A Pilot Study Assessing the Impact of a High Protein Supplementation Diet on Fat Mass in Obese Adolescents with and without Type 2 Diabetes
Abstract
Background:
Obese adults with Type 2 Diabetes (T2D) have shown differences in body composition and response to varying diets when compared to obese adults without T2D. This difference in body composition affects management options for obesity. Such studies have not been done on adolescents thus far.
Objectives:
To estimate differences in fat mass at baseline as well as following a two-week high protein liquid diet in obese adolescents with and without T2D.
Methods:
Thiswas a pilot study using a non-randomized, unblinded pre/post intervention design without a control group. We planned to recruit 40 subjects (20 per group, with and without T2D) from ages 10 – 19 years, with Body mass index (BMI) ≥ 95 percentile. Body fat mass was measured via the Bod Pod® at baseline and after two weeks of following the diet with Ensure High Protein shakes. Change in fat mass is presented as mean differences and 95% confidence intervals.
Results:
We recruited 28 subjects, 19 completed the study and 12 were compliant with the diet. The mean difference in fat mass percent from baseline in subjects who completed the study (n=19) and without T2D was 1.4 (95% confidence interval (CI): -2.1 to -0.8) and in subjects with T2D was 1.2 (95% CI: -3.6 to 1.24).
Conclusion:
We did not find any meaningful difference in fat mass at baseline or after following a two-week high protein diet between the groups. High dropout rate and noncompliance with the diet were major limitations.
Author Contributions
Academic Editor: Anthony DeMaria, Chiropractor
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2016 Meghana Gudala, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction:
Childhood obesity is an epidemic in the United States; data from 2012 indicate that 31.8% of children aged 2-19 years in the United States are overweight or obese, 1 with 20.5 % of children aged 12-19 years being obese. Adolescents are being afflicted with obesity-related comorbidities, including type 2 diabetes mellitus (T2D), hypertension, dyslipidemia, obstructive sleep apnea, nonalcoholic fatty liver disease and metabolic syndrome. 2,3T2D now constitutes about 25-45% of all the youth diagnosed with diabetes, with the mean age of onset being 13 years of age.4,5,6
Treatment options for T2D in the pediatric population are limited and weight loss continues to be an essential adjunct to whatever therapy is prescribed.7,8 The primary treatment for weight loss in obese children is lifestyle modification.9,10 However, dietary modification and exercise alone are insufficient for successful and long-term weight loss.11 Reasons for inability to lose weight and maintain weight loss include decreased motivation, 12 changes in resting energy expenditure, 13 and hormone changes that promote weight regain, including decreases in leptin,14 increases in ghrelin,15and alterations in thyroid function.16
Currently, lifestyle modification recommendations for morbidly obese adolescents are generally similar, irrespective of the presence or absence of T2D. Presence of T2D could impact not only how one loses weight, but also how successful one will be with different weight loss strategies. Studies in adults have assessed differences in body composition 17 and response to varying diets in the morbidly obese with and without T2D.18 Obese adults with T2D have been found to have more visceral and intramuscular adipose tissue than those without T2D.17 On very low calorie diets, subjects with T2D lost less fat mass per unit of weight compared to subjects who were obese without T2D.18 Such studies, to our knowledge, have not been reproduced in the obese adolescent population with and without T2D. Given the increased insulin resistance associated with puberty 19 and higher metabolic rates associated with younger age groups, 20 there could be clinically significant differences in body composition at baseline and following weight loss that could be used to guide more specific recommendations towards the adolescent obese, with and without T2D.
The purpose of this study was to assess the body composition in morbidly obese adolescents with and without T2D, at baseline and after a two-week high protein liquid diet. We hypothesized that subjects with T2D would lose less fat mass per unit of body weight lost than those without T2D.
Methods:
This pilot study, approved by the Baystate Institutional Review Board, was a non-randomized, unblinded pre/post intervention study without a control group. More specifically, the study cohort comprised adolescents who are obese, seen in our pediatric weight management and endocrinology clinics. Within this cohort two subgroups were defined: those with T2D and those without T2D. All patients in the study cohort received the standard high protein liquid diet, thereby enabling us to examine the influence of the presence or absence of T2D on the effects of a high protein diet.
Subjects:
Subjects between the ages of 10 and 19 years whose BMI was ≥95th percentile for age and gender were recruited for participation. In subjects with T2D, presence of T2D was confirmed by their pediatric endocrinologist prior to enrollment based on review of the electronic medical chart (fulfillment of American Diabetic Association (ADA) diagnostic criteria of HbA1C ≥6.5% OR fasting plasma glucose level ≥126 mg/dL (7 mmol/L) OR a 2-hour plasma glucose level of ≥200 mg/dL (11.1 mmol/L) during a 75-g oral glucose tolerance test (OGTT), OR a random plasma glucose of ≥200 mg/dL (11.1 mmol/L) in a patient with classic symptoms of hyperglycemia (polyuria, polydipsia, polyphagia, unintended weight loss); and, absence of anti-insulin, anti-glutamic acid decarboxylase (GAD) and anti-islet cell antibodies). Exclusion criteria were: history of kidney disease, elevated creatinine and blood urea nitrogen (BUN) levels for age, current pregnancy, genetic syndromes associated with obesity or T2D (i.e. Prader Willi Syndrome, Bardet-Biedl Syndrome, Klinefelter syndrome), or inability to provide consent or assent for the study.
Protocol:
Subjects were recruited from the pediatric weight management clinic and the pediatric endocrine clinic at Baystate Medical Center. Eligible subjects were invited to participate. Assent and/or consent were obtained from the patient and/or caregiver. A BUN and creatinine were drawn to confirm normal kidney function along with a urine pregnancy test in females to rule out pregnancy. Baseline evaluation included height to the nearest 0.1 cm measured on a standard wall stadiometer and weight to the nearest 0.1 kg (with subjects wearing the Lycra cap, top and shorts) obtained from the Bod Pod® (COSMED USA, Concord, CA) weighing scale. Body composition was assessed via the Bod Pod® (COSMED USA, Concord, CA), which measures body fat percent through air displacement plethysmography. Bod Pod® is a quick, comfortable, automated, noninvasive, safe measurement process that is comparably precise, reliable and valid when compared to alternate methods such as dual-energy X-ray absorptiometry (DXA) and hydrostatic weighing. 21,22
Subjects were then placed on a two-week high protein liquid diet, which consisted of 5 bottles of Ensure High Protein (25 g protein per 14 oz. bottle), plus unlimited water and non-caloric beverages and two cups of leafy greens daily. This diet regimen was chosen for two reasons: 1) Weight loss is achieved in obese patients following this diet regimen prior to Bariatric surgery procedures and 2) Compliance rates will likely be higher as the diet is only 14 days. Subjects were asked to keep a detailed log of any food and drink they ingested during the study, including the shakes. Subjects were asked to remain at their usual level of activity during the study. Within 10 days of completion of the two-week diet, subjects were asked to return for a reassessment of weight and body fat. Phone contact was made with the subjects at days 3, 7 and 10 of the study to assess compliance and provide reassurance and motivation. Subjects were compensated with $100 in appreciation of their time for participating in the study.
Study data were collected and managed using Research Electronic Data Capture (REDCap) tools hosted at Tufts Medical Center (Grant UL1TR001064).23 REDCap is a secure, web-based application designed to support data capture for research studies, providing: 1) an intuitive interface for validated data entry; 2) audit trails for tracking data manipulation and export procedures; 3) automated export procedures for seamless data downloads to common statistical packages; and 4) procedures for importing data from external sources.
Statistical Analysis:
The primary goal of this pilot study was to develop estimates for future research and assess feasibility of the protocol. Hypothesis testing was not the primary statistical goal. In general, continuous variables were characterized by means, standard deviations or medians and ranges. Categorical variables were characterized by frequencies and percentages. Baseline characteristics, feasibility and study outcomes were assessed across subjects with and without T2D. Feasibility was measured in terms of meeting recruitment goals, dropouts (subjects who intentionally left the study or could not tolerate a liquid diet) as well as compliance (subjects who went off the diet or missed some shakes). Estimates of outcomes included change in body fat mass percent, total fat mass in grams, and weight. Change was calculated as the post liquid diet measures minus the baseline measures two weeks prior. Estimates of the mean and mean difference along with 95% confidence intervals for these outcomes were calculated using Stata v14.1 (StataCorp, LP, College Station, TX). Due to the small sample size, we also calculated Cohen’s d effect sizes24 as a standardized way of estimating the practical significance of our findings. As a general guide, Cohen suggests small, medium and large effect sizes to be 0.2, 0.5 and 0.8 respectively.
Sample size was based on the stability of a 95% confidence interval (CI). 25 With a goal of recruiting 12 subjects per group and assuming some loss to follow up, we initially planned to recruit a total of 30 subjects (15 per group). Given this, we would expect the width of a CI around a proportion of 0.50 (a conservative estimate) to be nearly 52 percentage points for a sample of 15 and around 37 percentage points for a sample of 30. More important were the width of a CI for a continuous measure, such as fat mass. As we had unknown standard deviations, we characterized the width of the CI as a percent of the standard deviation (SD) 26 to be about 85% for a sample of 15 and 55% for a sample of 30. Partway through the study, we increased the sample size to 20 per group (40 total) to accommodate for unanticipated dropouts.
Results:
Twenty-eight subjects were recruited over 10 months, 19 obese without T2D & 9 obese with T2D. One patient was never able to schedule the baseline assessment and was not included in our analysis. No subjects were excluded due to pregnancy or impaired renal function. All subjects returned the next day of their diet completion date for repeat body composition testing. All patients tolerated the Bod Pod® procedures without any issue.
The overall mean (SD) age of the subjects at baseline was 16.3 (1.5) years and the majority (70%) were female (Table 1). Depression and/or anxiety were present in nearly 30% of the sample. Groups were relatively balanced at baseline in terms of gender, age, height and depression/anxiety. Of the subjects with T2D, glucose lowering agents used included metformin (n=4), glargine insulin (n=4), lispro insulin (n=1), sulfonylurea (n=1) and GLP1 agonist (n=1). No subject with T2D was being managed with lifestyle modification alone. Adiposity related comorbidities in the obese without T2D group included polycystic ovarian syndrome (n =3), dyslipidemia (n=5), vitamin D deficiency (n=7) and sleep apnea (n=2). In the obese with T2D group, comorbidities included dyslipidemia (n=7), non-alcoholic fatty liver disease (n= 1), hypertension (n=4), polycystic ovarian syndrome (n=1) and vitamin D deficiency (n=4).
Table 1. Subject Baseline Characteristics| Obese Only n=18 (66.7%) | Obese with Type 2 Diabetes n= 9 (33.3%) | |
| Male: n (%) | 6 (33.3) | 2 (22.2) |
| Age (y): Mean(SD) | 16.2 (1.7) | 16.7 (0.7) |
| Height (cm): Mean(SD) | 166.3 (8.0) | 164.8 (7.7) |
| Body weight (kg): Mean (SD) 95% CI | 106.1 (19.7) | 94.5 (21.2) |
| 96.3 to 115.9 | 78.1 to 110.8 | |
| Body Fat Mass (percent): Mean(SD) 95% CI | 44.1 (6.7) | 40.9 (8.8) |
| 40.8 to 47.4 | 34.1 to 47.7 | |
| Total Fat Mass (grams): Mean(SD) 95% CI | 47,486 (14,320) | 39,387 (14,678) |
| 40,365 to 54,608 | 28,105 to 50669 |
In terms of body weight and fat mass (percent and grams), the mean (95% CI) difference between groups at baseline showed the obese only group to have larger estimates than the obese T2D group (Table 1). The obese only group had an 11.6 kg larger body weight (95% CI = -5.3 to 28.6); 3.2% greater fat mass (95% CI = -3.1 to 9.4) and 8,100 gm higher fat mass (95% CI = -4,038 to 20,237) than the obese with T2D group. Distributional box and whisker plots for body weight and fat mass grams suggest a possible shift in the distributions between subjects with and without T2D (Figure 1).
Figure 1.Baseline body weight and fat mass in obese adolescent subjects with and without T2D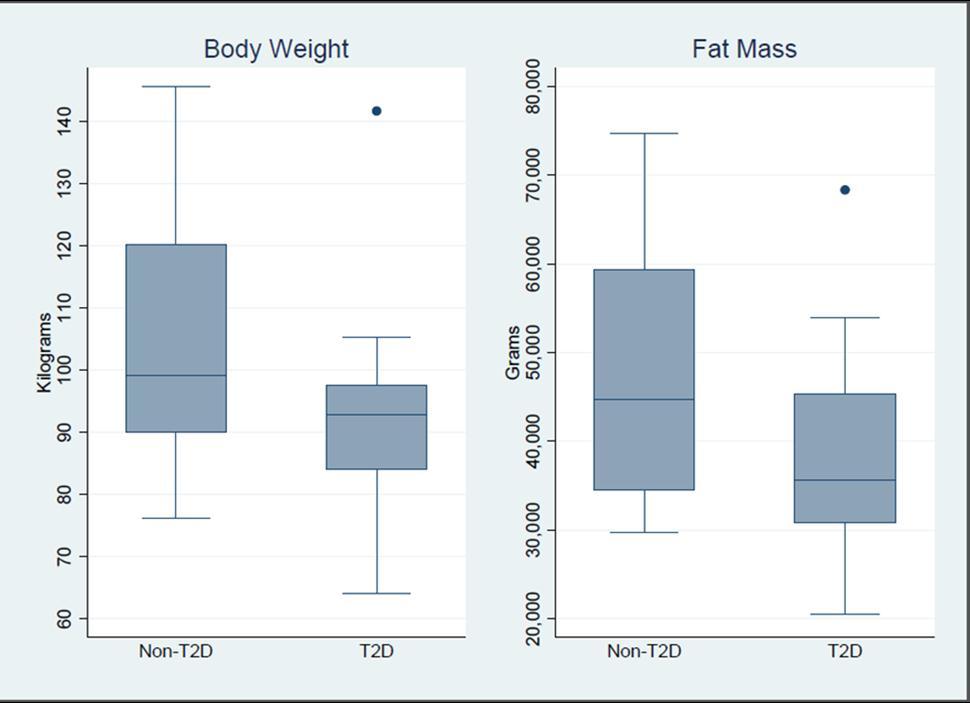
A total of 9/28 (32%) subjects dropped out following consent, 6 without T2D and 3 with T2D. Reasons for drop out were as follows: did not like the shakes 1, experienced vomiting 2, experienced mood changes 2, did not feel like they could complete the protocol 1, had a possible allergic reaction to the shakes 1, fell ill 1, and did not show up for a baseline appointment 1. On average, those that dropped out were 1 year younger and weighed approximately 5 kg less (with 1.52 kg less fat mass) than those that completed the study. In addition, rates of depression/anxiety were 30% higher among dropouts.
Of those that completed the study, a total of 7 (37%) subjects were non-compliant with the diet. Of these, 6 were obese without T2D and 1 was obese with T2D. Items ingested among those who were not compliant with the diet included cheese, hot dogs, crackers, chocolate, eggs, pasta and meatballs, cake, hamburger buns, oatmeal, tuna fish, soup and fruit punch. Total dropouts plus non-compliant participants were 57% (n=16) of the sample (63% (n=12) without T2D and 44% (n=4) with T2D). On average, those that were non-compliant were 1 year younger, 1.5 kg lighter (with 3.2 kg less fat mass). In addition, non-compliant subjects appeared to be nearly 40% more likely to be male.
Change from baseline among subjects completing the study as well as those compliant with the diet are presented in Table 2. Among those completing the study, the mean (95% CI) of the difference between groups for body weight was -2.8 kg (-3.9 to 0.9) compared to -1.6 kg (-5.3 to 2.1) among those who were compliant. The change in fat mass was -0.2% (-1.8 to 1.3) and -1.1% (-2.7 to 0.6) in the completers and compliant groups, respectively. The change in fat mass was -988 gm (-2937 to 961) in those completing the study and -1925 gm (-4324 to 473) among those who were compliant.
Table 2. Change from Baseline and 95% Confidence Intervals for Total Weight, Fat Mass Percent and Fat Mass, among subjects completing the study and subjects compliant with study diet. Effect size was calculated using Cohen’s d.| Completed Study n=19 | Compliant with Diet n=12 | |||||
| Obese Only n=13 | Obese with T2D n= 6 | Effect Size | Obese Only n=7 | Obese with T2D n=5 | Effect Size | |
| Body weight (kg): Mean (SD) 95% CI | -3.2 (2.2) -4.6 to -1.9 | -1.7 (2.5) -4.4 to 0.9 | 0.64 | -3.4 (1.1) -6.0 to -0.8 | -1.8 (2.8) -5.3 to 1.7 | 0.57 |
| Body Fat Mass Percent: Mean(SD) 95% CI | -1.4 (1.0) -2.1 to -0.8 | -1.2 (2.3) -3.6 to 1.24 | 0.16 | -1.4 (1.2) -2.6 to -0.3 | -0.4 (1.4) -2.1 to 1.3 | 0.83 |
| Total Fat Mass (grams): Mean(SD) 95% CI | -2820 (1420) -3678 to -1961 | -1831 (2660) -4623 to 960 | 0.53 | -2948 (1735) -4551 to 1343 | -1022 (1983) -3485 to 1440 | 1.05 |
Discussion:
This pilot study was designed to estimate differences at baseline in body composition between obese adolescents with and without T2D and change following a two-week high protein liquid diet. We hypothesized that adolescents with T2D would lose less fat mass percent than those without T2D, similar to adults. Although our estimates were variable, we found a clinically meaningful difference in body weight (11.6 kg heavier) and fat mass (8100 g greater) at baseline in the obese only group compared to the T2D group. However, no meaningful difference in body fat mass (988 gm greater) or fat mass percent (0.2% greater) after the 2 week high protein liquid diet was observed in the obese only group compared to the T2D group.
To the best of our knowledge, no studies in regards to baseline differences in body composition exist among obese adolescents with and without T2D. However, in the adult literature, a study done by Heshka et al. in over 1500 obese adults with and without T2D showed approximately 1.4 kg less fat mass in obese subjects with T2D (p< 0.001) when compared to obese subjects without T2D.27 Our study showed that subjects with T2D had an even larger difference with 8.1kg (95% CI -20.2 to 40 kg) less fat mass when compared to subjects without T2D at baseline. Although the general trend for lower fat mass in obese subjects with T2D was the same, our estimates suggested that this difference may be even greater among adolescents. However, as evidenced by our wide confidence intervals there was much variability and additional research would need to be conducted in a larger sample in order to make a more definitive statement.
Our study is also the first to date to assess body composition changes after a high protein liquid diet in adolescents. For comparison, in the adult literature Baker et al compared body composition changes in 27 obese adults without T2D to 24 obese adults with T2D after a low calorie diet and reported significantly less fat mass lost per unit of weight loss in obese with T2D when compared to obese without T2D. Change in fat mass index per unit change in BMI in the obese diabetics when compared with obese without diabetes was 0.574 vs 0.905 (P = .003).18 Our hypothesis was similar among the obese adolescent population with and without T2D, however, we did not find any clinically meaningful differences in fat mass percent, even among the compliant subjects. This could be due to the variability in our sample as well as the type of dietary intervention, but differences between these findings may also be due to metabolic differences between adults and children. However, given the increased metabolic rate in adolescents compared to adults, we would have expected a larger weight change in our subjects compared to the adult subjects in these studies. In the pediatric literature, there are no studies to date assessing the impact of a high protein diet on weight and body composition in adolescents with T2D; However, a study assessing the impact of a high protein diet on obese children at a summer camp indicated improvements in fat mass and weight over the 3-5 week period (3.8±5.4 kg and 5.5 ±2.9 kg, respectively). 28 Our findings for the obese only group showed a meaningful weight change over two weeks, but a nonmeaningful change in fat mass. Subjects in Gately et al’s study were in a supervised summer camp, 28 while our patients were unsupervised in their own home, a factor which could have certainly played a role in the compliance and outcomes of the diet. In addition, our study was of much shorter duration, only 14 days, compared to a mean of 27 days at camp, and we did not incorporate a strict exercise regimen in our study, while the subjects at the summer camp had daily exercise routines.
Our study had some strengths and limitations. We were able to look at adolescents with and without T2D to determine if body composition differences exist both before and after a dietary modification, which has not, to date, been performed. Additionally, through the use of Bod Pod®, we were able to obtain precise measurements of body fat without exposing subjects to minimal radiation if DXA was used and without concern for the ability to hold their breath if hydrostatic weighing was used.
Our largest limitations were around recruitment, retention and compliance. Recruitment was particularly difficult among subjects with T2D mainly because a larger proportion of our patients with T2D were between the ages of 19 and 21 years, ages outside of our eligibility criteria. Surprisingly, despite the short duration of the study, the prospect of weight loss and the participation stipend, we had a number of patients with T2D who did not want to participate in the study for reasons that were unspecified. In regards to retention and compliance, we had a 32% drop out rate in the obese subjects with T2D and a 33% dropout rate in the obese subjects without T2D group. We had an 11% noncompliance rate in the T2D arm and a 32% noncompliance rate in the obese only arm in our study. These factors were likely to have significantly affected our results, as with a larger sample size, we would have been able to better assess the impact of the dietary intervention on fat mass and weight loss in our study subjects. Larger studies should subdivide groups by gender, as we know males have more lean mass and lose weight faster than females do; subdividing the T2D group of subjects by the presence or absence of insulin therapy would also make the results more valuable, as insulin is an anabolic agent and might promote more fat storage and less fat loss in patients who are taking it and trying to lose weight.
High dropout rates associated with weight loss programs and the predictors of attrition in adolescents have been studied previously.29, 30 Younger age was one of the important predictors of dropout.31, 32 This was consistent with our observations as the mean age of our study patients was 16 years, and we found that subjects who dropped out were on average 1 year younger than subjects who completed our study. In our study, we attempted to recruit subjects as young as 10, however the youngest we actually enrolled was nearly 14 years old. Future studies may want to consider restricting to older age ranges and increasing the age limit to 21 years to improve retention.
In other studies, baseline depression levelaffected enrollment and duration of participation in a dietary weight loss trial and subjects with moderate to severe depression symptoms were more likely to drop out from the diet.33 A similar impression was suggested by Peyrot et al, in adults, wherein 41% of study subjects with diabetes had psychological problems and that affected adherence to diet and exercise as well as diabetes self-care.34 In our study, we found that rates of depression were nearly 30% higher among dropouts (we weren’t able to assess this among non-compliant subjects as most depressed/anxious patients had already left the study). While excluding patients who have mental health diseases from future studies may result in improved retention and/or compliance, many of our patients with T2D have mental health issues and thus, recruitment into the study would have been even more difficult than it already was. The issue of compliance with diet in weight loss studies has been seen in other studies as well. According to Leidy et al, in adults, dietary compliance was associated with improvements in weight loss, whereas those who did not adhere to the diet had no marked improvements.35 Behavioral and environmental factors contribute to the lack of compliance or adherence to weight loss diets, especially with the high protein diets.36 Many factors, including socioeconomic, cultural, cognitive and psychological made the process of adhering to diet difficult for obese youngsters. 37 One of the factors that has been found beneficial to increase adherence to diet and increase weight loss is counseling sessions. Attending group sessions in a long term weight loss trial had a positive impact on adherence. 38 While we were unable to provide counseling sessions during our short study, we did have phone contact with the study subjects on three occasions during the 14-day period.
Conclusion:
Our study sought to assess the difference in fat and weight loss in obese adolescents with and without T2D. Due in strong part to significant issues with recruitment, retention and compliance, we were unable to generate reliable estimates in body composition after a two-week high protein liquid diet. Steps to improve compliance and reduce attrition in studies in adolescents are needed, especially in this growing population of obese adolescents with and without type 2 diabetes.
Acknowledgements
Funding for our study was provided by Department of Pediatrics, Baystate Medical Center. We would like to thank Hilary Weiner RD, our dietitian for conducting Bod Pod measurements, counseling the patients regarding the high protein shakes and for following up with them during the two-week diet period. We would also like to thank all of our patients for participating in the study.
References
- 1.Ogden C L, Carroll M D, Kit B K, Flegal K M. (2012) Prevalence of obesity and trends in body mass index among US children and adolescents,1999–2010. , Journal of the American Medical Association 307(5), 483-90.
- 2.Sinha R, Fisch G, Teague B, Tamborlane W V, Banyas B. (2002) Prevalence of impaired glucose tolerance among children and adolescents with marked obesity. , N Engl J Med; 346, 802-10.
- 3.Weiss R, Dzuira J, Burgert T S. (2004) Obesity and the metabolic syndrome in children and adolescents. , N Eng J Med; 350, 2362-2374.
- 4.Rosenbloom A L, Silverstein J H, Amemiya S, Zeitler P, Klingensmith G. (2009) ISPAD Clinical Practice Consensus Guidelines 2007-2007. Type 2 diabetes mellitus in the child and adolescent. Pediatr Diabetes. 9, 512-26.
- 5. (2000) American Diabetes Association Type 2 diabetes in children and adolescents. , Diabetes Care; 23, 381-389.
- 6.Kaufman F. (2007) Type 2 diabetes in youth: rates, antecedents, treatment, problems and prevention. Pediatr Diabetes. 8, 4-6.
- 7.Rosenbloom A L. (2008) ISPAD Clinical Practice Consensus Guidelines 2006-2007. Type 2 diabetes mellitus in the child and adolescent. Pediatr Diabetes.
- 8.Meehan Colette.Treatment Options for Type 2 Diabetes in. , Youth Remain Limited The Journal of Pediatrics 170, 20-27.
- 9.S E Barlow, W H Dietz. (2002) Management of child and adolescent obesity: summary and recommendations based on reports from pediatricians, pediatric nurse practitioners, and registered dietitians. , Pediatrics 110, 236-238.
- 10.G P August. (2008) Prevention and treatment of pediatric obesity: an endocrine society clinical practice guideline based on expert opinion. , J. Clin. Endocrinol. Metab 93, 4576-4599.
- 11.Meehan C, Silverstein J. (2015) Treatment Options for Type 2 Diabetes in Youth Remain Limited. , Journal of Pediatrics
- 12.Elfhag K, Rossner S. (2005) Who succeeds in maintaining weight loss? A conceptual review of factors associated with weight loss maintenance and weight regain. , Obes. Rev 6, 67-85.
- 13.Leibel R L, Rosenbaum M, Hirsch J. (1995) Changes in energy expenditure resulting from altered body weight. , N Engl J Med; 332, 621-628.
- 14.Rosenbaum M, E M Murphy, S B Heymsfield, D E Matthews, Leibel R L. (2002) Low dose leptin administration reverses effects of sustained weight-reduction on energy expenditure and circulating concentrations of thyroid hormones. , J Clin Endocrinol Metab 87, 2391-2394.
- 15.D E Cummings, D S Weigle, R S Frayo. (2002) Plasma ghrelin levels after diet-induced weight loss or gastric bypass surgery. , N Engl J Med 346, 1623-1630.
- 17.Gallagher D, Kelley D E, Yim J E, Spence N, al et.(Mar2009) Adipose tissue distribution is different in type 2 diabetes. , Am J Clin Nutr; 89(3), 807-14.
- 18.L Baker ST, Jerums G, Prendergast L A, Panagiotopoulos S, Strauss B J.(Jun2002) Less fat reduction per unit weight loss in type 2 diabetic compared with nondiabetic obese individuals completing a very-low-calorie diet program. , Metabolism; 61(6), 873-82.
- 19.Caprio S, Tamborlane W V. (1994) Effect of puberty on insulin action and secretion. , Semin Reprod Endocrinol 12, 90-96.
- 20.Fukagawa N K, Bandini L G, Young J B.(Aug1990) Effect of age on body composition and resting metabolic rate. , Am J Physiol;259(2Pt1) 233-8.
- 21.David A Fields, Michael I Goran, Megan A McCrory.Body-composition assessment via air-displacement plethysmography in adults and children: a review. , Am J Clin Nutr 75(3), 453-467.
- 22.Dewit O, Fuller N, Fewtrell M, Elia M, Wells J. (2000) Whole body air displacement plethysmography compared with hydrodensitometry for body composition analysis. Archives of Disease in Childhood 82(2), 159-164.
- 23.Paul A Harris, Taylor Robert, Thielke Robert, Payne Jonathon, Gonzalez Nathaniel.(Apr2009) Research electronic data capture (REDCap) - A metadata-driven methodology and workflow process for providing translational research informatics support. , J Biomed Inform; 42(2), 377-81.
- 24.Cohen J. (1988) Statistical Power Analysis for the Behavioral Sciences. 2nd ed.Hillsdale,NJ:Erlbaum.
- 26.Rosner B. (2006) Fundamentals of Biostatistics. 6th ed” Duxbury,Press (The , Thompson Company): Belmont, CA 199-201.
- 27.Heshka S, Ruggiero A, Bray G A. (2008) Altered body composition. in type 2 diabetes mellitus International journal of obesity(2005); 32(5), 780-787.
- 28.P J Gately, N A King, H C Greatwood, L C Humphrey, Radley D.. Does a High-protein Diet Improve Weight Loss in Overweight and Obese Children? Obesity 15, 1527-1534.
- 29.Jelalian E, Hart C N, Mehlenbeck R S, Lloyd-Richardson E E, Kaplan J D. (2008) Predictors of attrition and weight loss in an adolescent weight control program. Obesity (Silver Spring). 16(6), 1318-23.
- 30.Skelton J A, Goff D C, Ip E, Beech B M. (2011) Attrition in a Multidisciplinary Pediatric Weight Management Clinic. Childhood Obesity. 7(3), 185-193.
- 31.Dalle Grave R, Calugi S, Molinari E, Petroni M L, Bondi M. (2005) Weight loss expectations in obese patients and treatment attrition: an observational multicenter study. , Obesity Research 13, 1961-1969.
- 32.Honas J J, Early J L, Frederickson D D, O’Brien M S. (2003) Predictors of attrition in a large clinic-based weight-loss program. , Obesity Research 11, 888-894.
- 33.Somerset S M, Graham L, Markwell K. (2011) Depression scores predict adherence in a dietary weight loss intervention trial. Clin Nutr. 30, 593-598.
- 34.Peyrot M, Rubin R R, Lauritzen T, Snoek F J, Matthews D R. (2005) Psychosocial problems and barriers to improved diabetes management: results of the Cross-National Diabetes Attitudes, Wishes and Needs(DAWN) Study. , Diabetic Medicine 22, 1379-1385.
- 35.Leidy H J, Clifton P M, Astrup A, Wycherley T P, Westerterp-Plantenga M S.(Apr2015,29) The role of protein in weight loss and maintenance. , Am J Clin Nutr
- 36.Ludwig D S, Ebbeling C B. (2010) Weight-loss maintenance–mind over matter?. , N Engl J Med; 363, 2159-61.
