.87 |
| Andra Pradesh | 910 | 2303 | 153.07 | 157 | 6.81 |
| Madhya Pradesh | 720 | 764 | 6.11 | 49 | 6.41 |
| Uttar Pradesh | 701 | 1744 | 148.78 | 142 | 8.14 |
| Rajasthan | 700 | 2976 | 325.14 | 74 | 2.48 |
| Karnataka | 500 | 2856 | 471 | 225 | 7.87 |
| Haryana | 250 | 1056 | 322.4 | 91 | 8.61 |
| Delhi | 197 | 1200 | 509.13 | 125 | 10.41 |
| Punjab | 95 | 400 | 321.05 | 18 | 4.5 |
| Chhattisgarh | 87 | 206 | 136.78 | 12 | 5.82 |
| Bihar | 56 | 562 | 903.57 | 76 | 13.52 |
| Tamil Nadu | 40 | 518 | 1195 | 17 | 3.28 |
| Kerala | 15 | 45 | 200 | 4 | 8.88 |
| Jharkhand | 27 | 96 | 255.55 | 26 | 27.08 |
| Goa | 12 | 24 | 100 | 10 | 41.66 |
| West Bengal | 12 | 45 | 275 | 11 | 24.44 |
The highest number of patients were reported from Maharashtra state, followed by Gujrat, Rajasthan and Karnatka. The lowest number of cases were reported from Goa followed by Kerala and West Bengal. The highest surge in mucormycosis patients were observed for TamilNadu (1159%), followed by Bihar (903%), Delhi (509%) and Karnatka (471%). The lowest surge was for Madhya Pradesh (6.11%) followed by Gujrat (54.52%). The highest percent mortality rate was observed for Goa (41.66%), followed by Jharkhand (27.08%) and West Bengal (24.44%). The lowest percent mortality rate was noted for Rajasthan (2.48%) followed by TamilNadu (3.28%).
The Maharashtra state stand first with 7359 cases and 653 deaths. In this state 2212 mucormycosis patients have recovered from disease and 4488 are under treatment. Five districts of the state recorded 57% of covid linked mucormycosis cases(4238 patients) viz. Pune(1216), Nagpur(1184), Aurangabad(700), Mumbai(596),Nashik(542) in their urban and rural parts and these districts are also among the worst hit places during the Covid’s second surge. In Rajasthan, the fatality rate of mucormycosis is 5 times than that of covid-19 in the state. While the covid mortality rate is below 1%, the death rate for mucormycosis is 4.8 % in the state. The increase in death numbers is driven, in part, by severe shortage of the key drug Amphotericin-B to treat affected patients. In view of the rising cases, the Maharashtra government has advised screening of a sizable chunk of hospitalized covid patients for mucormycosis immediately prior to or after the discharge. They include patients who have had steroid, oxygen therapy and intensive care unit (ICU) stay for more than 7 days at hospitals. Admitted patients with high blood sugar (above 200 mg/dl )or glycated haemoglobin(HbA1C) above 8 are also advised to be screened for the invasive fungal infection that primarily affect the sinuses, patient given the antirheumatic drug tocilizumab are also included in the list of factors for early screening.
Mucormycosis is considered to be a big, serious threat right now. The ICMR has now issued guidelines that not only could it affect a lot of COVID patients in recovery, it could also become extremely fatal if left unchecked. As per reports the black fungal infectious spores which is present in the air causes complications when an ill patient inhales these, which then spreads into the sinus cavities, lungs and chest cavities. It is believed that a high dependency on steroids (used to treat inflammation in COVID cases), pre-existing comorbidities, such as diabetes may make a person suffering from COVID at a higher risk for catching mucormycosis as well. For diabetes patients, this is life-threatening. A person affected with mucormycosis may need early detection and powerful diagnosis for treatment, which may, in turn, affect other organs such as the kidneys. People in an extreme immune-compromised state are likely to contract this infection. Diabetologists across the country raise caution against the increased prevalence of mucormycosis during COVID-19, urging people to keep their sugar levels under check to reduce the risk.
Predisposing Factors and Diagnosis for Mucormycosis
The various predisposing factors 91 for mucormycosis are: uncontrolled diabetes (particularly in patients having ketoacidosis), malignancies such as lymphomas and leukemias, renal failure, organ transplant, long-term corticosteroid and immunosuppressive therapy, cirrhosis, burns, protein-energy malnutrition, and acquired immune deficiency syndrome (AIDS). 92
In rhino-sinus mucormycosis, CT is the investigation of choice to study the invasion of bone and soft tissue abscesses, or hematoma, and extension to the central nervous system. MRI is more sensitive than CT for the investigation of possible cerebrovascular thrombosis. In pulmonary mucormycosis, chest radiograph or better chest CT typically show alveolar condensations sometimes excavated or nodular infiltrates frosted glass with or without halo sign. These nose and brain damage and lung characteristics are observed in the patients 89.
Since imaging lesions are not specific of mucormycosis, a mycological diagnosis is necessary. The reference method is the direct examination and culturing of the pathological product: puncture fluid (pus, serous fluid), tissue biopsy. Mucorales hyphae are short, little or non septate, thick-walled and often branched at right angles.
In addition to the mycological diagnosis, histological study 93 of biopsy fragments are useful for the diagnosis of mucormycosis and allows the confirmation in case of Mucorales filament presence in tissues and vessels where they are responsible for thrombosis with infarction and hemorrhage . In study conducted by Bellazreg 9, the Mucorales were isolated from pus obtained by percutaneous puncture or multiple swab samples in two cases, and on biopsy fragments in three cases. Culture was positive in all cases. The two isolated genera were Rhizopus and Lichteimia. The predominance of the latter was more in the male patients in their study.
Mucormycosis has poor prognosis with a mortality rate of 17–51% 94, 95. Mortality is higher in case of diagnostic delay of more than five days and monocytopenia in patients with active malignant blood diseases. Surgical treatment associated with antifungals improves rognosis 94.
Known and Unknown Factors
Diabetes Milletus and Compromised Patients More Prone to Mucormycosis
Diabetes mellitus tends to change the normal immunological response of body to any infection in several ways. Hyperglycemia stimulates fungal proliferation and also causes decrease in chemotaxis and phagocytic efficiency which permits the otherwise innocuous organisms to thrive in acid-rich environment. In the diabetic ketoacidosis patient, there is an increased risk of mucormycosis caused by Rhizopus oryzae as these organisms produce the enzyme ketoreductase, which allows them to utilize the patient's ketone bodies 96. It has been established that diabetic ketoacidosis temporarily disrupts the ability of transferrin to bind iron, and this alteration eliminates a significant host defense mechanism and permits the growth of Rhizopus oryzae97. Afroze et.al 98 studied a patient of mucormycosis who had uncontrolled diabetes for 4 months with fasting blood sugar level, 154 mg/dl (normal 70–110 mg/dl) and postlunch sugar level, 197 mg/dl (normal 70–140 mg/dl) . On biochemical investigation, an elevated fasting blood sugar level and decreased hemoglobin% (7 g %) was noticed and HbA1c level was 8.3%. To manage the infection two-unit blood transfusion was done and iron supplements were given (Venofer 800 mg which is iron sucrose solution, the drug has a pH of 10.5–11.5 which makes the alkaline environment, so the Rhizopus cannot multiply as they require acidic medium for their growth).
Bellazreg.et.al 9 in their studies observed the mucormycosis more often in immunocompromised patients (four out of five cases) and the most common sites were rhino-cerebral and rhino-orbital. Indeed, in 90% of cases, mucormycosis occur in immunocompromised patients, mainly diabetic ketosis or hematological malignancy with neutropenia. The naso-orbito-cerebral, lung and skin are the most common. A Tunisian retrospective study compiling 17 cases of mucormycosis between 1992 and 2007, diabetes and rhino-orbitofrontal cerebral localization was noted in all patients.
Use of Steriod for Covid Patients Increase the Risk of Mucormycosis
What is more worrying is that the use of steroids for treating certain cases of COVID-19 would shoot up sugar levels99 this coupled with lack of physical activity puts diabetic people at a higher risk of catching the black fungal infection. Steroids reduce inflammation in the lungs due to COVID-19 and appear to help curb some of the damage that can happen when the body’s immune system fights against the virus. But, they also reduce immunity and push up blood sugar levels in both diabetics and non-diabetic COVID-19 patients. One way to stem the surge of the fungal infection is to make sure COVID-19 patients, both in treatment and after recovery, are administered the right dose and duration of steroids. It is also important for people with diabetes, cancer, kidney and liver diseases, and organ transplant patients, to keep a check on their glucose levels from time to time.
Treatment of Mucormycosis
The treatment of mucormycosis is mainly based on antifungal and surgical debridement. The rapid equilibration of ketoacidosis in diabetics, transfusion of hematopoietic growth factors in long-term neutropenia and hyperbaric oxygen therapy may be useful 100. Reference antifungal therapy is liposomal amphotericin B, 5 to 10 mg / kg / day. However, Amphotericin B deoxycholate should no longer be used because of its nephrotoxicity 100. Other antifungal posaconazole or caspofungin can be used in combination with liposomal amphotericin B in case of treatment failure or as a substitute for serious side effects 100. Fluconazole, voriconazole and itraconazole have no activity on Mucorales 100.
In Tunisian studies 9 out of five patients treated with amphotericin B deoxycholate, two patients had surgical excision while Moderate renal impairment occurred in two patients indicating that there is no guarantee for cent percent cure of mucormycosis with the drug Amphotericin B. In India also inspite of use of Amphotericin B for the treatment of mucormycosis, at least 6.7 percent deaths occurred. This raises the question whether the drug is effective agains all the mucormycosis fungal species with the same dose.
Mucorales Species Responsible and their Drug Sensitivity
Mucormycosis is caused by at least 39 species of 12 generas of mucorales. In India, in the print media, the mucormycosis is reported due to black fungus, white fungus and blue fungus without quoting the scientific name of the fungus for which amphotericin B is used at present. It is not scientifically proved that the same drug is equally effective against these different fungi at the same dose.
India is a very diverse country in respect of climatic environment and fungal microflora. Different mucorales species responsible for mucormycosis may be present in different states. Their niches, frequencies, and spore loads may also be different. There sensitivity to drug may also be different. The scientific studies and data on these aspect are not available yet for the country. To have an effective control of mucormycosis in various Indian states, this data generation has an immense value. Until, this research is carried out with generation of adequate data, each state must, at least, confirm the genus and species of mucormycosis fungi in their state and study their sensitivity to the available antifungicidal drug and the effective drug found for their species should only be used.
Molecular identification based on ITS-sequences is the method of choice for Mucorales species, as it has been shown by numerous studies to reliably distinguish species 14, 37. Failure of the ITS region to discriminate R. microsporus and R. azygosporus can be explained by the fact that both species actually belong to the same species 31. In the Mucor circinelloides relationship, protein-coding genes such as tsr1 or rpb1 have a much higher resolution power than ITS, but reference sequences of these genes are usually lacking 48. A disadvantage of using ITS sequencing for identification is that ITS copies differ slightly in some taxa, such as Absidia or the Rhizopus stolonifer group, and direct sequencing becomes impossible 12. Also of note, in the genus Syncephalastrum some strains have two clearly differing types of ITS-sequences 101. Alternatively, LSU can be used for identification and has the advantage that direct sequencing is nearly always possible. This region resolves all mucoralean species, but the sequence differences among the species are relatively small.
In diagnostic facilities, matrix-assisted laser desorption ionization-time-of-flight mass spectrometry (MALDI-TOF MS) is being increasingly used for the identification of filamentous fungi. In the last few years, several studies have shown the potential of this method for a fast identification of the Mucorales 102, 103. Using the VITEK®MS v3.0 system, a species identification rate of 86% was achieved for 118 mucoralean isolates 104. However, only a species identification rate of 49.5% was possible using the Bruker library. The combination of the Bruker library with an in-house database allowed MALDI-TOF MS to identify 81.1% of 111 mucoralean isolates 105.
Numerous DNA-based assays have been developed to detect the aetiological agents of mucormycosis from fresh or formalin-fixed, paraffin-embedded clinical samples. The range of methods includes PCR-RFLPs 106, conventional semi-nested PCR 107, 108, or RT-PCR 109, 110, usually targeting nuclear rDNA (SSU, LSU, ITS) or less frequently cytochrome b 111, 112. A recent study used the gene of the mucoralean-specific spore coating protein homolog, cotH, as the target for PCR amplification 113. In some studies, PCR amplification was combined with another method of identification, such as reverse line blot hybridization or electrospray-ionization mass spectrometry 111. Pathonostics commercially distributes the MucorGenius assay, which is a multiplex, real-time PCR assay detecting Rhizopus spp., Mucor spp., Rhizomucor spp., Lichtheimia spp., and Cunninghamella spp. in respiratory tract samples and biopsies. A completely different approach for Mucorales diagnostics is the detection of Mucorales-specific T cells 114.
Last but not least, species identification based on morphology combined with growth measurement at different media and temperatures is possible in taxonomically revised taxa 48, 21, 14, 33.
Few antifungal drugs are active against mucorales. Considering the low efficacy of monotherapy, combination therapy strategies have benn described by Leonardelli et .al. 115. It is known that fungi are susceptible to zinc deprivation, thus in vitro effect of zinc chelator Clioquinol, Phenanthroline and TPEN combined with either Amphotericin B or posaconazole were studied against 25 strains of Mucorales. Clioquinol- posaconazole was the most active combination, although results were strain dependent.
Caetano et.al 116 studied the prevalence of mucorales in different indoor environment in Portugal. Mucorales was found as more prevalent in air conditioning filters and waste sorting fork lifters. In their studies among the mucorales isolates able to grow in Azol-supplimented media, 16 isolates of mucor species, Rhizopus sp, or Rhizomucor sp were not susceptible to 1mg/L Variconazole and 4 isolates of Mucor sp or Rhizopus sp were not susceptible to 4 mg/L Itraconazole. Although Amphotericin B is considered to be the drug of choice for mucormycosis, there are differences in in vitro susceptibility between species 117.
The mucorales species are characterized by high level resistance to most currently available antifungal drugs. Antifungal specificity within the group may be species specific 118. Amphotericin B, Posaconazole and Isavuconazole are currently the most active drug against mucorales, however, their activity remains sub-optimal and new therapeutic strategies are needed 118. Espinel-Ingroff et.al 119 proposed species- specific Amphotericin- B ECVs (Epidemiologic Cutoff Value) comprising > 95 % of the modelled population of 1ug/mL (for L. corymbifera and M. circinelloides) to 2 ug/mL (for R. arzhizus and R. microspores). Barnert et.al. 120 reported an Amphotericin-B resistant case of rhinocerebral mucormycosis. The disease advanced despite of early amphotericin-B therapy and extensive surgical intervention. The MIC of amphotericin-B for the isolated mucor species was 64 mg/L, meaning that in vitro resistance also existed. Only long term treatment with Ketoconazole (600 mg/day, perorally) was successful in curing the disease. Biswas et.al 121 reported Amphotericin-B resistant Apophysomyces elegans causing Rhino-oculo-Celebral mucormycosis in an immunocompetent host. Though the disease is usually managed with the surgical debridement of the affected tissues and medical therapy with Amphotericin-B, the fungal isolate recovered in their studies was found to be resistant to Amphotericin-B. Though treatment failure and relatively high MICs with Amphotericin-B have been reported in literature, this isolate of A. elegans was completely resistant to Amphotericin-B. Asghar et.al 122 reported Rhino-oculo-cerebral mucormycosis resistant to Amphotericin-B in a young patient with diabetic ketoacidosis. Although therapeutic approach includes aggressive surgical and medical interventions the rhino-oculo-cerebral mucormycosis remain a life threatening infection with poor prognosis in young patient with delayed diagnosis and resistant to Amphotericin-B contributing to dreadful outcome.
Nagy et.al 123 reported characterization of three pleiotropic drug resistance transporter genes and their participation in the Azole resistance of Mucor circinelloides. Their results suggest that the regulation of eight pdr (pleiotropic drug resistance) genus is interconnected and pdr 1 and pdr 2 participate in the resistance of the fungi to Posaconazole, Ravuconazole and Isavuconazole. By deletion of pfr 2 and pdr 6, the growth ability of the mutant strains decreased, while deletion of pdr 2 resulted in increased sensitivity against the antifungal drugs.
Now, it is bocoming increasingly evident that the exact knowledge of the genus and species is important from the standpoint of antifungal therapy as some of mucorales have distinct antifungal susceptible profile 117. Similarly, the knowledge of ecology and distribution of different species of mucorales in different parts of the nation will prove a milestone in developing strategies for types of drug to be used in its managements.
Ecology and Geographic Distribution
Mucorales are often considered to be cosmopolitan saprobes. However, for most species, the data are not sufficient to accurately assess their ecological niche or geographic distribution. For example, Actinomucor elegans was thought to be a saprotrophic soil fungus 124 until it was shown that it efficiently infects chafer beetle 125. Even from the existing data, it can be safely concluded that the Mucorales are ecologically highly diverse. Beside from saprobes, they contain parasites of plants (e.g., Choanephora), arthropods (e.g., Sporodiniella umbellata), mushrooms (Dicranophora, Spinellus, and Synzygites), and other Mucorales (Chaetocladium, Lentamyces, and Parasitella) 126. Several Mucorales have been isolated from the inside of plants that did not show any symptoms and were consequently considered as endophytes, e.g., Mucor endophyticus (127, as Rhizomucor endophyticus).
The most important habitats for saprotrophic Mucorales are soil, dead plant material, and dung. These grow on normal culture media, but show a distinct association to dung (e.g., mucormycosis causing genera Cokeromyces, Mucor flavus, M. mucedo, M. plasmaticus, and Thamnostylum 126, 128, 129. On the other hand, there are generalists such as Mucor circinelloides and M. racemosus that have been isolated from a broad range of substrates, including dung 130. For some mucoralean species, the evidence is sufficient to call them cosmopolitan, e.g., Rhizopus arrhizus, Lichtheimia corymbifera, or Mucor circinelloides, but for numerous species this is not yet the case. Most of our information on the ecology and distribution of species is based on the source provided by culture collections and clinical studies. The number of studies directly addressing the natural habitats of Mucorales 125, 130, 131 is very limited. Taxa such as Apophysomyces, Saksenaea, and Mucor circinatus (misapplied: Circinella simplex) seem to show a distribution in tropical and subtropical regions. Reports of Apophysomyces spp. and Saksenaea ssp. in colder regions are related to clinical isolates and might refer to imported clinical cases. Other taxa have been isolated from restricted areas: e.g., Lichtheimia sphaerocystis is only known in India, Ellisomyces anomalus has only been found in California, despite being widely distributed there 132. A better understanding of the biology of the opportunists, including their natural niches, reservoirs, dispersal, and geographic distribution, is a prerequisite for understanding the route of acquisition and consequently for the prevention of infections.
Monitoring of Mucormycosis Causing Fungal Spore Load in the Environment
Different climatic environments have different fungal load of mucorales. The control environments like hospital settings and residential places have less sporangiospore loads than the open damp places, composting sites, left-over field residues, spoiled and rotting agricultural produce. These places/settings are important sources of inhaling sporangiospores of mucormycosis fungi for the non-morbid and immunocompromised patient, particularly those having diabetes. The post covid patients having diabetes may be more prone to infection of mucormycosis and for their safety and benefits, the spore load data of mucormycosis fungi in their citi may prove beneficial for them to avoid such places and localities for their movements. Around the world different cities display the air quality data at different places in their cities for the citizens. On the same pattern, the air quqlity data for mucorales fungal load, if displayed, may prove beneficial for the safety of immunocompressed peoples in post covid era to avoid the infection of mucormycosis.
Breathing Exercise and Mucormycosis Infection
Inhalation of mucorales spores (sporangiospores of mucormycosis causing species) during the breathing is an important step for causing rhino-cerebral and pulmonary mucormycosis infection. During the regular breathing process, the nasal hairs filters the allergens and pathogenic entities and restrict their entry in to the respiratory tracts to cause infection or become infectious. In India, certain breathing exercises in pranayama, particulary lom-Vilom are recommended where air is forcely inhaled and exel. No scientific data is available whether forceful inhalation of air through nostrils does not allow the mucorales spores to enter into respiratory tract to cause infection or otherwise. Therefore, the diabetic and immunocompromised patient and particularly those of post covid patient should avoid the forceful breathing exercise in the mucorales fungi prone air.
Conclusion
The proper identification of Mucorales species causing Mucormycosis is important for its management as these species varies in their antifungal drug sensitivity and resistance to the drug. The factors which are responsible to succumb to mucormycosis infection are particularly, the diabetic condition of patients, steroid medications, and major surgeries which should be taken care of to avoid the mucormycosis fungal infection. Handling of issues like monitoring of air quality data for mucorales in metro cities and in hospitals and their displays at fortnight intervals will be worth to forecast the mucormycosis infections and management of etiological agent. Such forecast will prove a milestone for immunocompromised patients to avoid such places and thereby mucormycosis infection. The governments should make a policy for municipal corporations and hospitals to monitor and display the air quality data for mucormycosis fungi. WHO also should look and consider such important policy issues of monitoring air quality data for mucorales to deal with the emerging trends of Mucormycosis.
References
- 1.Mendoza L, Vilela R, Voelz K, Chan L. (2014) Human fungal pathogens of Mucorales and Entomophthorales. Cold Spring Harbor Perspective in. , Medicine 5(4), 1-33.
- 2.Ribes J A, CL Vanover Sams, Baker D J. (2000) Zygomycetes in human diseases. , Clinical Microbiology Reviews.13 2, 236-301.
- 4.Prakash H, chakrabarti A. (2019) Global epidemiology of mucormycosis. , Journal Fungi.(Basel) 5(1), 26.
- 5.Ramalingam S, Malathi N, Karthikeyan V, Janagaraj V D. (2019) Mucormycosis: A Brief Review. , Journal of Pure and Applied Microbiology 13(1), 161-165.
- 6.Roden M M, Zaoutis T E, Buchanan W L, Knudsen T A, Sarkisova T A et al. (2005) Epidemiology and outcome of zygomycosis: A review of 929 reported cases. , Clin. Infect. Dis 41, 634-653.
- 7.Fehr M, Cathomas G, Graber A, Makert E, Gaus E et al.Multi-fungal sepsis and mucormycosis of the central nervous system in a patient treated with ibrutinib, a case report and review of literatrure. Medical Mycol Case Report.27:. 14-16.
- 8.Q B Rahman, Rahman M M, Imon A A, Rabby A I, Chowdhary M J. (2015) Management of Maxillary Mucormycosis- A case report in Bangladesh. Medicine Today. 27(1), 38.
- 9.Bellazreg F, Hattab Z, Meksi S, Mansoun S, Hachti W et al. (2015) Out-come of Mucormycosis after treatment: A report of five cases. , New Microbes New Infect 6, 49-52.
- 10.Walther G, Wagner L, Kurzai O. (2019) Updates on the Taxonomy of Mucorales with an emphasis on clinically important taxa. , Journal of fungi 5, 106.
- 11.Hoffmann K, Pawlowska J, Walther G, Wrzosek M, Hoog G S de et al. (2013) The family structure of the Mucorales: A synoptic revision based on comprehensive multigene-genealogies. , Persoonia 30, 57-76.
- 12.Walther G, Pawlowska J, Alastruey-Izquierdo A, Wrzosek M, Rodriguez-Tudela J L et al. (2013) DNA barcoding in Mucorales: An inventory of biodiversity. , Persoonia 30, 11-47.
- 13.Mahmud A, Lee R, Munfus-McCray D, Kwiatkowski N, Subramanian A et al. (2012) Actinomucor elegans as an emerging cause of Mucormycosis. , J. Clin. Microbiol 50, 1092-1095.
- 14.Álvarez E, Stchigel A M, Cano J, Sutton D A, Fothergill A W et al. (2010) Molecular phylogenetic diversity of the emerging mucoralean fungus Apophysomyces: Proposal of three new species. , Rev. Iberoam. Micol 27, 80-89.
- 15.Bonifaz A, A M Stchigel, Guarro J, Guevara E, Pintos L et al. (2014) Primary cutaneous mucormycosis produced by the new species Apophysomyces mexicanus. , J. Clin. Microbiol 52, 4428-4431.
- 16.L J Ryan, Ferrieri P, R D Powell, C D Paddock, S R Zaki et al. (2011) Fatal Cokeromyces recurvatus pneumonia: Report of a case highlighting the potential for histopathologic misdiagnosis as Coccidioides. , Int. J. Surg. Pathol 19, 373-376.
- 17.Navanukroh O, Jitmuang A, Chayakulkeeree M, Ngamskulrungroj P. (2014) Disseminated Cunninghamella bertholletiae infection with spinal epidural abscess in a kidney transplant patient: Case report and literature review. , Transpl. Infect. Dis 16, 658-665.
- 18.García-Rodríguez J, Quiles-Melero I, Humala-Barbier K, Monzon A, Cuenca-Estrella M. (2012) Isolation of Cunninghamella blakesleeana in an immunodepressed patient. , Mycoses 55, 463-465.
- 19.Álvarez E, D A Sutton, Cano J, A W Fothergill, Stchigel A et al. (2009) Spectrum of zygomycete species identified in clinically significant specimens in the United States. , J. Clin. Microbiol 47, 1650-1656.
- 20.Yu J, Walther A D, Diepeningen AHg Gerrits Van, Ende ruo-Yu li Van Den, TAA Moussa.. OA Almaghrabi and GS De Hoog. (20150. DNA barcoding of clinically relevant Cunninghamella species. Medical Microbiology 53(2), 99-106.
- 21.Alastruey-Izquierdo A, Hoffmann K, Hoog G S de, J L Rodriguez-Tudela, Voigt K et al. (2010) Species recognition and clinical relevance of the zygomycetous genus Lichtheimia (syn. Absidia pro parte. , Mycocladus). J. Clin. Microbiol 48, 2154-2170.
- 22.Álvarez E, Cano J, A M Stchigel, D A Sutton, A W Fothergill et al. (2011) Two new species of Mucor from clinical samples. , Med. Mycol 49, 62-72.
- 23.Wagner L, S de Hoog, Alastruey-Izquierdo A, Voigt K, Kurzai O et al. (2019) A revised species concept for opportunistic Mucor species reveals species-specific antifungal susceptibility profiles. , Antimicrob. Agents Chemother 63, 00653-19.
- 24.L De Repentigny, St-Germain G, Charest H, Kokta V, Vobecky S. (2008) Fatal zygomycosis caused by Mucor indicus in a child with an implantable left ventricular assist device. , Pediatr. Infect. Dis. J 27, 355-357.
- 25.X L Lu, M J Najafzadeh, Dolatabadi S, Y P Ran, Ende Gerrits van den et al. (2013) Taxonomy and epidemiology of Mucor irregularis, agent of chronic cutaneous mucormycosis. , Persoonia 30, 48-56.
- 26.Hesseltine C W, Ellis J J. (1964) An interesting species. , of Mucor, M. ramosissimus. Sabouraudia 3, 151-154.
- 27.Trachuk P, Szymczak P Muscarella W A, U N Sarwar. (2018) A case of Invasive gastrointestinal Mycotypha infection in a patient with Neutropenia. Case Reports in Infectious Diseases.
- 28.P C Iwen, A G Freifeld, Sigler L, S R Tarantolo. (2005) Molecular identification of Rhizomucor pusillus as a cause of sinus-orbital zygomycosis in a patient with acute myelogenous leukemia. , J. Clin. Microbiol 43, 5819-5821.
- 29.Dolatabadi S, Hoog G S de, J F Meis, Walther G. (2014) Species boundaries and nomenclature of Rhizopus arrhizus (syn. , R. oryzae). Mycoses 57, 108-127.
- 30.Chakrabarti A, R S Marak, M R Shivaprakash, Gupta S, Garg R et al. (2010) Cavitary pulmonary zygomycosis caused by Rhizopus homothallicus. , J. Clin. Microbiol 48, 1965-1969.
- 31.Dolatabadi S, Walther G, Ende Gerrits van den, A H G, Hoog G S de. (2013) Diversity and delimitation of Rhizopus microsporus. , Fungal Divers 64, 145-163.
- 32.Weitzman I, D A McGough, M G Rinaldi, Della-Latta P. (1996) Rhizopus schipperae, sp. nov. a new agent of zygomycosis. , Mycotaxon 59, 217-225.
- 33.Álvarez E, Garcia-Hermoso D, D A Sutton, J F Cano, A M Stchigel et al. (2010) Molecular phylogeny and proposal of two new species of the emerging pathogenic fungus Saksenaea. , J. Clin. Microbiol 48, 4410-4416.
- 34.P W Crous, M J Wingfield, T I Burgess, Hardy G, P A Barber et al. (2017) Fungal Planet description sheets:. , Persoonia 38, 558-624.
- 35.Schlebusch S, D F Looke. (2005) Intraabdominal zygomycosis caused by Syncephalastrum racemosum infection successfully treated with partial surgical debridement and high-dose amphotericin B lipid complex. , J. Clin. Microbiol 43, 5825-5827.
- 36.Xess I, Mohapatra S, M R Shivaprakash, Chakrabarti A, G L Benny et al. (2012) Evidence implicating Thamnostylum lucknowense as an etiological agent of rhino-orbital mucormycosis. , J. Clin. Microbiol 50, 1491-1494.
- 37.Hermet A, Meheust D, Mounier J, Barbier G, J L. (2012) Molecular systematics in the genus Mucor with special regards to species encountered in cheese. , Fungal Biol 116, 692-705.
- 38.Hüttel W, Hoffmeister D. (2011) Fungal biotransformations in pharmaceutical sciences. In Industrial Applications. The Mycota (A Comprehensive Treatise on Fungi as Experimental Systems for Basic and Applied Research); Hofrichter. , M., Ed, Springer: Berlin/Heidelberg, Germanry 10.
- 39.Chung Kwon, KJ. (2012) Taxonomy of fungi causing mucormycosis and entomophthoramycosis (zygomycosis) and nomenclature of the disease: Molecular mycologic perspectives. , Clin. Infect. Dis 54, 8-15.
- 40.O’Donnell K, Lutzoni F, T J Ward, Benny G. (2001) Evolutionary relationships among mucoralean fungi (Zygomycota): Evidence for family polyphyly on a large scale. , Mycologia 93, 286-297.
- 41.J W Spatafora, Chang Y, G L Benny, Lazarus K, M E Smith et al. (2016) A phylum-level phylogenetic classification of zygomycete fungi based on genome-scale data. , Mycologia 108, 1028-1046.
- 42.L J Ma, A S Ibrahim, Skory C, M G Grabherr, Burger G et al. (2009) Genomic analysis of the basal lineage fungus Rhizopus oryzae reveals a whole-genome duplication. , PLoS Genet 5, 1000549.
- 43.L M Corrochano, Kuo A, Marcet-Houben M, Polaino S, Salamov A et al. (2016) Expansion of signal transduction pathways in fungi by extensive genome duplication. , Curr. Biol 26, 1577-1584.
- 44.V U Schwartze, Winter S, Shelest E, Marcet-Houben M, Horn F et al. (2014) Gene expansion shapes genome architecture in the human pathogen Lichtheimia corymbifera: An evolutionary genomics analysis in the ancient terrestrial Mucorales (Mucoromycotina). , PLoS Genet 10, 1004496.
- 45.J W Taylor, D J Jacobson, Kroken S, Kasuga T, D M Geiser et al. (2000) Phylogenetic species recognition and species concepts in fungi. , Fungal. Genet. Biol 31, 21-32.
- 46.H A Ariyawansa, K D Hyde, S C Jayasiri, Buyck B, Chethana K W T et al. (2015) Fungal diversity notes 111–252—taxonomic and phylogenetic contributions to fungal taxa. , Fungal Divers 75, 27-274.
- 47.G J Li, K D Hyde, R L Zhao, Hongsanan S, F A Abdel-Aziz et al. (2016) Fungal diversity notes 253–366: Taxonomic and phylogenetic contributions to fungal taxa. , Fungal Divers 78, 1-237.
- 48.Wagner L, J B Stielow, S de Hoog, Bensch K, Schwartze V et al. (2019) A new species concept for the clinically relevant Mucor circinelloides complex. Persoonia. Doi: 10.3767/persoonia.2020.44.03
- 49.Tibpromma S, K D Hyde, Jeewon R, Maharachchikumbura S S N, Liu J-K et al. (2017) Fungal diversity notes 491–602: Taxonomic and phylogenetic contributions to fungal taxa. , Fungal Divers 83, 1-261.
- 50.Loh L S, Nawawi A, Kuthubutheen A J. Institute of Biological Sciences, University of Malaya: Kuala Lumpur (2001) Mucoraceous Fungi from Malaysia (Kuala Lumpur);. , Malaysia
- 52.Schipper M A A. (1978) On certain species of Mucor with a key to all accepted species. , Stud. Mycol 17, 1-48.
- 53.O’Donnell K L, Ellis J J, Hesseltine C W, Hooper G R. (1977) Morphogenesis of azygospores induced in Gilbertella persicaria (+) by imperfect hybridization with Rhizopus stolonifer (-). , Can. J. Bot 55, 2721-2727.
- 54.W A Schell, O’Donnell K, J A Alspaugh. (2011) Heterothallic mating in Mucor irregularis and first isolate of the species outside of Asia. , Med. Mycol 49, 714-723.
- 55.Jeong W, keighley C, Wolfe R, Lee M A Slavin W, Kong D C M et al. (2019) The epidemiology and clinical manifestations of mucormycosis: A systematic review and meta-analysis of case reports. , Clinical Microbiol Inf 25, 26-34.
- 56.Schipper M A A. (1976) On Mucor circinelloides, Mucor racemosus and related species. , Stud. Mycol 12, 1-40.
- 57.X L Lu, Z H Liu, Shen X D She Y N, Lu P Zhan G X, Fu X L Zhang M H et al. (2009) Primary cutaneous zygomycosis caused by Rhizomucor variabilis. A new endemic zygomycosis. A case report and review of 6 cases reported from China. Clinical Infect. , Dis 49, 39-43.
- 58.Schipper M A A. (1973) A study on variability in Mucor hiemalis and related species. , Stud. Mycol 4, 1-40.
- 61.Abe A, Asano K, Sone T. (2010) A molecular phylogeny-based taxonomy of the genus Rhizopus. , Biosci. Biotechnol. Biochem 74, 1325-1331.
- 62.Vagvolgyi C, Heinrich H, Acs K, Papp T. (2004) Genetic variability in the species Rhizopus stolonifer, assessed by random amplified polymorphic DNA analysis. , Antonie Van Leeuwenhoek 86, 181-188.
- 63.Fischer A, Hauck F, Limpricht G, Luerssen C, Richter P et al. (1892) . Dr.L.Rabenhorst’sKryptogamen-Flora von Deutschland, Österreich und der Schweiz-Pilze-Phycomycetes 1, 1-505.
- 64.Went F A F C, Geerligs H C Prinsen. (1895) . Beobachtungen über die Hefearten und zuckerbildenden Pilze der Arackfabrikation. Verh. Kon. Ned. Akad. Wet Sect 2(4), 3-31.
- 65.Ellis J J. (1985) Species and varieties in the Rhizopus arrhizus-Rhizopus oryzae group as indicated by their DNA complementarity. , Mycologia 77, 243-247.
- 66.Abe A, Oda Y, Asano K, Sone T. (2007) Rhizopus delemar is the proper name for Rhizopus oryzae fumaric-malic acid producers. , Mycologia 99, 714-722.
- 68.A P Gryganskyi, S C Lee, A P Litvintseva, M E Smith, Bonito G et al. (2010) Structure, function, and phylogeny of the mating locus in the Rhizopus oryzae complex. , PLoS ONE 5, 15273.
- 69.M C Chibucos, Soliman S, Gebremariam T, Lee H, Daugherty S et al. (2016) An integrated genomic and transcriptomic survey of mucormycosis-causing fungi. , Nat. Commun 7, 12218.
- 70.A P Gryganskyi, Golan J, Dolatabadi S, Mondo S, Robb S et al. (2018) Phylogenetic and phylogenomic definition of Rhizopus species. , G3 (Bethesda) 8, 2007-2018.
- 71.R y Zheng, G Q Chen. (1998) Rhizopus microspores var. tuberosus var. nov . Mycotaxon.69: 181-186.
- 72.Ganesan A, Wells J, Shaikh F, Peterson P, Bradley W et al. (2019) Molecular detection of filamentous fungi in formalin-fixed paraffin-embedded specimens in invasive fungal wound infections is feasible with high specificity. , J. Clin. Microbiol 58(1), 01259-19.
- 73.Z U Khan, Ahmad S, Mokaddas E, Chandy R, Cano J et al. (2008) Actinomucor elegans var. kuwaitiensis isolated from the wound of a diabetic patient. Antonie Van Leeuwenhoek.94:. 343-352.
- 74.S C Jong, G F Yuan. (1985) Actinomucor taiwanensis sp. nov. for manufacture of fermented soybean food. , Mycotaxon 23, 261-264.
- 75.A, Guarro J, Guevara E, Pintos L, Sanchis M et al. (2014) Primary cutaneous mucormycosis produced by the new species Apophysomyces mexicanus. , Journal Clinical Microbiol.52; 4428-4431.
- 76.Ramani R, Newman R, Salkin L Li I F, Slim M, Arlievsky N. (2000) Cokeromyces recurvatus as a human pathogenic fungus: Case report and critical review of the published literature. Pediatr Infect Dis. , J 19, 155-158.
- 77.Hoffmann K, Discher S, Voigt K. (2007) Revision of the genus Absidia (Mucorales, Zygomycetes) based on physiological, phylogenetic, and morphological characters; thermotolerant Absidia spp. from a coherent group. Mycocladiaceae fam. nov. Mycol Res.111: 1169-1183.
- 78.Garcia-Hermoso D, Hoinard D, J C Gantier, Grenouillet F, Dromer F et al. (2009) Molecular and phenotypic evaluation of Lichtheimia corymbifera (formerly Absidia corymbifera) complex isolates associated with human mucormycosis: Rehabilitation of L. , ramosa. J. Clin. Microbiol 47, 3862-3870.
- 79.De A Santiago, Hoffmann A L C M, Lima K, Oliveira D X de, Vieira R J V et al. (2013) A new species of Lichtheimia (Mucoromycotina, Mucorales) isolated from Brazilian soil. , Mycol. Prog 13, 343-352.
- 80.Schwartze V U, Santiago A L, Jacobsen I D, Voigt K. (2014) The pathogenic potential of the Lichtheimia genus revisited: Lichtheimia brasiliensis is a novel, non pathogenic species. , Mycoses 57, 128-131.
- 81.Nottebrock H, H J Scholer, Wall M. (1974) Taxonomy and identification of mucormycosis-causing fungi I. Synonymity of Absidia ramosa with A. , corymbifera. Sabouraudia 12, 64-74.
- 82.E A Fenner. (1932) Mycotypha microspore, a new genus of the mucoraceae. , Mycologia 24(2), 187-198.
- 83.Padhye A A, Ajello L. (1988) Simple method of inducing sporulation by Apophysomyces elegans and Saksenaea vasiformis,”. , Journal of Clinical Microbiology 26(9), 1861-1863.
- 84.Labuda R, Bernreiter A, Hochenauer D, Schuller C, Kubatova A et al. (2019) Saksenaea dorisiae sp.nov. a new opportunistic pathogenic fungus from Europe. , International Journal of Microbiology
- 85.Baby Sabitha, T G Ramya, R K Geetha. (2015) Onychomycosis by Syncephalastrum racemosum; case report from Kerala, India. Dermatology Reports. 7(1).
- 86.Irshad M, Nasir N, U H Hashmi, Farooqi J. (2020) Invasive pulmonary infection by Syncephalastrum species. Two case reports and review of literature. IDCases.21. e 00913. Doi. 10.1016/j.idcr.2020e 00913.
- 88.Spellberg B, Edwards J, Ibrahim. (2005) A Novel perspectives on mucormycosis: Pathophysiology, presentation, and management. , ClinMicrobiolRev 18, 556-69.
- 89.Petrikkos G, Skiada A, Lortholary O, Roilides E, Walsh T J et al. (2012) Epidemiology and clinical manifestations of mucormycosis external icon. Clin Infect Dis. 54 Feb. , Suppl 1, 23-34.
- 90.Soare A Y, TNWatkins, Bruno V M. (2020) Understanding mucormycosis in the age of Omics. , Front. Genet. Doi 10.
- 91.Sarvestani A S, Pishdad G, Bolandparvaz S. (2013) Predisposing factors for mucormycosis in patients with diabetes mellitus: An experience of 21 years in southern Iran. , Bull Emerg Trauma 1(4), 164-170.
- 92.Neville W B, Damm D, Allen C M, Bouquot J E. (2001) . Text Book of Oral & Maxillofacial Pathology; 2nd ed. Philadelphia In: W.B.: Saunders 16.
- 93.Goel A, Kini U, Shetty S. (2010) Role of histopathology as an aid to prognosis in rhino-orbito-cerebral zygomycosis. , Indian J. of pathology and Microbiology 53(2), 253-257.
- 94.Nosari A, Oreste P, Montillo M. (2000) Mucormycosis in hematologic malignancies: an emerging fungal infection. , Haematologica 85, 1068-1071.
- 96.Marx R E, Stern D. (2003) . In: Inflammatory, Reactive and Infectious Diseases in Oral and Maxillofacial Pathology; editors. Carol Stream III, USA: 104-6.
- 97.Artis W M, Fountain J A, Delcher H K, H E Jones. (1982) A mechanism of susceptibility to mucormycosis in diabetic ketoacidosis: Transferrin and iron availability. Diabetes.31: 1109-1114.
- 98.S N Afroze, Korlepara R, G V Rao, Madala J. (2017) Mucormycosis in a diabetic patients: A case report with an insight into its pathophysiology. Contemporary Clinical Dentistry. 8(4), 662-664.
- 99.J L Hwang, R E Weiss. (2014) Steriod induced diabetes: a clinical and molecular approach to understanding and treatment. , Diabetes Metab Res.Rev 30(2), 96-102.
- 100.S M Trifilio, C L Bennett, P R Yarnold. (2007) Breakthrough mucormycosis after voriconazole administration among patients with hematologic malignancies who receive hematopoietic stem-cell transplants or intensive chemotherapy. , Bone Marrow Transplant 39, 425-429.
- 101.R G Vitale, Hoog G S de, Schwarz P, Dannaoui E, Deng S et al. (2012) Antifungal susceptibility and phylogeny of opportunistic members of the order Mucorales. , J. Clin. Microbiol 50, 66-75.
- 102.Patel R. (2019) A moldy application of MALDI: MALDI-ToF Mass Spectrometry for fungal identification. , J. Fungi 5, 4.
- 103.Dolatabadi S, Kolecka A, Versteeg M, Hoog S G de, Boekhout T. (2015) Differentiation of clinically relevant Mucorales Rhizopus microspores and R. arrhizus by matrix-assisted laser desorption ionization time-of-flight mass spectrometry (MALDI-TOF-MS). , J. Med. Microbiol 64, 694-701.
- 104.Rychert J, E s Slechta, A P Barker, Miranda E, N E Babady et al. (2018) system for the identification of filamentous fungi. Multicenter evaluation of the Vitek MS v3.0 , J. Clin. Microbiol 56(2).
- 105.Shao J, Wan Z, Li R, Yu J. (2018) Species identification and delineation of pathogenic Mucorales by matrix-Assisted Laser Desorption Ionization-Time of Flight Mass Spectrometry. J.Clin. Microbiol.56 (4)
- 106.Machouart M, Larche J, Burton K, Collomb J, Maurer P et al. (2006) Genetic identification of the main opportunistic Mucorales by PCR-restriction fragment length polymorphism. , J.Clin. Microbiol 44, 805-810.
- 107.Bialek r, Konrad F, Kern J, Aepinus C, Cecenas L et al. (2005) PCR based identification and discrimination of agents of mucormycosis and aspergillosis in paraffin wax embedded tissue. , J. Clin. Pathol 58, 1180-1184.
- 108.S P Hammond, Bialek R, D A Milner, E M Petschnigg, L R Baden et al. (2011) Molecular methods to improve diagnosis and identification of mucormycosis. , J. Clin. Microbiol 49, 2151-2153.
- 109.Bernal-Martinez L, M J Buitrago, M V Castelli, J L Rodriguez-Tudela, Cuenca-Estrella M. (2013) Development of a single tube multiplex real-time PCR to detect the most clinically relevant Mucormycetes species. , Clin. Microbiol. Infect 19, 1-7.
- 110.D J Hata, S P Buckwalter, B S Pritt, G D Roberts, L Wengenack 2008 N.Real-time PCR method for detection of Zygomycetes. , J. clin. Microbiol 46, 2353-2358.
- 111.Alanio A, Garcia-Hermoso D, Mercier-Delarue S, Lanternier F, Gits-Muselli M et al. (2015) Molecular identification of Mucorales in human tissues: contribution of PCR electrospray-ionization mass spectrometry. , Cli. Microbiol. Infect 21, 594-594.
- 112.Baldin C, Soliman S S M, H, Alkhazraji S, Gebremariam T et al. (2018) PCR-based approach targeting Mucorales-specific gene family for diagnosis of mucormycosis. , J. Clin. Microbiol 56(10), 00746-18.
- 113.Zhao Z, Li L, Wan Z, Chen W, Liu H et al. (2011) Simultaneous detection and identification of Aspergillus and Mucorales species in tissues collected from patients with fungal rhinosinusitis. , J. Clin. Microbiol 49, 1501-1507.
- 114.Potenza L, Vallerini D, Barozzi P, Riva G, Gilioli A et al. (2016) Mucorales-specific T cells in patients with hematologic malignancies. , PLoS ONE 11, 0149108.
- 115.D Macedo F, Dudiuk C, Theill L, Cabeza M, Gamarra S et al. (2019) In vitro activity of combination of Zinc-Chelators with Amphotericin B and Posaconazole against six mucorales species. Antimicrobial Agents and Chemotherapy. 63.
- 116.L A Caetano, Faria T, Springer J, Loeffler J, Viegas C. (2019) Antifungal resistant Mucorales in different indoor environment. , Mycology 10(2), 75-83.
- 117.Macedo D, Leonardelli F, Dudiuk C, Garcia-Effron G. (2018) Molecular confirmation of the linkage between Rhizopus oryzae CYP51A gene coding region and its intrinsic Voriconazole and Fluconazole resistance. Antimicrobial Agents and Chemotherapy. 62(8).
- 118.Dannaoui E. (2017) Antifungal resistance in mucorales. , International Journal of Antimicrobial Agents 50(5).
- 119.A Chakrabarti, Chowdhary A, Cordoba S, Dannaoui E, Dufresne P et al. (2015) Multicenter evaluation of MIC distribution for Epidemiologic cutoff value definition to detect Amphotericin B, Posaconazole and Itraconazole resistance among the clinically relevant species of Mucorales. Antimicobial Agents and Chemotherapy. 59(3), 1745-1750.
- 120.Barnert J, Behr W, Reich H. (1985) An Amphotericin B resistant case of Rhinocerebral mucormycosis. Infection.13: 134-136.
- 121.A Kotwal D, kakati B, Ahmad S. (2015) Amphotericin B resistant Apophysomyces elegans causing Rhino-oculo-Cerebral mucormycosis in an immunocompetent host. , Journal of Clinical & Diagnostic Research 9(8), 10-7860.
- 122.S A Asghar, Majid Z, Tahir F, L T Qadar, Mir S. (2019) Rhino-oculo-cerebral mucormycosis resistant to Amphotericin B in a young patient with Diabetic ketoacidosis. , Cureus 11(3), 4295-10.
- 123.S Kiss G, Verghese K Bauer R, Szebenyi C, Kocsube S, Homa L Bodai M et al. (2021) Characterization of three pleiotropic drug resistance transporter genes and their participation in the Azole resistance of Mucor circinelloides. Frontiers in Cellular and Infection Microbiology.
- 125.Karimi K, Arzanlou M, Ahari A B, Ghazi M M. (2015) Phenotypic and molecular characterization of the causal agent of chafer beetle mortality in the wheat fields of the Kurdistan province. , Iran. J. Plant Prot. Res 55(3).
- 126.G L Benny, R A Humber, J B Morton. (2001) Zygomycota: Zygomycetes. In: Systematics and Evolution;. , McLaughlin, D.J., McLaughlin, E.G., Lemke, P.A., Eds, Springer: Berlin/Heidelberg, Germary
- 127.Zheng R, Jiang H. (1995) Rhizomucor endophyticus sp. nov. an endophytic zygomycetes from higher plants. , Mycotaxon 56, 455-466.
- 128.G L Benny, M E Smith, P M Kirk, E D Tretter, M. (2016) Challenges and future perspectives in the systematics of Kickxellomycotina, Mortierellomycotina, Mucoromycotina, and Zoopagomycotina. In : Biology of Microfungi; , Berlin, Germany 65-126.
- 129.Zycha H, Siepmann R. (1969) . Mucorales. Eine Beschreibung aller Gattungen und Arten dieser Pilzgruppe I; Lubrecht & Cramer Ltd.: , Lehre, Germany .
- 130.Souza C A de, D X Lima, L M Gurgel, A L Santiago. (2017) Coprophilous Mucorales (ex Zygomycota) from three areas in the semi-arid of Pernambuco. , Brazil, Braz. J. Microbiol 48, 79-86.
Cited by (24)
This article has been cited by 24 scholarly works according to:
Citing Articles:
Frontiers in Microbiology (2024) Crossref
Bo Song, M. Raza, Lijuan Zhang, Bing-Qiang Xu, Pan Zhang et al. - Frontiers in Microbiology (2024) Semantic Scholar
Frontiers in Microbiology (2024) OpenAlex
World Academy of Sciences Journal (2024) Crossref
World Academy of Sciences Journal (2024) OpenAlex
Biologia (2024) Crossref
Insha Amin, Riasa Zaffar, Ruqeya Nazir, M. Rather - Biologia (2024) Semantic Scholar
Biologia (2024) OpenAlex
European Journal of Medical Research (2024) Crossref
Xiaobo Yang, Xue-hui Gao, Hongling Zhang, Jiqian Xu, You Shang - European Journal of Medical Research (2024) Semantic Scholar
European journal of medical research (2024) OpenAlex
ACS Omega (2024) Crossref
Valdemir Vargas-Junior, Ana Carolina Ramos Guimarães, E. Caffarena, Deborah Antunes - ACS Omega (2024) Semantic Scholar
ACS Omega (2024) OpenAlex
Brazilian Journal of Microbiology (2024) Crossref
Brazilian Journal of Microbiology (2024) OpenAlex
Sunita Panda, M. C. Sahu, Jyotimayee Turuk, Sanghamitra Pati - Brazilian Journal of Microbiology (2024) Semantic Scholar
International Journal of Science and Research Archive (2024) OpenAlex
Heliyon (2024) Crossref
Komal Tilwani, Drashti Patel, Prachi Soni, Shyam Wadhwani, Gayatri Dave - Heliyon (2024) Semantic Scholar
Heliyon (2024) OpenAlex
Expert Review of Molecular Diagnostics (2024) Crossref
Botany (2023) OpenAlex
Botany (2023) Crossref
C. Thornton - Expert Review of Molecular Diagnostics (2023) Semantic Scholar
Expert Review of Molecular Diagnostics (2023) OpenAlex
Energy and Earth Science (2022) OpenAlex
ChemistrySelect (2022) Crossref
ChemistrySelect (2022) OpenAlex
Journal of Infection and Public Health (2022) Crossref
N. Shah, Zaid Khan, Hashim Ahad, Abozer Y. Elderdery, M. Alomary et al. - Journal of Infection and Public Health (2022) Semantic Scholar
Journal of Infection and Public Health (2022) OpenAlex
