Nanotechnology Meets Immunotherapy: CAR-T Cells Technology and Beyond
Abstract
The crusade against cancer has a new army: immunotherapy. The rational design is very simple, but brilliant at the same time. Extract the patients T-cells, reprogram them in vitro for the expression of highly specific receptors against cancer, perfuse them back to the patient. As a result, T-cells are now instructed to selectively kill circulating tumor cells, while avoiding potential side effects. This ‘Fairy Tale’ however does not lack of drawbacks and limitations. First, malignant progression can be accompanied by profound immune suppression, which counteracts the immune system-mediated tumor elimination. Second, the immune cells modification does not match high standards in terms of safety for humans. Here, nanotech can fill these gaps, and help immunotherapy to be safer and more effective.
Author Contributions
Academic Editor: Mehrez El-Naggar, National Research Centre, Dokki, Giza, Egypt.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2018 Loris Rizzello. et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
A Brief History of Immunotherapy
The question of boosting the immune system activity towards the treatment and eradication of cancer is one of the most attractive topics of the last couple of years. This field is unanimously known as immunotherapy, and together with chemotherapy, radiation therapy, stem cells transplants, and hormones perfusion, it represents a brand new fifth pillar in the current available methods for cancer treatment. A correlation between cancer development and immune system dysfunctions is not a new discovery, as the first hypothesis of inflammation-induced cancer dates back to the 19th century. Similarly, a pioneering hypothesis on the potential use of the human immune system for cancer therapy has been tested in the 70s. Here, the tuberculosis vaccine Bacille Calmette-Guérin (BCG) has been infused to prevent the recurrence of non-muscle invasive bladder cancer, because of the observation that patients with cancer went into spontaneous remission after developing acute infection.1 In the same years, the newly discovered T-cells growth factor interleukin 2 (IL-2) has been demonstrated to be effective when administered to patients with established metastatic cancer. FDA approved then IL-2 therapy in 1991. At the same time, rituximab has been developed as the first monoclonal antibody-based therapy for non-Hodgkin’s lymphoma,2 and FDA approved its use in 1997.
Few decades passed until a brilliant idea turned into one of the most revolutionary and promising immunotherapy approaches for non-solid tumor therapy. In 2010, a 5-year old girl has been diagnosed with acute lymphoblastic leukaemia (ALL) at the Children’s Hospital of Pennsylvania, and despite the many cycles of chemotherapies, she experienced several relapses. Out of options, and with a fatal prognosis, one of the oncologist made the parents aware of a new experimental therapy. The parents opted to enroll her in this new study, and she became the first child ever receiving chimeric antigen receptor T-cells (CAR-T) cells infusion. Now she is not only 12-year old, but this success helped reversing that line of research that was close to fail, and in 2017 the FDA approved the first CAR-T cell technology. Results are now striking, and 83% of the 63 evaluated children who have received CAR-T cells infusion eliminated malignancies after 3 months.3 Hundreds of years passed after the Paul Ehrlich’s theory of a ‘magic bullet’ sailing within the human body, and selectively targeting the tumor. Now, the bullet is not a drug, or a nanotech-based delivery system anymore, but it is a natural killer T-cell (NK-T cell), which is instructed to chase and kill metastatic tumor cells. Being part of our immune system, the NK T-cells possess the intrinsic ability to avoid normal (self) cells, and they are thus the perfect candidate for a tumor-specific therapy. However, all that glitters is not gold. There are still some drawbacks in the methodology that have to be solved, and nanotechnology may again represent smart solution to the CAR-T cell technique.
CAR-T Cells Technology. What is it?
In CAR-T therapy, T-cells are extracted and purified from the patients’ blood (or from an allogeneic donor), and they are genetically engineered to express specific membrane receptors. These are called chimeric antigen receptors (CARs). CARs consist of an extracellular antigen-recognition domain, scFv (namely, an antibody single chain variable fragment), linked to one (or more) intracellular signaling domain(s) (Figure 1).4Upon binding of the scFv domain with the specific tumor antigen, the intracellular signaling domain will trigger the stimulation of T-cell towards proliferation, cytolysis and cytokines release. The final result is the cytokine-mediated elimination of the targeted tumor cells. Being isolated from the patients, there is a very low risk of graft-versus-host disease, and also non-specific binding is really unlikely to happen. Different CARs generations have been developed during the years. The first generation used only one CD3ζ (zeta, from T-cells) signaling domain (Figure 1, left), and the efficacy was limited in the clinical trials due to the lack of long term T-cells proliferation.5,6 A second CARs generation has been developed then, where the intracellular segment of the receptor includes an additional co-stimulatory domain for an enhanced T-cell activation (Figure 1, centre). This second generation of CARs showed a threshold limit in the persistence and expansion of circulating CAR-T cells in several clinical trials. Therefore, a third generation of CARs has been designed, which contain a CD3ζ domain and two co-stimulatory domains (Figure 1, right). This class demonstrated superior anti-tumour efficacy in several preclinical trials, with respect to the previous generations of CAR-T.7
Figure 1.CAR designs contain an antigen-recognition domain and a signaling domain that provides 'signal 1' to activate T cells. Only this signaling domain is present in first-generation CARs. By contrast, a co-stimulatory signaling domain that provides 'signal 2' is added in second-generation CARs, and in third-generation CARs two co-stimulatory signaling domains are added.4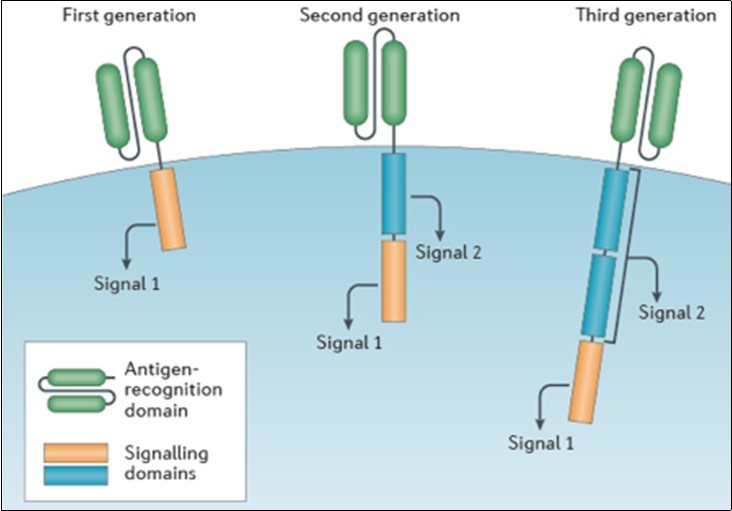
There is nowadays a plethora of new genetic approaches to improve the CAR-T efficacy, and they go far beyond the choice of a specific tumor target and/or stimulating factor. We redirect the readers to the comprehensive work of H. Jackson for extensive descriptions.4 There are however some intrinsic limits that still must be addressed, as well as methodological drawbacks that should be overcome to make this therapy more effective. The first issue concerns the way CAR-T cells are currently produced, and the second problem is the ability of the tumor to suppress the immune system. The next paragraph will explain the basics of these limits, and provide some guidelines on how nanotechnology can fill the gap and provide new biomaterials for addressing such question.
CAR-T Cells Immunotherapy. Its Limitations and the Possible Nanotech Solutions
One thing is clear is that CAR-T cells immunotherapy has been proved to work, and it is definitely one of the most promising candidate for defeating cancer nowadays. One important question to be asked is how CAR-T cells are produced. Everything starts with rather difficult and specialised manufacturing procedures and quality checks, which include leukapheresis (for removing blood from the patient’s body), followed by leukocytes separation and T-cells enrichment.8 The most crucial step is the T-cells activation process, where cells are incubated with a viral vector containing the CAR sequence. Usually this is a retrovirus (HIV-like lentivirus) which enters T-cells and insert its genetic material in the form of RNA. The RNA is then reverse-transcribed into DNA that permanently integrates within the genome of the patient’s cells. The final result is a T-cell expressing the desired CAR receptor. However, this stable insertional mutagenesis is one of the most dangerous procedure, as we do not know its potential effects on humans. Lentiviral or gamma-retroviral integration is permanent in the host cells, and a potential insertion nearby an oncogene is a serious concern. There is indeed a clear risk associated to the use of these viral vectors, and we need a safer replacement.
To this respect, magnetic nanoparticles (MNPs, Fe3O4) incorporated into the complexes of cell penetrating peptides (CPPs)-oligonucleotides have been proved to promote cell transfection for plasmid transfection, splice correction, and gene silencing efficiencies.9 The specific question of replacing lentiviral with nanoparticles-based transfection has been recently addressed in a refined work by Smith and co-workers at the Fred Hutchinson Cancer Research Center of Seattle.10 The authors demonstrated that DNA-carrying polymeric nanoparticles are able to effectively introduce CAR genes within the T-cells nuclei, which then recognise specifically leukaemia cells.10 The use of nanoparticles (NPs) for gene delivery, as a replacement of viral vectors, has another advantage. Recent evidences suggest that unspecific pro- or anti-proliferative, and therefore pro- or anti-inflammatory effects, could be conferred by NPs-mediated transfection.11 This means that such effect can even enhance a specific therapeutic approach towards a desired outcome. For example, Fe3O4NPs inhibit primary immune cells proliferation and induce a short-term cell cycle arrest, while reducing the expression of genes that are important for immune cell proliferation.11 This suggests that NPs can boost the therapeutic outcome by triggering anti-replicative effects, in case of gene therapy targeting highly proliferative cancer cells like CAR-T. Nanotechnology has provided us with a plethora of new nanoparticles that can be exploited to vehicle genetic material within T-cells.12,13,14 Such advancement in vector technology will lead to establish new protocols for the replacement of viral transfection in CAR-T production. In this framework, the size, shape, and surface moieties of NPs have been extensively explored to find the optimal conditions for an effective targeting and transfection efficiency.13
In addition to the viral-based transfection of T-cells, the second big limit in CAR-T therapy is the ability of tumor to bypass the human immune system, a crucial requirement for its spreading within body compartments that are far from the original growth site. Tumor cells are able indeed to highly express the programmed death-ligand 1 (PD-L1), which binds to programmed death-1 (PD-1) receptors on T-cells and inhibits their effectors and function.15 Tumor cells can also secrete cytokines such as IL-10 and TGF-β, which both directly inhibit the proliferation of cytotoxic T-cells. The final result is that the tumor microenvironment has a strong immunosuppressive behavior against the excessive activation of T-cell responses.
Also in this case, nanoparticles can help overcoming this issue, and facilitate T-cell activation. For example, 60 nm nanogels made of polysaccharide cholesteryl pullulan have been proved to prime anti-tumoral CD8+ T-cell responses, upon in vivo subcutaneous injection in mice.16 Similar enhanced cytotoxic CD8+ T-cell response, and consequent inhibition of tumor growth, has been observed by targeting nano-vaccines to tumor-draining lymph nodes.17 Gold nanoparticles coated with cationic antigen peptides are also demonstrated to boost antigen-specific CD8+ response in vivo (Figure 2).18
Figure 2.TLR3 agonist poly I:C and an antigen peptide were complexed onto gol nanoparticles via electrostatic interactions, and elicited more antigen-specific CD8+ T cells compared to the soluble vaccine.18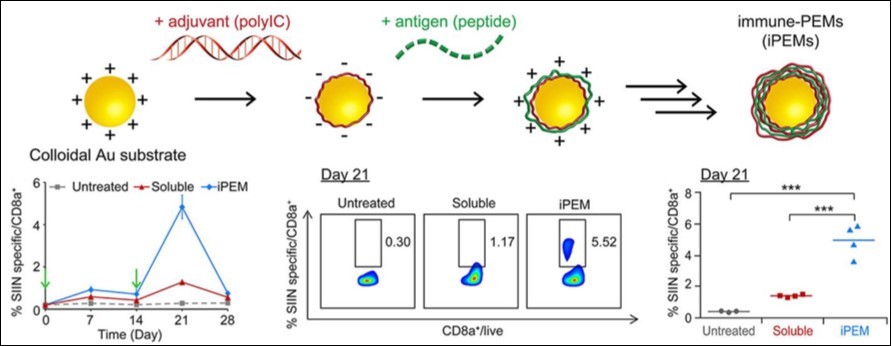
To conclude, it is time to rethink how nanoparticles can contribute in the biomedical research. The last decades showed us that they can be used as a drug delivery platform for cancer treatment or infection control. However, if we think them out of the box, we come to the conclusion that nanoparticles can have high impact implications also in immune-oncology. To this respect, CAR-T cells technology represents a new pillar in the current available strategies to defeat non-solid tumor. It is an extremely promising method indeed, and any strategy aiming to improve it will have strong and wide-range impact. CAR-T therapy comes however with some limitations, including the cells modification procedure, and the ability of the tumor to circumvent the immune system. This is the field where nanotechnology can help overcoming these drawbacks. Nanoparticles-based drug delivery platforms can be rationally insert in the production workflow of CAR-T cells, thus replacing the current methods of lentivirus transfections. Additionally, nanoparticles can help boosting the intrinsic activity of T-cells towards tumor clearance.
References
- 1.Morales A, Eidinger D, Bruce A W. (1976) Intracavitary Bacillus Calmette-guerin in the Treatment of Superficial Bladder Tumors. , The Journal of Urology; 116, 180-182.
- 2.Rudnicka D. (2013) Rituximab causes a polarization of B cells that augments its therapeutic function in NK-cell–mediated antibody-dependent cellular cytotoxicity. , Blood 121, 4694-4702.
- 3.Buechner J. (2017) Global Registration Trial of Efficacy and Safety of CTL019 in Pediatric and Young Adult Patients with Relapsed/Refractory (R/R) Acute Lymphoblastic Leukemia (ALL): Update to the Interim Analysis. Clinical Lymphoma, Myeloma and Leukemia;17,S263-S264.
- 4.Jackson H J, Rafiq S, Brentjens R J. (2016) Driving CAR T-cells forward. , Nature Reviews Clinical Oncology 13, 370.
- 5.Till B G. (2008) Adoptive immunotherapy for indolent non-Hodgkin lymphoma and mantle cell lymphoma using genetically modified autologous CD20-specific T cells. , Blood; 112, 2261-2271.
- 6.Jensen M C. (2010) Anti-Transgene Rejection Responses Contribute to Attenuated Persistence of Adoptively Transferred CD20/CD19-Specific Chimeric Antigen Receptor Re-directed T Cells in Humans. , Biology of Blood and Marrow Transplantation; 16, 1245-1256.
- 7.Carpenito C. (2009) Control of large, established tumor xenografts with genetically retargeted human T cells containing CD28 and CD137 domains. Proceedings of the National Academy of Sciences; 106, 3360-3365.
- 8.Smith J W. (1997) Apheresis Techniques and Cellular Immunomodulation. Therapeutic Apheresis;. 1, 203-206.
- 9.Dowaidar M. (2017) Magnetic Nanoparticle Assisted Self-assembly of Cell Penetrating Peptides-Oligonucleotides Complexes for Gene Delivery. , Scientific Reports 7, 9159.
- 10.Smith T T. (2017) In situ programming of leukaemia-specific T cells using synthetic DNA nanocarriers. , Nature Nanotechnology; 12, 813.
- 11.Przybylski S. (2017) Influence of nanoparticle-mediated transfection on proliferation of primary immune cells in vitro and in vivo. , PLOS ONE 12, 0176517.
- 12.Guan L, Rizzello L, Battaglia G. (2015) Polymersomes and their applications in cancer delivery and therapy. , Nanomedicine; 10, 2757-2780.
- 13.Lin G, Zhang H, Huang L. (2015) Smart Polymeric Nanoparticles for Cancer Gene Delivery. Molecular Pharmaceutics;. 12, 314-321.
- 14.Amreddy N, Babu A, Muralidharan R, Munshi A, Ramesh R. (2017) Polymeric Nanoparticle-Mediated Gene Delivery for Lung Cancer Treatment. Topics in Current Chemistry;. 375, 35.
- 15.Zou W. (2005) Immunosuppressive networks in the tumour environment and their therapeutic relevance. , Nature Reviews Cancer; 5, 263.
- 16.Muraoka D. (2014) Nanogel-Based Immunologically Stealth Vaccine Targets Macrophages in the Medulla of Lymph Node and Induces Potent Antitumor Immunity. , ACS Nano; 8, 9209-9218.

