First Lumbar Treatment of Chronic Mixed Low Back Pain with High Dose Capsaicin 8% Patch
Abstract
Background
Capsaicin 8% patch reduces peripheral neuropathic pain. Based on the concept of neuropathic pain (NeP) in mixed low back pain (LBP) it is hypothesized, that an exclusively lumbar capsaicin 8% patch is an effective treatment of mixed LBP. The aim is a proof of this concept and to identify predictors of responsiveness.
Methods
This prospective stratified study included 54 chronic, mixed, LBP patients with spontaneous pain >3/10 on the NRS (0-10) and a painDETECT Questionnaire (PDQ) score >12 meaning possible or likely (>18) NeP. Pain intensity, PDQ, and quantitative sensory testing (QST) were assessed at baseline. After a one-hour capsaicin 8% treatment on the low back, follow-up was carried out regularly over three months. Response was determined at one month (≥30% pain reduction) and predictors were compared accordingly.
Results
The average change in pain intensity at week four was -1.1 (-0.50;-1.71, 95%CI, p < 0.001). Twenty-one (39%) patients responded at one month with a mean pain reduction of -3.1 (-4.0;-2.3, 95%CI) and even 10 of the 21 responders showed a ≥ 50% pain reduction. No pain reduction was seen in 33 (61%) patients (p = 0.42). Responders and non-responders did not differ at any baseline parameter: NRS (p = 0.85), PDQ score (p = 0.47), duration of pain (median of 48 and 36 months) nor QST profiles.
Conclusions
Lumbar capsaicin 8% patch is an effective treatment in about 40% of chronic patients with mixed neuropathic LBP. However, predictors for response could not be identified.
Author Contributions
Academic Editor: Dimos-Dimitrios Mitsikostas, National & Kapodistrian University of Athens, Greece
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2017 Julian Prosenz et al
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
B. Gustorff received an unrestricted research grant from Astellas Austria, and honoraria for lectures and consultancy from Astellas Austria, Astellas Pharma Europe Ltd and Grunenthal, Germany. H. Kloimstein received honoraria for lectures from Astellas Austria. R. Likar received honoraria for lectures from Astellas Austria. J. Prosenz and S.M. Neuwersch have no conflicts of interests to declare.
Citation:
Introduction
Chronic low back pain (LBP) is one of the most common pain syndromes, with a reported lifetime prevalence of up to 84% 1, 2. A recent widely accepted hypothesis addresses underlying pain mechanisms in LBP to allow for individual appropriate treatment 3. This concept is based on the fact that LBP may present as nociceptive, neuropathic, or mixed pain, the latter combining both pain types 4. Up to half of LBP patients exhibit symptoms of neuropathic pain 5. Prevalence estimates the neuropathic component in the range of 4-13% and indicates worse pain and lower quality of life in these patients 3, 6, 7. One of the tools of identifying patients with a neuropathic pain component is the use of the painDETECT Questionnaire (PDQ), validated for neuropathic pain (NeP) in patients with back pain 3.
Despite considerable research effort, the treatment of neuropathic pain remains difficult and efficacy low 8. A more recent therapeutic option for peripheral NeP has emerged in the form of capsaicin 8% patch (QUTENZATM). The patch is applied once, delivering a high dose of locally active capsaicin, followed by a pain relief up to 12 weeks. Capsaicin patch has been tested mostly in post herpetic neuralgia (PHN) and HIV-associated neuropathy (HIV-AN) in randomized-controlled trials (RCTs) and in large non-interventional trials of various types of NeP with consistent, positive results 9, 10, 11. In a large variety of neuropathic patients of our study centers more than 40% responders were found one month after the treatment 12. However, at date, data on lumbar capsaicin for chronic low back pain, especially mixed low back pain, is scarce. Earlier repeated applications of low-dose capsicum patch in non-specific chronic LBP patients reported positive results 13, 14. However, application methods have changed since, and no data is available on the efficacy of a single high-dose application. It is generally accepted that capsaicin is applied to the peripheral neuropathic painful area. This has also been shown recently in painful radiculopathy 15. However, capsaicin treatment leads to a reduction of the area of secondary hyperalgesia 12, which suggests the possibility of a central effect, at least in part. Hence, capsaicin may also act via the dorsal branch of the lumbar nerve innervating the lumbar skin and a mere lumbar application may be sufficient for pain relief.
The aim of this study was to investigate the efficacy of exclusively lumbar topical capsaicin 8% patch in chronic, neuropathic and mixed, low back pain, as well as to establish possible predictors of response including quantitative sensory testing (QST) profiles. Our hypothesis was that capsaicin is an effective treatment in chronic LBP with a neuropathic pain component determined by the mean change of spontaneous pain intensity four weeks after treatment.
Methods
This was a prospective, unblinded, multicenter, single-treatment comparative pilot study conducted with patients’ written consent according to the Declaration of Helsinki. This study was registered with the European Clinical Trials Database (EudraCT) (EudraCT number: 2012-001420-35), approved by the ethics committee (Ethikkommission der Stadt Wien, EK-12-059-0412), and performed according to Good Clinical Practice.
The study was performed at two hospital-based outpatient pain clinics in Austria, at the Centre for Interdisciplinary Pain Therapy, Oncology and Palliative Care, Klagenfurt, Austria and at the department of Anesthesia, Intensive Care and Pain Medicine, Wilhelminenspital, Vienna, Austria.
Patients
Patients were screened at two hospital-based pain centers (Vienna and Klagenfurt). All patients, male or female, of at least 18 years of age, with low back pain of at least three months’ duration, with a numerical pain intensity of at least 4/10 during the 24 hours prior to inclusion, a painDETECT Questionnaire score of 13 or more, without analgesics or on stable (for least 1 week prior to inclusion) pain treatment were included, unless they met any of the exclusion criteria (see Table 1). All patients gave written informed consent prior to inclusion. Demographic information was documented at baseline.
Table 1. Exclusion criteria| Use of topic analgesics within 7 days prior to inclusion Prior use of capsaicin patch |
|---|
| Alcohol or drug abuse |
| Pre-existing psychiatric condition |
| Unstable arterial hypertension |
| Acute skin diseases in relevant areas, allergy to capsaicin or any other relevant component or medication |
| Scheduled intervention or surgery within 3 months |
| Pregnancy |
| Court proceedings, planned or ongoing requests for occupational disability or early retirement related to the low back pain of the subject |
Treatment
Treatment area was defined by determination of painful skin area based on mechanical hyperalgesia evoked by pin prick 256 mN (256 mN, The PinPrick, MRC Systems, Heidelberg, Germany). In cases where no pinprick hyperalgesia was found, allodynia was assessed with a brush (Somedic No. 5) or in case of absence the painful area was determined as indicated by the patient. In all cases, treatment area was restricted to the lower back. No lower extremities were treated.
This area was treated with the capsaicin 8% patch (Capsaicin 179 mg patch (14 cm x 20 cm); Astellas Pharma Europe, Leiderdorp, Netherlands) for one hour as indicated by the label and manufacturer. Before application, the area was treated with a topical anesthetic cream (EMLA 5% cream containing 2.5% lidocaine, 2.5% prilocaine; ASTRAZENECA Ltd., Germany) for 60 minutes according to the valid procedure at the time of study initiation. Afterwards, capsaicin patch was applied by a trained study nurse or medical doctor. After treatment, the area was cleaned with a special cleansing gel provided by the manufacturer and the skin was carefully washed with water and soap.
First line treatment for treatment related discomfort was cooling packs, which could be applied starting 30 minutes or later after start of application. Patients were informed of the availability of intravenous opioids (Piritramide, Dipidolor®; Janssen Cilag, Austria) as rescue medication. If requested, a medical doctor titrated increments of 3 mg intravenously.
Assessment of Pain
Throughout the study, pain was assessed using the Numeric Rating Scale (0-10, with 0 being “no pain” and 10 being “the most intense pain imaginable”). Pain was assessed before, during and after the capsaicin patch application and at the follow-up visits after 1 day, 1 week, 2 weeks, 1 month, 2 months, and 3 months, and via telephone every 2 weeks between the first and third month.
Assessment of Quality of Life, Anxiety and Depression
Quality of life was assessed with the Short Form (36) Health Survey (SF-36) questionnaire at baseline, 4 weeks, 2 months, and 3 months. Additionally, the EuroQol short version EQ-5D-3L questionnaire (EuroQol Research Foundation, Rotterdam, Netherlands) was filled out by patients at baseline and at each follow-up visit.
Anxiety and depression were assessed with the Hospital Anxiety and Depression Scale 16 with the German version (HADS-D) at baseline and follow up visits at 4 weeks, 2 months, and 3 months.
Quantitative Sensory Testing (QST)
The Quantitative Sensory Testing battery was carried out as already described previously (for details see Gustorff and others Scand. J. of Pain 2013 12), in an extended version of the protocol of the German Research Network on Neuropathic Pain (DFNS) 17, according to DFNS specifications 18.
The following quantitative sensory testing parameters were assessed: warmth perception threshold (WPT), heat pain perception threshold (HPPT), heat pain tolerance threshold (HPTT), cold perception threshold (CPT), cold pain perception threshold (CPPT), cold pain tolerance threshold (CPTT), pressure pain perception threshold (PPT), stimulus-response (SR) function to mechanical stimuli, mechanical pain threshold (MPT), area of pinprick hyperalgesia (as described above), and area of dynamic allodynia to brush. We used a Thermal Sensory Analyzer (TSA-2001, Medoc Ltd., Israel), von Frey filaments (OptiHair, Marstock, Germany), hand crafted calibrated pin pricks 8, 16, 32, 64, 128, 256, 512 mN (The PinPrick MRC Systems, Heidelberg, Germany), a brush (Somedic no. 5), cotton wool and a Q-tip. All measurements were performed on the painful area in the lumbar back. Patients were trained according to a standardized protocol before baseline assessments and the entire QST battery was repeated at follow up visits after 1 day, 1 week, 1 month, and 3 months.
Study Design and Data Analysis
This was a prospective trial; patients were treated with a single, one-hour capsaicin 8% patch application. The primary outcome parameter was defined as reduction of spontaneous pain four weeks after treatment. For further analysis, group allocation was undertaken as follows: to discern treatment effects we defined a “clinically meaningful response” as at least 30% pain reduction on the numeric rating scale from baseline, in accordance with general practice 15, 19, 20. This was done at four weeks after treatment, and patients were stratified into two groups – “responders” and “non-responders”. Secondary outcome parameters were defined as number and respective percentage of treatment responders at four weeks, mean change of pain intensity from baseline after one week, eight weeks, and 12 weeks, as well as change in quality of life (SF-36, EuroQuol, and HADS (anxiety and depression)). Baseline characteristics (pain intensity, duration of LBP, painDETECT Questionnaire score, and QST parameters) were explored for possible prediction of treatment response.
As a sub-analysis patients were also stratified according to painDETECT Questionnaire scores (≥19 meaning “likely neuropathic pain” and 13-18 meaning “possible neuropathic pain”) and percentage of responders in each group was assessed.
Statistical analysis was performed using SPSS Version 22 (IBM SPSS Statistics; IBM Corporation, New York, USA) for Windows and GraphPad Prism Version 7 (GraphPad Software, Inc.; La Jolla, CA, USA) for Windows. For missing data concerning the primary outcome parameter (NRS after four weeks), the last-observation-carried-forward (LOCF) method was used. Categorical data are described with absolute and relative frequencies, continuous data with mean and standard deviation for normally distributed data and median, quartiles, minimum, and maximum otherwise.
After testing for normality of data distribution (Shapiro-Wilk normality test), t-test or Mann-Whitney-U-test respectively were used for inter-group comparisons, paired t-test or Wilcoxon signed-rank test for intra-group testing. For multiple within group comparisons over time we used a repeated-measure ANOVA. Fisher’s exact test was used to compare group characteristics at baseline. A P-value of < 0.05 (2-tailed) was considered statistically significant. The main outcome parameter analysis was based on a one-way repeated-measures ANOVA with Dunnett’s multiple comparison test (baseline vs. 4 weeks after treatment)
Results
In total 54 patients were included in the data analysis. All patients suffered from low back pain on average over 3 years (1.5-10.0 years, median IQR) and presented with a mean pain intensity of 6.3 on the numeric rating scale (6 5.86.87.8, medi
an IQR). Patients were treated with an average of one patch. Adverse events occurred in 96% of patients, with erythema (96%) and pain (44%) occurring most frequently, 7% of patients complained of pruritus in the treated area. No serious adverse events occurred. Response groups were comparable concerning age, sex, duration of pain, NRS, or painDETECT score (see Table 2 for baseline characteristics).
Table 2. Baseline characteristics| Characteristic | Responders | Non-responders | |
|---|---|---|---|
| Number of Patients | 21 | 33 | |
| Age yr – mean (SD) | 59.2 (±13.0) | 59.7 (±12.8) | p=0.89 |
| Malen (%) | 9 (42.9%) | 20 (60.6%) | p=0.27 |
| Duration of Pain – months median (IQR) | 48 (18-120) | 36 (17-119) | p=0.72 |
| Baseline NRS – mean (SD) | 6.2 (±1.7) | 6.3 (±1.3) | p=0.85 |
| painDETECT Score – median (IQR) | 18 (13-21.5) | 20 (15.5-22.5) | p=0.47 |
Response to Treatment
Pain ratings were statistically significantly different after treatment (baseline, one week, two weeks, four weeks, eight weeks, 12 weeks) (F (5, 53) = 7.203, p < 0.001). The overall average change in pain intensity at week four was -1.1 (-0.50;-1.71, 95%CI, p < 0.001) (see Figure 1). Twenty-one patients (38.8%) showed a ≥ 30% pain reduction on the numeric rating scale four weeks after treatment (p < 0.0001). The mean difference in pain intensity in this responder group was -3.1 (-4.0;-2.3, 95%CI) points on the NRS (6.2 at BL vs. 3.1 at four weeks), hence pain was cut by 50% on average. Of these 21 responders, ten patients (47.6%; 18.5% of all patients) achieved even a pain reduction ≥ 50% compared to their baseline value. The change in mean values in this subgroup was -4.1 (6.1 at BL vs. 2 at four weeks).
The mean pain intensity at week one in the responder group (number of patients; change vs. baseline) was NRS 4.4 (n=21; -35.1%), at week four NRS 3.1 (n=21; -50.4%), week eight NRS 3.9 (n=20; -37.5%), and week 12 NRS 4.3 (n=19; -31.7%) (see Figure 1).
In the non-responder group (n=33, 61.2%) the mean change was an increase of +0.2 on the NRS (6.3 at BL vs. 6.5 at 4 weeks, p=0.42) (see Figure 1). Pain ratings four weeks after treatment differed significantly between groups (mean of 3.1 vs. 6.5 on the NRS, p < 0.0001).
The LOCF method was used in 14 patients (three patients of the responder group and eleven patients of the non-responder group) who did not attend the follow-up visit at four weeks.
Figure 1.Pain (NRS) from baseline up to 12 weeks after treatment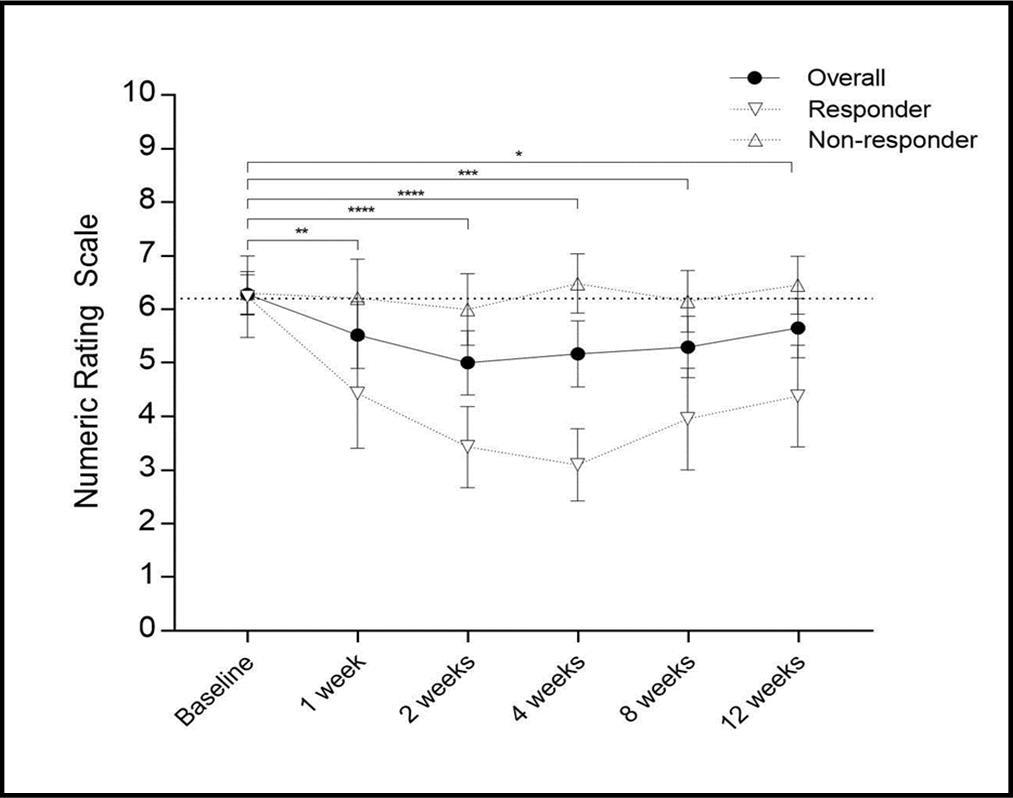
Quality of Life, Anxiety, Depression, and painDETECT
Quality of life, as assessed with the SF-36 questionnaire, was equally impaired in both groups at baseline. Both physical and mental health sum scores did not differ significantly (see Figure 2).
Figure 2.Results from the EuroQuol, SF-36, and HADS assessments at baseline and 4 weeks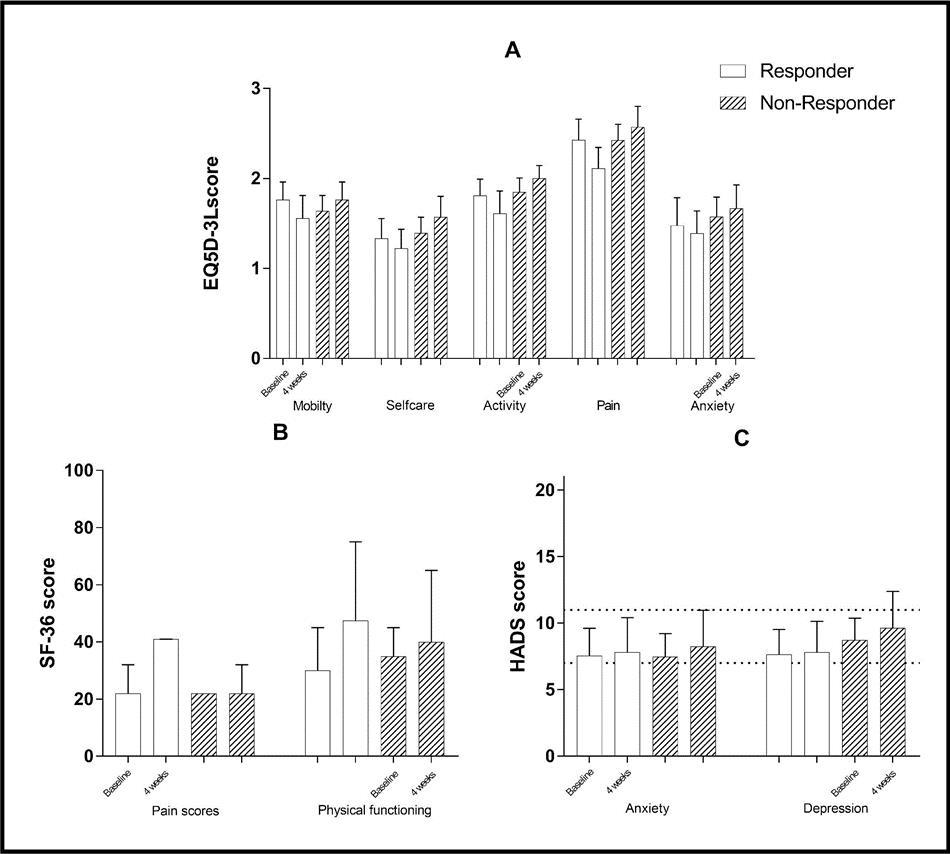
The subcategories “pain” and “physical functioning” were analyzed separately, with no significant differences in either subcategory at baseline (“pain”, 22 vs. 22, p = 0.48; “physical functioning”, 30 vs. 35, p = 0.59). The sum score for “pain” at four weeks was significantly higher in the responder group than in the non-responder group (41 vs. 22, p < 0.01). Comparison of the baseline scores with the scores at four weeks within the respective groups showed a significant improvement at four weeks in the responder group, with an increase by 19 points (22 vs. 41, p = 0.0017), whilst there was no change evident in the non-responder group (22 vs. 22, p = 0.19). Concerning “physical functioning”, at four weeks an improvement with a significant increase of 17.5 points was observed (p < 0.001) in the responder group. Meanwhile a 5 point (16%) non-significant increase had occurred in non-responders (p = 0.26).
The EQ5D-3L included the parameters “mobility”, “self-care”, “activity”, “pain”, “anxiety”, and “general health VAS”. Again, there were no differences between groups at baseline in any of the categories. At four weeks, patients in the responder group had significantly better values in the categories “self-care” (p <0.05), “activity” (p <0.01), and “pain” (p < 0.01). At four weeks, patients’ ratings concerning their general health (“general health VAS”) were significantly better among responders than non-responders (median 60 vs. 40, p < 0.05).
Analysis of the “Hospital Anxiety and Depression Scale” scores revealed no significant difference between the groups at baseline (anxiety scores 7.6 vs. 7.5, depression scores 7.7 vs. 8.7). Interestingly scores in the responder group had not changed at four weeks. Results from the non-responder group showed a slight increase of anxiety (0.8 points, n.s.) and depression (0.9 points, p = 0.027) levels.
PainDETECT Questionnaire scores were evenly distributed amongst the two groups (see Table 3). The median baseline score was 18 in the responder group and 20 in the non-responder group, with no significant difference between both groups (p = 0.47). After four weeks painDETECT scores in both groups had decreased significantly towards nociceptive pain values, with a median of 12 in the responder group and a median of 17.5 in the non-responder group (see Figure 3). At baseline 29 patients presented with “likely neuropathic pain” (score >18) and 25 patients with “possible neuropathic pain” (score 13-18). When keeping this group assignment and looking at pain scores at four weeks, 10 patients (34.5% of the group) in the “neuropathic pain” group showed a response to treatment, versus 11 patients (44% of the group) in the other group (p = 0.58). Also, mean pain scores (NRS) did not differ between the two painDETECT stratified groups at four weeks (p = 0.36).
Figure 3.painDETECT Questionnaire scores at baseline and after four weeks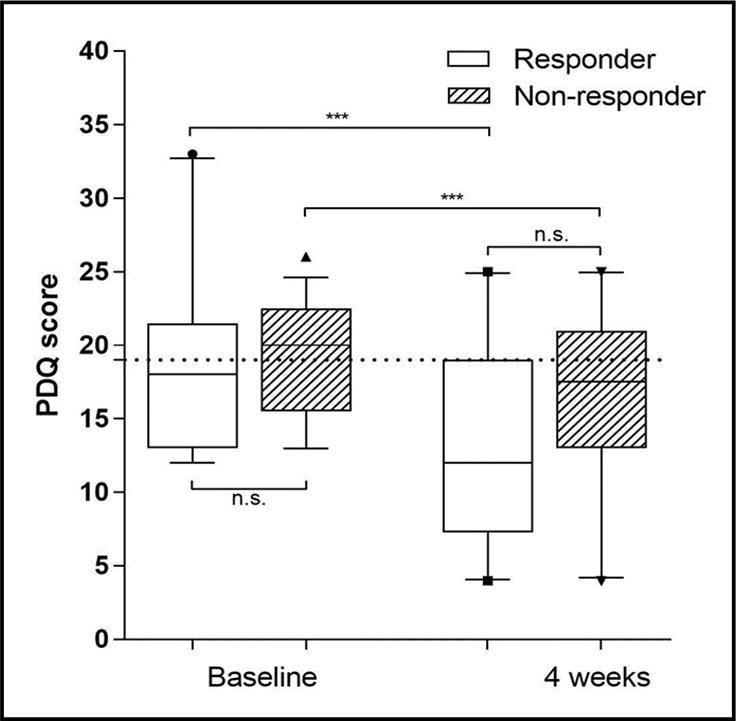
| Parameter | Responders n (%) | Non-responders n (%) | ||
|---|---|---|---|---|
| PDQ Score | ≥19 | 10 (19) | 19 (35) | p = 0.58 |
| 13-18 | 11 (20) | 14 (26) | ||
| NRS Score | ≥7 | 8 (15) | 15 (28) | p = 0.78 |
| ≤6 | 13 (24) | 18 (33) |
Taken together, PainDETECT Questionnaire scores could not predict responsiveness to treatment.
QST None of the quantitative sensory testing parameters differed between groups at baseline or four weeks after treatment (see Table 4).
Table 4. Quantitative sensory testing parameters at baseline and 4 weeks (mean, CI 95%)| Group | PPT kPa | MPT mN | WPT °C | HPPT °C | HPTT °C | CPT °C | CPPT °C | CPTT °C | ||
| Baseline | Responders | 361.5 | 129 | 35.2* | 40.1 | 44.8 | 27.9 | 21.2 | 6.8 | no significant differences |
| (252.5-470.5)) | (62-196) | (34.8-35.7) | (35.5-41.6) | (41.8-47.7) | (24.9-31.0) | (17.4-25.1) | (2.5-11.1) | |||
| Non-responders | 374.8 | 93 | 35.8 | 41.6 | 46.2 | 28 | 19 | 6.6 | ||
| (279.8-469.8) | (67-120) | (35.0-36.6) | (39.8-43.3) | (44.7-47.6) | (26.0-30.1) | (15.4-22.5) | (3.3-9.8) | |||
| 4 weeks | Responders | 350.2 | 94 | 36.0* | 40.4 | 46.1 | 29.2 | 20.9 | 6.2 | no significant differences |
| (263.0-437.4) | (39-148) | (35.2-36.9) | (38.8-42.0) | (44.6-47.6) | (28.4-30.0) | (17.1-24.8) | (1.5-11.0) | |||
| Non-responders | 366.6 | 72 | 35.9 | 41.2 | 46.6 | 28.6 | 18.5 | 5.4 | ||
| (261.9-471.4) | (55-90) | (35.0-36.8) | (39.2-43.3) | (45.1-48.0) | (27.7-29.4) | (14.0-23.0) | (2.1-8.8) |
Intra-group testing of averaged SR-function data (baseline versus four weeks) revealed a significant difference in the analysis of the responder group (p < 0.05) and the non-responder group (p < 0.0001), meaning responders and especially non-responders became more sensitive to painful stimuli (i.e. a trend towards mechanical hyperalgesia). In absolute values, the mean difference on the 101-point-Likert-scale between baseline and 4 weeks in the responder group was an increase of 2.6 (14.11 vs. 16.69), and in the non-responder group and increase of 6.5 (16.2 vs. 22.7). Except a trend towards an increase of the warm perception threshold (baseline versus four weeks, p = 0.073) no other sensory changes developed within the responder group.
Discussion
Response Rate, Pain Reduction and Improvement of QoL
This pilot study demonstrated for the first time that a single treatment with a capsaicin 8% cutaneous patch in the low back leads to a significant and ongoing pain reduction and improvement of QoL parameters in about 39% of chronic low back pain patients with a neuropathic component at one month after treatment. Even more so, almost half of the responders had a pain reduction greater than 50%, which is considered a good response 15. These response rates are in agreement with results of treatment of neuropathic pain. A meta-analysis of seven RCTs (PHN and HIV-AN) demonstrated a 46% (PHN) and 41% (HIV-AN) proportion of ≥30% response 11. Similar results are reported by a Cochrane systematic review 21 and recent data from a non-interventional study in 1000 patients 10.
In the past, Capsaicin has been applied rarely to irradiating neuropathic back and neck pain with comparable results to mere neuropathic pain, and further investigation had been suggested 15. Now, our results demonstrate for the first time that the topical dorsal approach is enough to reduce both lumbar and irradiating mixed low back pain and may therefore act via a common – more central than expected - pathway. This is in agreement with results from a QST-based study in pseudoradicular low-back pain and radicular pain patients, where data indicated similarities between the diseases, questioning the distinction as completely separate disease entities. 22
Other low-dose capsaicin application methods (patch for several hours a day for three weeks, and cream three to four times daily for at least three weeks) were used in musculoskeletal pain. In chronic low back pain capsicum showed a response rate of 67%, as measured by a ≥ 30% reduction on a composite pain score derived from the Arhus low back rating scale 13. However, presence or absence of a neuropathic pain component was not reported and capsaicin was applied differently to our study patch. A review 23 on topical capsaicin for the treatment in chronic musculoskeletal pain conditions reported a pooled treatment response rate (at least 50% reduction vs. baseline) of 38%. Mode of application has since then changed to the single high-dose application. Thus, a comparison is virtually impossible.
We assessed quality of life and psychological parameters with several tools, comprising the SF-36 questionnaire, EuroQol EQ5D-3L and HADS score. As with pain response all results emphasize the improvement at four weeks. Pain and physical functioning sub-scores of the SF-36 questionnaire as well as health self-rating (“general health VAS”) were significantly better in the responder group. These results are well in line with other trials 10, 13 and underline the importance of this essential long-term outcome in patients who are not pain-free 24, 25, 26.
Predictors of Response
The distinction of neuropathic from ambiguous or no neuropathic pain was not predictive of an improved response rate. It seems, capsaicin efficacy does not presuppose the presence of clear neuropathic pain at all or, in contrast to the accepted hypothesis, is not a specific marker of a single mechanism. To our knowledge, this is the first study reporting results on this particular issue. However, since phenotyping only was used for pain classification (i.e. PDQ and QST) further explanation for this is open.
A recent meta-analysis by N. P. Katz et al. 27 studying possible predictors of treatment response in patients with PHN and HIV-AN found mild pain intensity at baseline to be the most consistent predictor. Edwards et al. 28 found a correlation between higher baseline pain score and increased treatment response to pharmacotherapy. This stands in contrast to our findings. Baseline pain scores in the responder and non-responder group did not differ, nor did rates of treatment response. However, again, results are difficult to compare with different conditions (PHN and chronic mixed low back pain) and different treatments (topical capsaicin and oral pharmacotherapy) being studied.
Based on the sensitized nociceptor hypothesis 29, 30, 31 we expected to identify predictors of response to the TRPV1 receptor agonist capsaicin.
QST discerned different etiologies in neck pain syndromes, as well as altered central pain processing in patients with low back pain 32, 33. None of our quantitative sensory testing demonstrated any difference of responders at baseline. This is once again in agreement with an earlier study on neuropathic pain patients treated with capsaicin 8% patch conducted by our group, where no QST predictors could be identified 12. In contrast to results of a recent meta-analysis 27, reporting predictive value of absence of allodynia, we could not demonstrate any difference between the groups concerning allodynia. Nevertheless, the absence of allodynia in low back pain is a normal feature.
Another explanation may be, that the so-called peripheral defunctionalization following capsaicin application 34 is not the mode of action in our patients, since , as in our previous study 12, no loss of sensory function was demonstrated in our study.
Limitations of the Study
This is a pilot study and no control treatment was used for this proof of concept study. Further RCTs are needed for confirmation.
Of note, with the inclusion of patients with “possible neuropathic pain” according to the painDETECT Questionnaire our sample was quite heterogeneous. Yet, according to our results, a clear neuropathic component may not necessary for the efficacy of capsaicin 8% patch treatment in chronic low back pain. This raises the question whether “neuropathic pain” in itself is a prerequisite for treatment or relevant for the outcome respectively. Further prospective studies are necessary to elucidate this question.
The median duration of pain was more than three years in our trial and might explain the moderate overall pain relief, since chronic back pain is difficult to treat and only short pain duration of 6 months increased response in neuropathic pain 10.
The authors admit that the dropout rate, especially amongst the non-responders, may limit group comparison for later time points. Application of the LOCF method for the main outcome parameter is an accepted approach in these cases, and bias towards overestimation of treatment efficacy is less likely, since data of dropped out non-responders were carried forward in the analysis. Hence, we concluded that the LOCF method was warranted, risking underestimation of a beneficial effect, but becoming more robust against errone
ously reporting great efficacy. Nevertheless, it cannot be ruled out that resulting selection bias influenced assessment notably of QST parameters and predictors.
In conclusion, we demonstrate for the first time, that capsaicin patch applied to the lower back in patients suffering from chronic mixed low-back pain may result in significant pain relief for several weeks. Results from this trial justify further investigating the effects of capsaicin patch in a larger, controlled trial.
Acknowledgements
The authors thank N. Hacker und S. Demschar for help with recruitment and information of patients and collection of data.
Authors’ contributions: BG, RL, SMN and HK contributed to conception, design and data collection. JP contributed to analysis and interpretation of data and drafting/revision of the article. All authors approved the final manuscript. BG vouches for the integrity of the study.
References
- 1.Baron R, Binder A. (2004) [How neuropathic is sciatica? The mixed pain concept]. Orthopade. 33(5), 568-75.
- 2.B F Walker. (2000) The prevalence of low back pain: a systematic review of the literature from1966to1998. J Spinal Disord. 13(3), 205-17.
- 3.Freynhagen R. (2006) painDETECT: a new screening questionnaire to identify neuropathic components in patients with back pain. Curr Med Res Opin. 22(10), 1911-20.
- 4.Morlion B. (2011) The relevance of neuropathic components in chronic back pain. Curr Med Res Opin. 27(10), 2067-8.
- 5.Morlion B. (2011) Pharmacotherapy of low back pain: targeting nociceptive and neuropathic pain components. Curr Med Res Opin. 27(1), 11-33.
- 6.C O Schmidt. (2009) Modelling the prevalence and cost of back pain with neuropathic components in the general population. Eur.J Pain. 13(10), 1030-5.
- 7.A M McDermott. (2006) The burden of neuropathic pain: results from a cross-sectional survey. Eur.J Pain. 10(2), 127-35.
- 8.N B Finnerup. (2015) Pharmacotherapy for neuropathic pain in adults: a systematic review and meta-analysis. Lancet Neurol. 14(2), 162-73.
- 9.Backonja M. (2008) NGX-4010, a high concentration capsaicin patch, for the treatment of postherpetic neuralgia: a randomised, double-blind study. Lancet Neurol. 7(12), 1106-12.
- 10.Maihofner C, M L Heskamp. (2013) Prospective, non-interventional study on the tolerability and analgesic effectiveness over 12 weeks after a single application of capsaicin 8% cutaneous patch in 1044 patients with peripheral neuropathic pain: first results of the QUEPP study. Curr Med Res Opin. 29(6), 673-83.
- 11.Mou J. (2013) Efficacy of Qutenza(R) (capsaicin) 8% patch for neuropathic pain: a meta-analysis of the Qutenza Clinical Trials Database. Pain. 154(9), 1632-9.
- 12.Gustorff B.Treatment of neuropathic pain with the capsaicin 8% patch: Quantitative sensory testing (QST) in a prospective observational study identifies potential predictors of response to capsaicin 8% patch treatment. , Scand J Pain 4(3), 138-145.
- 13.Frerick H. (2003) Topical treatment of chronic low back pain with a capsicum plaster. Pain.106(1-2):. 59-64.
- 14.Keitel W. (2001) Capsicum pain plaster in chronic non-specific low back pain. Arzneimittelforschung. 51(11), 896-903.
- 15.R D Treede. (2013) Mechanism- and experience-based strategies to optimize treatment response to the capsaicin 8% cutaneous patch in patients with localized neuropathic pain. Curr Med Res Opin. 29(5), 527-38.
- 16.A S Zigmond, R P Snaith. (1983) The hospital anxiety and depression scale. Acta Psychiatr Scand. 67(6), 361-70.
- 17.Rolke R. (2006) Quantitative sensory testing in the German Research Network on Neuropathic Pain (DFNS): standardized protocol and reference values. , Pain 123(3), 231-43.
- 18.Rolke R. (2006) Quantitative sensory testing: a comprehensive protocol for clinical trials. Eur.J Pain. 10(1), 77-88.
- 19.J T Farrar. (2000) Defining the clinically important difference in pain outcome measures. Pain. 88(3), 287-94.
- 20.R H Dworkin. (2009) Interpreting the clinical importance of group differences in chronic pain clinical trials: IMMPACT recommendations. Pain. 146(3), 238-44.
- 21.Derry S. (2013) Topical capsaicin (high concentration) for chronic neuropathic pain in adults. Cochrane Database Syst Rev. 2, 007393.
- 22.Freynhagen R. (2008) Pseudoradicular and radicular low-back pain--a disease continuum rather than different entities? Answers from quantitative sensory testing.Pain.135(1-2):. 65-74.
- 23.Mason L. (2004) Systematic review of topical capsaicin for the treatment of chronic pain. BMJ. 328(7446), 991.
- 24.D C Turk, H D Wilson, Cahana A. (2011) Treatment of chronic non-cancer pain. Lancet. 377(9784), 2226-35.
- 25.Skevington S M. (1998) Investigating the relationship between pain and discomfort and quality of life, using the WHOQOL. , Pain 76(3), 395-406.
- 26.E J Dansie, D C Turk. (2013) Assessment of patients with chronic pain. , Br J Anaesth 111(1), 19-25.
- 27.N P Katz. (2015) Predictors of Response in Patients With Postherpetic Neuralgia and HIV-Associated Neuropathy Treated With the 8% Capsaicin Patch (Qutenza). Clin.J Pain. 31(10), 859-66.
- 28.Edwards R R. (2006) Basal heat pain thresholds predict opioid analgesia in patients with postherpetic neuralgia. Anesthesiology. 104(6), 1243-8.
- 29.Baron R. (2000) Peripheral neuropathic pain: from mechanisms to symptoms. Clin.J Pain. 16(2 Suppl): 12-20.
- 30.H L Fields, Rowbotham M, Baron R. (1998) Postherpetic neuralgia: irritable nociceptors and deafferentation. Neurobiol Dis. 5(4), 209-27.
- 31.Serra J. (2011) Double and triple spikes in C-nociceptors in neuropathic pain states: an additional peripheral mechanism of hyperalgesia. Pain. 152(2), 343-53.
- 32.Lewis C, Souvlis T, Sterling M. (2010) Sensory characteristics of tender points in the lower back. Man Ther. 15(5), 451-6.
Cited by (3)
This article has been cited by 3 scholarly works according to:
Citing Articles:
Health Research Policy and Systems (2023) OpenAlex
Health Research Policy and Systems (2023) Crossref
Sarika Parambath, Nathalia Costa, Carmen Huckel Schneider, Fiona M. Blyth - Health Research Policy and Systems (2023) Semantic Scholar
Research Square (Research Square) (2022) OpenAlex
