Drivers and Barriers to Medication Adherence in Patients with Chronic Myeloid Leukaemia: A Qualitative Study
Abstract
With the introduction of tyrosine kinase inhibitors (TKI), patients with chronic myeloid leukemia (CML) have obtained survival rates close to normal. It may appear paradoxical, then, that medication adherence is suboptimal in some health care settings. As the first of its kind, this study aimed to explore drivers and barriers to TKI treatment adherence in Danish CML patients. A literature study informed the design of qualitative interviews with 20 patients, individually and in focus groups, focusing on their disease perceptions of CML, their health-related quality of life (QoL) and medication adherence. The study showed that many participants had previously switched treatment due to lacking efficacy or intolerance but most felt their current disease burden was tolerable. Anxiety might, however, resurface if treatment stopped working or with the occurrence of infections or side effects, creating a state of ‘fragile peace’. To these patients, their role functioning – as professionals, spouses, parents and grandparents – was crucial to uphold a positive self-image and meaningful life. Whether treatment enabled or hindered this was thus decisive to their QoL and medication adherence. Our participants expressed high adherence rates with only one having intentionally non-adhered due to side effects and poor QoL. Most participants felt well-informed about CML and treatment and privileged to receive specialised personal care from the public health care system acting to motivate their medication adherence. As a novel finding, this study indicates that the prospect of treatment-free remission may positively affect ‘adherence’ suggest this should be explored in future studies.
Author Contributions
Academic Editor: Fernando Luiz Affonso, Fonseca Universidade Federal de São Paulo, UNIFESP
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2017 Gitte Lee Mortensen, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Chronic myeloid leukaemia(CML) constitutes approximately 15% of adult leukaemia cases. It affects slightly more men than women and has an average onset at 64-68 years. Most patients (60-85%) are diagnosed in the initial chronic phase (CP-CML) based on symptoms such as night sweats, abdominal pain, infections, fatigue, weight loss, or bruising 1, 2, 3. 15-40% are asymptomatic and often identified by routine blood tests. If untreated, CP-CML will progress to an accelerated phase (AP-CML) and a fatal blast phase (BP-CML) in three to five years 1, 4, 5.
With the introduction of the tyrosine kinase inhibitor (TKI) imatinib in 2001, CML patients have obtained revolutionary survival rates close to normal 6. 2nd generation TKIs dasatinib, nilotinib, and bosutinib have since been approved 7, followed by 3rd generation ponatinib 4, 8, 9. Some patients are initially treated with hydroxyurea 4. Prior to TKIs, patients could be treated with interferon-alpha (IFNa), and allogeneic stem-cell transplantation (SCT) remains an option with elevated mortality rates. As CML is a low-grade symptom disease, treatment often represents patients’ main disease impact 3, 10. Side effects are mostly considered mild to moderate but may include edema, diarrhoea, pleural effusions, nausea, muscosceletal or abdominal pain, rashes, fatigue, or headache 1, 2, 3, 4, 7, 9, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21.
“Three levels of TKI treatment response may be obtained: complete hematologic, cytogenetic, and molecular responses (CHR, CCyR, and CMR) reflecting successive decrease in leukemic cells 4. Efficacy is highest in CP-CML and certain milestone responses should occur within 3, 6, and 12 months after initiating treatment 4, 22. Treatment should be re-evaluated if milestones are not reached 23, 24. Response rates are deeper and faster with 2nd generation TKIs compared to imatinib, and patients with rapid response have better long-term outcomes than those with slower or no response to therapy 11, 13, 17, 20, 22. Yet imatinib is still the most widely used TKI due to cost-effectiveness and acceptable side effects. CML patients with long-lasting sustained CMR, amounting to 10-15% of patients, may be eligible for discontinuation 9, 12, 16, 18, 25.
Medication adherence, i.e. ‘the extent of conformity to the recommendations about day to day treatment by the provider with respect to timing, dosage and frequency’ 3 is crucial to achieve optimal treatment response 6, 7, 8, 11, 12, 17, 26, 27, 28, 29, 30, 31, 32. Yet the fact that CML has become a manageable chronic disease with much improved quality of life (QoL) may challenge adherence 4, 8, 26. Non-adherence in up to 25-30% of CML patients have been suggested 4, 7, 19, 26, 27, 28, 30, 33. Side effects are the most frequent reason for intentional non-adherence and has been associated with a negative impact on activities such as travels, other illness, psycho-social well-being and QoL 3, 4, 6, 7, 9, 10, 11, 13, 14, 15, 17, 26, 29, 31, 33, 34. Unintentional non-adherence is mostly due to careless slips. Non-adherence has also been linked to lacking knowledge about CML and treatment, co-payment, poor communication from health care providers and complacency due to sustained disease control 2, 3, 4, 6, 7, 10, 11, 12, 14, 17, 19, 26, 27, 28, 29, 30, 31, 32. Results diverge about adherence and type of TKI and may relate to the drug as well as patients’ experiences with earlier treatments 7, 8, 19.
The aim with the present study was to explore in depth the drivers and barriers to CML patients’ adherence in a Danish health care setting.
Materials and Methods
We used qualitative methods in order to examine patient perspectives on factors that may affect adherence behaviour in Danish CML patients. An initial literature search was carried out in PubMed, Embase and PsycInfo using the search terms ‘chronic myelogenous/myeloid/myelocytic leukemia/CML’ or ‘chronic granulocytic leukemia/CGL’ in combination with ‘patient’ and ‘perspectives/experiences/ preferences’ or ‘quality of life’ or ‘adherence/compliance/nonadherence’. From a total of 341 identified abstracts of journal articles published within the past ten years, 67 described QoL and treatment adherence in patients with CML and were thus selected for full-text analysis – five used qualitative methods 19, 29, 35, 37, 38.
The literature study informed the research design using focus groups to elicit discussions about a broad variety of patient perceptions of disease and treatment. The participants were encouraged to discuss convergent and divergent views and experiences on each topic. Four focus groups and three individual interviews with a total of 20 self-selected CML patients, 12 women and eight men, were carried out. Participants were invited via The Danish Patient Organisation for Lymphoma, Leukaemia and MDS (LyLe) that posted invitations in the LyLe and CML specific Facebook groups. Interested CML patients contacted the researchers directly for further information and screening. Due to the focus on subjective patient perspectives, no medical records were collected, and as participation was anonymous, ethical committee approval was not required. Written consent was obtained from all participants. The focus groups were set in conference rooms in four cities across Denmark to elicit potential regional differences. Each included 4-6 participants to create confidential settings for open discussions among peers about their perceptions of CML and experiences with treatment. Individual telephone interviews were carried out with two patients who were unable to attend a focus group and one focus group participant who wished to share additional information.
The focus groups lasted two hours and were moderated by G. Lee Mortensen using a semi-structured interview guide 40, 41. When introducing the focus group discussions, the participants were told of the aim to explore all possible perspectives and encouraged to voice converging as well as diverging views, provide examples and elaborate on their own and others’ experiences. The interviews began with descriptions of the participants’ CML diagnosis and then moved on to its subsequent impact on their QoL, i.e. their physical and psychosocial well-being and functioning. The patients then described their treatment experiences and perceptions of drivers and barriers to TKI treatment adherence (Table 2).
The interviews were transcribed verbatim and analysed using Nvivo 8 software (QSR International) and inductive content analysis 42. This involved an analysis of the participants’ statements in a manner to generate clusters of meaning about their perceptions of CML and treatment. First, the transcripts were read through numerous times (by two researchers independently) to get familiar with the data. Secondly, the data was coded into topics (categories) that were raised during the focus groups – mostly reflecting the topics in the question guide – and discussed among the researchers until agreement was reached. Some categories were collapsed; others were spilt up into separate categories. Thirdly, the main themes (sub-categories) within each topic were identified, and finally, recurrent connections between topics and themes were analysed. This generated a pattern of the relative significance that the topics and themes had for the participants, i.e. their perceptions of CML and drivers and barriers to treatment adherence.
Results
The 20 participants were aged between 36-75 years (mean 60.9) and had been diagnosed for 1-19 years (mean 6.23). 18 had a partner, all had children (four living at home) and 13 were grandparents. At the time of the interviews, they attended haematological clinics at seven hospitals across Denmark, 12 were retired, four worked reduced hours and five still had full-time work. Previously, six had worked reduced hours and six had taken early retirement due to the diagnosis (Table 1).
Table 1. Question guide for focus groups with CML patients| Opening question |
|---|
| First names and time of CML diagnosis |
| Quality of life (QoL) impact of CML |
| Reactions to the CML diagnosis |
| QoL impacts – whatever is important to your life and daily well-being (ensuring the following are covered): |
| Physical symptoms and their consequences |
| Side effects, if any, and their consequences |
| Emotional/psychological reactions and their impact |
| Disease impact on relationships and family life |
| Disease impact on social life or leisure activities |
| Professional consequences and their impact |
| Main QoL impact of CML |
| QoL changes since diagnosis |
| Experiences with CML treatment |
| Knowledge about types of CML treatment (TKI) and individual recommendations |
| Involvement in and preferences related to treatment decisions |
| Current, latest and previous CML treatments, if any |
| Reasons for switching treatment |
| Experiences with discontinuation of CML treatment (reasons, concordance, time, resumption) |
| Considerations about treatment discontinuation in those who never discontinued |
| Reasons for intentional or unintentional skipping one/few doses |
| Dose reduction – why and how often |
| Changes in CML treatment adherence over time |
| Dialogue with a hematologist about how to take medication, skipping or reducing a dose |
| · Perceptions of consequences of non-adherence |
| Knowledge about what is being measured at controls |
| Are milestone responses perceived as important |
| Do milestone responses affect adherence |
| Other reasons or factors affecting adherence and non-adherence |
| Experiences with treatment of symptoms or side effects |
| Overall experience with taking CML medication |
| Outro |
| Feeling of being sufficiently knowledgeable about CML and treatment, today and at diagnosis |
| Sources of information about CML and treatment |
| Changes in disease perceptions of CML over time |
| Do you feel there is anything we should have discussed but did not? |
Table 2. Participant characteristics
| FG 1Southern DenmarkM =maleF = female | Age(years) | Time of diagnosis | Treatments | Reasons for switching treatment | Personal status | Family status | Occupation |
| 1MA | 56 | Aug. 2013 | ImatinibDasatinib | After 2 months: SE | Married | CGC | Full time employment |
| 1MB | 68 | 2005-2006 | ImatinibNilotinib | After 6 months: LE | Partner | CGC | Flex job after dxCurrently retired |
| 1MC | 41 | June2015 | ImatinibDasatinib | After 2-3 months: SE | Married | Young C | Self-employedFull time |
| 1MD | 70 | 2011 | ImatinibNilotinibPlanned discontinuation since Jan. 2016 | SE and LE | Married | CGC | Self-employedFull time |
| 1FE | 73 | 2008 | NilotinibImatinib | After 2 years: SE | Married | CGC | Retired |
| 1FF | 59 | 2002 | ImatinibDasatinib | After 12 years: LE | Single | C | Full-time employment until 2007Self-employed (reduced hours) since 2007 |
| FG 2 Northern Jutland | |||||||
| 2MA | 75 | Marts 2009 | ImatinibDasatinibNilotinibNilotinibPonatinib (15 mg in trial) | After 1 year: LESESESE and LE | Married | CGCGGC | Retired at time of dx |
| 2FB | 64 | Dec. 2013 | ImatinibDasatinib | SEDosis reduction due to SE | Married | CGC | Retired at time of dx |
| 2FC | 61 | Nov. 2010 | ImatinibDasatinib | After 2 years: SEDosis reduction due to SE | Married | CGC | Flex job after dxCurrently retired |
| 2FD | 68 | Jan. 2011 | ImatinibDasatinibNilotinib | Imatinib => 'After 3 months...Dasatinib => 'After 2½ years.."Nilotinib => 'Discontinuation after ½ year...'Discontinuation after ½ year for 3-4 months due to liver complications. Restart on low dose working fine | Married | CGC | Retired since 2013Worked reduced hours after dx |
| FG 3 Zealand | |||||||
| 3FA | 75 | Aug.2014 | ImatinibDasatinibBosutinib | After 6 months: LEAfter 1½ years: LE | Single | CGC | Retired |
| 3FB | 57 | Dec. 2013 | Imatinib | Dosis reduction after 6 months due to SE | Cohabiting | C | Disability retirement since 2003 |
| 3MC | 63 | 2007 | ImatinibPlanned discontinuation since Dec. 2014 | Cohabiting | CGC | Worked full time until 2013Early retirement since 2013 | |
| 3FD | 75 | Jan. 2005 | Imatinib | Married | CGC | RetiredVolunteer work | |
| FG 4 Central Jutland | |||||||
| 4FA | 43 | 2009 | (stem sell harvest that may later be re-infused)Nilotinib (in trial at first) | Non-cohabiting partner | C (aged 19) | Self-employedFull-time | |
| 4FB | 58 | Marts 2012 | ImatinibPlanned discontinuation since 1½ months | Married | CGC | Self-employedFull-time | |
| 4MC | 69 | Marts 2014 | Dasatinib (in trial for 5 years of which one was in combination with peginferon injections) | Married | C | Retired since 2009 | |
| 4FD | 53 | Nov. 2013 | NilotinibImatinib | SE | Married | C (aged 16-28) | Self-employed16h flex job |
| In dividual interview | |||||||
| 3FDSupp. | 75 | Jan 2005 | Imatinib | Married | CGC | ||
| 5MA | 36 | Dec. 2013 | 1. HydroxyureaImatinibDasatinib Nilotinib | 2 weeksAfter one month: SEAfter 2-3 weeks: dosis reduction due to SE. After 1 year: LE | Married (temp. separated) | C (teen) | Unemployed at time of dxFlex job since 2015 (3h per week) |
| 5FB | 54 | 1997 | 1. Interferon-alfa2. Hydroxyurea3. Bone-marrow transplant (sister)4. T-cell infusion x 3 with ½ year intervals (sister) 2000-2001 due to relapse5. Imatinib (2003-2010)6. Nilotinib7. planned discontinuation for 3½ years | 5. SE and LE | Re-married | CGC | Retired shortly after dx (disability) |
Five main topics were discussed during the focus groups, namely the perceived QoL impact of getting CML, disease perceptions of CML, treatment experiences, adherence issues, and finally, the prospect of treatment discontinuation.
The impact of chronic myeloid leukaemia on psychosocial and physical quality of life
The QoL impact of CML had psychological, physical, social/relational, professional and treatment-related aspects that were inter-related. Emotionally, the diagnosis with CML came as a shock to the participants who thought only one fatal kind of leukaemia existed and therefore initially reacted with disbelief and fear of death. Subsequent coping was described as closely related to the treatment options at the time, especially whether and how many TKIs had been introduced: while patients diagnosed before or around 2001 had thus been very anxious, those more recently diagnosed were quickly reassured of the treatment options and good prognosis of CML (Patient quotes 1). Experiencing the efficacy of treatment was further comforting.
Patient quotes 1.Changed disease perceptions of CML: from fatal disease to a chronic condition
Patient quotes 2.The impact of severe side effects on QoL and motivation to adhere
While 14/20 patients had previously switched TKI due to lack of efficacy or side effects, most found the burden of their current treatment acceptable considering its life-saving effect. Also, being aware that treatment options are still limited, they expressed a preference for not ‘using up their lifelines’ too quickly. Three participants previously had depressive symptoms due to ineffective treatment or intolerable side effects, the latter leading one to discontinue medication for 1-2 months (5MA). These patients expressed markedly poorer social support and comorbidity than other participants, altogether leading them to loss of hope and motivation (Patient quotes 2). Although well-treated at the time of the interviews, many participants had physical symptoms such as musculoskeletal pain and fatigue, in particular. Pensioners and patients without children at home tended to find this less intrusive in their daily lives as they were more flexible to adjust their activities. In comparison, those with work or children at home found that such symptoms reduced their QoL and role functioning (Patient quote 3). Parenting, grand parenting, and working were described as particularly important to sustaining meaning, identity and normality. The participants described that, today, the main impact of CML was treatment-related side effects such as muscle pain and cramps, nausea, and fatigue. Side effects affecting the participants’ relational or professional role functioning – including severe headache, visible rashes or swelling around the eyes – were less acceptable than minor pain, palpitation or raised cholesterol levels, for instance.
Patient quote 3.The impact of fatigue on quality of life and role functioning
Disease Perceptions of CML
Most participants conveyed that at present, CML did not much affect their everyday lives. They had good social support and, with some prioritising and lowered expectations, did not have to entirely abstain from preferred activities. Some with few or acceptable symptoms hardly even felt sick and likened CML to other chronic diseases such as diabetes (Patient quotes 4). Disease related worries were, however, reactivated if treatment stopped working, severe side effects occurred or when getting infections that might be perceived as life threatening. A few with recurrent infections were very attentive to avoiding contagion and might therefore avoid physical contact with poorly loved ones, social gatherings or public spaces such as transport and shopping facilities (Patient quote 5).
Patient quotes 4.Adapting to CML as a chronic condition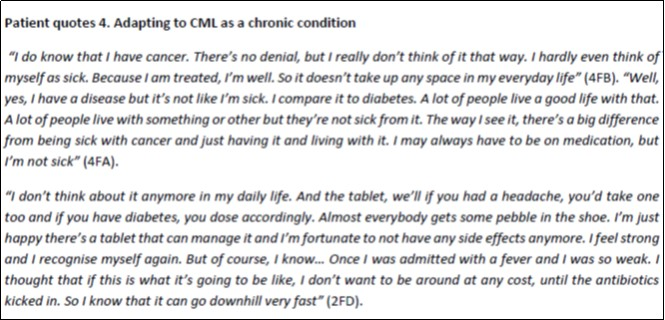
Patient quote 5.Fear of infections affecting intimacy and social participation
Treatment Experiences and Information Needs
Overall, the participants felt well-informed about CML and treatment options and regarded if they had any treatment preferences. Most attended one trusted hospital-based haematologist and felt reassured by the option of receiving telephone counselling at any time. Some described how this close communication enabled individually tailored support and treatment planning. At the time of the interviews, controls typically occurred at three month intervals with test results given by telephone every other time. Overall, the participants had no knowledge of, nor interest in, the details of the different treatment response types (CHR, CCyr, and CMR) but focused on their ‘numbers being within the normal range’, i.e. if the CML was under control. Women, however, tended to show more interest than men in the effects of concurrent medication and foodstuffs and in details of test results, e.g. by assessing them online prior to consultations.
Drivers and Barriers to Treatment Adherence
With one exception, the participants had been conscientious about adhering to their medication and had initially worried about forgetting or throwing up even a single tablet. They felt they received clear instructions and had established medication routines such as telephone alarms or visible medication boxes. Today, all believed that a rare slip was inconsequential but said this only occurred once or twice a year. They would only discontinue treatment in consultation with their haematologist, e.g. due to switch of medication or temporary co-morbid disease.
The participants explained that their adherence was mainly driven by knowledge about the importance of adhering, unwillingness to gamble with their lives and responsibility vis-à-vis their families. Some stated that not adhering would be disrespectful to the free Danish health care system that provided them with expensive lifesaving medication and specialist treatment. They speculated that in countries with co-payment, CML patients might be more tempted to skip medication if they felt well, if test results were good or if lacking expert supervision (Patient quotes 6). The one participant who consciously and entirely discontinued treatment without prior agreement did so because of severe side effects and depression: “It’s one or the other. Skipping a few pills doesn’t take away the side effects” (5MA). He finally resumed treatment but felt that the burden of CML treatment had been much understated.
Patient quotes 6.Assumed reasons for non-adherence in other CML patients
The Prospect of Treatment Discontinuation
Today, four participants – in three focus groups and one individual interview – had discontinued TKI in agreement with their haematologist due to sustained treatment response. They described this as creating immense relief at not being sick, of ‘returning to oneself’ and freedom from of a medicalised lifestyle including dietary restrictions, side effects, symptom management and constant telephone alarms (Patient quotes 7). These patients felt confident about suspending treatment due to the increased frequency of controls and the option of resuming treatment if needed. When discussing this, several other participants expressed hope in someday being able to discontinue too, while some felt safer on their haematologist.
Patient quotes 7.The impact of treatment discontinuation on psychological well-being
Discussion
This study is the first to qualitatively explore adherence in CML patients in a Danish health care setting. In-depth understanding of patient perspectives is crucial to provide locally relevant and individually tailored care and support throughout the disease course. Our participants’ disease perceptions had changed from fear of death at diagnosis to gradually adjusting to CML as a chronic condition. A large cross-cultural study has described this as a common five stage disease course in CML involving periods of crisis, hope, adaptation, new normal, and uncertainty. This basically linear process contains a cyclical component as patients experiencing uncertainty, due to treatment resistance or intolerance for instance, may return to earlier stages to then establish a new normal 35. This concurs with our finding that the participants were mostly able to live fairly normal lives with a tolerable disease burden but had reactivated worries when treatment stopped working, or side-effects or fear of infections reduced their QoL and functioning. Hence our description of the disease perception of CML as being in a state of fragile peace.
In CML, adherence has been measured using various methods, e.g. as percentage rates, using adherence measure scores, or categorising adherence into low, medium or high levels 31. Previous studies have been carried out in very different health care settings and sub-populations, e.g. CML patients receiving only one or three types of TKI. This variety hampers comparability as well as understanding of how CML patients may weigh the various factors and influence medication or simply unburdened by it. The participants’ attitudes to discontinuation were closely related that of influencing their medication adherence. In addition, discrepancies have been shown between patients´ self-reported adherence and physicians who may overestimate adherence if deducting this from clinical outcomes only 29, 37.
Most of our participants expressed very high adherence to TKI treatment in the sense that they only rarely forgot to take it – not intentionally or without agreement with the haematologist. When explaining their reasons to adhere, they recurrently mentioned ‘owing it’ to the fortune of having a manageable form of cancer and the privilege of receiving free specialised health care. Most were very satisfied with their care: the continuity with the same haematologist, easy accessibility, personal communication and treatment planning throughout the disease journey. All felt knowledgeable about CML and the importance of adherence, notably including the patient who discontinued treatment. Knowledge may thus be a precondition to adherence but not enough in itself. These factors are all known drivers to adherence along with longer disease duration, fear of the consequences of not adhering, using concurrent medication, having social support and using systems of reminders. Our results also confirm previous study results that relate higher levels of adherence to higher perceived functional status and QOL, including ability to work, and low burden of symptoms and side effects (between 3 and 36). Our participants described their role functioning as crucial to uphold a positive self-image and normality, which they felt was vital to a meaningful life. Symptoms thus became especially burdensome when negative affecting identity and social relations.
Only one participant had intentionally non-adhered to TKI treatment and few had considered it. Our study confirms that side effects were a main reason for this, exacerbated by comorbidity and poor social support. Side effects thus acted as a barrier to adherence due to its reduction in his psychosocial well-being and role functioning 3, 6, 9, 14, 33, 38. Several participants suggested that comprehensive patient care should include psychosocial support including information about peer support from patient networks. Though very rare, careless slips were the most common reason for unintentional non-adherence in our participants. Unlike other studies, our sample did not allow us to associate this with younger age, living alone and male gender, however (Between 2-39). While others have suggested that non-adherence may be caused by complacency and sustained disease control 4, 6, 29, 30, 31, 38, this was not the case in our participants although some speculated this might be the case in settings without public health care. Paradoxically then, higher QoL – stemming from successful treatment – may thus act as a barrier as well as a driver to adherence.
Only few studies have assessed patient perspectives on planned medication discontinuation due to sustained treatment response. Sanford and colleagues showed that side effects and daily adherence were reasons for some patients to want and feel safe discontinuing, while fear of relapse and disappointing others were reasons to reject discontinuation 16. In another study, half the patients dared not discontinue even if their treatment response was optimal, but 16% were interested to avoid side effects, and 20% were indifferent 12. In our explorative study, the participation of four patients in treatment-free remission allowed for unforeseen and for important novel insights into their experiences andthe other participants’ reflections on this topic. Most were diagnosed before the results from stop-trials were known and had only later heard of this option. Based some participants’ descriptions of positive feelings of freedom from medication, normality and wellness – and others’ hopes for this – we hypothesise that if informed about the prospect of treatment-free remission already at diagnosis, this may act an important driver to adherence in patients at risk of non-adherence and perhaps improve acceptance of side effects throughout the disease course. Medication adherence is not only influenced by the presence of side effects but as much by patients’ coping with them 6. Also, patients’ disease perceptions are influenced by the temporal perspective of disease and treatment, notably whether it is chronic, cyclical or transient 43. We therefore suggest that the prospect of treatment discontinuation and its perceived impact on health-related QoL and adherence should be further explored as results from clinical stop trials are gained. This finding may be significant in Denmark as well as other health care settings.
As described in the health belief model (HBM), patients’ medication taking behaviour is contingent on their evaluation of the gains and losses of taking it 744. A variety of factors may act as drivers and barriers to adherence in patients with CML. Individual factors, such as self-assessed QoL and burden of disease and side-effects are interrelated elements in an environment influenced both by the patient’s personal context (health status, work, familial and social roles and support) and the context of the specific health care system. In other words, the impact of side effects, for instance, may differ in a context with/without co-payment or with/without the prospect of planned discontinuation.
We used qualitative methods to explore patient perspectives on drivers and barriers to adherence. While previous quantitative studies have described various factors affecting adherence and their prevalence, our approach allowed for an in-depth understanding of ways in which these may be interrelated. For instance, how side effects or dietary restrictions may reduce patients’ QoL due to activity impairment in core role function domains such as parenting and work potentially leading to non-adherence. As such, our results have explanatory strength and are analytically valid but not statistically generalizable due to the small sample size. While we have shown that successful role functioning is closely related to one’s professional and family situation – factors that are related to age – a quantitative analysis of the impact on adherence of demographic factors such as work status and age was not possible. Likewise, our data did not allow relating adherence to exact age or number of years since diagnosis but rather to explain the importance of patients’ age to their overall life situation and the significance of treatment options at diagnosis to their hope and motivation.
Our study likely involves some inclusion bias as most participants were high-functioning and well-treated with good social support and relatively low current symptom burden. They participated in patient networks that may include fewer men and non-adherent patients less inclined to participate in study interviews. Due to the selection method of inclusion, men were underrepresented compared to the general CML patient population. The study indicates that women may sometimes need more detailed information than men, but this too should be confirmed by larger studies. Also, as our study did not include participants without children or who were diagnosed before the birth of their children, we were unable to gain knowledge about the potential impact of the CML diagnosis on the decision to have children or continue treatment during pregnancy. Finally, non-adherence may be associated with stigma potentially leading to social desirability bias with overstatements of adherence.
Conclusion
Overall, the health care of Danish CML patients supported these participants’ adherence to TKI treatment. Health care providers play a key role in addressing drivers and barriers to CML patients’ adherence throughout their disease journey. Continuous communication is required about their health-related QoL to allow for sustained symptom management and modification of treatment, if necessary. QoL and role functioning are closely related to adherence which is critical to ensure optimal treatment outcomes. Our study indicate that CML patients may benefit from being informed about the possibility of treatment discontinuation if they achieve stable complete molecular response and the impact of this prospect on adherence should be further explored.
Disclosures
GLM has conceived of the study, managed and carried out the literature study, designed the qualitative interview study, carried out the data collection and analysis and acted as main author of the manuscript. JM has provided medical expert advice to GLM, authored the medical parts of the manuscript and reviewed the entire manuscript. GLM has received an unrestricted research grant for the study from Pfizer, Denmark, which was not involved in the study, analysis or manuscript. JM has no conflicts of interest.
Acknowledgements
The authors wish to thank the study participants for their time and openness about their disease experiences. We are also indebted to the Danish patient organisation for Lymphoma and Leukemia (LyLe), and particularly its chair Rita O. Christensen, for her dedication to the project, her help including study participants and valuable counsel along the way. Finally, GLM is indebted to Ivan Bredbjerg Madsen for his invaluable assistance with the literature study and focus groups.
References
- 1.Simoneau C A. (2013) Treating chronic myeloid leukemia: Improving management through understanding of the patient experience. , Clinical Journal of Oncology Nursing 17(1), 13-20.
- 2.Mauro M J. (2014) The challenge of the 'C' in CML [Editorial]. Leukemia Research;. 38(3), 286-10.
- 3.Efficace F, Rosti G, Cottone F, Breccia M, Castagnetti F. (2014) Iurlo A et al. Profiling chronic myeloid leukemia patients reporting intentional and unintentional non-adherence to lifelong therapy with tyrosine kinase inhibitors. Leukemia Research. 38(3), 294-298.
- 4.Ault P S. (2012) Allen-Bard SL. Implications of improved survival in patients with chronic myeloid leukemia: A nursing perspective. Community Oncology. 9(2), 56-64.
- 5.Hehlmann R, Saussele S.Voskanyan A,Silver RT. , Management of CML blast crisis.BestPractResClinHaematol 29(3), 295-307.
- 6.Eliasson L. (2012) Treatment adherence in chronic myeloid leukemia: A systematic review of the literature. Clinical Practice. 9(1), 87-100.
- 7.Anderson K R, Chambers C R, Lam N, Yau P S, Cusano F et al. () Medication adherence among adults prescribed imatinib, dasatinib, or nilotinib for the treatment of chronic myeloid leukemia. , Journal of Oncology Pharmacy Practice: Official Publication of the International Society of Oncology Pharmacy Practitioners,2015; 21(1), 10-1177.
- 8.Santoleri F, Sorice P, Lasala R, Rizzo R C, Costantini A.Patient adherence and persistence with imatinib, nilotinib, dasatinib in clinical practice. PloS One 2013; 8(2) e56813. doi:. 10-1371.
- 9.Fava C, Morotti A, Dogliotti I, Saglio G, Rege-Cambrin G. (2015) Update on emerging treatments for chronic myeloid leukemia. Expert Opnion on Emerging Drugs. 20(2), 183-196.
- 10.Cella D, Jensen S E, Webster K, Du H, Lai J-S et al. (2014) Measuring health-related quality of life in leukemia: the functional assessment of cancer therapy – Leukemia (FACT-Leu) Questionnaire. 15, 1051-1058.
- 11.Bixby D L. (2013) Managing inadequate responses to frontline treatment of chronic myeloid leukemia: A case-based review. Cancer Treatment Reviews. 39(3), 241-251.
- 12.Breccia M, Efficace F, Sica S, Abruzzese E, Cedrone M et al. (2015) Adherence and future discontinuation of tyrosine kinase inhibitors in chronic phase chronic myeloid leukemia. A patient-based survey on 1133 patients. Leukemia Research. 39(10), 1055-1059.
- 13.Ferdinand R, Mitchell S A, Batson S, Tumur I.Treatments for chronic myeloid leukemia: a qualitative systematic review. , Journal of Blood Medicine 2012, 51-76.
- 14.Efficace F, Baccarani M, Breccia M, Cottone F, Alimena G et al.Chronic fatigue is the most important factor limiting health-related quality of life of chronic myeloid leukemia patients treated with imatinib. , Leukemia 2013, 1511-1519.
- 15.Russo D, Malagola M, Skert C, Fili C, Bergonzi C et al. (2013) Treatment of chronic myeloid leukemia elderly patients in the tyrosine kinase inhibitor era. Current Cancer Drug Targets. 13(7), 755-767.
- 16.Sanford D, Kyle R, Lazo-Langner A, Xenocostas A, Chin-Yee I et al.Hsia C.Patient preferences for stopping tyrosine kinase inhibitors in chronic myeloid leukemia.Curr Oncol 2014;21(2):e241-9. doi:. 10-3747.
- 17.Welch M A, Kaled E S. (2013) Ensuring optimal adherence to BCR-ABL1 tyrosine kinase inhibitor therapy for chronic myeloid leukemia. Community Oncology. 10(5), 138-146.
- 18.Hosoya K, Mochinaga S, Emoto A, Yokoo H, Tokushima H et al. (2015) Failure mode and effects analysis of medication adherence in patients with chronic myeloid leukemia. , International Journal of Clinical Oncology 10-1007.
- 19.Okumura L M, Antunes V D, Aguiar K S, Farias T, Andrzejevski V M et al. (2015) Tyrosine kinase inhibitors in patients with chronic myelogeneous leukemia: Defining the role of social risk factors and non-adherence to treatment. Pharmacy Practice. 13-2.
- 20.Yilmaz M, Abaza Y, Jabbour E. (2015) Selecting the best frontline treatment in chronic myeloid leukemia. Current Hematologic Malignancy Reports. 10(2), 145-157.
- 21.Pasic I, Lipton J H.Current approach to the treatment of chronic myeloid leukemia.LeukRes 2017;55:. 65-78.
- 22.Di Bella NJ, Bhowmik D, Bhor M, Yap M, Middlebrook B et al. (2015) The effectiveness of tyrosine kinase inhibitors and molecular monitoring patterns in newly diagnosed patients with chronic myeloid leukemia in the community setting. Clinical Lymphoma, Myeloma and Leukemia. 15(10), 599-605.
- 23.Radich JP.Monitoring response to tyrosine kinase inhibitor therapy, mutational analysis, and new treatment options. in chronic myelogenous leukemia.J Natl Compr Canc Netw 2013;11(5 Suppl): 663-666.
- 24.Baccarani M, Soverini S, C De Benedittis. (2014) Molecular monitoring and mutations in chronic myeloid leukemia: how to get the most out of your tyrosine kinase inhibitor.AmSocClinOncolEducBook. 167-75.
- 25.Richter J SaußeleS, Mahon FX HochhausA. (2016) The concept of treatment-free remission in chronic myeloid leukemia:Leukemia. 30(8), 1638-1647.
- 26.Abraham I, MacDonald K.Why are patients with chronic myeloid leukaemia (non-)adherent?. , British Journal of Cancer 107(6), 901-903.
- 27.Accordino M K, Hershman D L. (2013) Disparities and challenges in adherence to oral antineoplastic agents. , American Society of Clinical Oncology Educational Book / ASCO. American Society of Clinical Oncology Meeting 271-276.
- 28.Al-Barrak J, Cheung W Y. (2013) Adherence to imatinib therapy in gastrointestinal stromal tumors and chronic myeloid leukemia. Supportive Care in Cancer:. , Official Journal of the Multinational Association of Supportive Care in Cancer 21(8), 2351-2357.
- 29.Wu S, Chee D, Ugalde A, Butow P, Seymour J et al. (2015) Lack of congruence between patients' and health professionals' perspectives of adherence to imatinib therapy in treatment of chronic myeloid leukemia: A qualitative study [Abstract]. Palliative & Supportive Care. 13(2), 255-263.
- 30.MH de Almeida, Fogliatto L, Couto D. (2014) Importance of adherence to BCR-ABL tyrosine-kinase inhibitors in the treatment of chronic myeloid leukemia. Revista Brasileira De Hematologia e Hemoterapia. 36(1), 54-59.
- 31.Hall A E, Paul C, Bryant J, Lynagh M C, Rowlings P. (2015) Enjeti A et al. To adhere or not to adhere: Rates and reasons of medication adherence in hematological cancer patients. Critical Reviews in oncology/hematology. 1040-8428.
- 32.Marin D, Bazeos A, Mahon F X, Eliasson L, Milojkovic D et al.Adherence is the critical factor for achieving molecular responses in patients with chronic myeloid leukemia who achieve complete cytogenetic responses on imatinib. , Journal of Clinical Oncology: Official Journal of the American Society of Clinical Oncology 28(14), 2381-2388.
- 33.Hirji I, Gupta S, Goren A, Chirovsky D R, Moadel A B et al. (2013) Chronic myeloid leukemia (CML): Association of treatment satisfaction, negative medication experience and treatment restrictions with health outcomes, from the patient's perspective. Health and Quality of Life Outcomes. 11, 167-7525.
- 34.Noens L, van Lierde MA, R De Bock, Verhoef G, Zachee P et al. (2009) Prevalence, determinants, and outcomes of nonadherence to imatinib therapy in patients with chronic myeloid leukemia: The ADAGIO study. Blood. 113(22), 5401-5411.
- 35.Guilhot F, Coombs J, Szczudlo T, Zernovak O, Paolantonio M et al. (2013) The patient journey in chronic myeloid leukemia patients on tyrosine kinase inhibitor therapies: Qualitative insights using a global ethnographic approach. The Patient. 6(2), 81-92.
- 36.Dusetzina S B, Winn A N, Abel G A, Huskamp H A, Keating N L.Cost sharing and adherence to tyrosine kinase inhibitors for patients with chronic myeloid leukemia. , Journal of Clinical Oncology: Official Journal of the American Society of Clinical Oncology 32(4), 306-311.
- 37.Kekäle M, Talvensaari K, Koskenvesa P, Porkka K, Airaksinen M. (2014) Chronic myeloid leukemia patients' adherence to peroral tyrosine kinase inhibitors compared with adherence as estimated by their physicians. Patient Preference and Adherence. 8, 1619-1627.
- 38.Efficace F, Baccarani M, Rosti G, Cottone F, Castagnetti F et al. (2012) Investigating factors associated with adherence behaviour in patients with chronic myeloid leukemia: An observational patient-centered outcome study. , British Journal of Cancer 107(6), 904-909.
- 39.Noens L, Hensen M, Kucmin-Bemelmans I, Lofgren C, Gilloteau I et al. (2014) Measurement of adherence to BCR-ABL inhibitor therapy in chronic myeloid leukemia: Current situation and future challenges. Haematologica. 99(3), 437-447.
- 41.Kvale S Interviews. (1996) An introduction to qualitative research interviewing. Sage Publications, Thousand Oaks.
Cited by (7)
This article has been cited by 7 scholarly works according to:
Citing Articles:
European Journal of Oncology Nursing (2023) Crossref
European Journal of Oncology Nursing (2023) OpenAlex
A. Hewison, Eve Roman, Alex Smith, D. McCaughan, Rebecca Sheridan et al. - European Journal of Oncology Nursing (2023) Semantic Scholar
European Journal of Cancer Care (2023) Crossref
A. Hewison, Eve Roman, Alex Smith, D. McCaughan, Rebecca Sheridan et al. - European Journal of Cancer Care (2023) Semantic Scholar
European Journal of Cancer Care (2023) OpenAlex
Leukemia & lymphoma/Leukemia and lymphoma (2020) OpenAlex
Atalay Mulu Fentie, Fishatsion Tadesse, E. Engidawork, A. Gebremedhin - PLoS ONE (2019) Semantic Scholar