Abstract
Breast cancer has high incidence in women from both developed and developing countries. Approximately 2 million women are diagnosed with breast cancer in 2018. In Asia, unfortunately Pakistan leads the highest number of breast cancer patients. Various treatment strategies are present but they are not well developed. There is a great need to develop effective methods for early detection and treatment of the disease. For cancer treatment chemotherapeutic interventions have always been a method of choice. One of the mechanisms involved in cancerous cell proliferation is Mevalonate (MVA) pathway. It is hypothesized that arresting MVA pathway leads to cell death hence cancer cell growth is suppressed. Various inhibitors of MVA pathway have been studied that can suppress cell proliferation. Nitrogen containing bisphosphonates are MVA pathway inhibitor and clinically used for treatment of bone diseases. Their anticancer efficacy is also reported. Current study focuses on alendronate, a nitrogen containing bisphosphonate to examine their anticancer effect on breast cancer cell line. Results of this study may help in addition of new anticancer drug for breast cancer.
Author Contributions
Academic Editor: Aiping Zheng, China.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2019 Nida Syed Amber Ilyas, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Cancer is defined as a progressive group of disease characterized by abnormal clusters of cell mass that exceeds due to the uncontrolled growth 1, 2. Cancer is second major cause of death in the world. In 2015, 1600 deaths per day were anticipated in USA 3.Studies showed that in 2010 there are 1.6 million cancer cases have been diagnosed and by 2030 cancer burden will increase up to 2.3 million in United States 4. Asia contributes 60% of the world population. Half of the cancer incidences were reported from Asian countries. An estimated population of 3.6 million males and 4.0 million females were diagnosed with cancer in this region, in which lung cancer is most prevalent among men and breast cancer among women 5.
Breast cancer is one of the leading malignancy in women worldwide and in Pakistan. It accounts 41% of female cancers 6. According to the GLOBOCAN of WHO more than 1.38 million women have diagnosed with breast cancer in 2008 7.
According to recent statistics of American cancer society (ACS), in 2016 approximately 246,660 cases of invasive breast cancer and 61,000 cases of non invasive breast cancer will be reported. 40,730 deaths due to breast cancer will be reported in 2016 8 . In Asia, Pakistan has the highest prevalence of breast cancer. One in eighth womenis facing breast cancer in our country and annually 40,000 women are dying because of this particular cancer 9 .
Currently treatment methods are limited, that are mastectomy/lumpectomy followed by palliative care and radiation therapy. Palliative care is suggested in any stage and is a need of advanced stage breast cancer. Normally chemotherapeutic drugs are inhibitor of several metabolic pathways with severe side effects such hair loss, Nausea, fatigue, loss of appetite etc. Bisphosphonates are the synthetic derivative of the inorganic pyrophosphate that belongs to the family of drug that inhibits osteoclast mediated bone resorption by blocking essential enzyme of the mevolonate pathway. Bisphosphonates are the FDA approved remarkable therapeutic drug for the treatment of the osteoporosis, leg calve perthes disease and currently it is suggested as an adjuvant for the treatment of different cancers that results in metastasize of bone during the progression of disease 10.
Bisphosphantes are inhibitors of mevalonate pathway that is responsible for several vital functions within the cell leading to the formation of cholesterol through multi-step process 11 and to the post-translational modification by prenylation of Ras proteins. The mevalonate pathway can be interrupted by anticancer drugs at several levels 12. Therefore inhibiting mevalonate pathway is of great importance for cancer treatment. In several studies it is concluded that bisphosphonates prevent progression and invasion of cancer cells 13.
Materials and Methods
Cell Culture
Breast cancer cell line HTB132 was obtained from ATCC. Breast cancer cell line (HTB-132) was maintained in leibovitz’s L-15 medium (ATCC catalog no. 30-2008) supplemented with 10% fetal bovine serum (Sigma) and 100 ug/mL of penicillin and streptomycin (Life technologies Inc.) in a free gas exchange with atmospheric air at 370C.
Drug Treatment
Cell Proliferation Assay
~1000 cells/mL were seeded in 24-well plates (Corning) for 24 hours. After 24 hours fresh medium containing different concentrations of Alendronate (5µM, 10µM and 20µM) was added and incubated for 24, 48 and 72 hours. After each respective time, cell viability (MTS) assay was performed using CytoTox 96® Non-Radioactive Cytotoxicity Assay kit (Promega) as per manufacturer`s instructions. Absorbance was taken at 490 nm using microplate reader (Backmann Coulter). The cell viability was expressed as percentage against the control wells. All assays were performed in triplicate.
Statistical Analysis
Data were analyzed using SPSS software (SPSS® for Windows® 20.0). t-test was used to find statistically significant differences in cell viability.One way ANOVA was performed for comparison between different time intervals (24h, 48h, 72h).p<0.05 was consideredas statistically significant cytotoxicity.
Results
Effect of Alendronate in HTB-132 Breast Cancer Cell Line
Cell viability of HTB-132 Breast Cancer cells was measured after treatment with drugs using proliferation assay. CytoTox 96® Non-Radioactive Cytotoxicity Assay kit (Promega) was used which determines the quantity of formazan product as measured by absorbance at 490nm is directly proportional to the number of living cells in culture.Cell viability was measured at 24, 48, and 72h after treatment with 5µM, 10µM and 20µMALN as shown in Figure 1, Figure 2 and Figure 3. Figure 4 illustrated decreased cell proliferation with increasing time and drug concentration. ALN showed a significant time and dose dependent effect on cytotoxicity (Figure 4). Cell death was found to be 3%, 47% and 56% after 24, 48 and 72 hours incubation at 5µM concentrations. At 10µM concentration 11% increased in cell cytotoxicity was observed at 24 hours that reached up to 70% till 72 hours incubation period.A further increased in cellular toxicity was observed in HTB-132 cells at 20µMALN treatment. Maximum cell death (80%) was observed at 72 hours after 20µMALNtreatment (p<0.05).
Figure 1. HTB-132 breast cancer cells viability before and after 24 hrs alendronate treatment. A: Control; B, C & D: 5µM, 10 µMand 20 µM ALN respectively.
Figure 2. HTB-132 breast cancer cells viability before and after 48 hrs alendronate treatment. A: Control; B, C & D: 5µM, 10 µMand 20 µM ALN respectively.
Figure 3. HTB-132 breast cancer cells cytotoxicity before and after 72 hrs alendronate treatment. A: Control; B, C & D: 5µM, 10 µMand 20 µM ALN respectively.
Figure 4. The Cytotoxic effect of ALN on HTB-132 breast cancer cell line after 24, 48, and 72hrs treatment with 5 µM, 10 µM, and 20 µM concentration of drug. Each bar represents mean ± S.D for each concentration. Asterisk represents statistically significant values with reference to respective control (p< 0.05).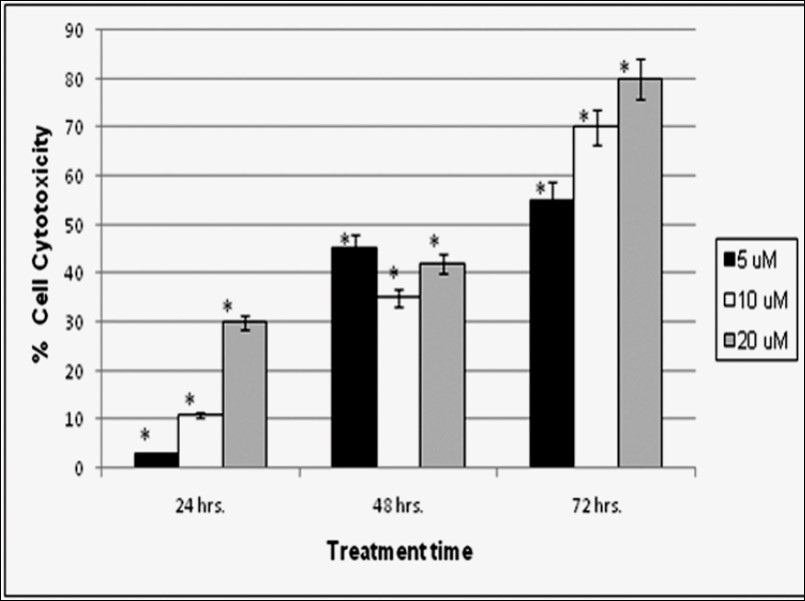
Discussion
Besides MVA pathway inhibitor, bisphosphonates (BPs)have inhibitory bone resorption property and studies also suggested there anticancer activity in various cancer including breast cancer 16, 17. BPs exert their effects through inhibition of bone resorption and bone-derived growth factors that ultimately results in inhibition of tumor cell invasion 18, proliferation 19 and increased apoptosis 20 in breast and prostate carcinoma.There are two major classes based on the antiresorptive activity that is amino based bisphosphonates and non-amino based bisphosphonates 21. Non amino bisphosphonates (e.g., etidronate, clodronate and tiludronate) are metabolizes to non-hydrolysable cytotoxic analogs of ATP. These analogs block ATP utilizing enzymes to inhibit osteoclast activity. Whereas amino bisphosphonates (N-BPs, e.g., alendronate, ibandronate, pamidronate, zolendronate and riserdronate) block the synthesis of farnesyl pyrophosphate synthase. 22 that causes the inhibition of small regulatory binding protein guanosine triphosphate (GTP) of Ras superfamily (e.g. Rac, Rho, Rabs , Rans, Raps, Ralsetc) by blocking the process of farnesylation and geranylgeranylation that causes cytosolic confiscation and cease biological activity 23, 24, 25. Alendronate is a member of BPs used to lower the risk of breast cancer in post-menopausal women26. Effect of alendronate on various cancer cell lines have been reported earlier 15, 27.Present study examined the effect of alendronate on HTB-132 that has not been reported earlier. In our results alendronate showed time and dose dependant pattern of cytotoxicity on each given drug concentration. We observed 47% cell death at 5𝜇M after 48 hours of treatment and this dose was found to be most effective in least time and concentration.In current work we have found anticancer activity of ALN on HTB-132 breast cancer cell line. This time and dose dependent cytotoxic potential of ALN is also an agreement with previous studies reported in other cancer15, 28.
Previous study has reported that N-BPs suppress GGPP synthetic pathway 29 without effecting cholesterol synthesis 30. It has also been studied that BPs exert changes in CpG-methylation state of gene promoter regions that is involved in the proliferation and cell death 31.Gambino etal., (2014) have suggested that BPs cause significant changes in gene regulation by modulating DNA methylation in osteonecrosis 32.
Conclusion
This study presents first report on cytotoxic potential of alendronate on HTB-132 breast cancer cell line. Our study provides a baseline cytotoxic efficacy of alendronate in breast cancer. Furthermore, bisphosphonates specifically ALN in less concentration is capable to induce cytotoxicityin breast cancer cells and might be used as chemotherapeutic agent in human cancer.
Acknowledgements
This study was supported by research grant (NRPU#20-2827) from Higher Education Commission Islamabad) and partial financial assistance for the work was provided by Dean`s grant, University of Karachi.