Trends of Diabetes in Children and Adolescents in Pediatric Endocrinology Clinic and Clinical Pediatric Diabetes Center at Al Baha Central Hospital in Saudi Arabia
Abstract
Objectives
This study describes and compares the epidemiological criteria of children with diabetes from 2007 to 2014.
Methods
A prospective study conducted with 372 children with diabetes mellitus. An access program was used for data collection, and SPSS 17 was used to analyses the results.
Results
A total of 372 cases with diabetes were studied and were divided into three groups according age. The first age group was between one day and six years and comprised 128 (34.4%) patients. The second group aged 7- 12 years of age comprised 174 (46.7%) patients. The third group aged 13-18 years of age comprised 58 (15.59%) patients. The average age was 8.32 years. Females constituted 52 % (194 cases) of all cases. T1DM constituted 95.4%, equal to 355 patients, T2DM constituted 1.88%, equal to seven cases. Monogenic diabetes affected three patients or 0.79%, with one case of monogenic neonatal diabetes confirmed by genetic study, and two other cases suspected to be MODY. The highest incidence was observed in the autumn months, with 105 children affected, constituting 27.8 % cases. The year 2012 had the highest incidence rate, with 59 children and adolescents affected at an incidence rate of 25.48 / 100000.Al-Baha region had the highest number of cases constituting 37.7 % of cases. Diabetic ketoacidosis was the first presentation and constituted 44.2% of cases.
Conclusion.
Diabetes in children is increasing significantly in recent years, requiring more detailed analysis of its epidemiological factors to find out any significant correlations which may help in its prevention.
Author Contributions
Academic Editor: Shemaila Saleem, Federal Medical and Dental College/ Shaheed Zulfiqar Ali Bhutto Medical University, Islamabad
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2016 Ahmed H.Alghamdi, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Many types of diabetes can affect children, the most common being type 1 diabetes mellitus (T1DM) which results from cellular-mediated autoimmune destruction of the B-cells of the pancreas. T1DM has multiple genetic predispositions and is also related to environmental factors that are poorly defined. The incidence of Type 1 diabetes has been increasing worldwide for decades at an average annual rate of 3% 1. It represents approximately 10% of all cases of diabetes, represents 90% of all diabetes affecting children and affects approximately 20 million people worldwide (American Diabetes Association, 2001) 11. It is rare in the first few months of life, with rising incidence starting from nine months of age until puberty. There are two age groups in the occurrence of T1DM, the first occurring at five years of age and the second at puberty. A seasonal variation has also been seen worldwide with a reduction of incidence in warm summer months.
Although the genetic susceptibility to type 1 diabetes is inherited, only 12–15% of type1 diabetes occurs in families. Girls and boys are almost equally affected. There is no apparent correlation with the poor socioeconomic status.DKA occurs in 20–40% of children with new-onset diabetes 1.
T1DM is characterized by the presence of auto antibodies such as GAD65, insulin, tyrosine phosphatases IA, and islet cell antibody. The other common form of diabetes is T2DM, which mainly affects pubertal obese adolescents. The incidence of T2DM is increasing due to the increasing incidence of obesity in children, and now constitutes approximately 10% of all cases of diabetes in children. In addition to obesity, T2DM is associated with a family history of type 2 diabetes in first- or second-degree relative, race/ethnicity (e.g., Native American, African American, Hispanic/Latino, Asian American, and Pacific Islander), signs of insulin resistance or conditions associated with insulin resistance (e.g., a canthosis nigricans, hypertension, dyslipidemia, poly cystic ovarian syndrome (PCOS) , or small for gestational-age birth weight), and a maternal history of diabetes or gestational diabetic mellitus (GDM) during the child’s gestation. The third type of diabetes is monogenic diabetes, which is increasingly being discovered because of the advances in genetic sciences, and now constitutes 1 -2 % of all diabetes in children. Monogenic diabetes is of two types: (1) Neonatal diabetes, which mainly affects infants below one year of age due to genetic mutations that impair insulin synthesis or excretion, and (2) Maturity onset diabetes of the young (MODY).
This study aims to describe some of the epidemiological criteria of diabetes and the trend of the disease in children followed in the Department of pediatrics at King Fahad Hospital (KFH) in the diabetes clinic and the diabetes pediatric clinic in Diabetic Center.
Research and Methods
Our study settings included The Pediatric Endocrinology Clinic in KFH in Al-Baha, a 400 bed capacity, and pediatric clinic in the Diabetes Center in AL-Baha region in Saudi Arabia. The inclusion criteria included all the children and adolescents with diabetes and followed in the above settings as out-patients. They included males and females with age group ≤ 18 years from the AL-Baha and nearby areas during the year 2007- 2014. The exclusion criteria included patients who had diabetes with age greater than18 years. A total of 372 patients were seen in our study. Depending on the computer based access program, data sheets contained the name of the patients, the file number, the age at diagnosis, gender, date of diagnosis, the first presentation with diabetes, the family history of diabetes, the family history of other autoimmune diseases, maternal education, area of residence ,socioeconomic status, duration of breast feeding, celiac profile results, associated autoimmune diseases, complications, other diseases, type of diabetes management regimen and the latest HbA1C level. This prospective study started in the year 2007 and finished in the year 2014.All the children and adolescents followed in both clinics were enrolled in the study after taking written approval from the administrative authority and a verbal approval from the parents to enroll their children in this study, and to collect the information and enter it into the computerized data collecting sheets. Most of the information was collected by direct questions to the parents during follow up visits with their children to the pediatric endocrinology clinic. The remaining information such as date of diagnosis, last HbA1C value, and celiac profiles, was taken from the medical records of the patients. Male and female patients were divided into three age groups: one month to six years of age, from seven to 12 years of age and from 13 to 18 years of age.(Note the age groups divided to this according to difference normal hormonal physiological change and category of international insulin dose etc)
Results
A total of 372 diabetic children and adolescents aged one month to 18 years were studied. The mean of age was 8.32 years, and the CI was 95% (+7.91 to + 8.73) with p value < 0.001. The diabetic center and the pediatric clinic were the main source of the target population. Therefore, the incidence rate and prevalence represent the incidence rate and the prevalence in the Al-Baha area. The average incidence rate was 8.6/100000, among a total population of 231,474 children and adolescents aged18 years and younger. The highest incidence rate was 59 children in 2012, and it was 25.48/100000, the lowest incidence rate was in 1996 and 1999, when one patient was affected, constituting 0.3%, as shown in figure 1. The highest incidence was in autumn, and constituted 27.8 % of all cases (105cases), while winter surprisingly had the lowest incidence i.e 19.6 % of all cases (75cases).
Figure 1.Cases distribution according to years and sex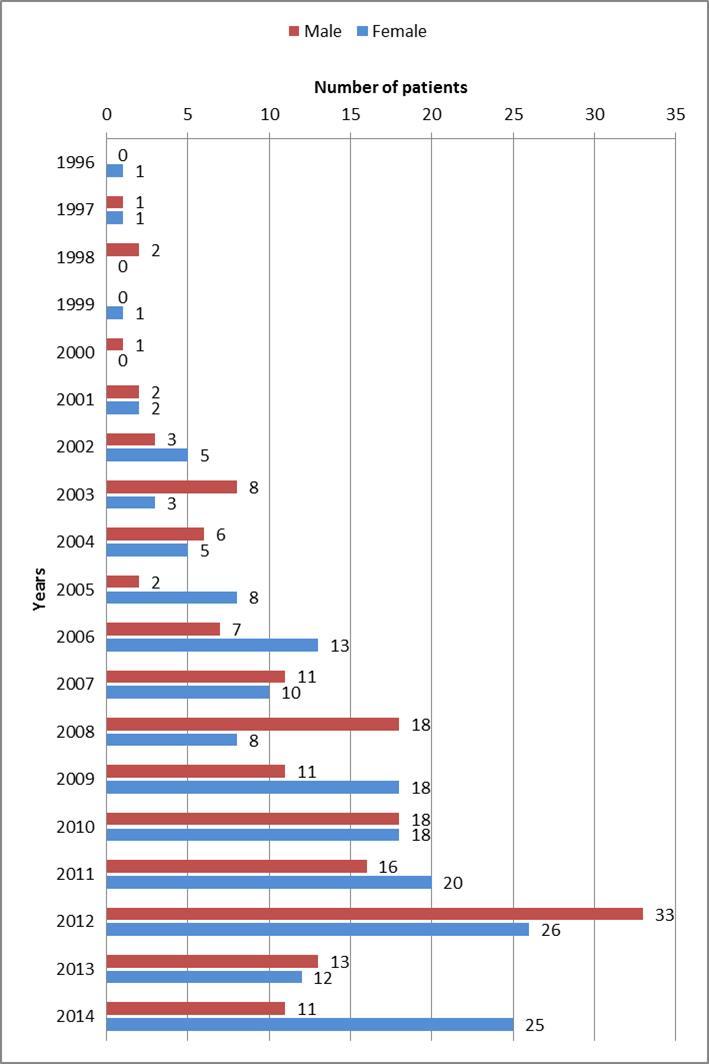
Our sample was divided into three age groups: the first age group, between one month and six years, constituted 34.4% cases (128), the second age group, 7- 12 years of age ,constituted 46.7% cases (174), and the third group, 13-18 years of age, constituted 15.59% cases (58 ). 2.9% were of unknown age (11cases).
In this correlation with age, the first age group was the most affected from 1996 until 2004, after which the second age group presented as the most prevalent age group, as shown in figure 1. Females were slightly more affected than males, constituting 52 % cases (194). Furthermore, in correlation to age groups, both females and males were more affected in the second age group.
T1DM constituted 95.4% cases (355), being the most common in the second age group. Of the remaining 4.6% cases 17 T2DM constituted 1.88% cases 7, which was most common in the third age group. Monogenic diabetes affected 0.79% cases (3 cases; one case being confirmed as monogenic neonatal diabetes by a genetic study, the other two cases were suspected to be MODY). The remaining 1.06% cases 4 were patients with stress hyperglycemia and one patient with an impaired oral glucose tolerance test (OGT), as shown in figure 2.
Figure 2.Cases distribution according to diabetes types and age groups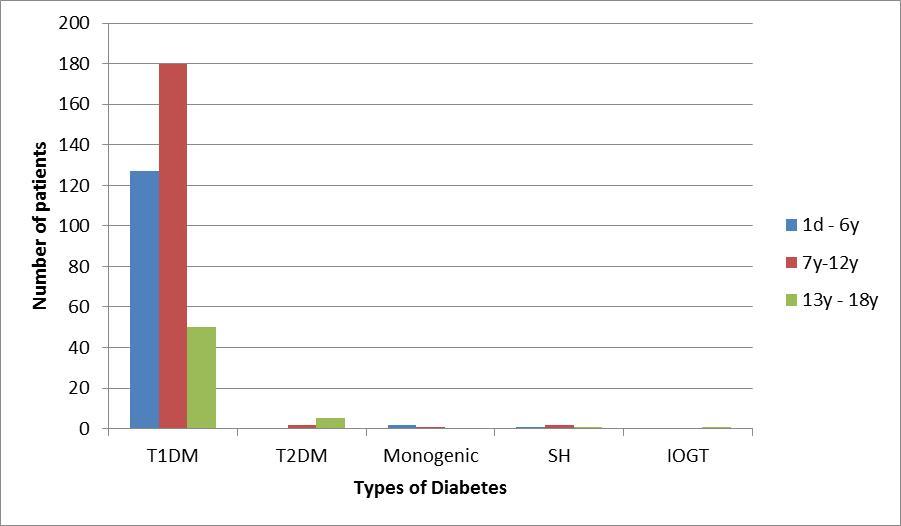
The duration of diabetes in 48.3% (180 cases) was less than five years, and in 49.6% (184 cases) was more than five years. The remaining 2.1 %( eight cases) were of unknown duration. Correlating the duration of diabetes with the most prevalent chronic complications of diabetes (diabetic nephropathy) showed an incidence of 17.02% (32 cases) in those who had diabetes for more than five years.
Al-Baha city had the highest number of cases, constituting 37.7% (140 cases).Second was the city of Al-Meqwa with 15.1% (56 cases). The city of Qelwa had the least cases, where there was 4.5 %( 17 cases).
Diabetic ketoacidosis (DKA) as the first presentation constituted 44.2% of patients (167 cases). Presentation other than DKA (polyuria, polydipsia, weight loss only) comprised 50% of the patients (189 cases) and was more common in all age groups, as shown in figure 3and was also more common in males, P value 0.003. Correlating the first presentation as DKA with the mothers education showed 18.5% (31cases) of children who had illiterate mothers, 17.3% (29cases) where mothers had primary education, 20.35% (34 cases) where mothers had intermediate and secondary school education and, surprisingly, 33.5% (56 cases) where the mother had college and postgraduates degrees. DKA as the first presentation was more common in children with mothers with postgraduate education, with 33.5% (56 cases) and least common in mothers who were illiterate with 18.5% (31cases). Mothers of unknown level of education having children with DKA as the first presentation were10% (17 cases), with the P value being non-significant (0.270), as shown in table 1.
Figure 3.Cases distribution according to 1stpresentation and age groups
| DKA as FIRST PRESENTATION correlated with MOTHER EDUCATION | |||||||||||
| MOTHER EDUCATION | |||||||||||
| DKA as 1st presentation | Illiterate | Primary | Intermediate & Secondary | University & Post Graduate | Unknown | Total | |||||
| No. | % | No. | % | No. | % | No. | % | No. | % | ||
| 31 | 18.5 | 29 | 17.3 | 34 | 20.35 | 56 | 33.5 | 17 | 10 | 167 | |
Correlating the first presentation with socioeconomic class in children with T1DM:74.2% (276 cases) had high socioeconomic class, 45.6%(126 cases) of which had DKA as the first presentation .19.9 % (74 cases) of patients had low socioeconomic class, of which 40.5 % (30cases) had DKA, with a significant p value 0.048, the correlation is significant at the 0.05 level (1-tailed), as shown in table 2.
Table 2. Correlation between 1st presentation & socioeconomic classes| First Presentation & Socioeconomic Class | ||||||||
| FIRST PRESENTATION | ||||||||
| Socioeconomic Class | DKA | OTHERS (polyuria, polydepsia, wt. loss) | Unknown | Total | ||||
| High | 126 | 45.60% | 144 | 52.2% | 6 | 1.8% | 276 | 100% |
| Low | 30 | 40.5% | 42 | 56.8% | 2 | 2.7% | 74 | 100% |
| Unknown | 11 | 52.4% | 3 | 14.3% | 8 | 36.3% | 22 | 100% |
| Total | 167 | 189 | 16 | 372 | 100% | |||
Consanguinity was present in 39.7% (148) of cases .Regarding family history of T1DM,41.1% (153cases) had positive family history of T1DM, p value 0.03,< 0.05.Regarding the family history of autoimmune diseases (thyroid diseases, celiac and adrenal),8.8% (33cases) had thyroid disease, 5.9% (22cases) had a family history of both T1DM and thyroid disease, and 4% were of unknown status (16 cases).The time of introduction to bottle feeding (cow’s milk) was known for 248 patients, 83.46 % (207cases) of which started cow’s milk before six months.10% (25cases ) started after the age of six months, and 6.45% (16cases) never received .The correlation was significant at the 0.01 level (2-tailed). Regarding associated autoimmune diseases; celiac disease in patients with T1DM occurred in10.4% (39cases), 64% of which were female. The celiac disease profile was not available in 14.7% (55cases). A duodenal biopsy was performed in six patients who had celiac disease, which was positive in three girls and one boy, as shown in figure 4. Associated thyroid diseases were present in 3.49% (13cases), 77 % of which were females; p value 0.001. The correlation is significant at the 0.01 level, as shown in Figure 5
Figure 4.T1DM and associated celiac disease according to sex
Figure 5.Thyroid problems in children with T1DM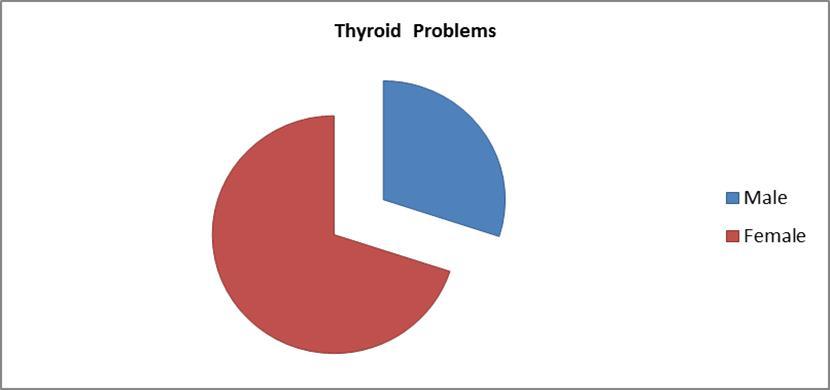
The chronic complications of diabetes in this study were 7.8 % ( 32cases) with nephropathy,7.2% (27cases) with neuropathy,1.3 % (five cases) with retinopathy,12.4% (46cases) were overweight or obese, and hyperlipidemia was present in 4.3% (16 cases),as shown in figure 6. Correlating the rate of complications with diabetes duration: those who had diabetes for more than five years,87.5% (28 cases)had nephropathy, p value < 0.01; 85.1% had neuropathy, p value 0.01; 85.7 % had retinopathy, with a significant p value < 0.01;and 81.25% had hyperlipidemia, also with a significant p value < 0.01 as shown in figure 7
. Correlations were significant for all apart from obesity and overweight at the 0.01 level. 36.95% of patients were obese or overweight, with non-significant p value of 0.4.
Figure 6.T1DM and associated complications according to sex
Figure 7.Chronic complications according to diabetes duration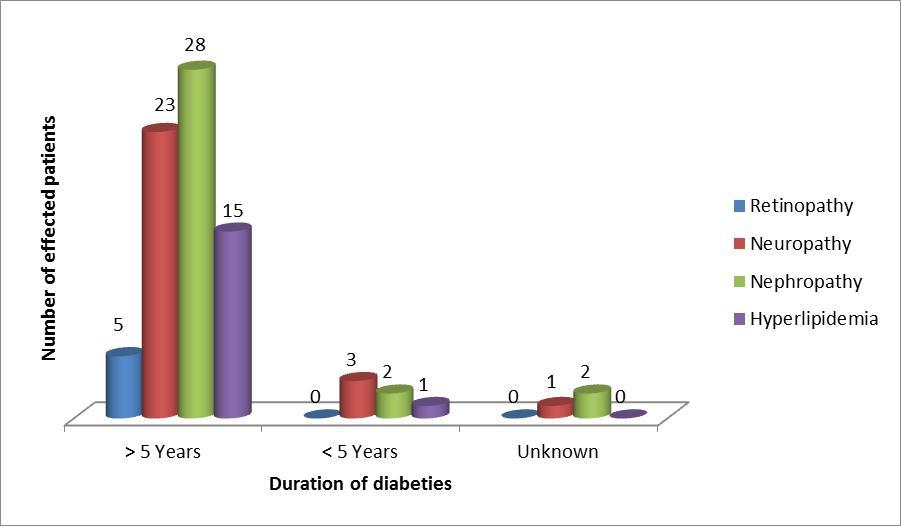
In this study, 19.3% (72 cases) had HbA1c values below 7.5%, of in which females constituted 61% (44 cases).HbA1c for 17.4% (65cases). The correlation of last HbA1c with insulin therapy regimens combined with oral metformin had the highest frequency 45.8% (170cases), 21.7% (37cases) had an intensive regimen, of which last HbA1c was less than 7.5%. 3.5% (six cases) had unknown HbA1C information. As shown in Figure 8, 28.5% (106 cases) had conventional regimen, 19.8% (21cases) of which had a last HbA1C less than 7.5%, 4.7% (five cases) had unknown HbA1c values, p value < 0.01. The correlation was significant at the 0.01 level (2-tailed), as shown in table 3.
Figure 8.Types of treatment regimens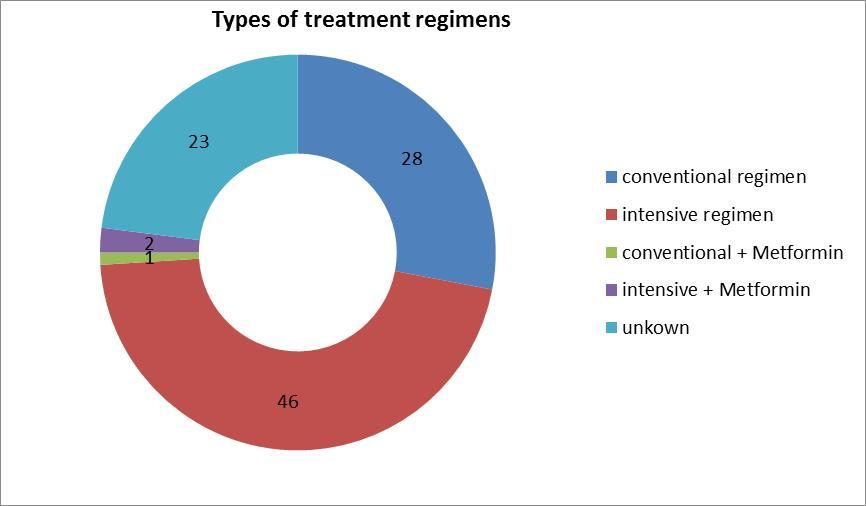
| LAST REGEMIN & LAST HbA1C | |||||
| LAST REGEMIN | LAST HBA1C | ||||
| Less than 7.5 | More than 7.5 | UNKNOWN | Total | ||
| C (CONVENTIONAL) | 19.8% (n.21) | 74.5% (n.79) | 5.6 % (n. 6 ) | 106 | |
| I (INTENSIVE) | 21.7% (n.37) | 75.2% (n.128) | 3% (n.5) | 170 | |
| C + MET (METFORMIN ) | 66% (n.2) | 33% (n.1) | 0 | 3 | |
| I + MET (METFORMIN ) | 14.2%(n.1) | 85.7(n.6) | 0 | 7 | |
| NO TREATMENT (TR) | 50% (n.1) | 0 | 50% (n.1) | 2 | |
| UNKNOWN | 12% (n.10) | 25% (n.21) | 63% (n.53) | 84 | |
| Total | 72 | 235 | 65 | 372 | |
Discussion
Our results from this study shows that T1DM is the most common type of diabetes in children and adolescence less than 18 years old. It is more common in females, as well as being more common in both sexes in ages between 7 and 12 years (second age group). These data are inconsistent with Kulaylat and Narchi11. As well controversy, in AL- Herbash study, who mentioned no differences between the sexes 7.Moreover, the results ofBell showed that males are more affected, in contrast to our results 6. In this study, the females were found to be more represented than males in the first presentation with ketoacidosis with a 1.49:1 female to male ratio, similar to the Habib study 4, who observed a 1.4: 1 female to male ratio. The mean age at presentation with T1DM was 8.324 years ranging from one month old to 18 years old, CI 95% (7.91 to + 8.73).The first presentation was with DKA in 44.2% of cases which is similar to the Habib study 4. This is a high percentage compared with other international studies, for example the study by Arleta Rewers 14.
Our study shows bimodal peaks through the year, with the first from mid-summer to the beginning of winter, and the second in spring. This result contrasts with the results of the Lorenzi study 3. Moreover, Moltchanova and DiMe 13, 27, showed a single peak, mainly in the winter months. In the USA, T1DM diagnosis is least in summer time 26, which supports exploring factors other than viral infection, which is common in winter that may affect the seasonal distribution of the disease in Al-Baha area.
The increasing annual percentage of new cases attending the diabetes clinics, especially in the last three years, may reflect the general trend of the disease in Al- Baha area which correlates with international trend, sin URODIAB study group 8, Furthermore, (DiaMond) Project Group 9, and Taplin 10, mention in their conclusion similar incidencesin Switzerland Italy as we report in this study28.
The DKA percentage was surprisingly higher in mothers with a university education and reached 33.5% in mothers with a high education level. High percentage of T1DM in children who started with cow’s milk at less than six months was 83.46 % (207cases).This may indicates that the breast feeding plays an important role in disease prevention, which is similar to the findings of the by Malcova16.
Parental consanguinity is high in our patients, at39.7%, and indicates that consanguinity may play an important role in the etiology of this disease. This is in contrast to the study by El Mouzan, 17 .74.1% of the children`s families had a good socioeconomic status, which is in agreement with international trends 18.10.48% of cases had a positive celiac disease profile and were in need of intestinal biopsies to rule out celiac disease. This was similar to the study by Bhadad et al, which reported11.1% 19.Our study shows 37.9 % (141cases),coming from Al-Baha, the second most cases from the Al- Meqwa area , with15.1% (56 cases),and the least in Qelwa with only 4.5% which the most hot area compared with others which have adopted weather .
Our study shows that nephropathy was the most prevalent chronic microvascular complications. This finding is in contrast the UKPDS study, which previously reported that retinopathy is the most common complication (20, 21). In our study, the obesity rate was 12.4% (46cases) which is less than study by Sandu, who reported an obesity rate of 29.5% 22. Finally, our study shows that the percentage of patients on intensive regimen withHbA1C 7.5and less was higher than those with a conventional regimen and is similar to the comparable studies by SEARCH and DCCT 24, 25.
Conclusion
In conclusion, the present study demonstrates the epidemiological features of T1DM. The principal focus of this study was to identify similarities with international. Although DKA is more prevalent at the first presentation in our study, this can be combined within a unified framework to answer scientific and clinical questions of diabetes in children. This study should be extended to involve all of the patients in other regional hospitals to give a better understand the epidemiology. Other factors, such as association with viral infection, vaccination history and vitamin D deficiency, could be included in this extension. Moreover, prevention of diabetic ketoacidosis and a reduction of its frequency among patients should be a goal in managing children with diabetes. This may in the future identify an effective treatment approach from the beginning before treatment even starts. Rising standards of medical information and general awareness of diabetes can contribute to this which may support the diagnosis of diabetes.
Acknowledgment
The authors gratefully acknowledge Dr Majida N. Nasaif for her valuable help.
References
- 1.Kliegman R, Stanton B, GemeJ. (2016) . Nelson textbook of pediatrics 20th edition ; Section 6 Diabetes mellitus in children PART XXVI 589.
- 2.Holt R I. (2004) Diagnosis, epidemiology and pathogenesis of diabetes mellitus: an update for psychiatrists. , Br J Psychiatry 47, 55-63.
- 3.Lorenzi M, Cagliero E, Schmidt N J. (1985) Racial differences in the incidence of juvenile-onset Type 1 diabetes: epidemiologic studies in southern CaliforniaDiabetologia. 28, 734-738.
- 4.Habib H S. (2005) Frequency and clinical characteristics of ketoacidosis at onset of childhood type 1 diabetes mellitus in. , Northwest Saudi ArabiaSaudi Med J 26, 1936-1939.
- 5.Joner G, Søvik O. (1981) Incidence, age at onset& seasonal variation of diabetes. in Norwegian children1973 -1977 ActaPaediatrica 3, 281-440.
- 6.Bell R A, Mayer-Davis E J, Beyer J W. (2009) . SEARCH for Diabetes in Youth Study Group Diabetes Care.32 Suppl 2, 102-11.
- 7.Herbish A, El-Mouzan M, Al-Salloum A. (2008) Prevalence of type 1 diabetes mellitus in Saudi Arabian children and adolescents Saudi Med. , J 29(9), 1285-8.
- 8.Patterson C, Dahlquist G, Gyrs E. (2009) Incidence trends for childhood type 1 diabetes in Europe during 1989-2003 and predicted new cases 2005-20:A multicentre prospective registration study Lancet. 373, 2027-2033.
- 9.Karvonen M, Viik-Kajander M, Moltchanova E. (2000) Incidence of childhood type 1 diabetes worldwide. Diabetes Mondiale (DiaMond) Project Group Diab care. 23, 1516-1526.
- 10.Taplin C, Craig M, Lloyd M. (2005) The rising incidence of childhood type 1 diabetes in New South Wales. , Med J Aust 183(5), 243-246.
- 11.Kulaylat N A, Narchi H. (2012) A twelve year study of the incidence of childhood type 1 diabetes mellitus in the Eastern Province of Saudi ArabiaDiabetologia. 55(11), 2878-2894.
- 12.Romesh K, Aneela N, Howard A. (2016) . Diabetes Mellitus-Type1ONLINE.emedicine.medscape.com/article/117739-overview
- 13.Moltchanova E V, Schreier N, Lammi N.(2009)Epidemiology Seasonal variation of diagnosis of Type 1 diabetes mellitus in children worldwideDiabeticMedicine. 26(7), 673-678.
- 14.Rewers A, Klingensmith G, Davis C. (2008) . Presence of Diabetic Ketoacidosis at Diagnosis of Diabetes Mellitus in Youth study Pediatrics 121(5), 1258-66.
- 15.American Diabetes Association (2010) Diagnosis and classification of diabetes mellitus Diabetes Care. 33(1), 62-9.
- 16.Malcova H, Sumnik Z, Drevinek P.(2006)Absence of breast-feeding is associated with the risk of type 1 diabetes: a case–control study in a population with rapidly increasing incidenceEuropean. , Journal of Pediatrics 165(2), 114-9.
- 17.El Mouzan MI, Al Salloum AA, Al Herbish AS. (2008) Consanguinity and major genetic disorders in Saudi children: a community-based cross sectional study Ann Saudi Med. 28(3), 169-173.
- 18.Zalloua P A, Terwedow H, Shbaklo H. (2003) Host and environmental factors defining the epidemiology of type 1 diabetes mellitus in a group of Lebanese children and young adultsJ. , PediatrEndocrinol 16(5), 759-69.
- 19.Bhadada S K, Kochhar R, Bhansali A. (2011) Prevalence and clinical profile of celiac disease in type 1 diabetes mellitus in north. , India J Gastroenterol Hepatol 26(2), 378-81.
- 20.Gross J L, de Azevedo MJ, Silveiro S P. (2005) Diabetic nephropathy: diagnosis, prevention, and treatment Diabetes Care. 28, 176-188.
- 21.Chaturvedi N, Bandinelli S, Mangili R. (2001) Microalbuminuria in type 1 diabetes: rates, risk factors and glycemic threshold. , Kidney Int 60, 219-227.
- 22.Sandhu N, Witmans M B, Lemay J F. (2008) Prevalence of overweight and obesity in children and adolescents with type 1 diabetes mellitusJ. 21(7), 631-40.
- 23.Robertson K J, Schoenle E, Gucev Z. (2007) . Insulin detemir compared with NPH insulin in children and adolescents with Type 1 diabetes Diabet Med.24 1, 27-34.
- 24.Pihoker C, Badaru A, Anderson A. (2013) Insulin regimens and clinical outcomes in a type 1 diabetes cohort: the SEARCH for Diabetes. in Youth study Diabetes Care.36 1, 27-33.
- 25.Rubin R R, Peyrot M. (1994) Implications of the DCCT. Looking beyond tight control Diabetes Care. 17, 235-236.
- 26.LaPorte R E, Matsushima M, Chang Y F. (1995) Prevalence and Incidence of Insulin-Dependent Diabetes;. , In M. Harris(Ed). Diabetes in America 95(1468), 37-46.
Cited by (9)
This article has been cited by 9 scholarly works according to:
Citing Articles:
Archives of Pharmacy Practice (2021) OpenAlex
N. Ahmed, A. AlQahtani, Nader Mousa Rubayyi Albalawi, Fawaz Khalifah M Alanazi, Faisal Mohammed Alharbi et al. - Archives of Pharmacy Practice (2021) Semantic Scholar
Archives Of Pharmacy Practice (2021) Crossref
South African Journal of Child Health (2021) OpenAlex
M. Karsas, A. Terblanche, T. Kemp, J. van Dyk - South African Journal of Child Health (2021) Semantic Scholar
Gene Reports (2019) OpenAlex
D. Al-Koofee, A. A. Jasim, Hosam A.A. Al-Issawi - Gene Reports (2019) Semantic Scholar
Gene Reports (2019) Crossref
International Journal of Pediatrics and Adolescent Medicine (2019) OpenAlex
International Journal of Pediatrics and Adolescent Medicine (2019) Crossref
A. Alaqeel - International Journal of Pediatrics and Adolescent Medicine (2019) Semantic Scholar
Journal of Preventive Medicine And Care (2016) OpenAlex
