Abstract
The safety of cyclooxygenase (COX)-2 inhibitors has been tested in patients who had cross-reactive hypersensitivity reactions (HSRs) to nonsteroidal anti-inflammatory drugs (NSAIDs). However, these studies have been mainly done before the current classification of NSAID hypersensitivity and cross-reaction between COX-2 inhibitors has been rarely reported.We aimed to assess tolerability of COX-2 inhibitors and to evaluate the cross-reactivity between them in cross-reactive phenotype of NSAID hypersensitivity. The diagnosis was based on clinical features, reliable history of HSRs to at least two chemically different NSAIDs, and/or positive provocation tests with implicated NSAIDs in 151 patients. Single-blind, oral challenges with 1/4 and 3/4 divided doses of placebo, nimesulide, meloxicam, and celecoxib, as COX-2 inhibitors, were performed. The most common cross-reactive phenotype was NSAID-induced urticaria/angioedema (56.3%). Positive reactions to meloxicam, nimesulide, and celecoxib challenges were observed in 23/140 (16.4%), 7/33 (21.2%), and none of six patients, respectively. Overall, 24 patients were tested with two, one was tested with three COX-2 inhibitors. Six (31.6%) of 19 patients with meloxicam intolerance reacted to nimesulide provocation. Nimesulide, meloxicam, and celecoxib appeared safe alternatives in cross-reactive phenotypes of NSAID hypersensitivity. Although celecoxib has the most favorable tolerability, cross-reactivity among COX-2 inhibitors seems to be possible.
Author Contributions
Copyright © 2022 Selcan Gültuna, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
In allergy practice, the NSAIDs family is probably one of the most intriguing drug families implicated in hypersensitivity reactions (HSRs) due to differences in chemical structures and pharmacological properties with wide-pattern symptoms, different organ involvements, different putative mechanisms, and cross-reactivity between chemically unrelated NSAIDs. The prevalence of HSRs to NSAIDs, which vary in severity, clinical presentations, and frequency has been reported between 0.6 and 5.7% in the general population 1, 2.In our experience, NSAIDs were the second most frequently involved drugs (28%) to the self-reported HSRs (4.7%) among medical students 3. Certain underlying conditions, such as asthma, nasal polyposis, and chronic urticaria, have been associated with increases in HSRs to NSAIDs 4, 5, 6.
The main putative mechanisms which are responsible for NSAID-induced HSRs are immunological (immunoglobulin E mediated, and T cell-mediated) and non-immunologically (cyclooxygenase (COX)-1 inhibition) mediated mechanisms. Nonimmunologically mediated HSR to NSAIDs, which are acute reactions, have cross-reactivity among chemically nonrelated COX-1 inhibitors 4. Kowalski et al. classified cross-reactive reactions into three subgroups based on the clinical manifestation and the presence or absence of underlying disease as a result of the need for a lingua franca on the definition of types of NSAID hypersensitivity due to previously used various terms. These renamed reactions are NSAIDs-exacerbated respiratory disease (NERD), NSAIDs-exacerbated cutaneous disease (NECD), and NSAIDs-induced urticaria/angioedema (NIUA). In more detail, NERD is defined as NSAID-induced respiratory symptoms in patients with underlying chronic respiratory diseases. Exacerbation of urticaria or angioedema after NSAIDs consumption in patients with chronic urticaria is defined as NECD. NSAID-induced urticaria/ angioedema in otherwise healthy cases is defined as NIUA 5.
Considering COX-1 inhibition as the main mechanism underlying the cross-reactive HSRs to NSAID, it is expected that COX-2 inhibitors would be suitable alternatives. Cyclooxygenase-2 inhibitors, which are the other isoform of the COX enzyme, have higher anti-inflammatory effects and fewer adverse reactions than COX-1 inhibitors 7. In keeping with that, a number of studies have been reported about the safety of COX-2 inhibitors in patients with NSAID hypersensitivity 8, 9, 10, 11, 12, 13, 14. However, these studies have been mostly done before the current classification of NSAID hypersensitivity and cross-reaction between COX-2 inhibitors has been rarely the subject of these studies 8, 15, 16. Therefore, in the present study, we aimed to evaluate the tolerability of COX-2 inhibitors, including nimesulide, meloxicam, and celecoxib, in the currently defined cross-reactive phenotype of NSAID hypersensitivity. Additionally, we aimed to evaluate the cross-reactivity between these COX-2 inhibitors.
Methods
Study Subjects
This study retrospectively evaluated the data of adult patients with cross-reactive hypersensitivity to NSAIDs who underwent drug provocation tests to find safe alternatives with meloxicam, nimesulide, and/or celecoxib between January 2014- December 2021 in a tertiary immunology and allergy clinic. The procedures in the present study were performed in accordance with the Helsinki Declaration and this study was approved by the local Ethics Committee (date-no: 25.01.2022-2022/48).
Patients’ age, gender, presence of characteristic underlying diseases such as asthma, rhinitis, chronic urticaria, and nasal polyposis, family history of allergy, and history of additional drug allergy were recorded. The features of HSRs to NSAIDs regarding the related symptoms and timing of reactions, the number of episodes, and the number of chemical subgroups of NSAIDs involved in the episode were detailed.
Patients who had nonimmunological (cross-reactive) HSRs to NSAIDs, as defined by the EAACI/ENDA classifications were included. Immunoglobulin (Ig) E-mediated reactions (urticaria/angioedema, bronchospasm, laryngeal edema, rhinitis, and anaphylaxis) to single NSAID or NSAIDs belonged to the same chemical group; and T cell-mediated reactions (maculopapular eruption, fixed drug eruption, photosensitivity reactions, contact dermatitis, Steven-Johnson Syndrome/toxic epidermal necrolysis, acute generalized exanthematous pustulosis, drug-induced hypersensitivity syndrome, other organ-specific reactions, and DRESS) were excluded 5.
Evaluation of Atopy
Atopy was assessed by the skin-prick test (SPT) which was performed by a common inhalant panel (ALK, Madrid, Spain or Allergopharma, Stockholm, Sweden) along with histamine at 10 mg/mL as positive controls, and normal saline as a negative control. A wheal diameter of 3 mm greater than the negative control was considered positive. The patients were classified as “atopic” if the skin tests were positive.
Drug Provocation Tests with Aspirin, Other Culprit Nsaids, And Alternative COX-2 Inhibitors
Confirming the diagnosis with aspirin or putative NSAID, and identifying the safe alternative COX-2 inhibitor, single-blind and placebo-controlled oral drug provocation tests (OPT) were carried out in the equipped hospital setting.
The oral provocation test with aspirin was performed according to the protocol of Nizankowska et al. 17. On the first day, three placebo tablets were administered at 90 minutes intervals. On the second day, four increasing doses (27 mg, 44 mg, 117 mg, and 312 mg) were given at 90 minutes intervals until the cumulative dose of 500 mg was reached. Drug provocation tests with other NSAIDs were done with the protocols reported previously 18, 19, 20.
For OPT with COX-2 inhibitors, on two consecutive days, placebo and active drug were given one-fourth and three-fourths of the therapeutic doses at 60 minutes intervals, respectively. As alternative COX-2 inhibitors; nimesulide (100 mg Nimes; Sanovel/Turkey), celecoxib (200 mg Celgyn/Turkey), and meloxicam (7.5mg Melox, Nobel/Turkey) were used.
During these provocation tests, patients were monitored in terms of skin, ocular, nasal, and bronchial reactions, blood pressure, and values of forced expiratory volume in one second (FEV1). Patients were monitored for two hours after the last challenge dose and were followed up for 24 hours to define a delayed reaction. If clinical symptoms or changes in vital signs (rhythm alterations, decrease in FEV1, or hypotension) were considered as positive reactions, the procedure was stopped, and symptoms. Test results were categorized as negative, upper airway reaction, classic reaction, systemic and cutaneous reaction. The presence of nasal symptoms was defined as upper airway reaction; more than 20% decrease in FEV1 associated with upper airway reaction as a classic reaction; and extra bronchial symptoms such as hoarseness, redness of the face and upper chest, ocular injection, and/or periorbital swelling, nausea, vomiting and stomach cramps, and hypotension with or without cutaneous/bronchial symptoms as a systemic reaction. The presence of urticaria/angioedema was defined as the cutaneous reaction.
In the lack of OPT, a clear history of at least >2 respiratory or cutaneous reactions, occurring up to 6 hours of administration of chemically unrelated NSAIDs was considered positive in favor of NSAID hypersensitivity 21.
Statistical Analysis
The statistical analysis was performed using the Statistical Package for Social Sciences (SPSS) for Windows, version 11.5 (SPSS Inc., Chicago, IL, US). Continuous variables were stated as mean ± standard deviation (S.D) or the median (minimum-maximum) considering normality tests. Categorical variables were summarized as counts (percentages). The Chi-square test or Fisher’s exact test, where appropriate, was used to compare these proportions in different groups. As age was not normally distributed, Kruskal-Wallis tests were conducted to compare this parameter between the groups. A two-sided p-value of < 0.05 was considered statistically significant.
Results
A total of 151 patients (female/male:95/56) with a mean age of 42.5±12.5 years (min:18-max:74) were included in the study. Diagnosis of cross-reactive hypersensitivity to NSAIDs was confirmed in 19 patients with OPT. 10/151 (6.6%) of the study participants had a positive aspirin provocation test, and 9/151 (5.9%) had a positive DPT to the culprit NSAID (metamizole: n=5 and paracetamol: n=4). In one patient (0.6%), the diagnosis of NSAID hypersensitivity was based on a severe systemic reaction after aspirin consumption. The remainder of the study group (n=131/151, 86.8%) had a clear clinical history of at least two episodes with at least two different chemical groups of NSAIDs. Aspirin provocation test symptoms included urticaria (n=3), asthma (n=2), rhinitis and asthma (n=2), urticaria with angioedema (n=2), and rhinitis (n=1). Culprit drug provocation test symptoms included urticaria and angioedema (n=2), asthma (n=1), rhinitis (n=1), and anaphylaxis (n=1) with metamizole, and asthma (n=1) and urticaria (n=3) with paracetamol.
The most common cross-reactive phenotype was NIUA (n=85, 56.3%). 41 (27.2%) patients were in the NECD group, and 25 (16.6%) patients were in the NERD group. There were not any significant differences between the phenotype groups regarding gender (the female percentage was 69.6% in NECD, 66.6% in NERD, and 61.1% in NIUA; p=0.660), and age (44.7±14.4 in NERD, 43.1±11.8 in NIUA, and 39.7±12.4 in NECD; p=0.218) (Table 1). The prevalence of underlying diseases was as follows; chronic urticaria (n=41, 27.2%), asthma and nasal polyposis (n=12, 48.5%), asthma (n=7, 28%), asthma, nasal polyposis and rhinitis (n=4, 16%), and asthma and rhinitis (n=2, 8%). Skin prick test to inhalant allergen was positive in 33 of 119 (27.2%) patients. Twenty-three (19.3%) patients were atopic with mite allergen being the most frequent inhalant allergen. Twelve (10.1%) patients were atopic with pollens, a patient (0.8%) with moulds and animal dander (n=1, 0.8%).
The most implicated NSAIDs among whole group study was flurbiprofen (n=76, 50.3%), followed by paracetamol (n=68, 45%), naproxen (n=55, 36.4%), diclofenac (n=49, 33.5%), metamizole (n=46, 30.5%), aspirin (n=44, 29.1%), ketoprofen (n=39, 25.8%), ibuprofen (n=18, 11.9%), etodolac (n=9, 6%), propifenazone (n=6, 4%), salicylsalicylic acid (n=3, 2%), etophenamate (n=2, 1.3%), acemetacin (n=1, 0.7%), indomethacin (n=1, 0.7%), and benzydamine HCL (n=1, 0.7%). The most frequently implicated NSAIDs were shown in the whole group and the subgroups in figure 1 (A-D), separately. Flurbiprofen was the most implicated compound in NIUA (n=41, 48.2%) and NECD (n=24, 58.5%), whereas aspirin was the most implicated compound in NERD (n=13, 52%) (Table 1). Among three cross-reactive phenotypes, the reaction rates of ketoprofen and aspirin were statistically significant in the NIUA phenotype (the rates were shown in figure 1 (A-D); p=0.033 and 0.010). 41 (25.2%) patients had additional drug allergies; antibiotic (n=27, 66.9%), muscle relaxants (n=5, 12.2%), anesthetics (n=1, 2.4%), antiepileptic (n=1, 2.4%), antidiabetic (n=1, 2.4%), proton pomp inhibitor (n=2, 4.8%), enoxaparin (n=1, 2.4%), vitamin B-C complex (n=1, 2.4%), and missing data (n=1, 2.4%).
Figure 1A-D. NSAID causing cross-reactive hypersensitivity reactions*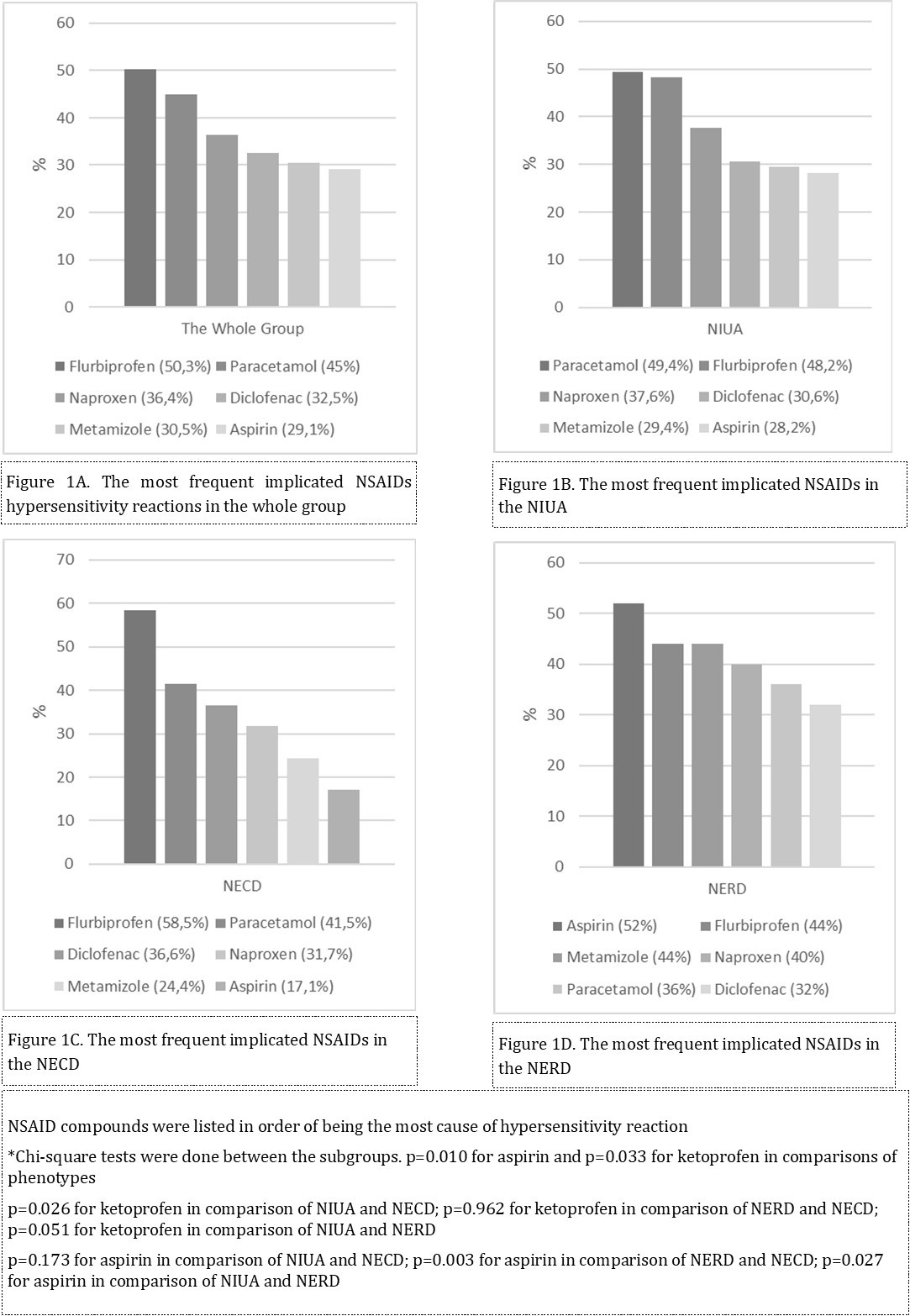
Meloxicam Provocation
Among the whole group, 117 of 140 (83.6%) challenged patients have perfectly tolerated meloxicam. All reactions on the meloxicam provocations occurred with a total dose of 7.5 mg of meloxicam. The rates of positive provocation tests among the phenotypes were as follows; 29.2% in NERD (n=7/24), 24.3% in NECD (n=9/37), and 8.9% in NIUA (n=7/79). Urticaria with/without angioedema (12/23, 52.2%) was the most observed reaction to meloxicam provocation (Table 1).
Patients with meloxicam intolerance were reactive to the following NSAIDs, ketoprofen (n=8/34, 23.5%), diclofenac (9/43, 20.9%), aspirin (9/44, 20.5%), metamizole (7/43, 16.3%), naproxen (7/54, 16.3%), flurbiprofen (11/71, 15.5%), paracetamol (9/65, 13.8%), etodolac (n=1/1, 100%), ibuprofen (n=1/1, 100%), benzydamine HCL (n=1/1, 100%), and salicylic acid (n=1/1, 100%).
Nimesulide Provocation
Among the whole group 26 of 33 (78.8%) nimesulide challenged patients were tolerant to nimesulide. Seven (21.2%) patients experienced an HSR with nimesulide, the majority of which were cutaneous reactions (Table 1).
Table 1. Frequencies of implicated NSAIDs according to cross-reactive subgroups, and tolerability of COX-2 inhibitors| NIUA(n=85) | NECD(n=41) | NERD(n=25) | P | |
| Age (mean±SD), years | 43.1 ±11.8 | 39.7±12.4 | 44.7±14.4 | 0.218 |
| Gender (female), n (%) | 51 (60) | 28 (68.3) | 16 (64) | 0.660 |
| Atopy, n (%) | 15/61 (24.6) | 10/36 (27.8) | 8/22 (36.4) | 0.138 |
| Paracetamol tolerability in history, n (%) * | 43/85 (50.6) | 24/42 (58.5) | 16/25 (64.0) | |
| Meloxicam tolerability to provocation, n (%) | 72/79 (91.1) | 28/37 (75.7) | 17/24 (70.8) | |
| Symptoms of Meloxicam provocation (n=23) | Urticaria±AO (3)Urticaria (3)AO (1) | Urticaria (6)Urticaria±AO (1)Asthma±rhinitis (1)AO (1) | Rhinitis (4)Asthma (2)Anaphylaxis (1) | |
| Nimesulide tolerability, n (%) | 11/13 (84.6) | 8/11 (72.7) | 7/9 (77.8) | |
| Symptoms of Nimesulide provocation (n=7) | Urticaria±AO (2) | Urticaria (2)AO (1) | Rhinitis (2) | |
| Celecoxib tolerability, n (%) | 2/2 (100) | 3/3 (100) | 1/1 (100) | |
| Meloxicam plus Nimesulide tolerability, n (%) | 2/6 (33.3) | -/6 | 2/9 (22.2) |
Patients with nimesulide intolerance were hypersensitive to the following COX-1 inhibitor compounds, naproxen (n=2/6, 33.3%), ketoprofen (n=4/13, 30.8%), diclofenac (n=4/16, 25%), paracetamol (n=2/11, 18.2%), metamizole (n=2/11, 18.2%), aspirin (n=1/8, 12.5%), flurbiprofen (n=1/16, 6.3%), and benzydamine HCL (n=1/1, 100%).
Celecoxib Provocation
Only 6 patients challenged with celecoxib and none reacted.
Meloxicam vs Nimesulide vs Celecoxib
A total of 24 patients were tested with two COX-2 and one patient was tested with three COX-2 inhibitors. Six (31.6%) of 19 meloxicam intolerant patients reacted to nimesulide provocation as well (Table 2). There was no patient with nimesulide positive but meloxicam negative on OPT and only one patient was challenged with three COX-2 inhibitors, meloxicam was reactive but tolerated nimesulide and celecoxib provocation.
Table 2. Characteristics of patients who reacted to at least two COX-2 inhibitors| Patient | Age, year (gender) | Atopy | Implicated NSAID | Phenotype | Meloxicam | Nimesulide | Celecoxib | Other Drug Allergies |
| 1 | 19 (F) | Negative | Aspirin* Metamizole | NERD (Asthma+NP) | Positive (rhinitis) | Positive (rhinitis) | Not tested | No |
| 2 | 35 (F) | Negative | Naproxen Ketoprofen* | NERD (Asthma+NP) | Positive (rhinitis) | Positive (rhinitis) | Not tested | No |
| 3 | 58 (M) | Negative | Aspirin Flurbiprofen Metamizole* | NERD (Asthma+NP) | Positive (rhinitis) | Negative | Not tested | No |
| 4 | 51 (F) | Positive (mite) | Aspirin Flurbiprofen Ketoprofen | NERD (Asthma+NP +rhinitis) | Negative | Negative | Not tested | No |
| 5 | 39 (M) | Positive (Pollen) | Aspirin* Flurbiprofen | NERD (Asthma+NP +rhinitis) | Positive (asthma) | Negative | Not tested | No |
| 6 | 42 (F) | Negative | Flurbiprofen Ketoprofen | NERD (Asthma+rhinitis) | Positive (asthma) | Negative | Negative | No |
| 7 | 34 (M) | Positive (mite) | Aspirin* Diclofenac Etodolac | NERD (Asthma+ NP) | Positive (anaphylaxis) | Negative | Not tested | No |
| 8 | 47 (M) | Negative | Diclofenac Ketoprofen | NECD | Positive (urticaria) | Positive (urticaria) | Not tested | Muscle relaxant |
| 9 | 63 (M) | Not tested | Diclofenac Metamizole | NECD | Negative | Not tested | Negative | No |
| 10 | 30 (F) | Positive (venom) | Diclofenac Flurbiprofen İbuprofen Ketoprofen | NECD | Not tested | Negative | Negative | No |
| 11 | 33 (M) | Positive (Cat dander) | Ketoprofen Diclofenac | NECD | Not tested | Positive (urticaria) | Negative | No |
| 12 | 31 (F) | Negative | Diclofenac Ketoprofen Metamizole | NECD | Positive (AO) | Positive (AO) | Not tested | No |
| 13 | 45 (F) | Negative | Diclofenac Metamizole | NECD | Positive (asthma+rhinitis) | Negative | Not tested | No |
| 14 | 36 (F) | Not tested | Diclofenac Flurbiprofen Metamizole | NECD | Positive (urticaria) | Negative | Not tested | No |
| 15 | 49 (F) | Positive (mite) | Aspirin Flurbiprofen | NECD | Positive (urticaria) | Negative | Not tested | No |
| 16 | 27 (F) | Negative | Diclofenac İbuprofen | NECD | Positive (urticaria) | Negative | Not tested | Antibiotic |
| 17 | 26 (M) | Negative | Metamizole Ketoprofen | NIUA | Positive (urticaria) | Negative | Not tested | No |
| 18 | 39 (F) | Positive (mite) | Flurbiprofen Naproxen | NIUA | Negative | Negative | Not tested | No |
| 19 | 28 (F) | Negative | Diclofenac Naproxen | NIUA | Negative | Negative | Not tested | No |
| 20 | 32 (M) | Negative | Aspirin Flurbiprofen | NIUA | Positive (urticaria) | Negative | Not tested | No |
| 21 | 26 (F) | Positive (mite) | Diclofenac Metamizole | NIUA | Positive (AO) | Negative | Not tested | No |
| 22 | 45 (M) | Not tested | Flurbiprofen Naproxen | NIUA | Positive (urticaria+AO) | Negative | Not tested | No |
| 23 | 32 (M) | Negative | Diclofenac Naproxen | NIUA | Positive (urticaria) | Positive (urticaria+AO) | Not tested | Antibiotic |
| 24 | 50 (F) | Positive (Pollen) | Benzidamin Hcl Flurbiprofen | NIUA | Positive (urticaria+AO) | Positive (urticaria+AO) | Not tested | Antibiotic |
| 25 | 48 (F) | Negative | Aspirin Diclofenac Naproxen | NIUA | Positive (urticaria+AO) | Negative | Not tested | No |
Discussion
In the present study, nimesulide, meloxicam, and celecoxib appeared safe alternatives in cross-reactive phenotypes of NSAID hypersensitivity. Positive reactions to the meloxicam, nimesulide, and celecoxib challenges were observed in 23/149 (16.4%), 7/33 (21.2%), and none of the patients, respectively. To the best of our knowledge, this is the first study comparing these drugs using the recent ENDA classification.
COX-2 inhibitors are classified as preferential or specific based on their selectivity to the COX-2 enzyme 22. In our country, nimesulide and meloxicam as preferential COX-2 inhibitors are available, and celecoxib as a selective COX-2 inhibitor became currently available. Because of the longer availability, meloxicam and nimesulide were the two most common COX-2 inhibitors evaluated as the safe alternative. Although being the least evaluated alternative, celecoxib was the most tolerable alternative in the present study. In terms of selective COX-2 inhibitors, in our previous study, only one patient (1.1%) presented urticarial-type cutaneous reaction to rofecoxib challenge, and the remaining patients (98.9%) with all types of NSAID hypersensitivity perfectly tolerated the drug challenge with rofecoxib 9. Similarly, none of the patients with a history of urticaria/angioedema, naso-ocular symptoms, bronchospasm, and/or anaphylactoid reactions induced by aspirin and/or NSAIDs had reacted to celecoxib provocation 23. The present study was consistent with these data and all patients within each cross-reactive group perfectly tolerated celecoxib. On the other hand, meloxicam was more tolerable than nimesulide. We have also evaluated the safety of two preferential COX-2 inhibitors in our previous studies and reported the safety of these drugs in NSAIDs hypersensitive subjects. In these studies, subjects with a history of different clinical presentations of HSRs to aspirin/NSAIDs received oral challenges with nimesulide and meloxicam. Positive reactions to the nimesulide and meloxicam challenges were observed varying from 8.3% to 14.3%, and from 3.7% to 8.1% respectively 8, 10, 11, 12. Patients in these studies had not been included based on the current classification of NSAID hypersensitivity, however, data were consistent with the results reported in the present study in which COX-2 inhibitors were tested on well-defined cross-reactive subgroups of NSAIDs hypersensitivity.
Evaluation of cross-reactivity between COX-2 inhibitors in the cross-reactive phenotype of NSAID hypersensitivity has been the subject of very limited studies. Our previous study was the first placebo-controlled report comparing nimesulide, meloxicam, and rofecoxib. In those days, celecoxib was not on the market in our country. In that study, 37 patients among a total of 127 subjects have been challenged with all three drugs. Three patients have reacted to more than one of the drugs tested, one of them reacted to all drugs and rofecoxib appeared to have the most favorable tolerability 8. Consistently, in the present study, selective COX-2 inhibitors celecoxib appeared to be the most tolerated drug by all cross-reactive groups, but the number of subjects tested with celecoxib was limited. In a Spanish study, 8 patients with COX-2 inhibitor intolerance have been retrospectively evaluated. All of the patients have reacted to meloxicam (angioedema or cutaneous rash), and 6 patients have reacted to celecoxib. They concluded that although their series was small, it is striking that all of the patients were women, a large percentage were also allergic to other drugs, and they all had mucosal or cutaneous involvement 15. In our study, only six patients had cross-reactivity to two COX-2 inhibitors, four were women, and none were atopic. In terms of phenotypes, patients were evenly distributed in pairs. Two patients with NIUA phenotype had an antibiotic allergy and one patient had DHR with a muscle relaxant.
Considering the cross-reactive phenotypes, the meloxicam and nimesulide tolerability was relatively higher in the NIUA group. The second most tolerability was observed in NECD for meloxicam, while NERD for nimesulide. The types of positive reactions during the provocation tests were consistent with the underlying phenotypes as in the previous study 8. The frequency of HSR to meloxicam and nimesulide was found relatively higher in the NECD phenotype attributing to the possibility of reactivation of preexisting urticaria 19. Remarkably, serious HSRs to COX-2 inhibitors were observed in the NERD phenotype that might be explained by the arachidonic acid metabolic pathway which may be induced by COX-2 inhibitors 24. We believe that these findings would shed light on the choice of safe alternatives. The tolerability and cross-reactivity of COX-2 inhibitors such as nimesulide, meloxicam, and celecoxib with relevance to NSAID hypersensitivity may be determined by the inactivation of critical genes that are important to immune system 25.
Our study outcomes regarding age, gender, the frequency of cross-reactive phenotypes of NSAIDs, and atopy ratio were compatible with the previous data from our country 8, 9, 26, 27 and also was similar to the general adult population of Turkey 28. Although no significant differences were found between groups, the atopy ratio was relatively higher in the NERD phenotype with mite sensitivity predominancy.
The culprit NSAIDs involved in the HSRs might vary among countries and different periods of time due to the preferences for the prescription. Metamizole and aspirin were the most culprit NSAIDs involved HSRs in the current report from our country 27. Dona et al. reported that propionic acid, aspirin, and pyrazolones were the most frequently involved NSAIDs to HSRs in Spain 19. Similarly, in our study, flurbiprofen, paracetamol, and naproxen were the most common NSAIDs involved in HSRs. Compatible with previous reports aspirin is still one of the most common causes of HSR, whereas the frequency of flurbiprofen has increased over the years 27. We found flurbiprofen was the most culprit drug in NIUA and NECD group, whereas aspirin was in NERD. Interestingly, aspirin and ketoprofen showed significant differences between the phenotypes. If further studies support this finding, underlying diseases might influence the usage of these drugs in certain conditions.
Because of the limited availability of in vivo and in vitro testing in the diagnosis of NSAID hypersensitivity, the controlled OPT with aspirin or a culprit drug is the only way to confirm aspirin/NSAID intolerance 5, 29, 30. We could do OPT with either aspirin or culprit drug in a total of 6.6%.and 5.9%, respectively, which is the major methodologic limitation of the present study. On the other hand, we included the patients with a history of at least 2 reactions to chemically different NSAIDs because this approach was found strongly indicate positivity in aspirin provocation 21, 31. The perfect correlation was reported between DPT with aspirin and patients’ history when reactions occur to more than two different NSAIDs in patients with NECD (30). Similar data were reported with the NERD subgroup. A study showed that patients with 2 or more prior aspirin and NSAID-associated respiratory reactions had an 89% chance of having a positive DPT with aspirin 21. Besides, the university-based hospital, we have been dealing with drug allergy for more than 20 years and have a good background experience in drug allergy in general and especially in NSAID hypersensitivity 8, 10, 11, 12. Therefore, we are quite confident in the appropriate selection of patients with NSAID hypersensitivity.
Conclusion
Nimesulide, meloxicam, and celecoxib appeared safe alternatives in cross-reactive phenotypes of NSAID hypersensitivity. Although celecoxib has the most favorable tolerability, cross-reactivity among COX-2 inhibitors seems to be possible. Data reported so far about cross-reactivity between COX-2 inhibitors comes from a limited number of studies performed on a small number of subjects. Finding risk factors associated with cross-reactivity with COX-2 inhibitors are important and more studies with a larger number of patients are needed to assess possible risk factors for cross-reactivity to COX-2 inhibitors.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.