Efficacy of Phytochemical Constituents of Castor Essential oil Towards the Mucor-Mycotic Mold Cunninghamella Bertholletiae
Abstract
The aim of this experiment is to study the efficacy of phytochemical constituents of Castor essential oil towards the mucor-mycotic mold Cunninghamella bertholletiae.The standard chemical analytical methods were used for the rapid study of the phytochemical constituents responsible for the antimicrobial efficacy of the procured castor essential oil. The standard antimicrobial assay technique employed to study the comparative values of the efficacy of the procured castor essential oil with that of the standard antifungal chemical agents against the clinical isolates obtained from the immune suppressed patients samples of Cunninghamella bertholletia mold mucor-mycotic infections. The best susceptibility values recorded in the standard antifungal agents against the clinical isolates of Cunninghamella bertholletiae was with Amphotericin B showing the average zone of inhibition diameter of 20.66 mm with the average MIC value of, 1.66 (µ/ml) but the antimicrobial assay results for the Castor essential oil showed better values with an average disc diffusion of 22.44mm zone of inhibition diameter with average MIC value of 1.72 µ/ml .This study has shown that the phytochemical compounds present in the Castor essential oil proves to be more an effective alternative antifungal substance towards the clinical isolates of Cunninghamella bertholletiae.
Author Contributions
Academic Editor: Zhe-Sheng Chenz, Professor, Department of Pharmaceutical Sciences, College of Pharmacy and Allied Health Professions, St. John’s University, United States.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2020 Muazzam Sheriff Maqbul, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
The mucor - mycosis infections one of the emerging potential dangerous infections among the immune-suppressed patients undertaking treatment for the leukemia, or diabetes with a high mortality rate ranging from 96% for disseminated mucor mycos,76% for pulmonary infections and 46% for synovial infections1, 2, 3. The mortality rate depends upon the condition of the patient and the site of infection. The mucor - mycosis was formerly known as zygomycosis which are rare fungal infections targeting the immuno compromised host among the humans 4, 5. In the recent past due to the abuse of antibiotic usage ignoring the constant alarming alert protocols of the WHO, these types of infections are paying way as a potential emerging threat to the human lives. The mucor-mycotic infections were due to the mucormycotic saprophytic fungal molds of the order Mucorales belonging to the family Mucaraceae6, 7, 8. The most common causative agents for these type of mucor –mycotic infections were species from the Mucor, Absida, Rhizopus and Cunninghamella. The rhinocerebral mucor-mycosis along with the cutaneous mucor- mycosis and the pulmonary mucor-mycosis were frequently found with the inhalation of spores of the Cunninghamella bertholletia mold which are heat resistant up to 500C posing a potential threats these opportunistic fungal infections are becoming common among the immune suppressed individuals9, 10. The spores of the mold Cunninghamella bertholletia been transported through the blood to the different parts of the human body causing the necrosis of the tissue among the immune suppressed patients. The infection of the eye ulceration or disfiguration of the face were also reported along with the gastrointestinal infections 1, 2, 3, 4, 5, 6, 7, 8. Though, the standard of antifungal-based therapy for Cunninghamella bertholletia infections is available for the treatment of this mold, only 33% of the recovery rate recorded. Hence, the need of the hour is to find an alternative potential prophylaxis therapy by revoking the forgotten ancient natural herbal medicine remedy to support the modern antifungal therapy in the treatment of the Cunninghamella bertholletia mold infections 5, 6, 7, 8, 9, 10. There are versatile of natural herbal products in the form of essential oils were used in the ancient medicine in the treatment of various dangerous pathogens of bacterial, fungal, and viral infections to a great results 11, 12, 13. This study is focused on the isolation and purification of the clinically procured aseptic samples of the fungal mold Cunninghamella bertholletia from the immune suppressed patients and to check its susceptibility with the efficacy of one such essential oils. The essential oil chosen for this study was Castor essential oil procured from the local market. The castor essential oil been extracted from the beans of the Ricinus communis belonging to the perennial flowering plant of Euphorbiaceae spurge family 14, 15, 16. The castor essential oil contains a rich source of phenolic compounds along with the other chemical constituents which possess to be a great antimicrobial agent14, 15, 16, 17, 18 and qualifies for this study. In this study the efficacy of the procured castor oil from the local market was tested against the clinical isolates obtained from the immune suppressed patients samples of Cunninghamella bertholletia mold mucor-mycotic infections 1, 2, 3, 4, 5, 6, 7, 8, 9, 10. The standard chemical analytical methods were used for the rapid study of the phytochemical constituents responsible for the antimicrobial efficacy of the procured castor essential oil. The standard antimicrobial assay technique employed to study the comparative values of the efficacy of the procured castor essential oil with that of the standard antifungal chemical agents against the clinical isolates obtained from the immune suppressed patients samples of Cunninghamella bertholletia mold mucor-mycotic infections 1, 2, 3, 4, 5, 6.
Materials and Methods
Materials
Castor essential oil procured from Jeddah local market, clinical skin scrape samples from the patient, Sabaraud's Dextrose agar, potassium hydroxide, peptone, lacto phenol. Standard antibiotics and standard Hi-Media were used. All the chemicals used during this investigation were of analytical grade.
Isolation and Purification of Cunninghamella Bertholletiae
The clinical sample from the patients was collected by employing the aseptic scraping technique and was inoculated on a sterile Sabaraud’s dextrose agar plate and incubated at 45ºC for 24-48 hours to observe the fungal mold rapidly growing white to tannish-gray color loose cottony colonies. The colonies mature upon further incubation for 96 hours 19, 20, 21. The high temperature incubation at 45ºC, the heat resistant Cunninghamella bertholletiae gets isolated from the other species of Cunninghamella which are all heat sensitive7, 8, 9, 10. The potassium hydroxide and lacto phenol test was performed to observe the mold hyphae under microscope by employing the wet mount technique as the confirmatory test. The microscopic observation reveals the sporangiophores, terminal vesicles with nonseptate or sparsely septate broad hyphae and the presence of oval shaped sporangioles with the sporangiospores with tuberculate projections4, 5, 6, 7, 8, 9, 10.
Antimicrobial Susceptibility Test
The antimicrobial susceptibility test for the isolated clinical specimens of Cunninghamella bertholletiae were evaluated for the efficacy of the standard synthetic chemical antifungal agents by performing the latest rapid e-test methodology where the clinical isolates were inoculated on Sabaraud’s dextrose agar plates separately and e-test plastic strips for the respective antibiotics were impregnated and incubated at 45ºC overnight to visualize the zone and ellipse and the results were tabulated by interpreting the observed results for the interaction of the ellipse as the Minimum inhibitory Concentration (MIC) whereas the zone as the susceptibility of the antibiotic towards the mold 3, 4, 5, 22. The traditional standard antibiotic assay methods such as Kirby-Bauer disc diffusion method was employed to observe the susceptibility of the clinical isolates of Cunninghamella bertholletiae the standard disc prepared from the Castor essential oil extract where the mold isolates were inoculated separately on the Sabaraud’s dextrose agar plates along with the impregnated discs for 24 hours at 45ºC to observe the zone formation determining the sensitivity of the mold towards the disc22, 23, 24, 25. The results were tabulated and interpreted. The MIC values along with Minimum Fungicidal Concentration (MFC) values for the efficacy antimicrobial activity of the Castor essential oil towards the mold was estimated by performing the standard tube dilution method where the clinical isolates were inoculated separately in the different sets of dilutions of the essential oil in the peptone water and incubated for 24 hours at 45ºC to observe the no turbidity determining the sensitivity of the mold towards the acid 22, 23, 24, 25. The last dilution with turbidity determines the MIC value of the acid towards the mold. The results were tabulated and interpreted. The MFC was determined by inoculating each dilution of MIC dilutions onto the separate agar plates for each clinical isolates of Cunninghamella bertholletiae for the MIC dilutions separately. The inoculated plates were incubated for 24 hours at 45ºC to observe the no growth determining the sensitivity of the mold towards the Castor essential oil. The first dilution with no growth determines the MFC of the essential oil towards the fungal mold. The results were tabulated interpreted.
Phyto-Chemical Analysis of the Castor Essential Oil
Biochemical Analysis for Alkaloids
Mayer’s Test
Castor essential oils were mixed with a drop of mercuric chloride and potassium iodide respectively resulting in the formation of a creamy substance indicating the presence of alkaloids.
Wagner’s Test
A drop of Castor essential oil was mixed with a drop of potassium iodide and iodine resulting in the formation of a reddish brown precipitate which indicates the presence of alkaloids in the oil.
Biochemical Analysis for Reducing Sugar
Fehling’s Test
2 ml of Fehling's reagents A and B were mixed with the Castor essential oil in a test tube and heated slightly to observe brick red color indicating the presence of reducing sugar.
Benedict’s Test
In a test tube 2 ml of Benedict’s reagent was treated with the Castor essential oil and heated gently heated to observe the formation of orange red precipitate indicates the presence of reducing sugar
Biochemical Analysis for Steroids
Salkowski’s Test
TheCastor essential oils were mixed with 2ml of chloroform along with 2 ml of concentrated sulphuric acid in a test tube and gently shaken to observe a reddish brown color which indicates the presence of steroids.
Chloroform and Sulphuric Acid with Acetic Acid Mixture Test
The Castor essential oil was treated with a mixture of 2ml of chloroform and concentrated sulphuric acid with 2 ml of acetic acid resulting in the formation of a green colour which indicates the presence of steroids
Biochemical Analysis for Proteins
Ninhydrin Test
The Castor essential oils were mixed with 2 ml of ninhydrin solution and heated gently to observe a violet color indicating the presence of protein
Xanthoproteic Test
The Castor essential oils were treated with a few drops of concentrated nitric acid resulting in the formation of a yellow colour which indicates the presence of proteins
Biochemical Analysis for Phenol
Litmus Test
A drop of Castor essential oil was added to the blue litmus paper which turns red in color due to acidic nature indicating the presence of phenol.
Phthalein Dye Test
The Castor essential oils were treated with conc. sulfuric acid after heating with phthalic anhydride results in the formation of colorless condensation compound and the addition of dilute sodium hydroxide solution results in the formation of a pink color fluorescent compound which indicates the presence of phenol.
Ferric Chloride Test
Castor essential oils were boiled with 10 ml of water in a test tubes. A few drops of ferric chloride was added to the 10 ml of heated Castor essential oil in a test tube to observe a blue black coloration which indicates the presence of phenol
Biochemical Analysis for Glycosides
Libermann-Burchard’s Test
The mixture of 2 ml of acetic acid with 2 ml of chloroform was treated with the Castrol essential oil in a test tube and few drops of concentrated sulphuric acid was added by placing the test tube on ice to observe the color change from violet to bluish green which indicate the presence of glycosides
Keller-kilani Test
Castrol essential oil was treated with 2ml of glacial acetic acid with 1 to 2 drops of ferric chloride solution in a test tube and 2 ml of conc. sulphuric acid was added to observe a brown ring at the interface which indicates the presence of cardiac glycosides.
Biochemical Analysis for Amino Acids
Ammonia Test
Dilute ammonia and conc. sulphuric acid treated with aqueous Castrol essential oil in a test tube to observe yellowish color formation indicating presence of amino acids.
Biochemical Analysis for Flavonoids
Ammonia and H2SO4 Mixture Test
The Castor essential oil was treated with dilute ammonia and conc. sulphuric acid resulting in the formation of a yellow colour which indicates the presence of flavonoids.
Biochemical Analysis for Iodine
Iodine Test
The Castor essential oil was determined added with a 2ml of iodine solution which results in the positive purple colored test which indicates the presence of iodine.
Biochemical Analysis for Terpenoids
Chloroform and H2SO4 Mixture Test
The Castor essential oil was treated with 2ml of chloroform and concentrated sulphuric acid resulting in the formation of a brownish red layer which indicates the presence of terpenoids
Results and Discussion
A Comparative analysis study was performed for the antimicrobial efficacy of Castor essential oil extract with that of the standard antifungal agents towards the clinical isolates of Cunninghamella bertholletiae 3, 4, 5, 6, 7, 8. The antimicrobial assay results for the Castor essential oil procured from the local market shown significant antimicrobial activity results against all the clinical isolates of Cunninghamella bertholletiae with an average disc diffusion of 22.44.mm zone of inhibition diameter determining the susceptibility obtained from performing the Kirby-Bauer technique with an average MIC value of 1.72 µ/ml and an average MFC value of 2..30 µ/ml. The best susceptibility for the clinical isolates of Cunninghamella bertholletiae towards the Castor essential oil was observed from the throat swab isolates of Cunninghamella bertholletiae sample with a zone diffusion of 28 mm with MIC of 2.25 µ/ml and MFC of 2 .5 µ/ml whereas the least susceptibility was observed from the nail scrape isolates of Cunninghamella bertholletiae sample with a zone diffusion of 18 mm with MIC of 1.75 µ/ml and MFC of 2 µ/ml respectively 12, 13, 17. The susceptibility with MIC and MFC results of the other isolates of Cunninghamella bertholletiae sample towards the Castor essential oil were also shown satisfactory results when compared with that of the standard antifungal agents against clinical isolates of Cunninghamella bertholletiae. The results of the other isolates of Cunninghamella bertholletiae towards the Castor essential oil obtained were ranged for the susceptibility with a zone diameter from 18 to 28 mm in disc diffusion method with MIC from 1 .5 to 2.5 µ/ml and MFC of 1.75 to 2.75 µ/ml respectively. The efficacy of the Castor essential oil extract against clinical isolates of Cunninghamella bertholletiae has shown excellent results when compared with that of the standard antifungal agents used in therapy. The average value of the zone of inhibition susceptibility value of the Castor essential oil extract against clinical isolates of Cunninghamella bertholletiae was 22.44 mm for all the clinical isolates compared to the standard antifungal agents values of 14.88 mm for Voriconozole, 10.11 mm for Itraconazole , 20.66 mm for Amphotericin B, 11.11 mm for Fluconazole, , 16.22 mm for Posoconazole, 16.11 mm for Metronidazole, 16.55 mm for Ketoconazole and 10.44 mm for Rifampin respectively for all the samples assayed. The average MIC value of the Castor essential oil extract against clinical isolates of Cunninghamella bertholletiae was 1.72 µ/ml for all the samples compared to the standard antifungal agents values of 1.30 (µ/ml) for Voriconozole, 1.47 (µ/ml) for Itraconazole , 1.66 (µ/ml) for Amphotericin B, 1.75 (µ/ml) for Fluconazole, 1.94(µ/ml) for Posoconazole, 2.11 (µ/ml) for Metronidazole, 2.30 (µ/ml)for Ketoconazole and 2.41 (µ/ml) for Rifampin respectively for all the clinicals isolates of Cunninghamell abertholletiae samples assayed. The best susceptibility values recorded in the standard antifungal agents against the clinical isolates of Cunninghamella bertholletiae was with Amphotericin B showing the average zone of inhibition diameter of 20.66 mm with the average MIC value of , 1.66 (µ/ml) but the antimicrobial assay results for the Castor essential oil showed better values with an average disc diffusion of 22.44mm zone of inhibition diameter with average MIC value of 1.72 µ/ml. The details of the obtained results were tabulated (Table 1, Table 2, Table 3) for the references. A detailed comparative analysis chart (Figure 1) was prepared for the antimicrobial activities of Castor essential oil extract with that of standard antifungal agents against clinical isolates of Cunninghamella bertholletiae for the references. The phytochemical analysis study was also been conducted for the procured Castor essential oil to determine the constituents which are responsible for the antimicrobial efficacy. The phytochemical analytical tests conducted for the Castor essential oil were as shown in Table 4. The obtained interpretation from the phytochemical analytical test results showed the presence of chemical compounds such as alkaloids, flavonoids, steroids, proteins, phenols, glycosides, reducing sugar, iodine, amino acids and terpenoids respectively. The phytochemical test results were tabulated for the reference (Table 4). The presence of the vital phytochemical component in the Castor essential oil is the phenolic compounds which serves as a potential antimicrobial activity and shown the promising results against the clinical isolates of Cunninghamella bertholletiae when demonstrated with the standard antimicrobial assay techniques14, 15, 16, 17. The presence of pheonolic and other miscellaneous constituents in the Castor essential oil extract contributes to its rich antimicrobial content and has shown the promising results in this study as well. Table 2, Table 3
Figure 1.Comparative analysis for the antimicrobial activities of Castor essential oil extract versus standard antifungal agents against clinical isolates of Cunninghamella bertholletiae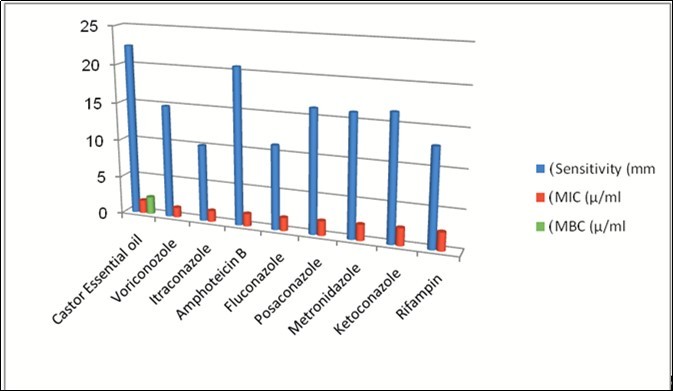
| Standard Antifungal agents | Specimens | Average Zone value | ||||||||
| Sinovialfluid | Nasal swab | Pleural effusion | Nail scrape | Abscess swab | Oral cavity | Throat swab | Ulcer swab | Wound swab | ||
| Voriconazole | 20mmS | 20mmS | 9 mmI | 11mmI | 25mmS | 22 mmS | 4mmR | 3mmR | 20mmR | 14.88 mm |
| Itraconazole | 12 mmI | 3mmR | 4mmR | 3mmR | 20mmR | 12mmI | 13mmI | 4mmR | 20 mmS | 10.11 mm |
| Amphotericin B | 25mmS | 14 mmI | 23 mmS | 26 mmS | 20 mmS | 22 mmS | 20mmS | 24 mmS | 12 mmI | 20.66 mm |
| Fluconazole | 10 mmI | 10mmI | 21 mmS | 11mmI | 9mmI | 13mmI | 12mmI | 12mmI | 2mmR | 11.11 mm |
| Posaconazole | 20mmS | 20 mmS | 9 mmI | 11mmI | 21mmS | 11mmI | 23mmS | 21 mmS | 11mmI | 16.22mm |
| Metronidazole | 11mmI | 21 mmS | 10mmI | 21 mmS | 20mmS | 20mmS | 9mmI | 11mmI | 22 mmS | 16.11mm |
| Ketoconazole | 14 mmI | 2 mmR | 11mmI | 21 mmS | 20mmS | 21mmS | 20mmS | 10 mmI | 20 mmS | 16.55 mm |
| Rifampin | 6mmR | 4 mmR | 12mmI | 23mmS | 6mmR | 4mmR | 4mmR | 14 mmI | 12 mmI | 10.44 mm |
| Total Sensitives | 3 | 3 | 2 | 4 | 5 | 4 | 3 | 2 | 3 | |
| Total Intermediates | 4 | 2 | 5 | 3 | 1 | 3 | 3 | 4 | 3 | |
| Total Resistance | 1 | 3 | 1 | 1 | 2 | 1 | 2 | 2 | 2 | |
| Standard Antifungal agents | Specimens | Average MIC Values | ||||||||
| SinovialFluid | Nasal Swab | Pleural Effusion | Nail Scrape | Abscess swab | Oral cavity | Throat swab | Ulcer swab | Wound swab | ||
| Voriconazole | 1.5 µ/ml | 1.25µ/ml | 1.75µ/ml | 1.25 µ/ml | 1.75 (µ/ml) | 1.5 µ/ml | 1.25 µ/ml | 1.5(µ/ml) | 1.25µ/ml | 1.30(µ/ml) |
| Itraconazole | 1.5 µ/ml | 1.25 µ/ml | 1.75 µ/ml | 1.25 µ/ml | 1.75 (µ/ml) | 1.5 µ/ml | 1.5 µ/ml | 1.5 (µ/ml) | 1.25 µ/ml | 1.47(µ/ml) |
| Amphotericin B | 1.75 µ/ml | 1.5 µ/ml | 1.75 µ/ml | 1.5 µ/ml | 2 µ/ml | 1.75 µ/ml | 1.5 µ/ml | 1.75(µ/ml) | 1.5 µ/ml | 1.66(µ/ml) |
| Fluconazole | 1.75 µ/ml | 1.5 µ/ml | 2 µ/ml | 1.5 µ/ml | 2.25 µ/ml | 1.75 µ/ml | 1.75 µ/ml | 1.75 (µ/ml) | 1.5 µ/ml | 1.75(µ/ml) |
| Posaconazole | 1.75 µ/ml | 1.75 µ/ml | 2.25 µ/ml | 1.75 µ/ml | 2.25 µ/ml | 2 µ/ml | 2 µ/ml | 2 (µ/ml) | 1.75 µ/ml | 1.94(µ/ml) |
| Metronidazole | 2 µ/ml | 2. µ/ml | 2.25 µ/ml | 1.75 µ/ml | 2.5 µ/ml | 2µ/ml | 2.25 µ/ml | 2.25 (µ/ml) | 2. µ/ml | 2.11(µ/ml) |
| Ketoconazole | 2.25 µ/ml | 2.25 µ/ml | 2.5 µ/ml | 2 µ/ml | 2.75 µ/ml | 2.µ/ml | 2.25 µ/ml | 2.5 (µ/ml) | 2.25 µ/ml | 2.30(µ/ml) |
| Rifampin | 2.25 µ/ml | 2.25 µ/ml | 2.5 µ/ml | 2 µ/ml | 2.75 µ/ml | 2.25 µ/ml | 2.5 µ/ml | 2.75 (µ/ml) | 2.5 µ/ml | 2.41(µ/ml) |
| Castor essential oil extract | |||
| Specimen | Disc Diffusion | MIC | MFC |
| Sinovialfluid | 19 mm S | 1.5 µ/ml | 1.75 µ/ml |
| Nasal swab | 25 mmS | 1.5 µ/ml | 1.75 µ/ml |
| Pleural effusion | 23 mmS | 1.75 µ/ml | 2 µ/ml |
| Nail scrape | 18 mmS | 1.75 µ/ml | 2 µ/ml |
| Abscess swab | 21 mmS | 2 µ/ml | 2.25µ/ml |
| Oral cavity | 25 mmS | 2 µ/ml | 2.25 µ/ml |
| Throat swab | 28 mmS | 2.25µ/ml | 2.5 µ/ml |
| Ulcer swab | 19 mmS | 2..25µ/ml | 2.5 µ/ml |
| Wound swab | 24 mmS | 2.5 µ/ml | 2.75 µ/ml |
| Average value | 22.44mmS | 1.72 µ/ml | 2.30µ/ml |
| Biochemical Analysis | Observed Result | Phytochemical constituents present |
| Mayer’sTest | Creamy Substance | Alkaloids. |
| Wagner’s test | Reddish brown precipitate | Alkaloids. |
| Fehling’sTest | Brick red color | Reducing sugar |
| Benedict’s Test | Orange red precipitate | Reducing sugar |
| Salkowski’sTest | Brown color | Steroids |
| Chloroform and Sulphuric acid with Acetic Acid mixture Test | Green color | Steroids |
| Ninhydrin | Violet color | Proteins |
| Xanthoproteic Test | Yellow colour | Proteins |
| Litmus Test: | Red color | Phenol |
| Phthalein Dye Test | Pink fluorescent compound | Phenol |
| Ferric chloride test | Blackish blue color | Phenol |
| Libermann-Burchard’sTest | Bluish green color | Cardiac glycosides |
| Keller-kilani test | Brown colored ring | Glycosides |
| Ammonia test | yellow colour | Amino acids |
| Ammonia and Sulphuric acid mixture Test | yellow colour | Flavonoids |
| Iodine Test | Purple color | Iodine |
| Chloroform and Sulphuric acid mixture Test | brownish red layer | Terpenoids |
Conclusion
The phytochemical compounds present in the Castor essential oil acts as an effective remedy towards the the clinical isolates of Cunninghamella bertholletiae compared to the standard antifungal agents. The interpretation of the observation and results for the Castor essential oil showed the promising study results regarding its efficacy as a potential antifungal agents when compared to that of the standard synthetic chemical agents used against the clinical isolates of Cunninghamella bertholletiae. This study recommends for more such of natural essential oils from the plant source as an alternative towards the synthetic chemical antimicrobial substances with more detailed studies need to be done in near future with the expectations that many dangerous infections can be cured with these types of phytochemical compounds. Thus, this study has shown that the phytochemical compounds present in the Castor essential oil proves to be more an effective alternative antifungal substance towards the clinical isolates of Cunninghamella bertholletiae.
Funding
None
Acknowledgements
We would like to acknowledge Ibn Sina National College, Jeddah, Kingdom of Saudi Arabia, for their constant support.
References
- 1.Rickerts V, Bohme A, Viertel A. (2000) Cluster of pulmonary infections caused by Cunningham Ella bertholletiae in immunocompromised patients,” Clinical Infectious Diseases. 31(4), 910-913.
- 2.D P Kontoyianis, Vartivarian S, E J Anaissie, Samonis G, G P Bodey et al. (1994) Infections due to Cunninghamellabertholletiae in patients with cancer: report of three cases and review,” Clinical Infectious Diseases. 18(6), 925-928.
- 3.Lemmer K, Losert H, Rickerts V. (2002) Molecular biological identification of Cunninghamella spec,”. , Mycoses 45(1), 31-36.
- 4.J A Ribes, C L Vanover-Sams, D J Baker. (2000) . Zygomycetes in human disease,” , Clinical Microbiology Reviews 13(2), 236-301.
- 5.Alvarez E, D A Sutton, Cano J. (2009) Spectrum of zygomycete species identified in clinically significant specimens in the United States,”. , Journal of Clinical Microbiology 47(6), 1650-1656.
- 6.Gomes M Z R, R E Lewis, D P Kontoyiannis. (2011) Mucormycosis caused by unusual mucormycetes, non-Rhizopus. , Mucor, and Lichtheimia species,” Clinical Microbiology Reviews 24(2), 411-445.
- 7.Motohashi K, Ito S, Hagihara M, Maruta A, Ishigatsubo Y et al. (2009) Cutaneous zygomycosis caused by Cunninghamellabertholletiae in a patient with chronic myelogenous leukemia in blast crisis,”. , American Journal of Hematology 84(7), 447-448.
- 8.M R, D H Walker, I E Dominy, Kaplan W. (1982) Zygomycosis caused by Cunninghamellabertholletiae: clinical and pathologic aspects,”. , Archives of Pathology & Laboratory Medicine 106(6), 282-286.
- 9.Zeilender S, Drenning D, F L Glauser, Bechard D. (1990) Fatal Cunninghamellabertholletiae infection in an immunocompetent patient. , Chest 97(6), 1482-1483.
- 10.A E Reed, B A, M B Austin, Jr H F Frierson. (1988) Cunninghamellabertholletiae and Pneumocystis carinii pneumonia as a fatal complicationof chronic lymphocytic leukemia,”. , Human 19(12), 1470-1472.
- 11.Ilavarasan R, Mallika M, Venkataraman S. (2006) Anti-inflammatory and free radical scavenging activity of Ricinuscommunis root extract. , J. Ethnopharm 103, 478-480.
- 12.Sandhyakumary K, Bobby R G M. (2003) Antifertility effects of Ricinuscommunis (Linn) on rats. , Phytother. Res 17, 508-511.
- 13.Shokeen P, P Y Anand, Murali K, Tandon V. (2008) Antidiabetic activity of 50% ethanolic extract of Ricinuscommunis and its purified fractions. Food Chem. , Toxicol 46, 3458-3466.
- 14.Tyagi K, Sharma S, Rashmi R, Kumar S. (2013) Study of phyto-chemical constituents of Ricinuscommunis Linn. under the influence of industrial effluent. , J. Pharm. Res 6, 870-873.
- 15.Scarpa A, Guerci A. (1982) Various uses of the castor oil plant (Ricinuscommunis L.): a review. , J. Ethnopharm 5, 117-137.
- 16.Jena J, A K Gupta. (2012) Ricinus Communis Linn: A Phytopharmacological Review. , Int. J. Pharm. Sci 4, 25-29.
- 17.Abraham Z, S D Bhakuni, H S Garg, A K Goel, B N Mehrotra et al. (1986) Screening of Indian plants for biological activity. Part XII. , Indian J.Experimental Biology 24, 48-68.
- 18.U G Akpan, Jimoh A, A D Mohammed. (2006) Extraction, Characterization and Modification of castor seed oil. , Leonardo J. Sciences 8, 43-52.
- 19.Muazzam S M, Khan A A, Tasneem M, SMS Iqubal, Shaikh I A et al. (2020) . Determination of Antioxidant Properties and Antimicrobial activity of Vinyl Phenolic compounds extractedfrom Saccharomyces cerevisiae Against Uropathogenic bacteria. Orient J Chem 36(1), 26-32.
- 20.CMS Bisht, SMS Iqubal, Khan A A, Tasneem M, Dawoud A et al. (2019) . Natural Products in Drug Discovery: Antibacterial and Antifungal Activity of Essential Oil of Compound Isolated from Senecio royleanus , J Pure Appl Microbio 13(3), 1611-17.
- 21.Bagewadi Z K, Muddapur U M, Madiwal S S, Mulla S I, Khan A A. (2019) Biochemical and enzyme inhibitory attributes of methanolic leaf extract of Datura inoxia Mill. Enviromental Sustainability. 2, 75-87.
- 22.Muazzam S M, Alshabi A M, Khan A A, SMS Iqubal, Tasneem M et al. (2020) Comparison of e-test Values for Standard Antibiotics and Conventional Antimicrobial Assay Values for Ethanoic Acids against Nosocomial Multidrug resistant Pseudomonas aeruginosa. , J Pure Appl Microbio 14(1), 255-260.
- 23.Muazzam S M, HMB AlHasel, Majid D H, Momen T N, HAM AlHazmi et al. (2019) Iqubal SMS. Chemical Analysis (GC-FID-MS) and Antimicrobial Activity of Parmotrema perlatum Essential Oil Against Clinical Specimens. , Orient J Chem 35(6), 1695-1701.
- 24.Gouse B S, Muazzam S M, Gokul S S, Ranjith M S. (2017) Isolation and characterization of actinomycetes from soil of ad-dawadmi, Saudi Arabia and screening their antibacterial activities. , Int J of Pharm and Pharmaceutical Sc 9(10), 267-79.
- 25.Muazzam S M, Yumna A B, Samaher G B, Shaden N A, MMO Bashair et al. (2020) A Comparative Study of Different Types of Thyme Essential Oils Against Streptococcus pyogenes to Determine their Biochemical and Antimicrobial Properties. , Orient J Chem 36(2), 220-228.
