Pure Small Cell Carcinoma of Prostate in a Patient Presenting with Acute Urinary Retention: A Rare Case Report
Abstract
Small cell carcinoma of the prostate is a rare and highly aggressive subtype of prostate cancers. In this case report, we evaluated a patient applied our outpatient clinic with acute urinary retention, whose pathology reported as pure small cell carcinoma of the prostate. A 73-year-old male patient was admitted to our outpatient clinic with acute urinary retention. Digital rectal examination was normal. The PSA value was 1.81 ng/dl. Prostate size was 101 cc. Open prostatectomy was performed. The pathology of the patient was reported as pure small cell prostate carcinoma. Four cycles of etoposide and cisplatin chemotherapy were administered to the patient. The patient died 13 months later after the first diagnosis. We evaluated the pathological and clinical findings of small cell carcinoma of the prostate.
Author Contributions
Academic Editor: Tag Keun Yoo, Eulji University, Korea.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2020 Sezgin Okçelik, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Small cell carcinoma of the prostate is a rare and highly aggressive subtype that accounts for approximately 0.4-2% of all prostate cancers 1, 2. This subtype is a neuroendocrine-transforming tumor of the prostate that resembles a small cell tumor of the lung 1. It was first described in 1977 by Wenk et al. 3. Patients frequently presented with symptoms of lower urinary tract symptoms and acute urinary retention 4. This tumor is predominantly predisposed to early metastases to the lymph nodes, visceral organs and central nervous system 5, 6. Small cell cancer of the prostate shows worse prognosis than conventional prostate cancer 7. Here, after getting the informed consent from the patient, we evaluated a patient applied our outpatient clinic with acute urinary retention, whose pathology reported as pure small cell carcinoma of the prostate.
Case Report
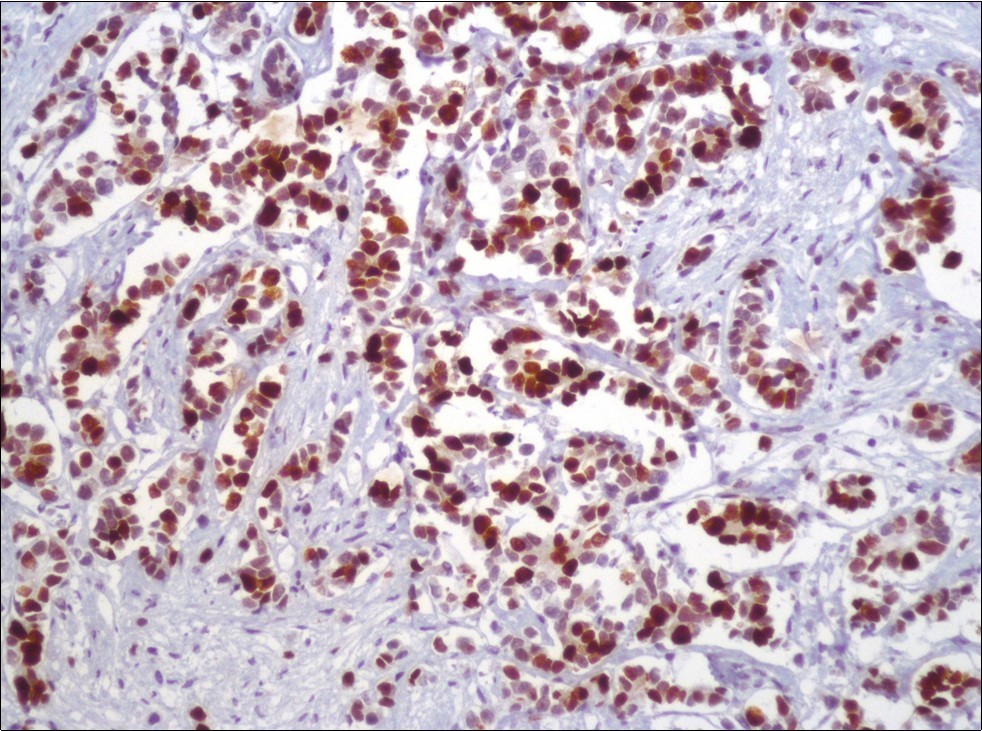
A 73-year-old male patient was admitted to our outpatient clinic with acute urinary retention. Digital rectal examination was normal. The PSA value was 1.81 ng/dl. Prostate size was 101 cc. Since the PSA value and digital rectal examination were normal, no biopsy was taken. Ultrasonography showed bilaterally ureterohydronephrosis. The creatinine level was 2.89 mg/dl. Hypertension has been presented as a comorbidity. An 18 Fr Foley catheter was inserted and alpha-blockers and 5-α reductase inhibitor medication was given. Creatinine level decreased to 0.91 mg/dl after one week. When the patient's catheter was pulled out, he could not urinate again. A catheter was inserted and waited one week more. After one week, the patient was unable to urinate again. The operation was decided due to this recurrent urinary retention. Because of prostate size was 101 cc and prostatic urethra was long, open prostatectomy was decided. When the prostate was removed during the operation, it was evaluated that the left side and the lower side were adhered to surrounding tissue. The prostate was removed as an en bloc. The catheter was removed after 5 days. The pathology of the patient has been reported as pure small cell prostate carcinoma. On the microscopic examination, the tumor was heavily infiltrated into the prostate parenchyma (Figure 1). Tumor cells were consist of atypical small-medium sized, mitotically active cells with a high nuclear to cytoplasmic ratio, hyperchromatic nucleus, nuclear molding, and inconspicuous nucleoli (Figure 2). On the immunohistochemically examination of synaptophysin showed diffusely and strong positivity while chromogranin A showed focal weak positivity with the absence of staining for PSA (Figure 3). Ki67 proliferative index was equal to 90% of the tumor cells (Figure 4). The results confirmed the diagnosis of prostatic small cell carcinoma. Metastatic lymph nodes were detected in the right perirectal region with metastatic multiple lymph nodes in the bilateral external iliac region, more on the left than in the PET CT. No metastasis was detected in the brain MRI and thorax computed tomography. Four cycles of etoposide and cisplatin chemotherapy were administered to the patient. Control PET CT taken after chemotherapy showed a significant decrease in size and metabolic activity of the right obturator and lymph nodes in the right perirectal area. The size and metabolic character of the left obturator, left external iliac and common iliac lymph nodes were increased. After a month, patient admitted to emergency outpatient clinic with left flank pain. There was an 8-cm mass in retroperitoneal area in the computed tomography. Supportive treatment was started because the patient could not tolerate chemotherapy. The masses reached to 13x11cm and fulfilled the retroperitoneal area. Multiple lung metastases were seen. The patient died 13 months later after the first diagnosis.
Figure 1.Infiltration of the tumor cells between prostatic ducts (H&E, x40)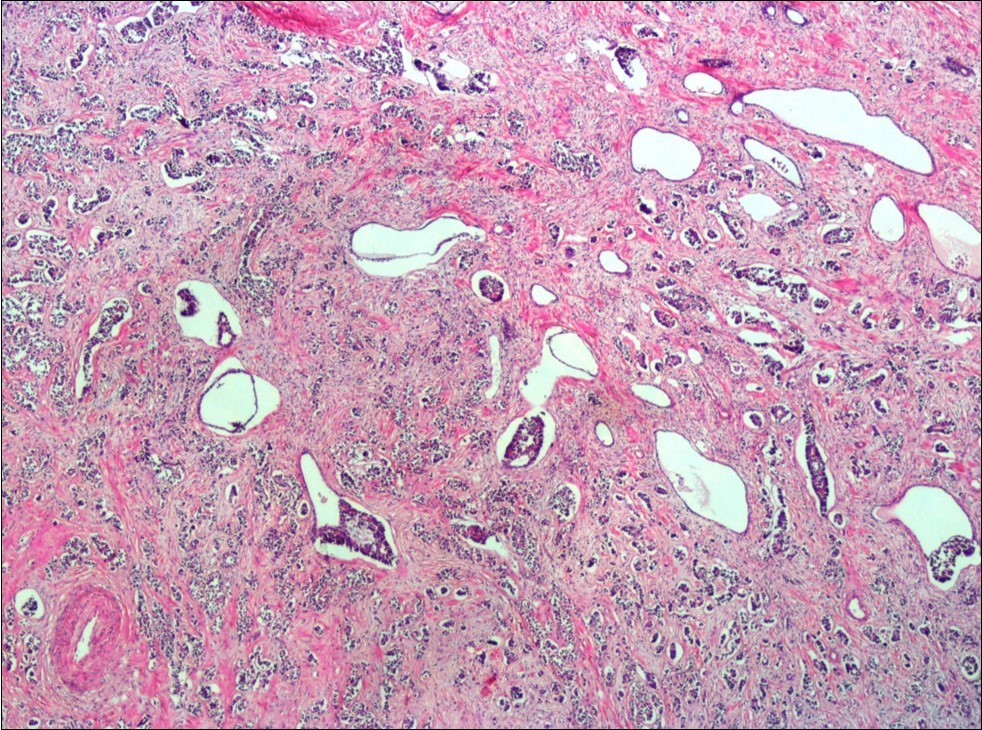
Figure 2.The tumor cells show rosette formation with hyperchromatic nuclei and nuclear molding (H&E, x200)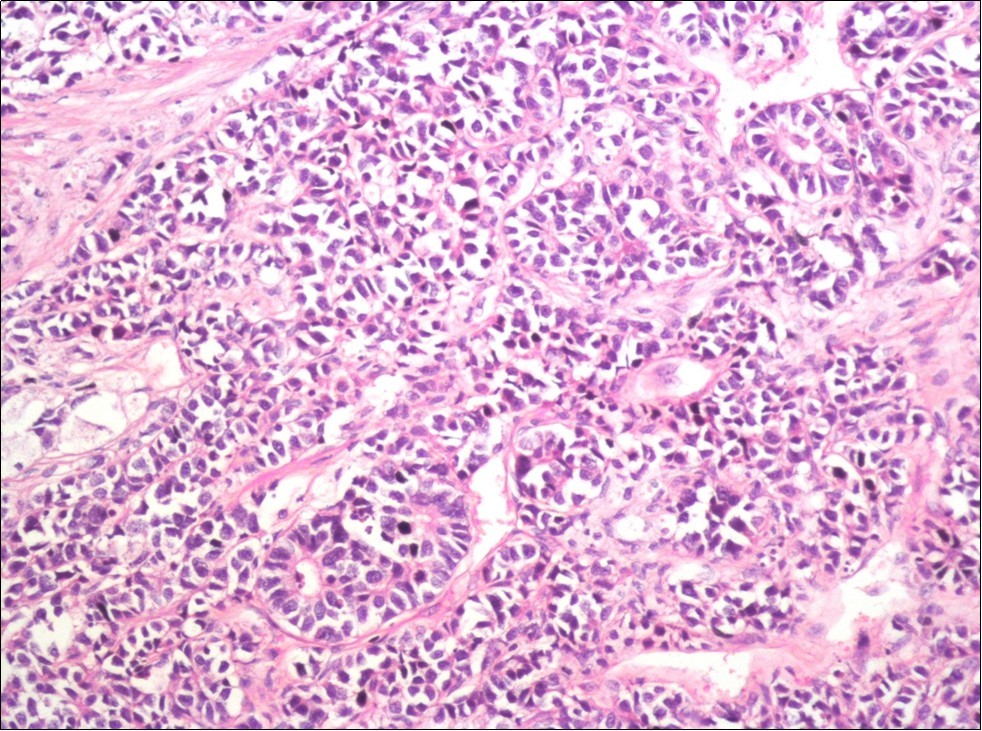
Figure 3.Diffusely and strong immunostaining for synaptophysin (Synaptophysin, x200)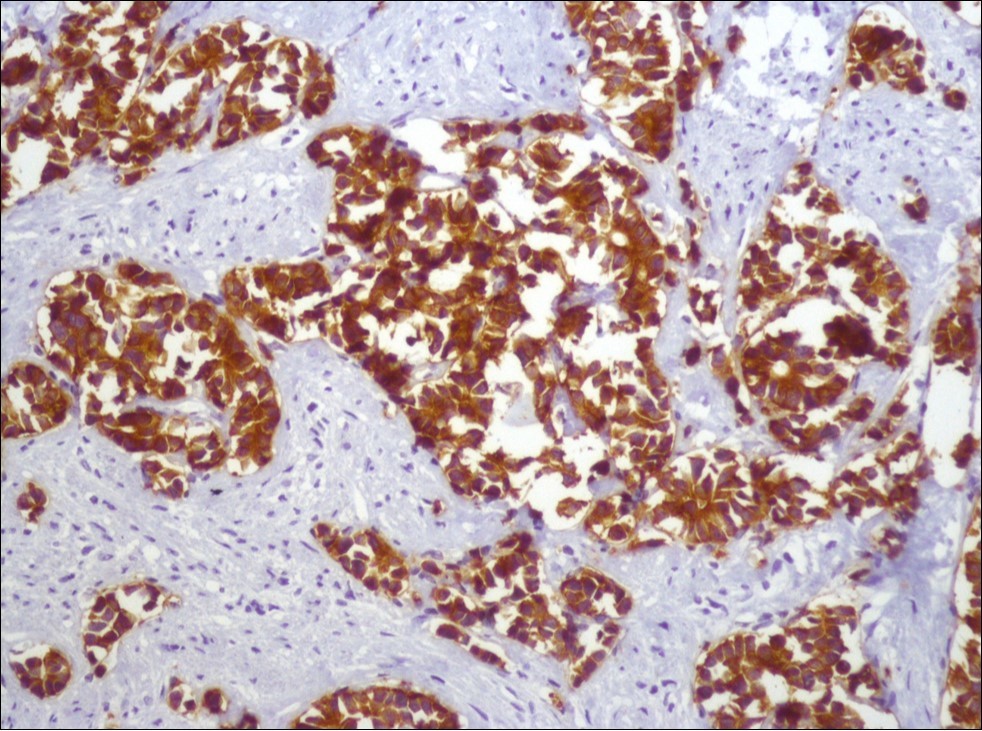
Discussion
Small cell prostate cancer is a rare, aggressive prostate tumor. It can be seen primarily or can be seen with adenocarcinoma 1, 5, 8. In pure ones, increased PSA cannot be seen. Therefore, it is usually detected in transurethral resection or open prostatectomy materials. In mixed types, PSA elevation can be seen and detected in prostate biopsy materials 1. The pathology of our patient was pure small cell carcinoma and there was no PSA elevation.
As patients may present with lower urinary tract symptoms and acute retention, and also may present bone pain, hydronephrosis, hematuria, hematochezia and abdominal pain at advanced stages 3, 8. They can also refer to paraneoplastic syndrome findings such as inappropriate ADH syndrome or ectopic ACTH production due to the nature of the tumor 3. Our patient here presented with acute retention and acute renal failure. This proved that the small cell prostate cancer can be silent.
The majority of patients at the time of diagnosis are in advanced levels 6. It has also been reported that Lymph node, liver, bone, lung and brain metastases can be seen. Our patient also had lymph node metastasis.
In clinical oncological applications, radiation therapy is recommended to deprive cancer cells of their potential to reproduce, to shrink tumor and achieve local tumor control. Also, chemotherapy can be used with radiation therapy 9, 10. Although initial treatment response is good in patients with small cell prostate cancer, prognosis is poor. Median time of survival following diagnosis is 10 months and less than 5% of patients survive for 2 years 11. In accordance with this information, Our patient was survived for 13 months.
Albisinni et al reported a case similar to ours. Their patient had no PSA elevation. Acute urinary retention was the reason for application. Also the biopsy result was pure small cell of carcinoma 4. In 2013, Demirtaş et al reported a case different from ours. Patient’s PSA level was high and his pathology was small cell carcinoma and adenocarcinoma 6. Furthermore, this patient died after 6 months. The patient's survival was shorter than the patient presented by Albisinni. Therefore, we find it very important to discuss whether PSA has an effect on survival.
In pure small cell prostate carcinoma, the PSA level does not usually increase. The prognosis is also poor 4, 6. However, the level of PSA in adenocarcinoma generally increases and the prognosis is good. Both of the tumors can reveal rectal examination findings.
As a result, pure small cell carcinoma of the prostate is a rare and poor prognostic tumor. Although it can be detected histopathologically, its the treatment results are very poor. Therefore, we think that there is a need for specific markers that can be used to detect the tumor earlier.
Disclosure
The authors declare no conflict of interest.
References
- 1.di Sant'Agnese PA. (1992) Neuroendocrine differentiation in carcinoma of the prostate. Diagnostic, prognostic, and therapeutic implications. , Cancer. (1 Suppl) 70, 254-268.
- 2.Zhang Y, Ouyang W, Sun G, Ding B, Yan L et al. (2018) Pure Small Cell Carcinoma of Prostate: A Report of 8 Cases. , Urol Int 101(3), 263-268.
- 3.Wenk R E, Bhagavan B S, Levy R, Miller D, Weisburger W. (1977) Ectopic ACTH, prostatic oat cell carcinoma, and marked hypernatremia. , Cancer 40(2), 773-778.
- 4.Albisinni S, C De Nunzio, Tubaro A. (2012) Pure small cell carcinoma of the prostate: A rare tumor. , Indian J Urol 28(1), 89-91.
- 5.Capizzello A, Peponi E, Simou N, Ntaskagiannis D, Tasiou I. (2011) Pure small cell carcinoma of the prostate: a case report and literature review. Case Rep Oncol. 4(1), 88-95.
- 6.Demirtas A, Sahin N, Ozturk F, Akinsal E C, Demirtas T. (2013) Small cell prostate carcinoma: a case report and review of the literature. Case Rep Urol. 387931.
- 7.Cohen R J, Glezerson G, Haffejee Z, Afrika D. (1990) Prostatic carcinoma: histological and immunohistological factors affecting prognosis. , Br J Urol 66(4), 405-410.
- 8.Wang L, Davidson D D, Montironi R, Lopez-Beltran A, Zhang S et al. (2015) Small cell carcinoma of the prostate: molecular basis and clinical implications. , Histol Histopathol 30(4), 413-24.
- 9.Spiess P E, Pettaway C A, Vakar-Lopez F, Kassouf W, Wang X. (2007) Treatment outcomes of small cell carcinoma of the prostate: a single-center study. , Cancer 110(8), 1729-1737.