Abstract
Gene therapy has entered a new era with the dawn of CRISPR/Cas9 technology which though were always available in nature but rediscovered to tame into a real-tlife genome editing tool. With the modernization upsurge and changes in ways the “homo sapiens” survived on this planet from hunger to current era of exuberance has led to multiple metabolic issues like type-2 diabetes. Notwithstanding the rapid emergence of medication to suppress the hyperglycemia and insulin resistance associated with this menace, need has definitely emerge to find more personalized and curative dimensions to therapeutics of type-2 diabetes mellitus. Gene therapy is one more addition to Type-2 Diabetes Mellitus (T2DM) therapy, where multiple options have emerged in the shape of microRNA, direct knocking out of cellular structures like proteins and enzymes and very recently the precision nucleases associated with CRISPR technologies. This mini-review attempt to summarize some of the recent examples of gene therapy with major focus on CRISPR/Cas technologies.
Author Contributions
Academic Editor: Qianqian Song, Wake Forest School of Medicine, Wake Forest Baptist Comprehensive Cancer Center, Medical Center Boulevard, Winston-Salem, NC 27157, United States.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2019 Sikandar Hayat Khan
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Background
The world struggles with the current onslaught of metabolic diseases with type-2 diabetes mellitus (T2DM) leading the race with more 451 million people currently living with the disease. 1 Developed and developing countries are and will probably be facing one of the worst hits with this disease of “sweetness” with one in six predicted to suffer from T2DM. 2 While we had evolved the conventional medicine from metformin, sulfonylurea, alpha-glucosidase inhibitors, and (Peroxisome Proliferator Activated Receptor) PPAR-agonist drugs to some new market entrants like (Glucagon Like Peptide) GLP-1agonists, (Dipeptidyl Peptidase- IV) DPP-IV inhibitors and (Sodium Glucose Like Transporter-2) SGLT-2 inhibitors with better hope for T2DM management, still literature search identifies minimal reduction in (Glycated Hemoglobin) HbA1c levels i.e., <1-2% as targetable after treatment with modest cardiovascular disease risk reduction. 3, 4 Furthermore, genetic studies have identified no glucose related factors as important contributors to diabetic complications like nephropathy. 5 Similar to commoner’s knowledge about glucose markers, Sunjaya et al have also demonstrated lipids and renal factors as real culprits in defining a downhill course for patients in the development of both micro and macro vascular complications. 4 Another study by Ishibashi et al have demonstrated a differential effect on diabetic complications type like retinopathy and nephropathy again questioning the current day diabetic management pharmacotherapy. 6 The discussion above highlights that modest and differential benefits is achievable by available anti-diabetic drugs with variable complications. Emerging evidence in pursuit to provide more personalized approaches with optimal management endeavors to provide short-term hyperglycemic crises to long-term micro and macro vascular complications. Therefore it is the need of time to have newer discoveries which could lead us towards better management of diabetes. 7 Linked to our desire to reach perfection in diabetes management and possibly ultimate eradication of this epidemically surging type-2 diabetes in world community with major burnt being shared by developing economies, the scene is set for innovation. Molecular medicine including gene therapy and genome editing provides innovative and creative ideologies with now some evidence in hand to allow us to visualize an improvement in diabetes biostatistics and associated morbidity and mortality. Cluster Regularly Interspaced Short Palindromic Repeat (CRISPR)/ CRISPR associated protein (Cas) technologies is one of the new incomers in genome editing technologies which after some research have generated some light in the darkening world of diabetic patients. This short communication intends to discuss the promise, shortcoming and possible future usage of CRISPR/Cas technologies in management of diabetes.
CRISPR/Cas Technology
In pursuit of pre-existing technologies using nucleases like Transcription activator like effector nucleases (TALEN), Zinc Finger Nucleases (ZFNs) a need was realized for more powerful, efficient and clinically acceptable tools for genome editing where CRISPR-Cas technology appear as hope and provided multiple avenues for clinical application. 8 The CRISPR/Cas system based upon a primitive acquired immune system in archaea and bacteria depends upon viral and non-viral taxying of Cas nucleases and short guide RNA (sgRNA) are entered into cell and later nuclei to identify the DNA target by sgRNA. Further to this, the Cas9 scissor the non-desirable structure and the blank is filled by desirable codon sequence which is allowed to heal non-homologous or homologous repair system. 9 The mechanistic details are presented in Figure 1.
Figure 1. Overview of CRISPR/Cas9 function from cellular entry to recognition by sgRNA and nuclease function.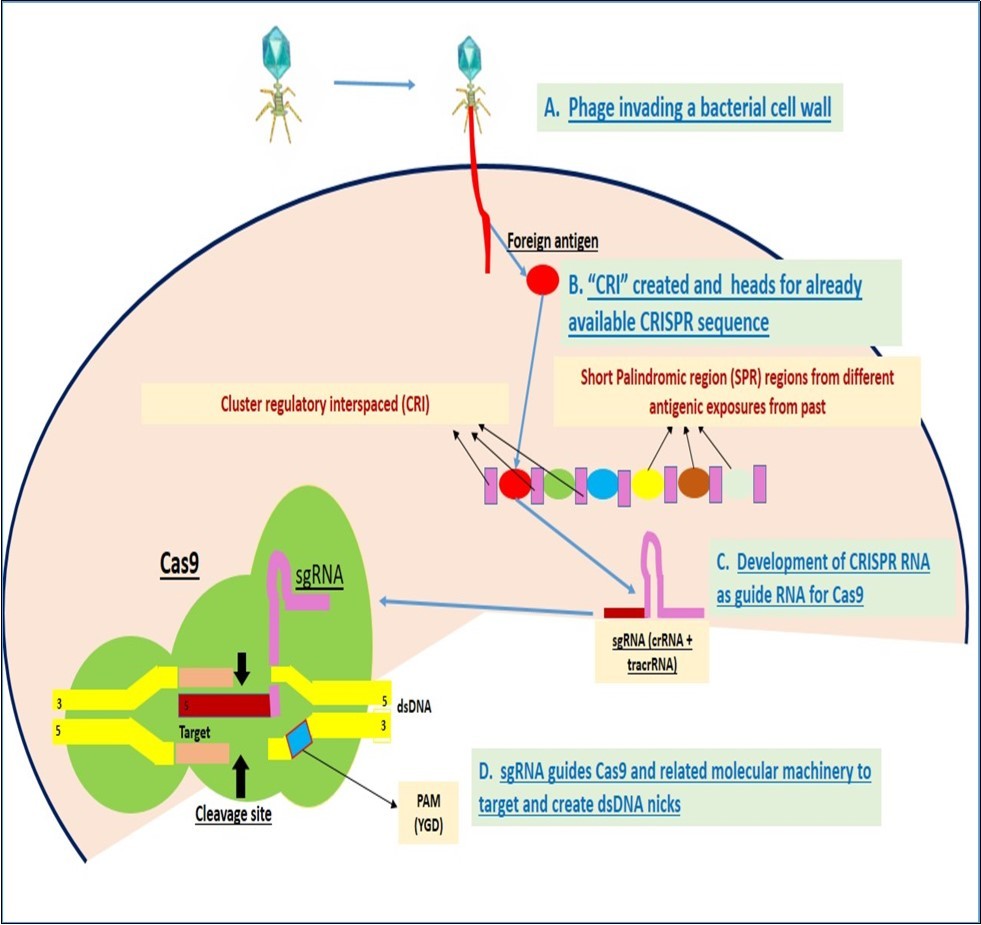
Over the last five years the technology has undergone major modifications from cellular entry modes, nucleases function, optimizing the role of sgRNA and repair mechanisms for new insert all leading to reducing in various off-target effects, making the biotechnology more efficient and cytotoxicity. However, the basic mechanism has not changed.
Literature Review on T2DM Gene Therapy
Literature data search through PubMed and Cochrane library provided only 18 articles dealing directly or indirectly with use of CRISPR Cas technology in management of type-2 diabetes mellitus indicating that a detailed systematic review on the subject is not yet possible. However, we did found some preliminary work related to CRISPR Cas techniques experimentation in animal subjects with type-2 diabetes mellitus. Chao et al (2019) in a proof of concept animal study demonstrated that Non- Liposome Cas9-SgRNA (NL@Cas-RNP) CRISPR payload carrier for inhibiting DPP-IV to boost incretin effect in comparison to DPP-IV inhibitors found the former to be superior in reducing hyperglycemia, providing higher GLP-I levels and reduce insulin resistance with minimal off target effects on hepatocytes and kidneys. 10 Furthermore, the CRISPR Cas treatment provided therapeutic effect lasted 4 weeks than the usual daily dosage need of sitaglibin. Roh et al (2018) used CRISPR-Cas 9 technology to create knock out (KO) mice models to generate loss of function mutations in both leptin gene (ob/ob) for obesity and leptin receptor gene (db/db) for diabetes. The resulting CRISPR modified mice developed characteristic diabetes abnormalities like insulin resistance, hyperglycemia, obesity and hepatic steatosis /NAFLD highlighting that these receptors can undergo reverse genetics through CRISPR modifications to allow researchers to cure genetic abnormalities in type-2 diabetes mellitus. 11 Stafeey et al (2019) have evaluated GLP-1, Leptin and adiponectin genes as targets for gene therapy through viral odes of cell entry. 12 Improta et al (2018) and Mittal et al (2019) have identified exercise induce miRNA cluster changes to help identify potential gene therapy targets for metabolic states like type-2 diabetes and hypertension. 13, 14
CRISPR’s Promise for T2DM in Future
The “aye and nay” of gene therapy utilizing CRISPR/Cas technology are questioned by every day more often than ever. We just can’t spiritualize the CRIPR T2DM promise home till we do not elucidate the wholesome interactive association between nano technology’s offshoots and spinoffs. The CRISPR/Cas technology, though climbing the staircase to glory and optimization in pursuit of personalized and curative medicine for T2DM in specific, still the downsides of the techniques including off-target mutagenesis (OTMs), the need for improvising delivery payload into cellular structures and overall effectiveness are key impediments to clinical application. 8 Apart from this the biotechnology has to show supremacy over conventional but new anti-diabetic drugs in terms of efficacy, being more patient specific, cost-effective and most importantly curative or providing long-term cure. The technology needs to be pre-emptive, predictive, and being capable to segregate the heterogeneity of the type-2 diabetes mellitus in order to replace the in vogue clinical pharmacotherapy. 15, 16.
Conclusion
CRISPR/Cas technologies present a real promise for the life-long pathology of type-2 diabetes mellitus; however, research only seems exuberant by scarcity of data on the subject and needs major efforts for long-term animal trials on the subject. It is anticipated that CRIPSR/Cas techniques have the futuristic potential to provide long, most beneficial, cost-effective and side-effect free personalized therapeutics for this incoming plague haunting mankind.