Abstract
A previously healthy 25 year old Chinese male presented with left eye blurring of vision and was diagnosed to have left eye branch retinal vein occlusion. Initial blood investigations and thrombophilia screen were negative. The patient subsequently improved with observation and conservative management, with no further events over a 2 year follow up period.
The blood investigations were repeated 2 years later as part of a health check-up and he was then tested to be heterozygous for the factor V leiden mutation. This was confirmed by sequencing of his genome that identified the mutation. The laboratory was contacted to provide details regarding the testing methods and was noted to have performed the two tests via different methods.
While false negative rates in genetic testing are low, we believe that there is greater need to standardize testing methods as ascertaining genetic conditions play a great role in clinical diagnosis, treatment and prognosis. Clinicians should be aware of the limitations of these tests. When clinical suspicion is high, there may be a role for repeat tests with different methods or in different laboratories.
Author Contributions
Academic Editor: Zheng Jiang, Johns Hopkins University School of Medicine
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2015 Yan Tong Koh,et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Retinal vein occlusion (RVO) is a common ocular disease that results in acute visual loss. It is multifactorial in origin; while its exact mechanism and pathogenesis remains unclear, conditions like glaucoma, hypertension, arteriosclerosis and diabetes mellitus are commonly known associations with RVO especially in the older population. These conditions contribute to RVO either by affecting flow in the vasculature or causing abnormalities in vessel walls.1, 2, 3, 4, 5 As part of Virchow’s triad, thrombophilic conditions also increase the risk of thrombosis. 6, 7 Examples of such conditions include resistance to activated protein C (APC) or factor V Leiden, hyperhomocysteinaemia, and deficiencies in the anticoagulation system (protein C, protein S, or antithrombin).
In the normal coagulation cascade, protein C is a major component in anticoagulation mechanisms, as activated protein C (APC) inactivates highly procoagulant factors in the generation of thrombin such as activated factor V (factor Va) and factor VIII (factor VIIIa). Activated factor V is normally fully inactivated by an initial cleavage of a peptide bond on the carboxyl side of Arg506 followed by a second cleavage at Arg306. Subsequent cleavage also occurs at Arg679. [8] In the setting of factor V leiden mutation, there is a 1691 G to A mutation in the factor V gene, leading to an R506Q substitution in the factor V protein. This results in inability of cleavage at Arg506, which results in reduced cleavage at the Arg306 site. This results in factor V resistance to activated protein C (APC) inactivation and therefore increases risks of thrombosis. While it is the most common inherited thrombophilic condition, it is less commonly seen in the Asia.[9] The presence of factor V Leiden predisposes a genetic risk for thrombosis, which is mainly venous. [10] Heterozygous mutation increases the risk of thrombosis 3- to 7-fold, while homozygous mutation increases the risk to approximately 80-fold. [11] It however, remains debatable whether these thrombophilic conditions are aetiological factors for RVO as studies have shown conflicting conclusions.[12]
Currently, there are various ways to test for the presence of factor V Leiden in the laboratory, and there is lack of agreement on the best laboratory method for its detection. Previous studies have demonstrated the possibility of false negative results in factor V leiden testing, and that different test methods can give rise to different results. [13]
Here, we report a case report on a young male with branch retinal vein occlusion (BRVO) that initially tested negative for factor V leiden, but on a different test method, turned out to have a heterozygous mutation.
Case Report
A 25 year-old healthy Chinese male presented with blurring of vision of both eyes, affecting the left more than the right, of a few days duration. It was not associated with any trauma or injury, and there were no associated headaches, giddiness, floaters, fever, chills or rigors. He had no medical history of glaucoma, vascular risk factors like hypertension, hyperlipidemia, diabetes mellitus, smoking, as well as autoimmune conditions which can predispose to vasculitis. He had no personal or family history of thrombotic tendencies.
On examination, distant Snellen visual acuity was 6/6 unaided bilaterally. Eyelids, conjunctiva, cornea, anterior chamber, pupils, lens and extraocular range of motion were normal. Assessment of the posterior segment showed inferior BRVO of the left eye (Figure 1). Figure 1 shows dilatation and tortuosity of the affected venous segment, with flame-shaped and dot/blot haemorrhages, retinal edema and cotton wool spots in the section of the retina drained by the obstructed vein. Humphrey visual field testing showed no abnormalities. Fundus flourescein angiography showed features suggestive of left branch retinal vein occlusion (BRVO) with delay in the venous phase. There was no significant capillary fallout.
Figure 1. The figure shows evidence of BRVO in the left eye. There is dilatation and tortuosity of the affected venous segment, with flame-shaped and dot/blot haemorrhages, retinal edema and cotton wool spots in the section of the retina drained by the obstructed vein.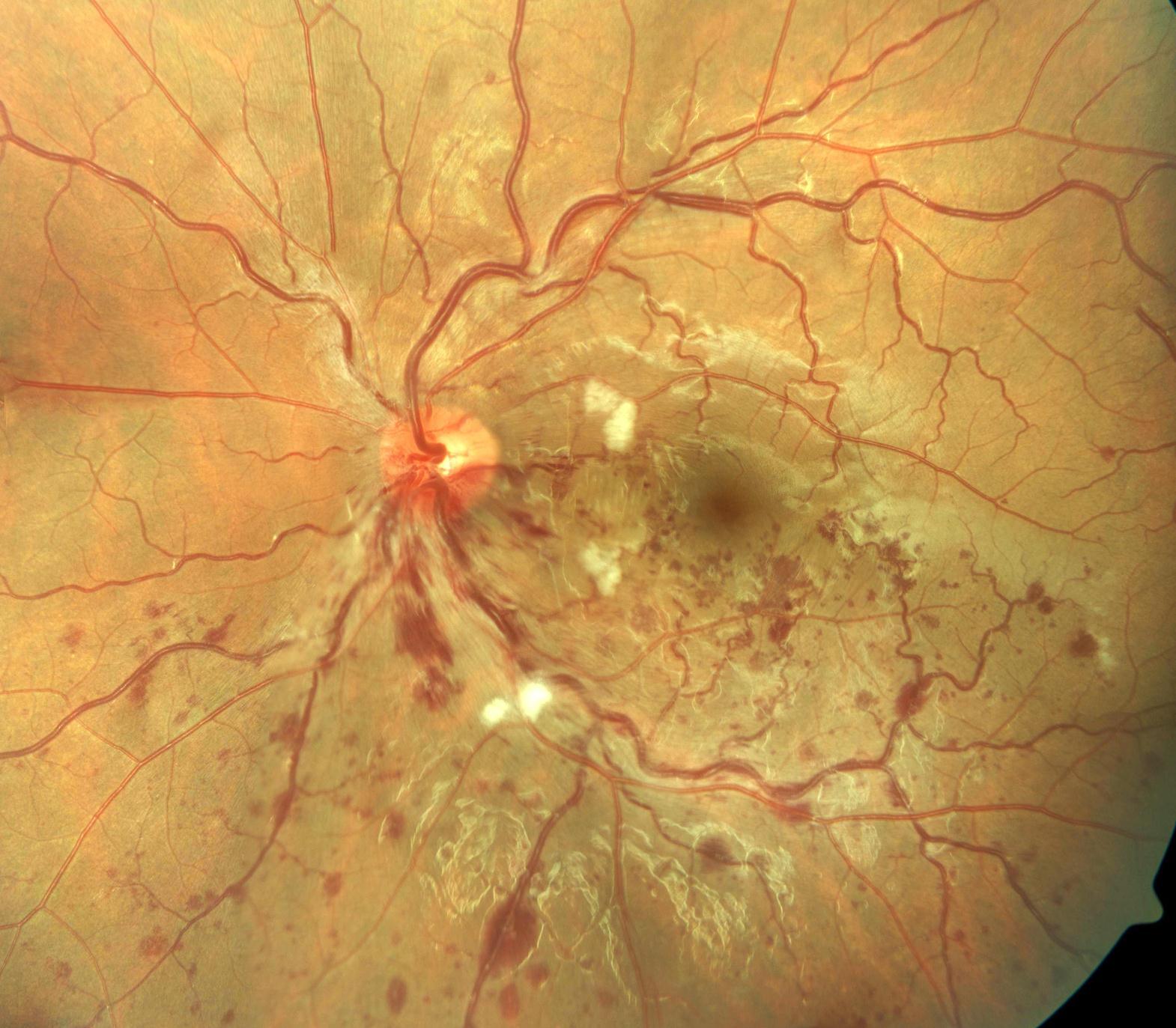
Initial investigations done included a full blood count, prothrombin time (PT), activated partial thromboplastin time (APTT), erythrocyte sedimentation rate, c-reactive protein, protein C, protein S, anticardiolipin antibody, antilupus anticoagulant, antinuclear antibody, anti-dsDNA antibody, rheumatoid factor, syphilis screen, homocysteine, anti-thrombin III, factor V Leiden, fasting lipids and plasma glucose. All venous blood samples were drawn on the same day and sent to the hospital laboratory for processing. The above investigations were negative. A cardiology referral was made to evaluate for any sources of cardiac emboli, which was negative. A haematology referral was also made for advice regarding the need to start on anti-platelets or anti-coagulation to prevent the onset of RVO in the other eye, and decision was made to hold off such medications as no definite prothrombotic etiology was established. The patient was managed conservatively with observation and his blurring of vision remained stable, with collateral formation noted within one year. There was no further development of RVO for a follow up of 2 year time period.
Two years later, as part of a health check-up, the above tests were repeated to confirm the absence of any pro-thombotic tendencies, and blood samples were sent to the same laboratory for processing. The patient was reported to have a heterozygous factor V leiden mutation, a result discordant from the previous test. The laboratory was contacted and asked to review the results of the earlier test and to describe the technical methods used.
In the first test performed two years ago, an allele specific polymerase chain reaction (PCR) targeting the mutant allele followed by gel electrophoresis test method was used. The patient was reported to be negative for the mutation. The original sample was no longer available for retesting.
In the second test, a different method was used. Deoxyribonucleic acid (DNA) was extracted from the peripheral blood sample and the factor V Leiden gene mutation was identified by PCR amplification followed by restriction endonuclease (Mn/l) digestion of the PCR product. The presence of the factor V leiden mutation is shown by the absence of an Mn/l site at position 1691, and the amplification by PCR allows heterozygosity or homozygosity to be ascertained. In this patient, he was found to be heterozygous for the mutation. Subsequent DNA sequencing identified the 1691 G to A mutation and confirmed the presence of factor V leiden mutation in our patient.
Discussion
In Factor V Leiden, a point mutation in factor V renders protein C resistant to the normal inactivation by activated protein C, thereby producing a mild thrombophilic state. While inherited coagulation disorders are established risk factors for thrombosis, the potential impact of these on retinal vasculature occlusive diseases remains unclear.12 Further, there is suggestion that the presence of factor V Leiden affects the prognosis of RVO, as its presence seems to enhance the risk of developing neovascular complications at least over a one year period. In a study by Hvarfner et al, there was an almost threefold risk of developing neovascular complications after central retinal vein occlusion (CRVO) with factor V leiden mutation present.14 Currently, there are currently no studies in the literature on the risk of neovascularisation with regards to BRVO with factor V leiden mutation. Our case report is the first that addresses this. The FFA in our patient did not show significant capillary fallout to warrant sectoral pan-retinal photocoagulation and during his 2 year follow-up, no neovascularisation was noted. This can suggest that factor V leiden mutation in the setting of BRVO might not predispose to increased risk of neovascularisation compared to CRVO. Until this has been studied with a larger population, the presence of factor V leiden mutation should still prompt closer follow-up intervals and more aggressive evaluation with a fundal fluorescein angiogram (FFA) in all such patients to evaluate the extent of retinal ischemia. Given that there is usually an interval of a few months before the retinal haemorrhages resolve to allow a FFA to be performed, early diagnosis of factor V leiden mutation will be useful in patient counseling so as to educate them on the utility on the FFA and need for closer follow-ups.
Through this case, we hope to highlight the importance of screening for thromphilic conditions in patients with retinal vascular occlusive disease, especially in those who are young and without any cardiovascular risk factors. Other than establishing a cause, such thrombophilic conditions may affect prognosis and therefore affect the frequency of followup and allow for more patient-specific therapy.
A spectrum of different approaches is used to detect mutations in clinical laboratories, and can give rise to sometimes contradictory results. A consensus statement published by the American College of Medical Genetics in 2001 does not recommend a specific technical approach for testing.[15] Previous publications have reported a 95-100% accuracy in detecting the heterozygous factor V leiden mutation, and there is a lack of standardization for genetic testing among laboratories16,17 Quality assurance publications in several countries have also demonstrated discordant test results and failure to detect the factor V leiden mutation in approximately 3-6%.17,18 In the literature, there are two prior case reports of discordant factor V leiden test results; one involved a patient whose blood genotype changed after stem cell transplant for acute myelogenous leukaemia [19], and the other could not be attributable to any definite cause.[12] In our patient, we believe that the discordance arose from a different in laboratory testing methods. The first PCR performed did not involve the use of the restrictive enzyme digestion and was therefore unable to identify the absence of an Mn/l site at position 1691. The second PCR was able to elucidate this, and therefore identify the 1691 G to A mutation suggestive of Factor V leiden mutation.
Further work should advance towards standardizing such genetic tests, in view of the implications that they have on diagnosis, management and prognosis. Other possible reasons for the discordant result include sample misidentification or errors in result interpretation.
Therefore, clinicians should be aware of the limitations of testing methods and should not accord genetic testing as fool-proof as they are also subject to errors in technique and interpretation. Repeat confirmatory testing may be appropriate if clinical suspicion is high, and can be performed with different test methods in a different laboratory.