Abstract
Background
The origin of osteoarthritis, the most common disabling disorder of older adults remains uncertain.
Aim
This mini review examines the possible role played by various neuromuscular deficits in the osteoarthritis etiopathogenic process.
Methods and Procedures
Articles that haveaddressed the current topic of interest and were located in the PUBMED,Google Scholar, and Web of Science electronic data bases as of 2017-2022 were carefully sought and examined.
Results
Various neuromuscular mechanisms may have a bearing on the development and progression of osteoarthritis, but the data are largely unrelated and observational in nature.
Conclusion
More research to examine this issue is sorely needed and could have considerable merit in light of the growing populations of older adults at risk for disabling osteoarthritis in the face of few intervention options to help them to achieve an optimal functional level.
Author Contributions
Copyright © 2022 Ray Marks.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction and background to the problem
Osteoarthritis, involving the destruction of one or more freely moving body joints such as the knee and hip is the among the oldest and most common disabling disorders affecting older adults, as well as many animal species, avians, non weight bearing mammals and marine reptiles 1, 2, 3. A chronic inflammatory disease frequently associated with some form of biomechanical derangement, wherein the articular cartilage lining of the joint is progressively destroyed, the disease is one where little progress has been made to date to mitigate, prevent, or reverse this condition successfully, and safely, despite decades of research 4, 5, 6, 7.
In this regard, although opinions differ, attention to the possible interaction of neuromuscular mechanisms and osteoarthritis, and a host of biochemical, molecular, functional, epigenetic, inflammatory, and structural changes that are commonly progressive and irreversible and that was documented almost 40 years ago, still remains open for examination. A potentially promising line of inquiry, wherein a variety of abnormal neuromuscular mechanisms may fail to protect one or more joints from damage, but may be challenging to detect, this mini review elected to examine some of the key current findings and their implications as published over the last five years, with a focus on the current year 2022. Since quality of life may be severely jeopardized by chronic osteoarthritis of one or more joints, and pain accompanying this disease is often intractable and causes unwarranted opioid related deaths, it was believed line of inquiry would prove highly fruitful to examine.
Indeed, this idea of a muscle-osteoarthritis linkage is not novel, and contrary to the ‘wear and tear’ theory so long propounded to explain osteoarthritis pathology, it has been proposed for some time. Thus, today one can find more than 5000 articles dedicated to this topic posted on PUBMED alone. These data have shown that it is quite feasible to believe that various manifestations of neuromuscular function if subnormal are sufficient possibly salient explanatory factors in this regard, and are hence of great interest to unravel as they may hold unique insights, plus multiple noteworthy clinical implications, for example fostering drug delivery efficiency and targeting, and cartilage chondrocyte genomics, while heightening protection against excessive mechanically derived inflammation 6, 8.
Aims
In light of the importance of achieving optimal health for all, and enhancing wellness opportunities for older populations, this mini review sought to establish if there is solid evidence to support further research to examine neuromuscular deficits as these may impact osteoarthritis risk among the elderly, and if so, whether this information can help to mitigate some of the excess health burden that is being evidenced among the elderly in all parts of the globe, regardless of locality.
A parallel aim was to establish whether any attributes in particular if strengthened or intervened upon through the application of one or more related therapeutic approaches would be more efficacious than not, regardless of the older adult’s physical health and socioeconomic status. Many current premature deaths due to opioid use by osteoarthritis sufferers might also be impacted favorably.
Rationale and Relevance
By 2030, the numbers of older adults in the United States and elsewhere will soar. However, not all will age successfully, even if they have no distinctive co-morbid health condition. On the other hand, this rapidly increasing number of older adults will probably have high rates of chronic diseases, such as osteoarthritis, which may be barriers to the achievement of a fully functional life, even if this is not evidenced by all sufferers or all older adults. However, without some more insightful approaches that can be applied universally or individually, at low cost, and with no side effects, the principle and vision of ‘successful aging’ for all is likely to fail. In this regard, muscle mechanical and metabolic factors that commonly interact with the static and/or dynamic features of a joint and that may be undermined in some way, can possibly be important to examine and mitigate or remediated in the context of osteoarthritis onset and progression.
Materials and Methods
To clarify and examine the extent of support for the aforementioned premises, we attempted to locate salient data currently housed on PUBMED, GOOGLE SCHOLAR, and the WEB of SCIENCE repositories using the key terms, Osteoarthritis and Muscle/Neuromuscular Deficits. All modes of publication identified between the years 2017-2022 were deemed acceptable in this regard if they were published in English as full length reports and addressed the topic of interest from a pathogenic perspective rather than a reactive outcome perspective and regardless of methodological approaches. The most salient articles related to the current theme were duly downloaded and examined and those deemed somewhat new and noteworthy are highlighted here in narrative form. All modes of muscle dysfunction were accepted as valid, as were all definitions of osteoarthritis and their various manifestations. The article builds on previous works, thus was designed to serve as a general one to introduce the topic, as well as an update, rather than any systematic review. The goal was to present a descriptive snapshot of the prevailing trends in this realm and the potential of this construct for advancing osteoarthritis clinical understandings and preventive approaches. The article excluded post surgical observations and studies, as well as all intervention studies that have been reviewed elsewhere and studies in animal models of osteoarthritis.
The term neuromuscular deficit is used throughout to encompass or represent one or more muscle impairments including, but not limited to muscle imbalances, muscle activation deficits, poor muscle coordination, deficient muscle responsiveness, low muscle mass, muscle atrophy, muscle weakness and poor endurance, muscle spasm and inflammation, plus muscle fat cells and sarcopenia, as these may independently or interactively influence bone modeling, joint alignment and stability, especially impact loading. See Figure 1 below. 9, 10, 11, 12, 13, 14, 15, 16, 17
Figure 1. Schematic representation of some anticipated muscular abnormalities and their impacts and outcomes in the presence of muscle weakness and other forms of muscle dysfunction coupled with the absence of efforts to identify and mitigate these attributes specifically and in a targeted manner Adapted from references 9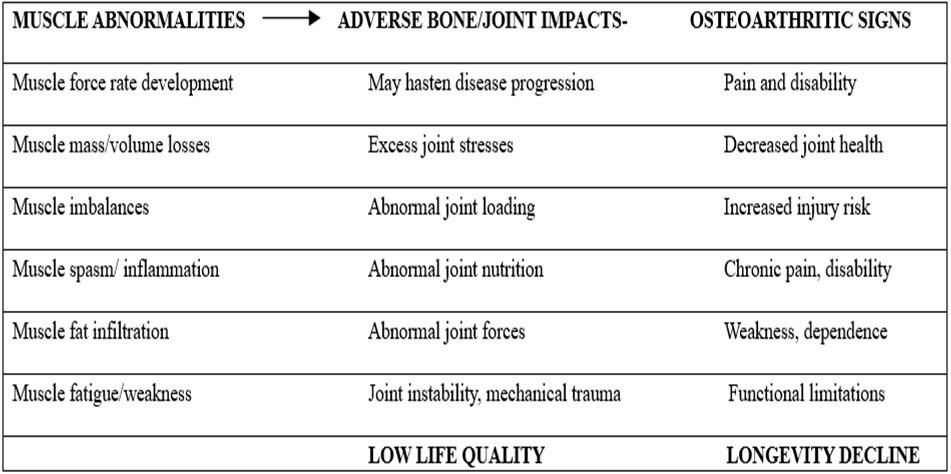
Osteoarthritis, now considered a disease of the whole joint 18, 19 including muscle, is a topic of high interest among those seeking to better understand the etiology of the disease, plus potential remediable associations of its structural features with osteoarthritis risk or progression including malalignment, and lower muscle strength/quality 20.
To this end this narrative review examines some key findings regarding muscle and its inclusion as a potential osteoarthritis risk factor. The review draws on prior research and reviews, with a focus on new research and emerging topics reported since 2017, as well as seminal studies.
Search Results
As of April 30, 2022 there were 1993 articles listed in PUBMED and classified as representing the combined themes of muscle and osteoarthritis. Of these listings, 1156 tended to focus on knee osteoarthritis and muscle factors, 425 on hip osteoarthritis and muscle, and 144 on hand osteoarthritis and muscle as documented over the years of current interest of 2017-2022. Other joints studied sporadically in the past five years in this regard included those of the foot, ankle, mandible, and spine.Yet, in spite of these fairly robust publication numbers, as with prior analyses of this topic of muscle and osteoarthritis by previous authors, a topic studied since 1929, most currently cited citations failed to examine one or more muscle factors as a potential osteoarthritis determinant in the preclinical or early osteoarthritis realm, and the majority of the listed works tended to explore the execution of various exercise interventions, the examination of limited varieties of possible muscle deficits in isolation relative to established osteoarthritis and with no conclusion concerning any cause-effect association being reached. Data listed on GOOGLE SCHOLARand the WEB OF SCIENCE were largely duplicates and repetitive in this regard.
In examining the prevailing data in more detail, and despite a lack of any current emergent uniform set of conclusions, it appeared most authors who examined one or more neuromuscular attributes among various manifestations of osteoarthritic as well as healthy joints, do tend to agree that one or more neuromuscular abnormalities can be quite consistently observed in the realm of osteoarthritis pathology, and that muscle status may have a bearing on one or more osteoarthritis disease features such as cartilage and bone attrition, synovial inflammation, joint instability, pain, and functional deficits.
At the knee, the most well studied joint, multiple authors have indeed observed muscle strength deficits, problems in voluntary muscle activation, muscle mass or volume declines/deficits, deficits in muscle flexibility, excessive muscle tone, generalized muscle weakness of the affected limb, muscle endurance losses, muscle power and force control ability 21, 22, 23, 24, 25, 26, 27, 28. Along with associated alterations in neuromuscular communication processes one or more of these attributes may be instrumental in inducing various molecular and cellular adaptations within both muscle as well as cartilage tissues that further provoke pathological joint changes 29, while altering dynamic joint stability that appears to impact osteoarthritis progression 28.
Other data reveal that cases diagnosed as having symptomatic mid-foot osteoarthritis, a less well studied topic, are also found to exhibit weakness of the foot and leg muscles when compared to those of asymptomatic controls 30, while cases with hip osteoarthritis reportedly exhibited atrophic, bilateral hip and knee muscle weakness and volume deficits that were not evident in control subjects 13, 14, wherein the most seriously affected muscles may surprisingly be the knee extensor muscles 22. At the temporomandibular joint site, osteoarthritis is also possibly influenced by muscle imbalances among other factors, thus suggesting the role of muscle in joint attrition is a possible generalizable finding, rather than solely an isolated or spurious one 31.
Studies also show the presence of age-associated muscle mass losses may engender both the onset or degree of disability associated with knee osteoarthritis 32, including a decline in the rate of muscle force development 33, while knee muscle weakness may have a role to play in mediating radiographic osteoarthritic changes, especially in women who are obese 23, as well as pain 34 and falls injuries that may lead to joint damage 11. Carvalho et al. 35 show cases with patella femoral osteoarthritis exhibited impairments regarding muscle capacity of the hip and knee, higher level of pain and stiffness as well as compromised physical functioning in comparison with healthy controls, while Andrews et al. 36 concluded that measures of grip strength declines may incrementally induce osteoarthritic joint changes at the knee joint, as well as knee pain among men.
Other data indicate that muscle asymmetries from side to side as well as between muscle groups may also have a bearing on lower limb biomechanics and hence the degree of prevailing knee osteoarthritis 37, 38. The pathogenic role of muscle co contraction abnormalities and their impact on joint biomechanics as this pertains to osteoarthritis pathology has also been discussed 39 as has the fact that the presence of osteoarthritis may indeed affect muscle strength adversely and significantly, even if it is not a causative factor 40.
Additional research, albeit currently non-conclusive, also supports a role for muscle wasting 17, 29, 41, 42, neuromuscular deficits 43, and muscle activation abnormalities 44 that might tentatively be remediated through carefully construed intervention 12, 45. Other newer findings reveal a role for multiple muscular attributes in the osteoarthritis disability cycle including the possible combined influence of a related musculo-osteo-arthro-neuropathic syndrome that may underscore the discomfort and pain associated with the osteoarthritic condition, and that warrants a holistic treatment approach 46. Other data are portrayed in Table 1.
Table 1. Sample of key findings published in 2022 selected at random to depict the spectrum of findings in relationship to the interaction of muscle and osteoarthritis pathology.| ATTTRIBUTE ASSESSED | KEY FINDINGS | RESEARCH GROUP |
|---|---|---|
| Knee muscles | There was a positive correlation between vastus lateralis stiffness measures and the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) scores in patients with knee osteoarthritis compared with healthy, asymptomatic subjects | Chang et al. 24 |
| Skeletal muscle molecular attributes | Muscle inflammatory and molecular catabolic gene expression attributes associated with end-stage osteoarthritis differ between the contralateral and surgical limbs as well as between the skeletal muscle surrounding the diseased hip versus knee joints | Drummer et al. 47 |
| Knee muscles | Higher quadriceps and hamstring forces suggest that co-contraction with the gastrocnemius could lead to higher joint contact forces Combined with the excessive loading due to a high external knee adduction moment this may exacerbate joint destruction | Ghazwan et al. 39 |
| Hamstring muscles | The hamstring shear modulus measures were significantly higher in the knee osteoarthritis group than in the control group The hamstring shear modulus in the osteoarthritis group was moderately positively correlated with pain, shear modulus, and physical function score | Li et al. 25 |
| Glenohumeral muscles | Patients with eccentric glenoid wear in the setting of primary glenohumeral osteoarthritis and an intact rotator cuff appear to have both larger posterior rotator cuff musculature and higher rates of infraspinatus fatty infiltration Although the temporal and causal relationships of these associations remain ambiguous, muscle atrophy and fatty infiltration should be considered 2 discrete processes in the natural history of glenohumeral osteoarthritis | Moveman et al. 48 |
| Knee extensor muscle strength | There is low quality evidence that knee extensor muscle weakness is associated with incident symptomatic and radiographic knee osteoarthritis in women and men Optimising knee extensor muscle strength may help to prevent knee osteoarthritis | Oiestad et al. [ 16 ] |
| Knee muscles | Altered neuro-muscular control around knee interferes with normal load distribution and facilitates osteoarthritis disease progression in knee joint | Sharma et al. 49 |
| Hand muscles | In the early stages of hand osteoarthritis, there is a functional deficit associated with a reduced muscle activity of the wrist muscles during manual activities | Tossini et al. 50 |
| Vastus medialis muscle | Muscle weakness is associated with non-contractile muscle tissue of the vastus medialis muscle in knee osteoarthritis | Van den Noort et al. 51 |
| Aged/damaged muscle cells | Muscle cellular senescence may contribute to abnormal osteoarthritis signaling pathways, sarcopenia, excess mechanical exposure to trauma, osteoarthritis initiation and progression | Wu et al. 41 |
| Knee extensors | Knee osteoarthritis patients have lower maximal knee extension strength, but intermuscle group differences prevail, but both are associated with cartilage thickness measures and perceived pain | Yagi et al. 52 |
In addition to those studies depicted in Table 1, adding to the diversity of information retrieved is the fact that, as with prior research 53, almost all additional studies reviewed employed differing osteoarthritis samples, research questions, assessment approaches, and durations, and even if a similar variable or idea was examined, basically no two conclusions were alike. Indeed, some reported associations between reduced rates of torque development and osteoarthritis features, while others noted a propensity for fat cells in muscle to increase the risk for osteoarthritis [see Figure 1]. Yet, others noted the importance of abnormal dynamic muscular force adaptations, afferent sensory dysfunction, along with diminished strength capacity, co-ordination impairments, and problems with reduced muscle endurance.
Loureiro et al. 26 conclude, pre-existing deficits in hip muscle strength in individuals with mild-to-moderate hip osteoarthritis do appear to be accentuated over a 12-month period, despite a lack of any parallel changes in symptoms or joint structure measures. According to their synthesis of the literature, Tayfur et al. 27 found neuromuscular deficits to be consistently evident across different structural knee osteoarthritis severities including those of muscle strength, voluntary activation, muscle size, and force control ability, wherein women have tended to exhibit these alterations to a greater extent than men. Ghazwan et al. 39 note the possible role of an increased lateral muscle co-contraction in mediating hip joint osteoarthritis progression, whereas an increased medial muscle co-activation could potentially differentiate between whether osteoarthritis affects one- or more joint compartments in their view. As well, Lijema et al. 54 report that their recent findings of an increased medio-lateral trunk movement asymmetry in adults with knee osteoarthritis may be an indicator of impairment, rather than an adaptation. While requiring validation by future study, these authors argue that paying close attention to medio-lateral trunk movement asymmetry may be key to our understanding of osteoarthritis-related pain and disability. However, a longer follow-up period is clearly required to establish whether muscular factors do drive clinical and structural declines in these patients, as well as whether interventions to prevent or slow such abnormalities or declines may be relevant in the management of osteoarthritis.
In the interim-as outlined above, consistent with the view that osteoarthritis may result from a number of different interrelated factors and is not merely a disease of a single tissue 55, muscular differences do appear to discriminate osteoarthritic muscle patterns from those of controls 44, 56, and to thereby possibly influence or explain the severity of the observed osteoarthritis manifestations 56. Moreover, while not necessarily comprehensive, those observations depicted in Table 1, and others further indicate a variety of muscle impairments may well differ among muscles of the same individual at both the early, as well as the later stages of osteoarthritis, as well as in muscles unrelated to the diseased joint, hence the possibility that some cases of osteoarthritis represent the outcome of a clinical neuromuscular set of subnormal mechanisms cannot be ignored. Lee et al. 57 who found leg muscle asymmetry patterns in men with knee osteoarthritis, regardless of obesity level, showed these patterns to be significantly associated with both knee pain as well as radiographic indicators of osteoarthritis severity. Lower limb strength asymmetry has been associated with increased odds of secondary osteoarthritic development, with a 1% increase in quadriceps symmetry resulting in a 4% lower odds of developing clinical osteoarthritis 58. Bird et al. 59 note hip abductor muscle quality and hip osteoarthritis to be related, while Drummer et al. 60 found evidence of differential inflammatory and catabolic gene expression between the contra lateral and surgical limbs along with differences between the skeletal muscles surrounding the diseased hip versus knee joints.
As such, these aforementioned muscle abnormalities clearly indicate that they may well have considerable independent as well as collective influence on joint status, including, but not limited to, multiple forms of strength loss and aberrant motor control attributes, pathological alterations in muscle molecular mechanisms and muscle structure, muscle temporal and contractile characteristics, muscle proprioception and fat content, plus muscle imbalances. However, even though it appears neuromuscular factors are being more accepted as possible osteoarthritis pathogenic determinants 61, which attributes are of specific clinical relevance, or of greatest import in the context of different phases of the disease process and a variety of joint sites remains impossible to discern with any clarity. Indeed, the lack of uniformity in cohorts studied, the unknown effects of age, weight, health and comorbid disease effects, differing research designs and questions, and small samples, along with confusion as to whether muscle strength or muscle balance or muscle power is a salient variable, and if so, in what way-precludes any meaningful synthesis of this research even though considerable recent research prevails. Moreover, even with prospective analyses, which are more supportive of a temporal relationship of the muscle and joint disturbances than not, attempts to isolate sub categories of muscle dysfunction for purposes of a systematic analysis persist in remaining extremely challenging at best to aggregate, because keywords selected to describe a specific topic, do not always comport with or yield the desired data. As well, which if any of the muscle function assays represent protective reactions, rather than pathological correlates or adaptations, is also impossible to discern with any clarity at the present time.
Discussion
While aging is inevitable, increasing evidence suggests aging as a negative state does not have to be a foregone ordained experience of downward spiraling events and perceptions. This review explored the degree to which one or more muscle factors may be implicated in the pathology of osteoarthritis, a disabling disease, commonly believed to originate in the articular cartilage lining of one or more freely moving joints, and that affects the entire joint, including the associated muscles surrounding the joint. This brief highlights the research published in this respect since 2017, an area of research that is not well documented when compared to related studies of cell biology, tissue engineering, and molecular in vitro studies, among others. To this end, to update a prior analysis, PUBMED, GOOGLE SCHOLAR and the WEB of SCIENCEs indices were searched for information specifically regarding this topic using key words: Osteoarthritis and Muscle.
Among these studies, and bearing in mind, most were cross-sectional studies, based on knee osteoarthritis, these included a very broad array of thematic topics, rather than any uniform body of content. These topics included observations of how various forms of muscle dysfunction appear to interact with various forms of osteoarthritis pathology, various measures of pain, and various aspects of function, although very few tested any possible hypothesis that might prove revealing and most did not explore the underpinnings of the observed muscle alterations specifically. Others described the relationship between muscle power measures, the rates at which selected muscles contract 15, 62, muscle relaxation associated deficits and their association with the extent of the prevailing pathology 23, 63, and/or osteoarthritis disease progression 15, but not other overlapping factors, determinants of these impairments, or possible molecular or inflammatory joint associated influences.
However, since most results did not negate an important role for muscle in the osteoarthritic disease process, more carefully construed biomechanical, radiological, molecular, biochemical, and neuromuscular oriented prospective studies of adequate duration in the future may help to solidify the current observations, which at a minimum are supportive of a role for muscle strength as a salient remediable factor in the osteoarthritic disease process as outlined by others some time ago 61, 64. Newer research may also help to more clearly discern if muscle dysfunction may be a causative or pre existing, rather than a reactive disease factor 65, 66, as well as how several differing muscle factors are possibly associated with either the onset or progression of osteoarthritis at various joints.
In this regard, in addition to strength training, possible cited benefits that might accrue from other muscle oriented interventions, especially those cited below may prove revealing and should be examined in comparative studies to evaluate their possible impact on chondrogenic repair, muscle cell structures and mechanisms, and overall mobility, pain, and function 12, 17, 41, 66, 67, 68, 69, 70, 71, 72, 73, 74, 75, 76, 77, 78, 79. Box 1
Box 1. Selected approaches other than exercise that might be examined more specifically in diverse populations of adults of a broad spectrum of ages as regards their preventive, maintenance, and disability reducing capacity. Key Sources: (12, 17, 41, 67-79)| Electrical Muscle Stimulation |
| Exercise + Minimally Invasive Injection Therapy |
| Graded Motor Imagery and Muscle Relaxation |
| Kinesio Taping |
| Local Muscle Vibration |
| Minimize Sarcopenia |
| Modulation of Muscle Micro RNAs |
| Muscle Stretching and Low Level Laser |
| Neuromuscular Training |
| Strategies to Counter Muscle Cell Senescence |
| Pequi Oil Applications |
| Proprioceptive Training |
| Protein Supplements |
| Whole Body Vibration and Strengthening Exercises |
In short, contrary tothe enormous amount of available literature on osteoarthritis related topics of articular tissue biology, genetics, pharmacologic and surgical options for osteoarthritis, plus stem cell research and tissue bioengineering, where almost no singular approach has proven viable to date as either a preventive or treatment approach, a conceptual framework for guiding clinical practice remains elusive at best, but is one likely to fail if muscular factors are ignored.
Limitations to this review are its narrow focus, the sole use of three key data bases and not others and a lack of any systematic attempt to aggregate or critique the research approaches, that were deemed salient and their possible shortcomings. Moreover, not all articles may have been accessed or reported, and those that have may yet appear to support a muscle osteoarthritis association due to issues of publication bias, rather than the undoubted weight of the supportive evidence currently housed electronically in the world’s leading data bases.
However, at the very least, and consistent with past reports 53, 64, 74, the weight of the evidence and present results do appear to provide a reasonable body of current science based evidence that shows efforts to identify one or more possible attributes of muscle structure and/or function that may be subnormal in some way may well further our understanding of the etiology of some forms of osteoarthritis, as well as its tendency to flare or wax and wane, but ultimately to progress rather than undergo any disease reversal. Additionally, while not conclusive, a second feature is that a variety of alterations in muscle quality, function, metabolism, and/or estimates of muscle bulk or vibratory acuity, do appear to correlate temporally with selected features of the osteoarthritic pathological processes and extent of disability, regardless of age, even though long term studies and efforts to identify important muscle associated correlates are commonly lacking. Third, rather than exercise alone, multiple treatment approaches directed towards improving muscle structure and function in those with symptomatic osteoarthritis appear to hold promise for potentiating more favorable results than not. Conversely, a failure to identify what component of the muscle system may be specifically implicated in the osteoarthritis disease process may produce either null results or more negative clinical outcomes than not, for example if exercise induces fatigue.
Further research to examine the interaction of joint alignment, neurology, geometry, stability, and bone mass and the interplay of muscle metabolism, structure, mechanics and function on articular cartilage signaling mechanisms in a broad range of adults deemed at risk for osteoarthritis and that employ advanced biomechanical, electrophysiological, and imaging technologies is however strongly recommended in this regard. As well, as proposed by Becker et al. 65 in 2004, whether muscle dysfunction occurs not only at singular joint sites but represents a generalized presence of neuromuscular dysfunction is advocated as well. Examining the possible long term interactions of one or more neuromuscular attributes and the processes of joint attrition is also strongly warranted in this regard in our view, and could prove highly valuable in many spheres. In particular efforts to carefully explore the role of muscle mechanics and its mechanisms of control, and how one or more of these factors may interact with the multiple structural features of various joints affected by osteoarthritis is likely to prove insightful. Efforts to tease out the impacts of muscle on all joint structures affected by osteoarthritis, and particularly on cartilage content, genetics, molecular and structural organization features is also likely to prove of high clinical relevance and utility.
At the same time clinical efforts to examine the attributes of muscle strength, endurance, rate of force development, vibratory acuity, and possible inflammation and fat mass, along with muscle activation profiles, as well as sarcopenic indicators on the disease progression using advanced biological as well as biomechanical technologies is highly recommended in order to identify what interventions might be most helpful in retarding joint degenerative processes and why.
Conclusion
While not conclusive, in light of many current innovative findings and years of collective and current research, we conclude a case for rejecting muscle as an important osteoarthritis determinant cannot be advanced.
We further conclude:
1. At least some osteoarthritis sufferers may well have underlying muscle related impairments that may be challenging to detect or unrecognized and diagnosed.
2. Varying muscular deficits do appear to prevail for some considerable duration in many osteoarthritis cases post surgery, despite pain relief and successful surgery, thus may have been present pre-clinically.
3. Muscle deficits may occur in both limbs even in the case of a unilateral disease diagnosis and in non traumatic cases.
4. Aging does not explain why some older adults with normal muscle function do not exhibit osteoarthritis degeneration and implies more attention to muscular factors and their specific remediation, versus generic based approaches, are indeed urgently indicated.
5. To establish how these facts interconnect at the development of osteoarthritis, as well as its progression and to apply this knowledge in a carefully and insightfully targeted manner consistent with chondrocyte physiology is a realm of great promise for many in our view and is strongly advocated.
Indeed, despite a lack of consensus on the evolutionary impact of neuromuscular mechanisms on osteoarthritis, we conclude further insightful and carefully designed research may prove highly valuable in explicating those muscular mechanisms that may yet impact the immense disability associated with unrelieved osteoarthritis.