Capsaicin: A Potential Therapy Adjuvant for Intestinal Bowel Disease
Abstract
Most of the patients with inflammatory bowel disease avoid pepper or spicy food, alleging that this condiment causes anal sensation of burning and accelerates intestinal movements. Capsaicin is the main bioactive component of peppers responsible for the pungent flavor that characterizes red peppers. Capsaicin has been related to several biological effects, including decreased body fat, antianti-inflammatory, anticarcinogenic, antioxidant activites and modulator of intestinal motility. These actions mostly are due to its role as an agonist of the transient receptor potential vanilloid 1 (TRPV1), expressed in the mesenteric nervous system and epithelial cells of the colon. Nonetheless, the anti-inflammatory action of capsaicin is also related to its role in activating the peroxisomal proliferator-activated receptor gamma (PPAR-γ). Topical capsaicin formulations are already used for pain management, but oral administration of capsaicin is rare. Here, we discuss the main actions of capsaicin that could interfere with the symptoms and severity of IBD. Although animal experiments suggest a beneficial effect of capsaicin on colitis, clinical studies exploring the potential analgesic and anti-inflammatory of capsaicin on Crohn or Ulcerative Colitis are scarce. We concluded that there is no evidence that capsaicin aggravates IBD symptoms or severity. On the opposite, experimental studies suggest that capsaicin could reduce intestinal inflammation by a mechanism that could involve not only the TRPV1 receptor but also PPAR γ. However, clinical studies are still scarce, and data regarding capsaicin concentrations, routes of administration, and long-term side-effects need to be better understood before its use.
Author Contributions
Academic Editor: Qiang Cheng, Biomedical Informatics Institute, and Computer Science Department, China.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2019 Elandia A dos Santos, et al
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Inflammatory bowel diseases (IBD) comprise two disorders: Crohn's disease (CD) and ulcerative colitis (UC). The inflammatory process seen in UC extends from the rectum continually forward colon, affecting the superficial layers of the mucosa. The inflammation in CD involves all intestinal layer of any part of the gastrointestinal tract, alternating healthy and inflamed tissue in a non-contiguous pattern 1. IBD may start at any age, being men and women equally affected with a first peak incidence between 20 to 40 years and the second peak in old age 2. However, an increasing number of children and adolescents are also being diagnosed 1.
The etiology of IBD is not entirely understood yet. It is triggered by genetic and environmental factors that lead to disruption of the intestinal mucosa barrier with bacterial (or bacteria components) translocation, leading to exacerbated immune responses and chronic inflammation 3, 4. The most common symptoms of IBD are tiredness, abdominal pain, fever, diarrhea with blood or mucus, rectal bleeding, anemia and weight loss 5. Extraintestinal manifestations involve the dermatologic and musculoskeletal systems 6. Multiple sclerosis, psoriasis, rheumatoid arthritis and colon cancer are frequently associated with IBD 7, 8. Both UC and CD are mostly treated with a combination of aminosalicylates and immunosuppressants. The treatment should be maintained for life long due to frequent recurrences. The use of nutraceuticals could support conventional treatment, reducing the use of anti-inflammatory and immunosuppressants drugs and the consequent side effects 2.
Innate and adaptative immune responses are involved in the development and progression of IBD. The imbalance between regulatory T cells (Treg) and effector T cells (Th1, TH2, and TH17) is related to the development of UC and CD 9, 10. Pro-inflammatory cytokines such as tumor necrosis factor (TNF), interleukin (IL)-1β, IL-6, IL-17A, and interferon (INF)-γ play essential roles in the control of intestinal inflammation and clinical symptoms of IBD. Interleukin (IL)-1β increases the recruitment of granulocytes and activation of innate lymphoid cells (ILCs), participating in the initiation of the colon inflammation 4. IL-23-responsive ILCs are responsible for intestinal inflammation through secretion of IL-17A and INF-γ, in addition to the recruitment of inflammatory cells 11. On the other hand, the presence of anti-inflammatory cytokines such as IL-10, IL-37, and transforming growth factor (TGF)-β, are involved with the control of disease progression 12.
Pain is a recurrent IBD manifestation, which is present in more than 80% of patients 5. The hyperalgesic state in the colon is initiated by the release of cytokines and other inflammatory mediators, such as serotonin, bradykinin, prostaglandins that activates sensory neurons 13. One of the peripheral mechanosensors in afferent neuronal fibers is the transient receptor potential vanilloid 1 (TRPV1). TRPV1 is an ionic channel that permeates sodium and, preferentially, calcium (Ca2+). It is expressed in several organs, including the mesenteric nervous system and involved in intestinal motility, visceral nociceptive behavior, and intestinal hyperalgesia 14, 15. The role of TRPV1 in IBD-related pain is reinforced by studies showing that TRPV1 inhibitors block the hyperalgesia related to colon inflammation. Moreover, the hypersensitivity related to intestinal distension and with inflammatory mediator's release is absent in TRPV1-deficient (KO) mice 16, 17. One of the TRPV1 natural binders is the capsaicin, making this dietary component a potential adjuvant in IBD treatment.
Pepper, Capsaicin, and IBD
Pepper, together with foods rich in fiber, is the most cited avoided foods for IBD active and in remission due to the association of such condiment with burning sensation and increased bowel movement 18.
Capsaicin (trans-8-methyl-N-vanillil-6-Nonenamide) and dihydrocapsaicin are the main capsaicinoids from peppers (Capsicum genus), responsible for the pungent flavor that characterize red peppers. Capsaicin, an alkaloid highly volatile and hydrophobic, is the main bioactive and strongest capsaicinoid 19, 20. The content of capsaicinoids in peppers is about 0.1 to 2.5 mg/g pepper and the consumption of capsaicin in the human diet is estimated at 0.5 to 4 mg/kg/day 21. The Scoville organoleptic test is used to measure the degree of the pungency of several peppers (Figure 1).
Figure 1.Scoville Scale of Pungency 58.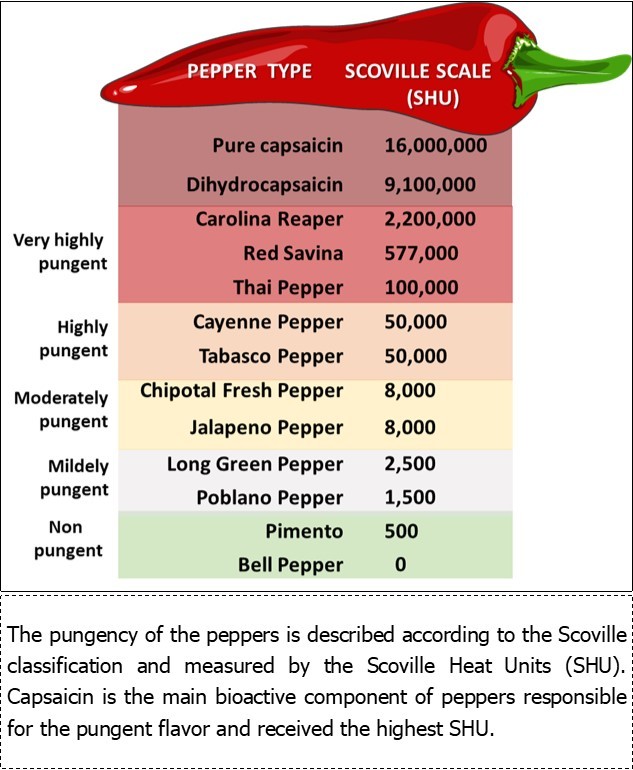
Studies in rats showed that the absorption of capsaicin (isolated) occurs mainly in the proximal portion of the gastrointestinal tract. Following intragastric administration, approximately one-third to one half of the capsaicin dose was absorbed from the stomach. The total intestinal absorption amounted to almost 90% 22. After absorption, capsaicin is passively absorbed and metabolized in the liver by cytochrome P450 system, generating metabolites such as vaniline, vanillamine, vanyl alcohol 23. Nonetheless, none of the capsaicin metabolites presented metabolic activity 24.
The amount of capsaicin reaching colon to be absorbed is not well known. It is possible that pure capsaicin has a faster and more proximal absorption than capsaicin contained in the spicy foods. It is well known that interactions with other food components such as dietary fibers can slow the absorption rate of some nutrients. In this way, a more significant portion of capsaicin from natural food could reach the colon compared to pure capsaicin. Some individuals allege that capsaicin containing foods induce an anal sensation of burning, suggesting that capsaicin reaches the distal parts of the colon. Nonetheless, studies with healthy individuals receiving capsaicin capsules showed that burning sensation during bowel movements was not related during the experiment 25.
Capsaicin has been related to several biological effects, including reduction of body fat 26, 27, 28, anti-inflammatory, anticarcinogenic, antioxidant activites and modulator of intestinal motility 29, 30, 31, 32, 33, 34, 35. Most of the capsaicin actions are due to its role as TRPV1 agonist. TRPV1 can be activated by acidity and high temperatures and is described as a molecular integrator of chemical and physical stimuli that provoke pain. In the inflammatory process, the stimulation of sensory neurons leads to a response in peripheral tissues and transmission of nociception to the central nervous system. In this context, TRPV1 mediates the transmission of sensory neuron signaling involved in thermal hyperalgesia and visceral sensitivity. In the human colon, TRPV1 is overexpressed in the epithelial cells and afferent nerves during inflammation 36.
TRPV1 activation leads to the release by sensory fibers of neuropeptides such as substance P (SP) and the calcitonin gene-related peptide (CGRP). These neuropeptides, besides their vasodilator effects, drive the transmission of nociception in the colonic mucosa. In this way, activation of TRPV1 contributes to the generation of inflammatory responses through the recruitment and migration of leukocytes to the site of inflammation, a process that is accompanied by visceral hypersensitivity 37, 38.
The transient activation of TRPV1 leads to the influx of calcium ions, leading to the membrane depolarization that results in an action potential that propagates the signaling to the brain. However, when a stable and persistent agonist, such as capsaicin, is present, TRPV1 significantly increases the influx of calcium to the intracellular environment of sensitive fibers. Organelles expressing TRPV1 also release calcium into the intracellular environment. The persistently elevated levels of intracellular calcium cause dysfunctionality of nerve fibers due to temporary loss of membrane potential. In this way, chronic capsaicin exposition results in inhibition of pain transmission by inducing persistent calcium influx and nerve fiber dysfunctionality. Capsaicin also downregulates the synthesis, storage, transport, and release of SP and CGRP, both messengers of peripheral pain impulses to the central nervous system (Figure 2). The depletion of SP in nerve terminals reduces or blocks the propagation of pain impulses toward the brain 31. In summary, although in short‐term capsaicin can excite afferent neurons and induce burn sensation (by stimulating TRPV1), chronic exposure causes a block of sensory neurons, attenuating the noxious stimuli 25.
Figure 2.The proposed mechanism for analytical and anti-inflammatory actions of capsaicin in the colon. Capsaicin is a natural agonist of transient vaniloid receptor potential 1 (TRPV1). TRPV1, when activated, leads to the influx of calcium ions, leading to membrane depolarization that results in a potential action that propagates signaling to the brain. However, when a stable and persistent agonist such as capsaicin is present, persistently high intracellular calcium levels cause afferent nerve fiber dysfunction due to loss of membrane potential, inhibiting pain signal spread. Capsaicin also decreases the release of the P component and calcitonin gene-related peptide (CGRP), messengers of peripheral pain impulses in the central nervous system. Capsaicin is also a peroxisome proliferator-activated receptor (PPAR-γ) agonist that regulates inflammation of the colon. When activated, PPAR-γ binds to other nuclear receptors, such as retinoid receptor X (RXR), forming a heterodimer that in turn binds PPAR response elements (PPRE) in DNA. As a result, a protein expression encoded by the target gene will be regulated up or down. PPAR-γ also regulates gene expression by interacting with other transcription factors, such as activating protein (AP) -1, signal transducers and transcription activators (STAT), and nuclear kappa factor B (NF-κB). rules the proinflammatory gene expression.
Some studies have shown that TRPV1 channels are involved in the induction and progression of colitis in humans and experimental models 39, 40, 41. However, the participation of TRPV1 channels in inflammatory bowel diseases is still controversial, showing evidence of pro-inflammatory or anti-inflammatory stimuli 42, 13, 44, 45.
Utsumi et al. (2018) investigated the contributions of TRPV1 and TRPA1 in a model of sodium dextran sulfate (DSS) induced colitis. They observed that pretreatment with high doses of capsaicin (100 mg/kg), worsened rectal bleeding, and other inflammatory signs of colitis. In agreement, DSS-induced colitis in TRPV1 KO mice was less intense than in wild-type controls, suggesting that TRPV1 is associated for the colitis progression 46.
However, previous studies of Massa and colleagues showed that colitis induced by dinitrobenzene sulfonic acid was aggravated in TRPV1KO mice 36, suggesting that these receptors could be protective against colon inflammation. Studies with sensory deafferentation induced by high doses capsaicin also exacerbated the inflammation in trinitrobenzene sulfonic acid - induced colitis 47. In contrast with high doses, chronic administration of lower doses of capsaicin attenuated intestinal inflammation in several studies 48, 49, 50.
Few studies have evaluated the role of TRPV receptors in human colitis. A study comparing colon biopsies of patients with active IBD and healthy controls showed that TRPV1 was increased in IBD patients. TRPV1 was also highly expressed in infiltrating inflammatory cells of patients with active disease. Nonetheless, TRPV1 expression was not associated with the disease severity 51. Another clinical study evaluated the expression of TRPV1 to TRPV4 in colonic mucosa biopsies of patients with UC or healthy controls 52. Interestingly, unlike the previous study, patients with UC had a lower expression of TRPV1 channels in colon epithelial cells, suggesting different associations between TRPV1 and colon inflammation. Once again, TRPV1 expression was not associated with IBD clinical manifestation or with the disease severity 52.
The reduction of proinflammatory cytokines, chemokines, and adhesion molecules related to capsaicin activity seem to involve pathways not mediated by TRPV1. Capsaicin is also an agonist of the peroxisome proliferator-activated receptor (PPAR), especially of PPAR-γ 14, 15, 19, 53, 54. PPAR-γ is part of the nuclear hormone receptor superfamily and expressed in epithelial cells, macrophages and lymphocytes, and other immune cells and plays an essential role in the homeostasis of the intestinal mucosa 15. PPAR γ regulates colon inflammation, as demonstrated by animal and clinical studies 55. To exert its action, PPAR-γ binds another nuclear receptor such as retinoid X receptor (RXR)-α, forming a heterodimer that binds to PPAR response elements (PPRE) in DNA 53, 56. As a result of PPAR γ action, the expression of proteins codified by the target genes will be up or downregulated 57. PPAR-γ can also regulate gene expression by interacting with other transcription factors such as activator protein (AP)-1, STAT (signal transducers and activators of transcription), and NF-κB, all factors that also regulate gene expression 57 (Figure 2).
Although there are no specific studies with IBD patients or colitis models, the participation of PPARγ in the capsaicin actions was confirmed by studies with macrophages cell line (RAW) that do not express TRPV1. In this cell line, capsaicin inhibited the lipopolysaccharide (LPS) induced production of pro-inflammatory cytokines (TNF, IL-6, and IL-1β). The mechanism was dependent on PPARγ since a PPARγ agonist intensified the cytokine inhibition, and PPARγ antagonist reverts the inhibition caused by capsaicin 53. The anti-inflammatory effect of capsaicin is possibly the consequence of PPARγ interaction with the transcription factor NF-κB. The NF-κB is closely related to the production of inflammatory cytokines, and it is the primary target for PPAR-γ to suppress inflammatory cytokine production 57 (Figure 2).
In conclusion, studies suggest that capsaicin containing foods unlikely aggravate the symptoms or the severity of inflammatory bowel diseases. On the opposite, some studies in animal models show promising results with the use of capsaicin in the control of intestinal inflammation by a mechanism that could involve not only the TRPV1 receptor but also PPARγ. However, clinical studies are still scarce, and data regarding capsaicin concentrations, routes of administration, and long-term side-effects need to be better understood before its use.
Acknowledgments
The authors are fellow of Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq).
References
- 1.Day A S, Ledder O, Leach S T, Lemberg D A. (2012) Crohn’s and colitis in children and adolescents. , World J Gastroenterol 18(41), 5862-9.
- 2.Rahimi R, Nikfar S, Abdollahi M. (2013) Induction of clinical response and remission of inflammatory bowel disease by use of herbal medicines:Ameta-analysis.WorldJGastroenterol.19(34):. 5738-5749.
- 3.Antoni L, Nuding S, Wehkamp J, Stange E F. (2014) Intestinal barrier in inflammatory bowel disease. , World J Gastroenterol 20(5), 1165-79.
- 5.Perler B, Ungaro R, Baird G, Mallette M, Bright R et al. (2019) Presenting symptoms in inflammatory bowel disease: Descriptive analysis of a community-based inception cohort. BMC Gastroenterol. 19(1), 47.
- 6.Levine J S, Burakoff R. (2011) Extraintestinal manifestations of inflammatory bowel disease. Gastroenterol Hepatol (N Y). 7(4), 235-41.
- 7.de Lange KM, Barrett J C. (2015) Understanding inflammatory bowel disease via immunogenetics. J Autoimmun. Nov;64: 91-100.
- 8.Geremia A, Biancheri P, Allan P, Corazza G R, Sabatino A Di. (2013) Innate and adaptive immunity in inflammatory bowel disease. PT First Department of Medicine , Centro per lo Studio e la Cura delle Malattie Infiammatorie. Autoimmun Rev .
- 9.Imam T, Park S, Kaplan M H, Olson M R. (2018) Effector T Helper Cell Subsets in Inflammatory Bowel Diseases. Front Immunol.9:. 1212.
- 10.Ueno A, Jeffery L, Kobayashi T, Hibi T, Ghosh S et al. (2018) Th17 plasticity and its relevance to inflammatory bowel disease. , J Autoimmun 87, 38-49.
- 11.KAG Buela, Omenetti S, Pizarro T T. (2015) Cross-talk between type 3 Innate lymphoid cells and the gut microbiota in inflammatory bowel disease. Current Opinion in Gastroenterology. Lippincott Williams and. , Wilkins 31, 449-55.
- 12.Tatiya-Aphiradee N, Chatuphonprasert W, Jarukamjorn K. (2019) Immune response and inflammatory pathway of ulcerative colitis. Journal of Basic and Clinical Physiology and Pharmacology. De Gruyter; 30, 1-10.
- 13.Bielefeldt K, Davis B, Binion D G. (2009) Pain and inflammatory bowel disease. Inflammatory Bowel Diseases. 15, 778-88.
- 14.Bouyer P G, Tang X, Weber C R, Shen L, Turner J R et al. (2013) Capsaicin induces NKCC1 internalization and inhibits chloride secretion in colonic epithelial cells independently of TRPV1. , Am J Physiol - Gastrointest Liver Physiol 304(2).
- 15.Chen D, Xiong Y, Lin Y, Tang Z, Wang J et al. (2015) Capsaicin alleviates abnormal intestinal motility through regulation of enteric motor neurons and MLCK activity: Relevance to intestinal motility disorders. Mol Nutr Food Res. 59(8), 1482-90.
- 16.RCW Jones, Xu L, Gebhart G F. (2005) The Mechanosensitivity of Mouse Colon Afferent Fibers and Their Sensitization by Inflammatory Mediators Require Transient Receptor Potential Vanilloid 1 and Acid-Sensing Ion Channel 3. J Neurosci. 25(47), 10981-9.
- 17.Szallasi A, Cortright D N, Blum C A, Eid S R. (2007) The vanilloid receptor TRPV1: 10 years from channel cloning to antagonist proof-of-concept. Nature Reviews Drug Discovery.
- 18.Bergeron F, Bouin M, D’Aoust L, Lemoyne M, Presse N. (2018) Food avoidance in patients with inflammatory bowel disease: What, when and who? Clin Nutr. 37(3), 884-9.
- 19.Sharma S K, Vij A S, Sharma M. (2013) Mechanisms and clinical uses of capsaicin. , European Journal of Pharmacology 720, 55-62.
- 20.O’Neill J, Brock C, Olesen A E, Andresen T, Nilsson M et al. (2012) Unravelling the mystery of capsaicin: A tool to understand and treat pain. Pharmacological Reviews.
- 21.Chanda S, Bashir M, Babbar S, Koganti A, Bley K. (2008) In vitro hepatic and skin metabolism of capsaicin. Drug Metab Dispos.
- 22.Donnerer J, Amann R, Schuligoi R, Lembeck F. (1990) Absorption and metabolism of capsaicinoids following intragastric administration in rats. Naunyn Schmiedebergs Arch Pharmacol.
- 23.Ludy M J, Moore G E, Mattes R D. (2012) The effects of capsaicin and capsiate on energy balance: Critical review and meta-analyses of studies in humans. Chem Senses.
- 24.Suresh D, Srinivasan K. (2010) Tissue distribution & elimination of capsaicin, piperine & curcumin following oral intake in rats. Indian J Med Res.
- 25.Hammer J. (2006) Effect of repeated capsaicin ingestion on intestinal chemosensation and mechanosensation. Aliment Pharmacol Ther. 24(4), 679-86.
- 26.Zheng J, Zheng S, Feng Q, Zhang Q, Xiao X. (2017) Dietary capsaicin and its anti-obesity potency: from mechanism to clinical implications. , Biosci Rep 37(3).
- 27.Kim J, Park J, Lim K. (2016) Nutrition Supplements to Stimulate Lipolysis: A Review in Relation to Endurance Exercise Capacity. , J Nutr Sci Vitaminol (Tokyo) 62(3), 141-61.
- 29.Walker J, Ley J P, Schwerzler J, Lieder B, Beltran L et al. (2017) Nonivamide, a capsaicin analogue, exhibits anti-inflammatory properties in peripheral blood mononuclear cells and U-937 macrophages. Mol Nutr Food Res. 61(2).
- 30.Clark R, Lee S H. (2016) Anticancer properties of capsaicin against human cancer. Anticancer Research. International Institute of Anticancer Research; 36, 837-44.
- 31.Anand P, Bley K. (2011) Topical capsaicin for pain management: Therapeutic potential and mechanisms of action of the new high-concentration capsaicin 8 patch. , British Journal of Anaesthesia. Elsevier 107, 490-502.
- 32.Srinivasan K. (2014) Antioxidant Potential of Spices and Their Active Constituents. Critical Reviews in Food Science and Nutrition 54, 352-72.
- 33.Adaszek Ł, Gadomska D, Mazurek Ł, Łyp P, Madany J et al. (2019) Properties of capsaicin and its utility in veterinary and human medicine. Res Vet Sci. Apr;123: 14-9.
- 34.Smith A S, Smid S D. (2005) Impaired capsaicin and neurokinin-evoked colonic motility in inflammatory bowel disease. J Gastroenterol Hepatol. 20(5), 697-704.
- 35.Yang M H, Jung S H, Sethi G, Ahn K S. (2019) Pleiotropic pharmacological actions of capsazepine, a synthetic analogue of capsaicin, against various cancers and inflammatory diseases. , Molecules
- 36.Massa F, Sibaev A, Marsicano G, Blaudzun H, Storr M et al. (2006) Vanilloid receptor (TRPV1)-deficient mice show increased susceptibility to dinitrobenzene sulfonic acid induced colitis. J Mol Med. 84(2), 142-6.
- 37.Akbar A, Yiangou Y, Facer P, Brydon W G, JRF Walters et al. (2010) Expression of the TRPV1 receptor differs in quiescent inflammatory bowel disease with or without abdominal. 59(6), 767-74.
- 38.Lapointe T K, Basso L, Iftinca M C, Flynn R, Chapman K et al. (2015) TRPV1 sensitization mediates postinflammatory visceral pain following acute colitis. , Am J Physiol - Gastrointest Liver Physiol 309(2), 87-99.
- 39.Engel M A, Becker C, Reeh P W, Neurath M F. (2011) Role of sensory neurons in colitis: Increasing evidence for a neuroimmune link in the gut. Inflammatory Bowel Diseases. 17, 1030-3.
- 40.Kihara N, SG De La Fuente, Fujino K, Takahashi T, Pappas T N et al. (2003) Vanilloid receptor-1 containing primary sensory neurones mediate dextran sulphate sodium induced colitis in rats. Gut. 52(5), 713-9.
- 41.Kimball E S, Wallace N H, Schneider C R, D’Andrea M R, Hornby P J. (2004) Vanilloid receptor 1 antagonists attenuate disease severity in dextran sulphate sodium-induced colitis in mice. Neurogastroenterol Motil. 16(6), 811-8.
- 42.Csekő K, Beckers B, Keszthelyi D, Helyes Z. (2019) Role of TRPV1 and TRPA1 ion channels in inflammatory bowel diseases: Potential therapeutic targets?. , Pharmaceuticals 12.
- 43.Beckers A B, ZZRM Weerts, Helyes Z, AAM Masclee, Keszthelyi D. (2017) Review article: transient receptor potential channels as possible therapeutic targets in irritable bowel syndrome. Aliment Pharmacol Ther. 46(10), 938-52.
- 44.Boesmans W, Owsianik G, Tack J, Voets T, Vanden Berghe P. (2011) TRP channels in neurogastroenterology: Opportunities for therapeutic intervention. , British Journal of Pharmacology 162, 18-37.
- 45.Parenti A, F De Logu, Geppetti P. (2016) Benemei S. What is the evidence for the role of TRP channels in inflammatory and immune cells?. , Br J Pharmacol 173(6), 953-69.
- 46.Utsumi D, Matsumoto K, Tsukahara T, Amagase K, Tominaga M et al. (2018) Transient receptor potential vanilloid 1 and transient receptor potential ankyrin 1 contribute to the progression of colonic inflammation in dextran sulfate sodium-induced colitis in mice: Links to calcitonin gene-related peptide and substance P. , J Pharmacol Sci 136(3), 121-32.
- 47.Eliakim R, Karmeli F, Okon E, Rachmilewitz D. (1995) Ketotifen ameliorates capsaicin-augmented acetic acid-induced colitis. Dig Dis Sci. 40(3), 503-9.
- 48.Okayama M, Tsubouchi R, Kato S, Takeuchi K. (2004) Protective effect of lafutidine, a novel histamine H 2-receptor antagonist, on dextran sulfate sodium-induced colonic inflammation through capsaicin-sensitive afferent neurons in rats. Dig Dis Sci. 49(10), 1696-704.
- 49.Goso C, Evangelista S, Tramontana M, Manzini S, Blumberg P M et al. (1993) Topical capsaicin administration protects against trinitrobenzene sulfonic acid-induced colitis in the rat. , Eur J Pharmacol 249(2), 185-90.
- 50.Kato S, Tanaka A, Kunikata T, Umeda M, Takeuchi K. (2000) Protective effect of lafutidine against indomethacin-induced intestinal ulceration in rats: Relation to capsaicin-sensitive sensory neurons. , Digestion 61(1), 39-46.
- 51.Luo C, Wang Z, Mu J, Zhu M, Zhen Y et al. (2017) Upregulation of the transient receptor potential vanilloid 1 in colonic epithelium of patients with active inflammatory bowel disease. , Int J Clin Exp Pathol 10(11), 11335-44.
- 52.Rizopoulos T, Papadaki-Petrou H, Assimakopoulou M. (2018) Expression Profiling of the Transient Receptor Potential Vanilloid (TRPV). , Channels
- 53.Park J-Y, Kawada T, Han I-S, Kim B-S, Goto T et al. (2004) Capsaicin inhibits the production of tumor necrosis factor alpha by LPS-stimulated murine macrophages, RAW 264.7: a PPARgamma ligand-like action as a novel mechanism. FEBS Lett. Aug;572(1–3): 266-70.
- 54.Mittelstadt M L, Patel R C. (2012) AP-1 mediated transcriptional repression of matrix metalloproteinase-9 by recruitment of histone deacetylase 1 in response to interferon β. PLoS One. 7(8), 42152.
- 55.Dubuquoy L, Rousseaux C, Thuru X, Peyrin-Biroulet L, Romano O et al. (2006) PPARγ as a new therapeutic target in inflammatory bowel diseases. 55, 1341-9.
- 56.Tang J, Luo K, Li Y, Chen Q, Tang D et al. (2015) Capsaicin attenuates LPS-induced inflammatory cytokine production by upregulation of LXRα. , International Immunopharmacology 264-9.
Cited by (11)
This article has been cited by 11 scholarly works according to:
Citing Articles:
Sherly Usman, Sri Nabawiyati Nurul Makiyah, Wiriasti Danantika - Berkala Kedokteran (2024) Semantic Scholar
Current Issues in Molecular Biology (2023) OpenAlex
G. I. Robinson, Dongping Li, Bo Wang, Yeva Zahoruiko, M. Gerasymchuk et al. - Current Issues in Molecular Biology (2023) Semantic Scholar
Current Issues in Molecular Biology (2023) Crossref
G. I. Robinson, Dongping Li, Bo Wang, Tahiat Rahman, M. Gerasymchuk et al. - Life (2023) Semantic Scholar
Microorganisms (2022) OpenAlex
A. Shehata, S. Yalçın, J. Latorre, S. Basiouni, Y. Attia et al. - Microorganisms (2022) Semantic Scholar
Microorganisms (2022) Crossref
A. Roșca, M. Ieșanu, C. Zahiu, S. Voiculescu, A. Pâslaru et al. - Molecules (2020) Semantic Scholar
