Gastric Pneumatosis: the tale of two late preterm infants with Necrotizing Enterocolitis
Abstract
Gastric pneumatosis though rare, when seen is cause for concern. It has been associated with fulminant necrotizing enterocolitis (NEC) and gastric outlet obstruction, although other associations have been identified. It is not to be confused with pneumoperitoneum and can be managed conservatively as it is not always a harbinger of intestinal perforation. This case report highlights gastric pneumatosis which occurred in association with necrotizing enterocolitis in two late preterm infants. NEC accounts for 5.4% of our Neonatal Intensive Care Unit admissions, however these are the first two cases of gastric pneumatosis seen in the history of our unit. It is crucial for clinical staff to be cognizant of this pathognomonic sign so that cases are not missed, and for the feeding policy that governs the management of late preterm infants to be revisited. That which specifically predisposes a baby with NEC to developing gastric pneumatosis remains unknown and warrants further research.
Author Contributions
Academic Editor: Syed ali, Diplomate National Board, DCH (Glasgow), MNAMS, Fellow in HIV Medicine &FIAP.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2023 Shivon Belle Jarvis
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Gastric Pneumatosis, defined as air within the wall of the stomach 18, continues to be rare with less than 40 cases being described in the literature since the 1960s 18. To our knowledge there has been no published case across the Caribbean region. It has been associated with but not limited to the following: necrotizing enterocolitis (NEC) 16, gastric outlet obstruction (GOO) 12, steroid exposure in a premature infant 4, lactobezoars 6, duodenal obstruction 3, intramural placement of a feeding tube 2, the use of indomethacin in a neonate on synchronized nasal intermittent positive pressure ventilation 17 and cardiac disease prior to and post cardiac surgery 18. Herein we describe the first two cases of gastric pneumatosis that occurred in late preterm infants with NEC in the Neonatal Intensive Care Unit (NICU) at the Sir Lester Bird Medical Center (SLBMC) in Antigua and Barbuda. The SLBMC has just over 1000 deliveries annually and offers both routine and critical newborn care; managing neonates of gestational ages 26 weeks and above. Over the past 10 years (2013 to 2022), the NICU has had 3447 admissions, of which 5.4% were accounted for by NEC (C.Richardson 2023, personal communication, 21 July).
Case A
A late preterm (gestational age 35+2/40) male neonate was born to a 25-year-old multiparous woman with minimal antenatal care (5 visits). Her booking parameters were unremarkable and her antenatal period reportedly uneventful. She presented with preterm, prolonged, pre-labour rupture of membranes (PPPROM) and was therefore induced with Cytotec® (Misoprostol); liquor was said to be clear. There were no signs of chorioamnionitis and no antibiotics or steroids were given during delivery. He was delivered via spontaneous vaginal delivery (SVD), and was suctioned; nil other active resuscitation was required. APGARs were 9 and 9 at 1 and 5 minutes respectively. Essential newborn care was offered. Initial examination was unremarkable with temperature of 36.8◦C. He was of low birth weight at 2.46 kg. He was allowed to room in with his mother on the postnatal ward; however by 6 hours of life he was notably distressed characterized by grunting and accessory muscle use, despite a normal respiratory rate of 56. Pre and post ductal saturations were acceptable (97% respectively).Given the presence of danger signs and his history of prematurity on a background of PPPROM, he was admitted to the Neonatal Intensive Care Unit (NICU), nursed in an isolette, attached to cardiac monitor and was screened. Blood and urine culture were taken, complete blood count documented and Chest X-ray (C-XRAY) requested. He was started on ampicillin and gentamycin which are first line antibiotics based on our local protocol. He required mixed gases via nasal cannula on admission and oxygen saturations were maintained between 88 – 92%. A cow’s milk protein base formula was offered due to the unavailability of breastmilk. He was fed via the orogastric tube (OGT) due to respiratory distress, however due to minimal coffee ground secretions subsequently being noted via OGT, he was kept nil per oral (NPO). C-XRAY findings were in keeping with Congenital Pneumonia, and abdominal XRAY (A- XRAY) was unremarkable. The coffee ground secretions resolved spontaneously, and was attributed to mucosal irritation by the feeding tube. Arterial blood gas done showed compensated metabolic acidosis and hyperoxia. Blood and urine cultures were sterile. By day 3 of admission he was no longer distressed, and respiratory support was discontinued. He was able to resume feeds after being NPO for 48 hours. By day 7 of life however, where discharge was imminent he was noted to be irritable and lethargic, with subsequent abdominal distension. He then had regurgitation episodes and appeared very ill looking. Abdominal examination was significant for mild distension, however his abdomen remained non-tender, with no skin changes or visible bowel loops. Ileus secondary to sepsis was entertained. A-XRAY revealed linear serpiginous radiolucency circumferentially around gastric bubble and within bowel wall primarily within the right lower quadrant see Figure 1.
Figure 1.Anterior posterior abdominal radiograph showing gastric pneumatosis (arrow shown) and pneumatosis intestinalis.
Bowel wall was notably thickened with air in the rectum. As gastric pneumatosis was a finding new to the managing team, intestinal perforation was initially considered; therefore a cross table lateral was requested, which confirmed gastric pneumatosis and pneumoperitoneum was ruled out, see Figure 2.Thrombocytopenia was noted. The lowest count observed was 70x103/uL (x109/L). Radiological and clinical findings therefore were in keeping with NEC stage 2a. He was re-screened and placed on Amoxicillin/Clavulanate potassium and Ceftazidime (second line as per local protocol) and Metronidazole was added.
Figure 2.Cross table lateral radiograph showing gastric pneumatosis (arrow shown)
The patient was then referred to the surgical service. Conservative management continued. Repeat cultures including cerebrospinal fluid were sterile. He completed 10 days of triple therapy, after which he recommenced feeds. He had an uneventful outcome with discharge by day 25 of life. On review one month post discharge, he was thriving with no symptoms/signs suggestive of malabsorption or the presence of a stricture. There were no parental concerns.
Case B
A late preterm (34+2) female infant, twin 2 of a diamniotic dichorionic gestation was delivered to a 23 year old multiparous female with minimal antenatal care, and unremarkable booking parameters. She presented with PPPROM with breech presentation and received 1 dose of dexamethasone and 2 doses of Ampicillin, one of which was administered during the intrapartum period. The baby was delivered via SVD and was suctioned and stimulated at birth. She had a birth weight of 2.11 kg. APGARs were 8 and 9 at 1 and 5 minutes respectively. Subsequent to essential newborn care, she was admitted to the NICU, screened to include blood and urine culture, complete blood count (CBC), and CXRAY and commenced on first line antibiotics. Feeds were commenced at target for age using a cow’s milk protein based formula as breast milk was unavailable. By day three of life baby was noted to have temperature instability, poor feeding, with a markedly distended firm full abdomen, with visible bowel loops. Frank blood was also noted via the nasogastric tube; however she remained haemodynamically stable. She was therefore re-screened, stepped up to second line antibiotics and an urgent A-Xray was done as shown in Figure 3. This revealed gastric pneumatosis, which was promptly recognized given the recent ‘findings of the previous case’, and pneumatosis intestinalis of small bowel. Nil portal venous gas was seen. Thromocytopenia was also confirmed; with the lowest platelet count of 93 x 103/uL(x 109/L) being observed. She was transfused with whole blood as the hemoglobin dropped from 18.0 g/dL to 14.3 g/dL. She remained NPO and received triple therapy for 14 days. Thrombocytopenia resolved. All cultures done were sterile. She recommenced feeds on Day 18 of life, subsequent to a repeat A-XRAY which confirmed radiological resolution of NEC. She subsequently had an uncomplicated outcome and was notably thriving when reviewed one month post discharge.
In both cases there were no adverse or unanticipated events noted during follow up, and no further radiological investigations were required.
Figure 3.Anterior posterior Chest and abdominal radiograph showing gastric pneumatosis (arrow shown) and pneumatosis intestinalis.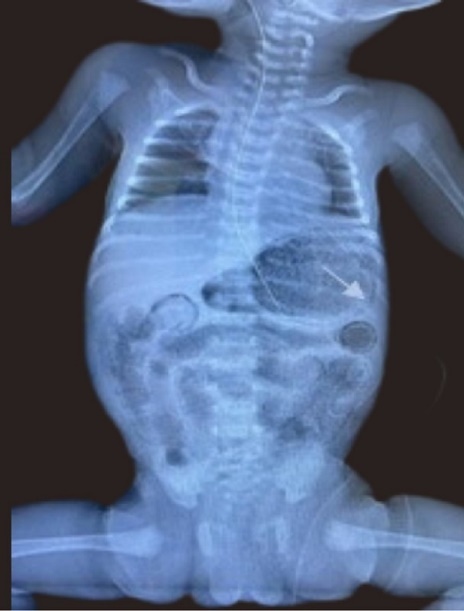
Discussion
Necrotizing enterocolitis (NEC) is classified into suspected (stage 1), confirmed (stage 2), or advanced disease (stage 3) based on systemic, abdominal and radiographic signs 1. Each stage can be further divided into type A or B as per the modified Bell classification 1. Pneumatosis intestinalis (PI), a feature which marks at least stage 2 disease, is characterized by bacterial overgrowth and translocation through mucosal micro perforations, with subsequent gas accumulation within the bowel wall due to bacterial fermentation 8. This is a radiologic pathognomonic sign of NEC 7. Although pneumatosis intestinalis can occur anywhere from oesophagus to rectum 5, gastric pneumatosis is rare and is limited to the stomach.
The pathophysiology of gastric pneumatosis is based on whether it is due to an infectious or noninfectious cause. Cases associated with infection, also known as emphysematous gastritis are characterized by inflammation and damage of the gastric mucosal wall with subsequent diffusion of gas through the necrotic mucosal breaks 17. Escherichia coli, Enterobacter species and Streptococcus are the commonly associated organisms. This proposed pathophysiology however was mostly seen in adults who misused alcohol. In cases associated with obstruction, it is proposed that the mechanical obstruction results in an increase in intraluminal pressure and overdistension which leads to either diffusion of gas into the wall through intact mucosa or escape of intraluminal gas through minor tears in mucosa that is otherwise normal 17. This has also been termed gastric emphysema 18. It is important to note however, that although the radiographic pattern of the intramural gas may be associated with the mechanism by which it occurred, that this is not pathognomonic. Cystic distribution is commonly associated with GOO such as pyloric stenosis, whilst a linear distribution may be seen in mucosal trauma; a bubbly appearance like that seen in PI in NEC is associated with emphysematous gastritis; however it may also be linear 18.
That which seems to be less clear however is whether the presence of gastric pneumatosis is always a harbinger of intestinal perforation. Although gastric pneumatosis is suggestive of widespread and fulminant NEC, with gastrointestinal tract perforation likely being imminent, there have been cases that have been managed conservatively 5, 18, 11, as our cases would have been. Pneumoperitoneum and gastric pneumatosis however have been confused as was the initial scenario in Case A, it is important to note that gastric mural gas remains constant irrespective of position, and that a lateral X-ray can be used to differentiate the two 18. This has significant implications in the ultimate management of the neonate, as the presence of intestinal perforation would dictate either an exploratory laparotomy as is the standard traditional management for bowel perforation, or the consideration of a less invasive modality, that of primary peritoneal drainage which is dependent on the neonate’s clinical state and weight, and the input of the surgeon 14. In our local resource limited context, we have a visiting pediatric surgeon on staff. The presence of intestinal perforation therefore would trigger escalation of care to include the transfer of the pediatric surgeon across borders. In his absence, however as was the scenario in the aforementioned cases, management was offered by the adult surgical team. The outcome of these 2 cases therefore may serve as a useful reference for other resource limited countries, enabling the implementation of conservative management, with judicious monitoring instead of immediate surgical intervention when gastric pneumatosis is seen.
This report is limited as we are unable to establish a cause-effect relationship between the feeding practice chosen and the occurrence of NEC; we are also unable to generalize the use of conservative management when gastric pneumatosis is seen. Bowel ultrasound a promising study that lends to improving the diagnostic accuracy in NEC 10 which is available to us, was a modality that could have been utilized in both cases. The genetic predisposition to NEC continues to garner attention 13 and therefore serves as potential scope for further review of NEC cases. The review of these two cases offers the additional advantage of adding to the scientific data available on a rare radiological occurrence. It still remains unclear that which uniquely predisposes a neonate to gastric pneumatosis associated with NEC and the factor/s that contribute to this being a rare finding.
It is therefore recommended that the policy that governs feeding strategies for the preterm infant be reviewed to include early versus later initiation of enteral feeding, the use of human milk versus formula and the concept of minimal enteral feed 15. The benefits of human milk to include reduced rates of sepsis and necrotizing enterocolitis 15, may prompt units to review the benefits of having human milk bank sites or accessible networks. Lastly, it may be prudent for all clinical and radiological staff to be aware of gastric pneumatosis as a radiological sign, though rare, as once identified as was in our case, it is unlikely to be missed in future cases.
Conclusion
Gastric pneumatosis remains rare but may sporadically occur in clusters within a NICU. Although cases associated with NEC can be managed conservatively, as our cases were, care must be taken to vigilantly monitor, as perforation may be imminent. Gastric pneumatosis is not to be confused with pneumoperitoneum and can be differentiated through the use of a lateral X-ray. Awareness of clinical and radiological findings, as well as potential differential diagnoses is crucial to ensure that appropriate and timely management is offered. Protocols governing the feeding strategies for the preterm infant should be reviewed.
Declarations
Ethical Approval
Not applicable
Funding
No external funding was received
Availability of data and materials
The data presented in this study are available on request from the corresponding author. The data are not publicly available due to privacy restrictions.
Acknowledgements
Acknowledgements The authors would like to acknowledge the nurses, doctors and ancillary staff of the Sir Lester Bird Medical Center for their continued service.
References
- 1.Aydemir G, Cekmez F, Tanju Asya, Emre Canpolat I, Alparslan Gene F et al. (2012) Increased fecal calprotectin in preterm infants with necrotizing enterocolitis. , Clinical laboratory 58(7), 841.
- 2.Bonitatibus G, G L Paterlini, Zanzucchi M, Perotti V, P E Villani. (2022) Gastric Pneumatosis in a Preterm Infant: a Case Report and Review of Literature.
- 4.Chesley P, Stark R, McAdams R, Effmann E, Drugas G. (2014) Isolated gastric pneumatosis in a premature infant following steroid exposure. , Journal of Pediatric Surgery Case Reports 2(9), 436-438.
- 5.Duran R, Vatansever Ü, Aksu B, Acunas B. (2006) Gastric pneumatosis intestinalis: an indicator of intestinal perforation in preterm infants with necrotizing enterocolitis?. Journal of pediatric gastroenterology and nutrition. 43(4), 539-41.
- 6.Fléchelles O, Daudens-Vaysse E, François-Coridon H, Pignol J, Locatelli-Jouans C et al. (2019) . Cluster of Gastric Lactobezoar in One NICU: 7 Cases in 37 Days, Consequence of Aggressive Enteral Nutrition. Ann Clin Case Rep 4, 1767.
- 9.Kasivajjula H, Maheshwari A. (2014) Pathophysiology and current management of necrotizing enterocolitis. , The Indian Journal of Pediatrics 81, 489-497.
- 10.J H Kim, Sampath V, Canvasser J. (2020) Challenges in diagnosing necrotizing enterocolitis. Pediatric research. 88, 16-20.
- 11.Lapillonne A, Claris O, Lachaux A, Miguet D, B L Salle. (1992) Neonatal gastric necrosis. , Archives Francaises de Pediatrie 49(10), 891-893.
- 12.J C Leonidas. (1976) Gastric pneumatosis in infancy. , Archives of Disease in Childhood 51(5), 395-398.
- 13.R M Moonen, M J Huizing, G E, Cavallaro G, Mosca F et al. (2020) Risk of necrotizing enterocolitis associated with the single nucleotide polymorphisms VEGF C-2578A, IL-18 C-607A, and IL-4 receptor α-chain A-1902G: a validation study in a prospective multicenter cohort. Frontiers in pediatrics. 8, 45.
- 14.Nguyen H, C H Lund. (2007) Exploratory laparotomy or peritoneal drain?: management of bowel perforation in the neonatal intensive care unit. , The Journal of Perinatal & Neonatal Nursing 21(1), 50-60.
- 15.R J Schanler, R J Shulman, Lau C. (1999) Feeding strategies for premature infants: beneficial outcomes of feeding fortified human milk versus prete formula. , Pediatrics 103(6), 1150-1157.
- 16.Springer A, C A Reck, Hoermann M, Messerschmidt A, Horcher E et al. (2010) Gastric pneumatosis in necrotizing enterocolitis. Klinische Pädiatrie. 273-273.
