Abstract
Multiple organ dysfunction syndrome or failure is one of the major concerns against healthcare services in order to maintain the normal function. The present study aimed to explore the impact of the Biofield Energy Treated test formulation on the function of vital organs such as bones, heart, liver, lungs, and brain using standard activity parameters in specific cell-based assays. The test formulation and cells medium was divided into two parts, one untreated (UT) and other part received the Biofield Energy Treatment remotely by a renowned Biofield Energy Healer, Ariadne Esmene Afaganis, Canada and was labeled as the Biofield Treated (BT) test formulation/media. The test formulation was tested for cell viability, and the data suggested that the test formulation was found safe and non-toxic against all the cell lines. Cytoprotective activity among the experimental groups showed a significant improved activity by 94.4% at 1 µg/mL in untreated medium (UT-Med) + Biofield Treated Test Item (BT-TI) group in human cardiac fibroblasts cells (HCF) cells, while 84.4% at 10 µg/mL in BT-Med + BT-TI groups in human hepatoma cells (HepG2), and 124% increased cytoprotective action at 1 µg/mL in UT-Med + BT-TI group in adenocarcinomic human alveolar basal epithelial cells (A549) cells as compared with the untreated test group. ALP activity in MG-63 cells was significantly increased by 85.9% at 10 µg/mL in the UT-Med + BT-TI group, while in Ishikawa cells showed maximum increased ALP activity by 59.2% at 0.1 µg/mL in BT-Med + BT-TI groups as compared to the untreated group. The percent protection of HCF (heart) cells (decreased of LDH activity) was significantly increased by 53% and 40.5% at 1 and 10 µg/mL concentrations respectively, in UT-Med + BT-TI group, while BT-Med + UT-TI group showed increased protection by 68.5%, 70.7%, and 16.8% at 0.1, 1, and 10 µg/mL respectively, and 86.5%, 62.5%, and 34.2% improved cellular protection at 0.1, 1, and 10 µg/mL respectively, in BT-Med + BT-TI group as compared to the untreated test group. The percent protection of HepG2 (liver) cells (decreased of ALT activity) was reported by 33.5%, 63.2%, and 99.2% at 10 µg/mL in the UT-Med + BT-TI, BT-Med + UT-TI, and BT-Med + BT-TI groups, respectively compared to the untreated group. Cellular protection of A549 (lungs) cells (increased of SOD activity) in terms of percentage was increased by increased by 39.8% (at 10 µg/mL), 44% (at 25.5 µg/mL), and 59.7% (at 25.5 µg/mL) in the UT-Med + BT-TI, BT-Med + UT-TI, and BT-Med + BT-TI groups, respectively compared to untreated group. Serotonin level was significantly increased by 59.2% (at 0.1 µg/mL), 190.3% (at 0.1 µg/mL), and 201% (at 1 µg/mL) in the UT-Med + BT-TI, BT-Med + UT-TI, and BT-Med + BT-TI groups, respectively compared to untreated in human neuroblastoma cells (SH-SY5Y). However, the relative quantification (RQ) of vitamin D receptor (VDR) was significantly increased by 159.1% (at 50 µg/mL), 212.7% (at 1 µg/mL), and 278.3% (at 10 µg/mL) in the UT-Med + BT-TI, BT-Med + UT-TI, and BT-Med + BT-TI groups, respectively as compared to the untreated in MG-63 cells. Thus, the present data concluded that the overall multiple organ health using various standard biomarkers in specific cell lines were significantly improved with respect to health of bones, heart, liver, lungs, and brain after treatment with the Biofield Energy treated test formulation (The Trivedi Effect®). Thus, it can be used as a complementary and alternative therapy approach against many multiple organ disorders such as coronary artery disease, arrhythmias, congenital heart disease, cardiomyopathy, cirrhosis, liver cancer, hemochromatosis, asthma, chronic bronchitis, cystic fibrosis, osteoporosis, etc.
Author Contributions
Academic Editor: Jun Wan, Department of Medical and Molecular Genetics, Indiana University School of Medicine, USA.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2019 Ariadne Esmene Afaganis, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Herbal product based test formulations using vital phytonutrients or nutraceuticals are showing immense importance around the world due to its high acceptance rate against various health care challenges with respect to health of major vital organs such as bones, heart, liver, lungs, and, brain 1. More than 80% of the world populations (~ 4 billion peoples) rely on herbal product for their respective health challenge in the developing world 2, 3. Thus, for measuring the biomarkers for overall health, specific cell lines were selected for various activities such as for bone health MG-63 cells (Human Osteoblasts like Cells) were used for estimation of ALP activity (phenotypic marker for the early differentiation and maturation of osteoblasts). The study data suggested that the response of MG-63 cells for 1, 25-dihydroxyvitamin D3 (1,25(OH)2D3) administration has been found to be very similar to the normal human osteoblast cells 4. MG-63 cell line has been widely reported for studying the potential of any test compounds to improve bone health 5. For estimation of menstrual pain dysmenorrheal, Ishikawa (Human Endometrial cancer cell line) were used for estimation of ALP activity (represents estrogenic activity). These cells are derived from human endometrium that are found to be very significant in female reproductive functions along with fertility-determining factor 6, 7, 8. Similarly, lung health was determined using estimation of cytoprotection SOD activity in A549 (Human lung adenocarcinoma cell line) 9. Liver health was determined using HepG2 (Human liver cell line) by estimation of cytoprotection ALT action 10, 11, 12. Heart health was determined using estimation of cytoprotection LDH in Human Cardiac fibroblasts cells 13, 14. Memory and cognitive health was detected using estimation of serotonin release using SH-SY5Y (Human Neuroblastoma Cells) 15, 16. Similarly, Vitamin D metabolism was detected using estimation of VDR expression in MG-63 (Human Osteoblasts like Cells), as these are the widely used biomarkers to study VDR expression as an in-vitro model 17, 18. The scientific data suggested that the death of more than 17 million people worldwide was due to cardiovascular disorders, multiple organ failure, and all are somehow related with the generation of free radicals, which are responsible for the onset of these health issues 19, 20.
Thus, the present study was aimed to detect the overall health status using novel test formulation, which included a combination of 11 ingredients, calcium chloride, panax ginseng extract, vitamin B12, beta carotene, vitamin D3, zinc chloride, magnesium gluconate, sodium selenate, ferrous sulfate, ascorbic acid and vitamin B6. Further, the effect of test formulation was tested on Ishikawa, MG-63, A549, SH-SY5Y, HepG2, and Human Cardiac Fibroblast (HCF) for respective activities after Biofield Energy Healing Treatment. Biofield Energy Healing therapies are accepted worldwide as one of the best Complementary and Alternative Medicine (CAM) concept for altering overall quality of life. Various forms of Energy Healing Therapies have been reported with significant clinical and non-clinical outcomes 21, 22. Energy Healing Therapies have been practiced and accepted by the U.S. population and is well defined by National Center for Complementary and Alternative Medicine (NCCAM) 23, 24. CAM therapies such as external qigong, Johrei, Reiki, therapeutic touch, yoga, Qi Gong, polarity therapy, Tai Chi, pranic healing, deep breathing, chiropractic/osteopathic manipulation, guided imagery, meditation, massage, homeopathy, hypnotherapy, progressive relaxation, acupressure, acupuncture, special diets, relaxation techniques, Rolfing structural integration, healing touch, movement therapy, pilates, mindfulness, Ayurvedic medicine, traditional Chinese herbs and medicines in biological systems both in vitro and in vivo. The Trivedi Effect®-Consciousness Energy Healing therapies have been widely accepted worldwide in nonliving materials and living organisms. Consciousness Energy Healing Treatment found to be significant to improve the metal physicochemical properties 25, 26, improved crop yield in agriculture science 27, microbiology 28, 29, biotechnology 30, 31, improved bioavailability of many compounds 32, 33, improved skin health 34, 35, improved properties of nutraceuticals 36, cancer science research 37, improved overall bone health 38, 39, 40, human health and wellness. Due to the continued outcomes and wide applications of Biofield Energy Healing Treatments, the test formulation was studied for impact of the Biofield Energy Healing Treated test formulation on the function of vital organs such as bones, heart, liver, lungs, and brain specific biomarkers in different cell-lines.
Materials and Methods
Chemicals and Reagents
The constituents of test formulations such as ferrous sulfate, vitamin B6, vitamin D3, vitamin B12, calcium chloride, naringenin, trimetazidine (TMZ), 3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide (MTT), and ethylenediaminetetraacetic acid (EDTA) were collected from Sigma Chemical Co. (St. Louis, MO). In addition, zinc chloride, magnesium gluconate, β-carotene, and calcitriol were procured from TCI chemicals, Japan. Panax ginseng extract obtained from panacea Phytoextracts, India. Sodium selenate and ascorbic acid were obtained from Alfa Aesar, India. Silymarin and curcumin were obtained from Sanat Chemicals, India and quercetin was purchased from Clearsynth, India. Reverse Transcription Kit, RNeasy Mini Kit, and Syber Green PCR kits were procured from Qiagen, India. All the other chemicals used in this experiment were analytical grade procured from India.
Biofield Energy Healing Treatment
Biofield Energy Healing was done on the test formulation, which was the combination of eleven ingredients viz. calcium chloride, panax ginseng extract, vitamin B12, β-carotene, vitamin D3, zinc chloride, magnesium gluconate, sodium selenate, ferrous sulfate, ascorbic acid, and vitamin B6. The test formulation/media was divided into two parts, one portion was considered as the untreated group, where no Biofield Energy Treatment was provided. Further, the untreated group was treated with a “sham” healer for comparison purposes with other experimental groups. The “sham” healer did not have any knowledge about the Biofield Energy Healing Treatment. The other portion of the test formulation/media received the Biofield Energy Treatment (The Trivedi Effect®) remotely by Ariadne Esmene Afaganis, under laboratory conditions for ~3 minutes through healer’s unique Biofield Energy Transmission process and was referred as the Biofield Energy Treated test formulation/media. The Biofield Energy Healer was located in the Canada, however the test items were located in the research laboratory of Dabur Research Foundation, New Delhi, India. Biofield Energy Healer in this experiment did not visit the laboratory, nor had any contact with the test samples. After that, the Biofield Energy Treated and untreated test items were kept in similar sealed conditions and used for the study as per the study plan.
Assessment of Cell Viability Using MTT Assay
All the cells were counted for cell viability using hemocytometer in 96-well plates at the specific density as mentioned in the Table 1. The cells were then incubated overnight under standard growth conditions to allow cell recovery and exponential growth. Following overnight incubation, cells were treated with different concentrations of test formulations (BT/UT). After respective treatments, the cells were incubated in a CO2 incubator at 37°C, 5% CO2, and 95% humidity. After incubation, the plates were taken out and 20 µL of 5 mg/mL of MTT 3-(4,5-dimethylthiazol-2-yl)-2, 5-diphenyl tetrazolium bromide solution was added to all the wells followed by additional incubation for 3 hours at 37°C. The supernatant was aspirated and 150 µL of DMSO was added to each well to dissolve formazan crystals. The absorbance of each well was read at 540 nm using Synergy HT microplate reader. The percentage cytotoxicity at each tested concentration was calculated using Equation 1:
% Cytotoxicity = ((R-X)/R)*100……………(1)
Where, X = Absorbance of treated cells; R = Absorbance of untreated cells
The concentrations exhibiting percentage cytotoxicity <30% was considered as non-cytotoxic 41.
Table 1. Information related to six cell lines with their plating density and time-point.| S. No. | Cell Line | Plating | Time Point |
| 1 | MG-63 (Bone) | 3x104 cells/ well, 96-well plate | 5 days |
| 2 | Ishikawa (Uterus) | 3x104 cells/ well, 96-well plate | 5 days |
| 3 | A549 (Lung) | 10x104 cells/ well, 96-well plate | 24 hours |
| 4 | HepG2 (Liver) | 1x104 cells/ well, 96-well plate | 24 hours |
| 5 | Human Cardiac fibroblasts (Heart) | 1x104 cells/ well, 96-well plate | 24 hours |
| 6 | SH-SY5Y (Neuronal cell) | 10x104 cells/ well, 96-well plate | 24 hours |
Evaluation of the Cytoprotective Effect of the Formulation
Cytoprotective effect of the test formulation in various cells such as human cardiac fibroblasts-HCF; human hepatoma cells-HepG2; and adenocarcinomic human alveolar basal epithelial cells-A549 were counted and plated in suitable medium followed by overnight incubation. Further, the cells were then treated with the test items/positive control at the non-cytotoxic concentrations for 24 hours. After 24 hours, the oxidative stress using 10 mM t-BHP for 3.5 hours was given to the cells. The cells treated with 10 mM of t-BHP alone served as negative control. After 3.5 hours of incubation with t-BHP the above plates were taken out and cell viability was determined by MTT assay. The percentage protection corresponding to each treatment was calculated using equation 2:
% Protection = ((Absorbancesample-Absorbancet-BHP))
100/ (Absorbanceuntreated-Absorbancet_BHP ) ……(2)
Assessment of Alkaline Phosphatase (ALP) activity
For the estimation of ALP, the cells (human bone osteosarcoma cells-MG-63 and human endometrial adenocarcinoma cells-Ishikawa) were counted using a hemocytometer and plated in 24-well plates at the density corresponding to 1 X 104 cells/well in phenol-free DMEM supplemented with 10% CD-FBS. Following the respective treatments, the cells in the above plate were incubated for 48 hours in CO2 incubator at 37°C, 5% CO2, and 95% humidity. After 48 hours of incubation, the plates were taken out and processed for the measurement of ALP enzyme activity. The cells were washed with 1 X PBS and lysed by freeze-thaw method i.e., incubation at -80°C for 20 minutes followed by incubation at 37°C for 10 minutes. To the lysed cells, 50 µL of substrate solution i.e. 5 mM of p-nitrophenyl phosphate (pNPP) in 1M diethanolamine and 0.24 mM magnesium chloride (MgCl2) solution (pH 10.4) was added to all the wells followed by incubation for 1 hour at 37°C. The absorbance of the above solution was read at 405 nm using Synergy HT microplate reader (Biotek, USA). The absorbance values obtained were normalized with substrate blank (pNPP solution alone) absorbance values. The percentage increase in ALP enzyme activity with respect to the untreated cells (baseline group) was calculated using Equation 3:
% Increase in ALP = {(X-R)/R}*100……………(3)
Where, X = Absorbance of cells corresponding to positive control and test groups
R = Absorbance of cells corresponding to baseline group (untreated cells)
2Estimation of Lactate Dehydrogenase (LDH) in Human Cardiac Fibroblasts (HCF) Cells
HCF cells were counted and plated at the density of 0.25 X 106 cells/ well in 24-well plates in human cardiac fibroblast specific medium followed by overnight incubation. The cells were then treated with the test formulation combinations/positive control at the non-cytotoxic concentrations for 24 hours. After 24 hours, oxidative stress was given to the cells using 10 mM t-BHP for 3.5 hours. The untreated cells were served as control group, which did not receive any treatment and were maintained in cell growth medium only. Cells treated with 10 mM of t-BHP alone served as the negative control. After 3.5 hours of incubation with t-BHP, the above plates were taken out and LDH activity was determined using LDH activity kit as per manufacturer’s instructions. The percent increase in LDH activity was calculated using Equation 4.
% Increase = ((LDH activitysample-LDH activityt-BHP))*100/ (LDH activityuntreated-LDH activityt_BHP )……………(4)
Estimation of ALT in Liver Cells (HepG2)
The human hepatoma cells (HepG2) were counted and plated at the density of 5 X 104 cells/well in 48-well plates in DMEM media followed by overnight incubation. The cells were then treated with the test formulation/positive control at the non-cytotoxic concentrations for 24 hours. After 24 hours, oxidative stress was given to the cells using 400 µM t-BHP for 3.5 hours. The untreated cells served as control that did not receive any treatment and were maintained in cell growth medium only. Cells treated with 400 µM of t-BHP alone served as negative control. After 3.5 hours of incubation with t-BHP, the above plates were taken out and ALT activity was determined using ALT activity kit as per manufacturer’s instructions. The percent increase in ALT activity was calculated using Equation 5.
% Increase = ((ALT activitysample-ALT activityt-BHP))*100/ (ALT activityuntreated-ALT activityt_BHP )……………(5)
Estimation of Superoxide Dismutase (SOD) in Lung (A549) Cells
The adenocarcinomic human alveolar basal epithelial cells (A549) were counted and plated at the density of 1 X 104 cells/well in 24-well plates in DMEM followed by overnight incubation. The cells were then treated with the test formulation/positive control at the non-cytotoxic concentrations along with 100 µM t-BHP to induce oxidative stress. The untreated cells served as control that did not receive any treatment and were maintained in cell growth medium only. Cells treated with 100 µM of t-BHP alone served as negative control. After 24 hours of incubation with t-BHP the above plates were taken out and SOD activity was determined using SOD activity kit as per manufacturer’s instructions. The percent increase in SOD activity was calculated using equation 6:
% Increase in SOD activity = ((X-R)/R)*100……………(6)
Where, X = SOD activity corresponding to test item or positive control
R = SOD activity corresponding to Control group.
Estimation of Serotonin in Neuronal Cells (SH-SY5Y)
The human neuroblastoma (SH-SY5Y) cells were counted and plated at the density of 10 X 104 cells/well in 96-well plates followed by overnight incubation. The cells were then treated with the test formulation/positive control at the non-cytotoxic concentrations. The untreated cells served as control that did not receive any treatment and were maintained in cell growth medium (DMEM + Ham’s F12) only. The treated cells were incubated for 24 hours. Serotonin release was determined by ELISA as per manufacturer’s protocol. The percent increase in serotonin levels was calculated using equation 7.
((X-R)/R)*100……………(7)
Where, X = Serotonin levels corresponding to test item or positive control,
R = Serotonin levels corresponding to control group.
Effect of Test Formulation on Vitamin D Receptor (VDR) in Bone (MG-63) Cells
The effect of test formulation on vitamin D receptor (VDR) activity in bone (MG-63) cells were counted using the hemocytometer at density 2 X 105cells/well in 6-well plates followed by overnight incubation. The cells were then sera starved for 24 hours and treated with the test formulation/positive control at the non-cytotoxic concentrations, while control group did not receive any treatment, which were maintained in cell growth medium only. The treated cells were incubated for 24 hours and VDR expression was determined by qPCR using VDR specific primers. Cells were harvested by scrapping and washed with PBS. Cell pellets obtained were analyzed for VDR gene expression using human VDR specific primers: Forward: 5’-GCTGACCTGGTCAGTTACAGCA-3’, Reverse: 5’-CACGTCACTGACGCGGTACTT-3’. VDR gene expression was normalized using House-keeping (HK) reference. Relative quantification (RQ) of VDR gene in Biofield Energy Treated cells was calculated with respect to the untreated cells using equation 8:
RQ = 2-N……………(8)
Where, N is the relative Threshold Cycle (CT) value of treated sample with respect to the untreated sample.
Statistical Analysis
All the values were represented as percentage.
Results and Discussion
Cell Viability Using MTT Assay
MTT assay data in each cell line showed various percentage of cell viability. The criteria for non-cytotoxic test formulation concentration and the positive controls by MTT cell viability assay was resulted in less than 30% cytotoxicity or greater than 70% cell viability were considered as non-cytotoxic concentrations. Thus, data suggested that the percent viable cells in six tested different cell-lines viz. MG-63, Ishikawa, A549, HepG2, HCF, and SH-SY5Y. Based on the percent cell viability data, it was observed that the test formulation and positive controls were found safe and non-toxic at the tested concentrations in six different cell lines and selected for other parameters analysis.
Evaluation of Cytoprotective Effect of the Test Formulation
Cytoprotective effect was checked using induced oxidative damage in various cells, which depicts the protection of vital organs and is functioning viz. heart, liver, and lungs. The activity of the test formulation was examined in the in vitro cell-based assays under the stimulation of oxidative stress in different cell-lines using tert-butyl hydroperoxide (t-BHP) 41, 42. The cytoprotective activity results with respect to the cellular restoration, which were determined against t-BHP induced cell damage and the data was presented in the Figure 1. Trimetazidine (TMZ), positive control group in human cardiac fibroblasts cells (HCF) showed significant restoration of cell viability by 60% and 98.3% at 10 and 25 µg/mL, respectively as compared to the t-BHP induced group. Besides, the maximum restoration of cell viability among the tested groups by the test formulation was reported as 94.4% (at 1 µg/mL), 21.5% (at 25.5 µg/mL), and 42% (at 25.5 µg/mL) in the UT-Med + BT-TI, BT-Med + UT-TI, BT-Med + BT-TI groups, respectively as compared to the UT-Med + UT-TI group. Similarly, silymarin was used as positive control in HepG2 cells, which resulted in significant cellular restoration by 38.4%, 56.6%, and 72.6% at 5, 10 and 25 µg/mL, respectively as compared to the t-BHP induced group. Besides, the test formulation showed maximum restoration of cell viability by 25.4% (at 1 µg/mL), 58.2% (at 1 µg/mL), and 84.4% (at 10 µg/mL) in the UT-Med + BT-TI, BT-Med + UT-TI, BT-Med + BT-TI groups, respectively as compared to the UT-Med + UT-TI group. In addition, quercetin was used as positive control in adenocarcinomic human alveolar basal epithelial cells (A549) resulted, restoration of cell viability by 76.7% and 86.1% at 10 and 25 µg/mL, respectively compared to the t-BHP induced group. Besides, the test formulation showed maximum restoration of cell viability by 124% (at 1 µg/mL), 21.9% (at 10 µg/mL), and 7.6% (at 10 µg/mL) in the UT-Med + BT-TI, BT-Med + UT-TI, BT-Med + BT-TI groups, respectively as compared to the UT-Med + UT-TI group 43. Oxidative stress has been reported as the major contributor for cellular damage and high incidence has been found with respect to the initiation of many liver diseases. Increasing evidence has indicated that cellular damage mediated by oxidative stress contributed to the initiation and progression of many liver diseases 44 along with various age-related disorders like diabetes, cardiovascular, autoimmune diseases, and cancer 45, 46, 47. The results suggest that Biofield Energy Healing Treatment significantly protects t-BHP induced oxidative stress with respect to cardiotoxicity, hepatotoxicity, and lung cell toxicity which could be due to The Trivedi Effect®. Therefore, the Biofield Energy Healing Treatment could be successfully used for the management of cardiovascular, liver, and various lung disorders.
Figure 1. Assessment of cytoprotective effect of the test formulation in human cardiac fibroblasts cells (HCF), human hepatoma cells (HepG2), and adenocarcinomic human alveolar basal epithelial cells (A549) against tert-butyl hydroperoxide (t-BHP) induced damage. Trimetazidine (µM), silymarin (µg/mL), and quercetin (µM) were used as positive control in HCF, HepG2, and A549 cells, respectively. UT: Untreated; Med: Medium; BT: Biofield Treated; TI: Test item.
Assessment of Alkaline Phosphatase (ALP) Activity
ALP activity was used against two cell lines, MG-63 and Ishikawa cells. Calcitriol (nM) was used as positive control for MG-63 cells, and the results suggested significant increased ALP level by 13.2%, 21.4%, and 35.4% at 0.1, 1, and 10 nM, respectively as presented in Figure 2. However, higher concentrations of calcitriol were also tested, which showed upto 97.3% increased ALP activity at 500 nM. However, the test groups showed maximum increased ALP activity by 85.9%, 85.2%, and 87.4% at 10 µg/mL in the UT-Med + BT-TI, BT-Med + UT-TI, BT-Med + BT-TI groups, respectively as compared to the UT-Med + UT-TI group in MG-63 cells. Similarly, naringenin was used as positive control for Ishikawa cells, and the data showed significant improved level of ALP by 9.5%, 23.7%, and 130.2% at 0.1, 1, and 10 nM, respectively. In the experimental tested groups, the maximum ALP percent was significantly increased by 8.5%, 28%, and 59.2% at 0.1 µg/mL in UT-Med + BT-TI, BT-Med + UT-TI, BT-Med + BT-TI groups, respectively as compared to the UT-Med + UT-TI group. ALP is considered as one of the major bone maker and bone health, responsible for bone mineralization 48. Thus, these bone markers would be the best clinical significance with respect to the progression of any bone diseases 49. The present experimental data suggested that the Biofield Energy Healing Treated test formulation significantly increased the level of ALP expression that can be significantly recommended against any bone-related disorders.
Figure 2. The effect of the test formulation on alkaline phosphatase (ALP) in human bone osteosarcoma cells (MG-63) and human endometrial adenocarcinoma cells (Ishikawa).Calcitriol and naringenin were used as positive control in MG-63 and Ishikawa cells, respectively. UT: Untreated; Med: Medium; BT: Biofield Treated; TI: Test item.
Assessment of Lactate Dehydrogenase (LDH) Activity in Human Cardiac Fibroblasts (HCF)
HCF cells were used for the testing of LDH activity and the data was reported in terms of increased percentage protection, which suggests decreased LDH activity. The positive control, trimetazidine (TMZ) exhibited 43.1%, 85.9%, and 107.9% protection of HCF cells (decreased of LDH activity) at 10, 50, and 100 µM concentration as compared to the t-BHP group. The effect of test items on the percent protection of HCF cells in terms of decreased level of lactate dehydrogenase (LDH) activity is presented in Figure 3. Among the tested groups, the maximum percent protection of HCF cells (decreased of LDH activity) was significantly increased by 53% and 40.5% at 1 and 10 µg/mL concentrations respectively, in the UT-Med + BT-TI group, and 68.5%, 70.7%, and 16.8% improved cellular protection (decreased of LDH activity) at 0.1, 1, and 10 µg/mL respectively in the BT-Med + UT-TI group, while 86.5%, 62.5%, and 34.2% improved cellular protection (decreased of LDH activity) at 0.1, 1, and 10 µg/mL respectively, in BT-Med + BT-TI group as compared to the UT-Med + UT-TI group. The lactate dehydrogenase (LDH) has been reported mainly for anaerobic respiration of cells 50, 51, 52 and found in mostly the heart and skeletal muscle, which are responsible for various conditions such as tissue injury, necrosis, hypoxia, hemolysis or malignancies. Thus, the overall data found that there was a significant reduction of LDH level after Biofield Energy Treatment and protect HCF cells, which might be helpful to resist against various pathological conditions. This Biofield Energy Treated test formulation would be the best alternative method for improved cellular function of heart under any type of oxidative stressful situations.
Figure 3. The effect of the test formulation on the percent protection of HCF cells in terms of decreased lactate dehydrogenase (LDH) activity against tert-butyl hydroperoxide (t-BHP) induced damage. TMZ: Trimetazidine; UT: Untreated; Med: Medium; BT: Biofield Treated; TI: Test item.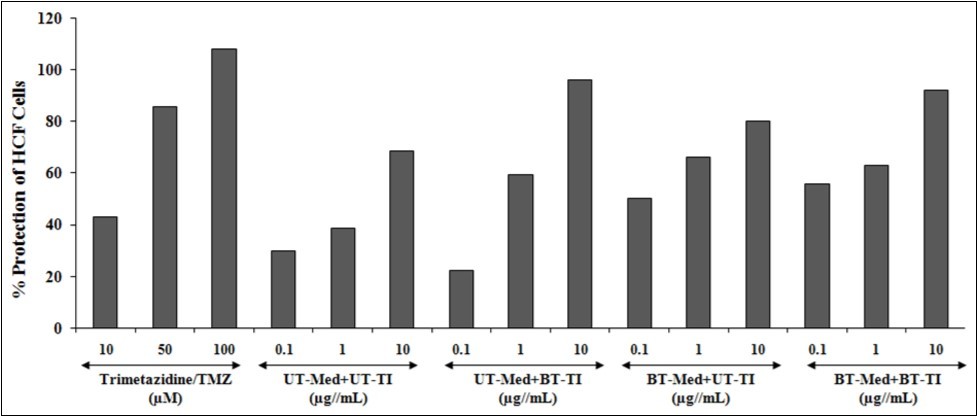
Estimation of Alanine Amino Transferase (ALT) Activity in HepG2 Cells
Alanine amino transferase (ALT) activity of the test formulation was evaluated on the protection of HepG2 cells in terms of decrease ALT activity is shown in Figure 4. The positive control, silymarin was selected in ALT activity and the data suggested increased percentage cellular protection (decreased ALT activity) by 40.2%, 63.4%, and 103.7% at 5, 10, and 25 µg/mL concentrations respectively. Similarly, the test formulation groups showed improved cellular protection of HepG2 cells (decreased of ALT activity) by 33.5%, 12%, and 24.5% at 10, 25.5, and 63.8 µg/mL respectively, in the UT-Med + BT-TI group, while increased cellular protection of HepG2 cells (decreased of ALT activity) by 30.4%, 63.2%, 40.9%, and 44.6% at 1, 10, 25.5, and 63.8 µg/mL respectively, in the BT-Med + UT-TI group, and increased cellular protection of HepG2 cells (decreased of ALT activity) by 99.2%, 49.6%, and 38.7% at 10, 25.5, and 63.8 µg/mL respectively, in the BT-Med + BT-TI group as compared to the UT-Med + UT-TI group (Figure 4). ALT is one of the enzyme exist mostly in liver and kidney, while largest internal organ of the human body is liver and maintain its health would control the overall quality of life. Thus, ALT would directly reflect the hepatocellular injury and death 53. Increased hepatoprotective effect was suggested by enhanced viability of cells against oxidative damage, while ALT increased level displays the chance of getting liver disorders 54. Therefore, the experimental data suggested Biofield Energy Treatment significantly protects the liver hepatocytes with reduced ALT enzyme as compared to the t-BHP inducing group.
Figure 4. Effect of the test formulation on the percent protection of human liver cancer (HepG2) cells in terms of decreased alanine amino transaminase (ALT) activity under the stimulation of tert-butyl hydroperoxide (t-BHP). UT: Untreated; Med: Medium; BT: Biofield Treated; TI: Test item.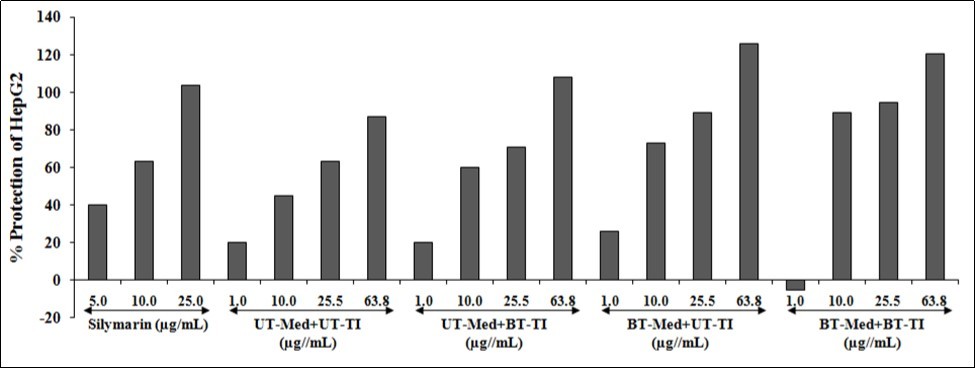
Estimation of Superoxide Dismutase (SOD) Activity in Adenocarcinomic Human Alveolar Basal Epithelial Cells (A549)
SOD activity was estimated in A549 cells and improved activity represents increased cellular protection and the data was presented in Figure 5. The positive control, quercetin showed improved percentage increase in the SOD activity with respect to the t-BHP by 62.1%, 80.3%, and 93.9% at 10, 25, and 50 µg/mL respectively. However, the percent protection of A549 (lungs) cells (increased of SOD activity) was significantly increased by 39.8%, 38.8%, and 17.4% at 10, 25.5, and 63.8 µg/mL respectively, in the UT-Med + BT-TI group, while increased SOD activity by 43.2%, 44%, and 5.1% at 10, 25.5, and 63.8 µg/mL respectively, in the BT-Med + UT-TI group, and increased SOD activity by 3.2%, 59.7%, and 10.3% at 10, 25.5, and 63.8 µg/mL respectively, in the BT-Med + BT-TI group as compared to the UT-Med + UT-TI group (Figure 5). Oxidative stress occurred due to many factors and results in severe damage, which play a pivotal role in case of ageing, many degenerative diseases and cancer. However, it has been reported that various factors such as endogenously or exogenously such as air pollutants, cigarette smoke, etc. were exposed to the lungs causing respiratory illness and other inflammatory disorders. SOD is one of the important antioxidant defense mechanism and also a powerful anti-inflammatory. However, their SOD enzyme based supplement significantly reduced the ROS generation, oxidative stress and also inhibits the endothelial activation 55. The experimental data showed that Biofield Energy Treatment has the significant capacity to improve the level of SOD enzyme that would be highly significant to resist against various pathological respiratory conditions like oxidative stress and related adverse effect.
Figure 5. Effect of the test formulation on the percent protection of lungs cells (A549) in terms of increased SOD activity under the stimulation of tert-butyl hydroperoxide (t-BHP). UT: Untreated; Med: Medium; BT: Biofield Treated; TI: Test item.
Effect of Test Formulation on Serotonin in Human Neuroblastoma (SH-SY5Y) Cells
The effect of test formulation on serotonin level was assessed in SH-SY5Y cells after 24 hours of treatment by ELISA and the results are presented in Figure 6. The positive control, curcumin showed 96.1%, 137.2%, and 169.6% increased the level of serotonin at 0.1, 1, and 5 µg/mL respectively, compared to the vehicle control (VC) group. The experimental test groups showed significant increased serotonin by 59.2% and 10.3% at 0.1 and 10 µg/mL respectively, in the UT-Med + BT-TI, while significant increased serotonin by 190.3%, 51.4%, and 20.9% at 0.1, 1, and 10 µg/mL respectively, in the BT-Med + UT-TI, and BT-Med + BT-TI group showed significant increased serotonin by 74.3%, 201%, and 120.8% at 0.1, 1, and 10 µg/mL respectively as compared to the UT-Med + UT-TI group (Figure 6). 5-HT has been found in the brain, bowels, and blood platelets, while it act as a transmit messages among the nerve cells, it is supposed to be active in the constricting smooth muscles, as it helps in wellbeing and happiness. Serotonin role has been well defined in the many neuronal functions like sleep, feeding, pain, sexual behavior, cardiac regulation, and cognition. Various neuropsychiatric disorders were reported due to serotonin imbalance such as depression, Alzheimer's disease, loss of ability of thinking, memory loss, cognitive health, etc. 56. Overall, the data suggested that Biofield Energy Healing Treated test formulation significantly improved the serotonin level, which would be highly useful against various neurodegenerative diseases and improved brain functioning.
Figure 6. Effect of the test formulation on percent increase in 5-hydroxy tryptamine (5-HT) or serotonin in human neuroblastoma cells (SH-SY5Y). UT: Untreated; Med: Medium; BT: Biofield Treated; TI: Test item
Effect of Test Formulation on Vitamin D Receptors (VDRs)
Human bone osteosarcoma cells (MG-63) were used for the estimation of VDR activity after treatment with the test formulation in different experimental groups. Vitamin D receptors expression and activity of the test formulation was determined using quantitative-polymerase chain reaction (qPCR) amplification. Using real time PCR, different VDR-relative threshold cycle (VDR-CT) values were obtained after complete amplification cycles. Relative quantification (RQ) was calculated from the VDR-CT and house-keeping (HK)-CT values in MG-63 cells. The values after treated with the test formulation and positive control are represented in Figure 7. The RQ of VDR was increased in a concentration-dependent manner by 59.1%, 93.2%, and 131.3% in positive control group (calcitriol) at 1, 10, and 100 nM, respectively. The experimental test groups showed increased RQ of VDR by 37.2% and 159.1% in the UT-Med + BT-TI group at 10 and 50 µg/mL respectively, while 190.7%, 212.7%, 190.3%, and 164.1% at 0.1, 1, 10, and 50 µg/mL respectively, in the BT-Med + UT-TI group, and increased RQ of VDR by 60.2%, 93.5%, 278.3%, and 215.6% at 0.1, 1, 10, and 50 µg/mL respectively, in the BT-Med + BT-TI group as compared to the UT-Med + UT-TI group. Calcitriol, one of the active form of vitamin D (1, 25-dihydroxyvitamin D), which regulates the calcium homeostasis, immunity, and overall cellular growth and differentiation 57. In addition, the calcitriol acts via. VDR, as it regulates transcription the rates that are involved directly or indirectly in calcium and phosphate regulation 58, 59. The data suggested that VDR activity was significantly improved by Biofield Energy Treated test formulation that could be significantly useful to bind more active vitamin D3 metabolites and that ultimately can improve overall bone cell growth and development.
Figure 7. Effect of the test formulation on percent increase in relative quantification (RQ) of vitamin D receptors (VDRs) gene in human bone osteosarcoma cells (MG-63).UT: Untreated; Med: Medium; BT: Biofield Treated; TI: Test item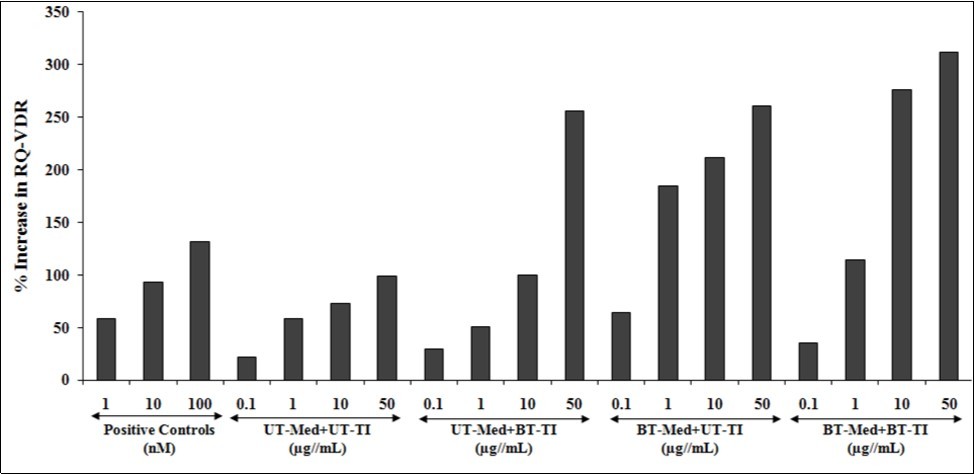
Conclusions
The experimental study outcomes showed that the test formulation treated with the Biofield Energy was found safe and non-toxic, which was tested using standard MTT cell viability assay in all the cell lines used for various activity. Cytoprotective activity was tested using human cardiac fibroblasts cells (HCF), which showed maximum restoration of cell viability by 94.4% at 1 µg/mL in UT-Med + BT-TI group, while in HepG2 cells the maximum restoration of cell viability was 84.4% at 10 µg/mL in the BT-Med + UT-TI group, and the test formulation in A549 cells showed maximum restoration of cell viability by 86.1% at 25 µg/mL in the BT-Med + BT-TI group as compared with the untreated test group. Similarly, ALP activity was significantly increased by 85.9%, 85.2%, and 87.4% at 10 µg/mL in the UT-Med + BT-TI, BT-Med + UT-TI, BT-Med + BT-TI groups respectively, as compared to the untreated test group in MG-63 cells, while 28%, and 59.2% increased ALP activity was found at 0.1 µg/mL in the BT-Med + UT-TI, BT-Med + BT-TI groups, respectively as compared to the untreated test groups in Ishikawa cells. Similarly, the LDH activity was decreased using increased percentage cellular protection data, which showed 53% and 40.5% increased protection at 1 and 10 µg/mL concentrations respectively in the UT-Med + BT-TI group, and 68.5%, 70.7%, and 16.8% improved cellular protection at 0.1, 1, and 10 µg/mL respectively in the BT-Med + UT-TI group, while 86.5%, 62.5%, and 34.2% improved cellular protection at 0.1, 1, and 10 µg/mL respectively, in BT-Med + BT-TI group as compared to the untreated test group. ALT activity was studied and data showed improved cellular protection of HepG2 cells (decreased of ALT activity) by 33.5%, 63.2%, and 99.2% at 10 µg/mL in UT-Med + BT-TI, BT-Med + UT-TI, and BT-Med + BT-TI groups respectively as compared with the untreated test group. SOD activity was significantly increased in A549 (lungs) cells by 39.8% (at 10 µg/mL), 44% (at 25.5 µg/mL), and 59.7% (at 25.5 µg/mL) in UT-Med + BT-TI, BT-Med + UT-TI, and BT-Med + BT-TI groups respectively as compared with the untreated test group. Serotonin level was significantly increased in SH-SY5Y cells by 59.2% (at 0.1 µg/mL), 190.3% (at 0.1 µg/mL), and 201% (at 1 µg/mL) in UT-Med + BT-TI, BT-Med + UT-TI, and BT-Med + BT-TI groups respectively as compared with the untreated test group. However, VDR expression was tested in MG-63 cells, which showed increased RQ of VDR by 37.2% and 159.1% in the UT-Med + BT-TI group at 10 and 50 µg/mL respectively, while 190.7%, 212.7%, 190.3%, and 164.1% at 0.1, 1, 10, and 50 µg/mL respectively, in the BT-Med + UT-TI group, and increased RQ of VDR by 60.2%, 93.5%, 278.3%, and 215.6% at 0.1, 1, 10, and 50 µg/mL respectively, in the BT-Med + BT-TI group as compared to the untreated test control group. Overall, it can be concluded that the Biofield Energy based test formulation would be the best alternative treatment strategy for improved functioning of heart, liver, bones, neuronal, and lungs parameters against any oxidative stress or damage induced by free radicals. Thus, results suggested that Biofield Energy Treatment can be used as a complementary and alternative treatment for the prevention of various types of cardiac disorders such as stroke, thromboembolic disease, congestive heart failure, congenital heart disease, peripheral artery disease, rheumatic heart disease, valvular heart disease, and venous thrombosis, etc. Besides, it would also protect against many hepatic disorders (cirrhosis, liver cancer, hemochromatosis, Wilson disease), lungs disorders (asthma, chronic bronchitis, emphysema, cystic fibrosis, and pneumonia), and many immune system related disorders. In addition, this novel test formulation can also be utilized for organ transplants (i.e., kidney, liver, and heart transplants), hormonal imbalance, aging, and various inflammatory and immune-related disease conditions like Alzheimer’s Disease (AD), Ulcerative Colitis (UC), Dermatitis, Asthma, Irritable Bowel Syndrome (IBS), Hashimoto Thyroiditis, Multiple Sclerosis, Aplastic Anemia, Graves’ Disease, Diabetes, Parkinson’s Disease, Myasthenia Gravis, Atherosclerosis, etc. to improve overall health and Quality of Life.
Acknowledgements
Authors gratefully acknowledged to Trivedi Global, Inc., Trivedi Science, and Trivedi Master Wellness for their support. In addition, authors are thankful for the support of Dabur Research Foundation for conducting this study.