Monk Fruit is a Safe, Natural, Non-Caloric Sweetener in Individuals with Type 2 Diabetes
Abstract
Non-nutritive sweeteners are used as adjuncts in treating patients with diabetes to reduce carbohydrate intake. Monk fruit is an FDA-approved1 natural NNS2-. There is no information regarding the use of monk fruit in individuals with T2DM3. The primary objective was to compare the glycemic response to sucrose and monk fruit in individuals with and without T2DM. Twenty-six individuals with T2DM and 29 without DM4 underwent two oral glucose tolerance tests5 after a 250-ml sucrose- or monk fruit-sweetened beverage. Glucose levels were measured at 0, 30, 60, 90, and 120 minutes. An appetite scale was used before and after the procedure. Insulin levels were measured in ten randomly selected individuals from the control group. The glucose levels at 30 and 120 min were significantly lower in both groups in the monk fruit-OGTT. There was a peak response in the T2DM group at 90 min (median, IQR, 247 mg/dL, 203-293) with the sucrose OGTT, while no peak response was seen in the monk fruit OGTT. There were no differences between sweeteners in terms of appetite evaluation. The postprandial effect on insulin levels was flat with the monk fruit-sweetened beverage. In contrast, there was a significantly higher spike with the sucrose-sweetened beverage. In conclusion, monk fruit does not increase glucose levels in individuals with T2DM; thus, it could be a safe alternative as a substitute food sweetener.
Author Contributions
Academic Editor: Kavitha Menon, Public Health Foundation of India| Indian Institute of Public Health Gandhinagar (IIPHG) Sardar Patel Institute Campus, Drive-In Road, Thaltej, Ahmedabad- 380 054, Gujarat, India.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2023 Fernando Javier Lavalle-Gonzalez, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Author note
Co-author credentials: Adriana Gabriela Rios-Ortega (MD); Leonardo G. Mancillas-Adame (MD); Alan Contreras-Arce (MD); Adan F. Díaz-Hernández (MD).
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Diabetes mellitus is a common disease and one of the biggest public health problems worldwide. It is currently an important cause of mortality and reduced life expectancy, with a prevalence that has increased in recent decades. The reported prevalence of DM in 2017 was 476,000 per 1000 inhabitants 1. The International Diabetes Federation projects that the number of adults living with diabetes will increase from 537 million to 783 million by 2045 if effective prevention methods are not adopted. 2
One of the main strategies for preventing and controlling diabetes is to moderate the consumption of high-calorie and high-carbohydrate foods. Non-nutritive sweeteners are an alternative for reducing the intake of simple carbohydrates due to their low caloric content 3.
Eight non-nutritive sweeteners have been approved by the US Food and Drug Administration 4. Siraitia grosvenorii, commonly known as "monk fruit," is one of two natural NNS approved by the FDA. It is a perennial vine of the Cucurbitaceae family, native to southern China. The sweetening effect of S. grosvenorii is up to 500 times greater than sucrose or common sugar 5. The compounds responsible for the sweetening effect are glycosylated cucurbitan-type triterpenes and mogrosides, which are not absorbed in the upper gastrointestinal tract and do not add calories to our diet 6. There has been a significant increase in international demand in the last few years; however, little is known about its effect on glycemic excursion in humans. Only a few studies have been conducted, and these have been performed in healthy young males 7, 8. No evidence is available on monk fruit use in patients with T2DM. We conducted a prospective, randomized, double-blind clinical trial to assess the effect of monk fruit on glucose excursion in individuals with and without type 2 diabetes.
Material and Methods
Participants
The study protocol was approved by the Institutional Research and Ethics Committee of the "Dr. José Eleuterio González" University Hospital. Each volunteer provided written informed consent before the study procedure.
Individuals from the Endocrinology outpatient clinic of the "Dr. José E. González" University Hospital were invited between November 2021 and June 2022 to participate. Subjects were divided into two groups. Eligible patients for the case group were adults ≥18 years of age with previously diagnosed T2DM according to the criteria of the American Diabetes Association 9, a recent HbA1c between 7.0 - 10%, and the absence of any acute or chronic complications. Individuals with fasting capillary glucose ≥200 before the OGTT, pregnant women, or with insulin treatment were excluded. Eligible individuals for the control group were adults ≥18 years of age without diabetes. Individuals with fasting capillary glucose ≥100 before the OGTT or pregnant women were excluded. No distinction was made regarding gender, and patients who did not complete the protocol were eliminated.
Methods
Eligible individuals underwent a physical exam and a full clinical history. Height, weight, waist circumference, hip circumference, and blood pressure were measured at the initial visit. The clinical history was intended to assess data concerning demographic characteristics, comorbidities, medications, and paraclinical variables.
Two OGTTs were performed on all participants with a 15- to 30-day period between sessions. They received a 75-g glucose load or a 0.51-g monk fruit load in a random manner. A blinded randomization procedure was designed to assign the order of the sweetener used. The participants were advised to fast for 8-12 hours the night before the test and to skip their antihyperglycemic drugs the morning before the test. A 250-ml glass of lemonade sweetened with either sucrose or monk fruit was given to the patient in a covered recipient. Blood samples were taken via venous punctures at baseline (0 min) and 30, 60, 90, and 120 minutes after receiving the glucose or monk fruit load. Ten individuals within the healthy group were randomly assigned through a random number table to measure insulin levels from the same blood sample at 0, 30, 60, 90, and 120 minutes. The blood samples were further analyzed by spectrophotometric analysis using the glucose oxidase technique (Byosystems® model BTS350 Spectrophotometer). A capillary glucose test was performed before the OGTT to guarantee the subject's safety in the T2DM individual group; if the result was ≥200 mg/dl, the individual was excluded from the study. We also monitored symptoms such as nausea, vomiting, abdominal pain, and consciousness. The patient received care if any signs or symptoms occurred during or after the protocol. Participants were simultaneously asked to rate their appetite before and 2 hr after the OGTT using an approved analog visual scale composed of 100 mm lines and 8 questions ranging from “not at all” to “very much.” Subjects were asked to make a mark across the line corresponding to their feelings. The response was quantified by measuring the distance from the left end of the line to the mark 10. (Supplementary Table 1).
Objective
The primary objective was to compare the glycemic response between sucrose and monk fruit in individuals with T2DM. The secondary objectives were to compare the insulin response to monk fruit and sucrose in healthy individuals and assess differences in appetite changes with both sweeteners.
Statistical Analysis
We used the data from SL Tey et al.'s study 8 to calculate the minimum sample for a significance level of 0.05, and a power of 80%, was 20 participants per group. Variables were tested for normality using the Kolmogorov-Smirnov test. The median and interquartile range (IQR) are reported as central tendency and dispersion measures for continuous variables. The Mann-Whitney U test for independent samples was performed to determine glucose distributions at baseline to compare across the sweeteners. A Kruskal-Wallis test compared the responses at 0, 30, 60, 90, and 120 minutes to the two sweeteners. Friedman’s Two-Way Analysis of Variance by Ranks was performed. A p-value ≤ 0.05 was considered significant for all the inferential tests.
Results
The study included 29 individuals in the control group and 26 in the T2DM group. The mean age was 29 years (IQR 26-31) in the control group and 50 (IQR 37-55) in the T2DM group. The female population represented 48% and 61%, respectively. The mean BMI6 was 25 kg/m2 (IQR 21-27) in the control group and 34.1 kg/m2 (IQR 32-38) in the T2DM group. The mean HbA1c in the T2DM group was 7.3% (IQR7-10). Eighty-five percent were treated with metformin, 15% with sulfonylurea, 9% with iDPP4, and 12% with iSGLT2. None of the participants used thiazolidinediones or GLP-1 analogs. (Table 1).
Table 1. General Baseline Characteristics and Anthropometric Measurements of participants.| T2DM n=26 | Control n=29 | |
| Female sex (%) | 61.5 | 48.3 |
| Age (year) * | 50 (37.0-55) | 29.0 (26.5-31.5) |
| Body weight (kg) * | 89.5 (79.7-97.6) | 69.8 (62.2-79.2) |
| BMI (kg/m2) †* | 34.1 (32.3-38.1) | 24.9 (21.7-27.3) |
| Waist circumference (cm) * | 108.5 (101.5-116.2) | 84.0 (75.6-91.2) |
| Systolic blood-pressure (mmHg)* | 130.0 (117.5-142.5) | 110.0 (110.0-120.0) |
| Diastolic blood-pressure (mmHg)* | 80.0 (71.5-88.5) | 75.5 (70.0-80.0) |
Comparison of Glucose Levels in monk fruit OGTT vs. sucrose-OGTT
a) T2DM Group:
The baseline glucose levels were not different between the two OGTTs (sucrose 116.5 mg/dL (IQR 99-136) and monk fruit 116.5 mg/dL (IQR 103-138) p=0.58). Figure 1 shows the glucose levels in response to the two sweeteners used. The sucrose-OGTT showed a peak maximum glucose response at 90 min (Median, IQR) of 247 mg/dL (203-293), while the monk fruit-OGTT showed stable glucose levels from 0-120 minutes without a peak response. Glucose levels at 30, 60, 90, and 120 min with the monk fruit-OGTT were significantly lower than the sucrose-OGTT. (Figure 1). Additionally, there was also a difference in the AUC7 with Monk fruit with a median of 226.3 (IQR 203-267) vs. AUC with sucrose 417.8 (IQR 365-492) (p<0.001). (Supplementary Table 2).
Figure 1.Blood glucose response to the monk fruit and sucrose-OGTT in the Type 2 diabetes mellitus group.
b) Control Group:
The results were similar to those observed in the T2DM group. Baseline glucose levels were not different between sucrose and monk fruit-OGTT (p=0.29). Similarly, during the monk fruit-OGTT, stable glucose levels were observed without a maximum peak response, unlike the OGTT with sucrose, in which a peak maximum glucose response was observed at 30 min (median 135 mg/dL; IQR 115-155). Glucose levels at 30, 60, 90, and 120 min and the AUC were significantly lower with the monk fruit-sweetened beverage vs. the sucrose-OGTT (Figure 2 and Supplementary Table 2).
Figure 2.Blood glucose response to the monk fruit and sucrose-OGTT in the control group.
Insulin Levels in the Sucrose vs. Monk Fruit OGTT in healthy individuals
Baseline serum insulin levels were no different between the sweeteners (Figure 3). The OGTT with the monk fruit-sweetened beverage, as with glucose, had a flat response with stable insulin levels without a maximum peak response and lower levels than the sucrose-sweetened beverage. Conversely, the sucrose-sweetened beverage had elevated insulin levels with a peak response at 30 min, as was seen with the glucose levels. Also, the AUC was significantly lower with the monk fruit-sweetened beverage (Supplementary Table 3).
Figure 3.Insulin response to the monk fruit and sucrose-OGTT in the control group.
Appetite Evaluation.
a) T2DM Group:
The appetite analysis before and after the monk fruit and sucrose-sweetened beverage-OGTT was performed within group and intergroup. No difference was observed in the results of the different points evaluated using the visual analog appetite scale. A decrease in the desire to eat sweet foods after the sweetened beverage was observed with both sweeteners; however, it was not statistically significant compared to the evaluation before the beverage (monk fruit p=0.079 and sucrose p=0.153). There were no differences between sweeteners regarding hunger and appetite (Figure 4a, 4b, 4c, 4d).
Figure 4a.Appetite scores before and after monk fruit-OGTT in the T2DM group. 4b. Appetite scores before and after sucrose-OGTT in the T2DM group. 4c. Scores for desire for specific types of food before and after monk fruit-OGTT in the T2DM group. 4d Scores for desire for specific types of food before and after sucrose-OGTT in the T2DM group.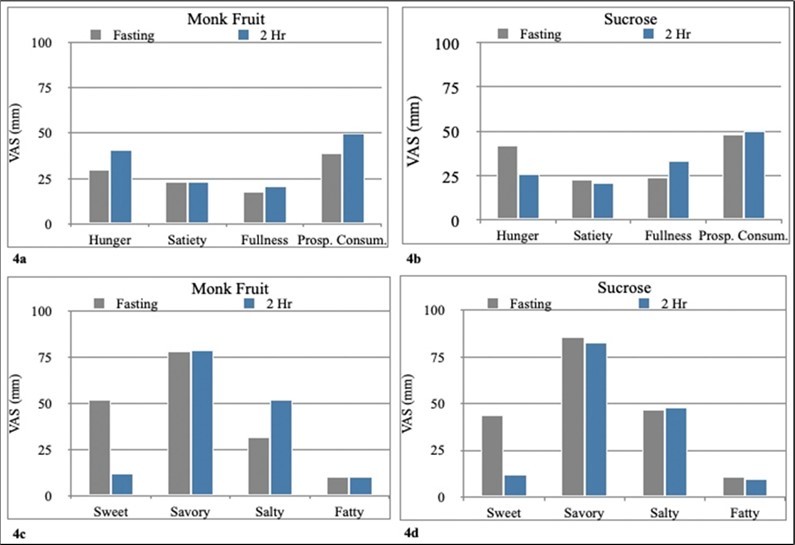
b) Control Group:
In contrast to the group with T2DM, the control group showed less desire for appetizing or eating salty foods after the sucrose-OGTT than the monk fruit-OGTT (Figure 5a, 5b, 5c, 5d). No differences were observed between both sweeteners regarding hunger, appetite, or desire to consume sweet foods.
Figure 5a.Appetite scores before and after monk fruit-OGTT in the control group. 5b. Appetite scores before and after sucrose-OGTT in the control group. 5c. Scores for desire for specific types of food before and after monk fruit-OGTT in the control group. 5d Scores for desire for specific types of food before and after sucrose-OGTT in the control group.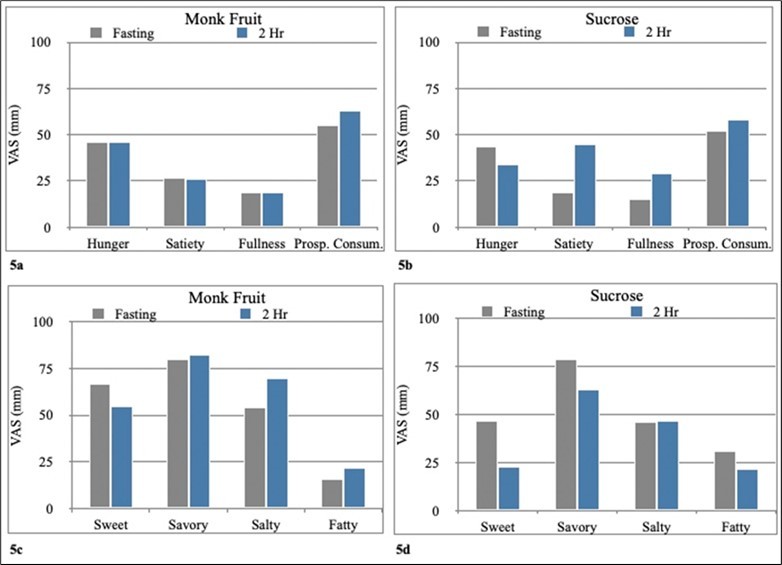
Discussion
This prospective randomized, double-blind clinical trial compared the effect of monk fruit and sucrose-sweetened beverages on postprandial glucose in patients with T2DM and non-T2DM volunteers. Our study is the first to evaluate the glycemic response of monk fruit in individuals with T2DM and without DM, regardless of the individual’s BMI. In this study involving individuals with T2DM and a control group, the monk fruit oral glucose tolerance test demonstrated significantly lower glucose levels at 30, 60, 90, and 120 minutes compared to the sucrose oral glucose tolerance test. In addition, with the sucrose-sweetened beverage, a peak maximum glycemic response was observed at 90´ in the T2DM group and 30´ in the control group, unlike the monk fruit-sweetened beverage intervention, with which glucose levels remained steady with a flat response and a tendency to decrease compared to basal glucose in both groups.
Our results agree with previous studies. Tey et al. 7 examined the impacts of consuming preloads that were sweetened with sucrose, an artificial non-nutritive sweetener (aspartame), and natural non-nutritive sweeteners (monk fruit and stevia) in healthy male individuals. They reported a maximum glycemic response peak 30 minutes after consuming sucrose-sweetened beverages, with no elevation of glucose levels observed 60 min after a monk fruit-sweetened beverage. These trial findings align with our findings, which support monk fruit as a safe alternative food and beverage sweetener.
Another study 8 that evaluated glucose levels by CGM on the consumption of a monk fruit-sweetened beverage during the day concluded that there was no difference in mean glucose or 24-hour AUC between sucrose and NNS; however, it was assessed with a single beverage in 24 hours without limiting the carbohydrate or calorie intake, which is one of the objectives of the use of NNS, to replace the consumption of foods high in carbohydrate, mainly simple carbohydrates, as part of a comprehensive diet without caloric compensation for decreases in energy intake which allows weight loss and improves glycemic control, both important parts of diabetes nutrition therapy 11. Thus, assessing greater exposure to the NNS and a real substitution of sucrose with NNS over the long term would be useful.
One of the major concerns about using NNS is that they may increase the risk of cardiometabolic and endocrine diseases such as obesity and T2DM; however, this remains a topic of debate. The proposed mechanism is that sweet-tasting compounds, including caloric and non-caloric sweeteners, activate the heterodimeric sweet taste receptor T1R2/T1R3, including pancreatic or intestinal sweet taste receptors that lead to insulin or glucagon-like peptide 1 release. It has thus been hypothesized that the sensation of sweetness without the delivery of calories via NNS may disrupt appetite regulation and impair metabolic signaling, leading to increased energy intake and weight gain 12; though, this concept is supported primarily by rodent studies. Monk fruit, despite its increasing popularity as a sweetening agent, has received comparatively less attention in the scientific literature when compared to other NNSs concerning its effects on human health. Some observational studies have found a link between NNS and T2DM development 13, 14. However, in a meta-analysis evaluating only RCTs, it was concluded that neither of the NNS was shown to be associated with elevated blood glucose levels 13, 15.
Nonetheless, owing to the distinctive structures and metabolic activities inherent in each sweetener, be it artificial or natural, it is not feasible to extrapolate the metabolic and endocrine effects of a singular artificial sweetener to all other artificial sweeteners 16. Only one study has evaluated the postprandial insulin response in healthy men volunteers to monk fruit. Tey et al. 7 found large spikes in insulin responses within the first hour following sucrose-sweetened beverage consumption, whereas no such changes were seen with monk fruit intake; a limitation of this study was the methodology for measuring insulin levels (capillary insulin levels). Our study measured serum insulin levels in ten individuals without DM, including a spectrum or BMI and both genders. Our findings showed a flat insulin response with the monk fruit-sweetened beverage; conversely, the sucrose-sweetened beverage showed a spike in insulin levels with a peak response at 30 min. Our findings are consistent with a recent systematic review and network meta-analysis, which concluded that NNS-sweetened beverages did not affect postprandial glucose, insulin, GLP-1, GIP, PYY, ghrelin, and glucagon responses 17.
The controversial issue surrounding the potential link between non-caloric sweeteners and increased appetite, which could lead to overcompensation in caloric intake, has been the subject of extensive investigation. A study involving monk fruit found that food desire and hunger were higher after consuming NNS preloads compared to a sucrose preload; however, there was no evidence of excessive caloric intake during titration of the 24-hour intake with the consumption of NNS 7. We used a validated questionnaire to assess appetite before and after each OGTT (Supplementary Figure 1). Unlike the previously described results, in the group of patients with T2DM, no differences were observed between both sweeteners in terms of appetite assessment, without a greater desire for food intake with monk fruit. In both groups, a tendency to decrease sweet food consumption was statistically insignificant.
In the control group, a preference for the consumption of appetizing and salty foods was observed after using monk fruit compared to sucrose; however, no increase or differences in appetite were observed between both sweeteners. These results favor consuming monk fruit, which, together with the beneficial effect on glucose excursion, does not seem to have a compensatory effect on appetite without causing excessive food intake.
One of the key strengths of our study includes a prospective study design, along with the inclusion of a larger patient population. Additionally, it is the first study to evaluate individuals with T2DM and without DM, irrespective of their BMI. Certainly, our study has some limitations, other potential confounding factors might affect our results and were not accounted for, such as body weight, the use of antihyperglycemic medication, and insulin resistance. However, the sucrose-OGTT demonstrated the typical pattern of glucose excursion seen in patients living with T2DM, even in the presence of antidiabetic medication. we found that despite the results that favor the use of monk fruit, the sweeteners were evaluated in a single sample; thus, their long-term effect cannot be concluded.
Conclusions
In conclusion, our findings suggest that monk fruit, as a non-caloric natural sweetener, is an alternative for individuals living with T2DM as an adjuvant to decrease carbohydrate consumption since it does not raise postprandial glucose levels. In addition, it does not increase appetite or hunger. On the other hand, we observed that monk fruit intake has no metabolic effect on postprandial insulin levels in healthy individuals. Our study establishes a foundation for future studies that focus on the long-term safety evaluation of monk fruit use.
Supplementary Material
Acknowledgments
The authors would like to thank BSc Carlo Bonilla by his help on performing the laboratory assessments and Sergio Lozano MD for manuscript edition review.
Keyterms
1Food And Drug Administration
2Non-nutritive sweeteners
3Type 2 Diabetes Mellitus
4Diabetes Mellitus
5Oral Glucose Tolerant Test
6Body mass index
7Area under the curve
Affiliations
1Endocrinology Service, Department of Internal Medicine, Facultad de Medicina y Hospital Universitario “Dr. José Eleuterio González”, Universidad Autónoma de Nuevo León, Monterrey, Mexico
References
- 1.GBD. (2017) Disease and Injury Incidence and Prevalence Collaborators. , Lancet; 392(10159), 10-1016.
- 2.Cho N H, Shaw J E, Karuranga S, Huang Y, JD da Rocha Fernandes. (2018) IDF Diabetes atlas: global estimates of diabetes prevalence for 2017 and projections for. DOI: 10.1016/j.diabres.2018.02.023. Diabetes Res Clin Pract 131, 271-281.
- 3.Laviada-Molina H, Almeda-Valdés P, Arellano-Montaño S, Gómez-Llanos A, Cervera-Cetina M. (2017) Posición de la Sociedad Mexicana de Nutrición y Endocrinología sobre los edulcorantes no calóricos. Rev Mex Endocrinol Metab Nutr. , (Internet) 4, 24-41.
- 4.U S Food, Administration Drug. (2018) Additional Information about High-Intensity Sweeteners Permitted for Use in Food in the United States.
- 5.Soejarto D D, Addo E M, Kinghorn A D. (2019) Highly sweet compounds of plant origin: From ethnobotanical observations to wide utilization. DOI: 10.1016/j.jep.2019.112056. , Journal of Ethnopharmacology 243, 112056.
- 6.Murata Y, Ogawa T, Suzuki Y A, Yoshikawa S, Inui H. (2010) Digestion and absorption of Siraitia grosvenori triterpenoids in the rat. DOI:. 10-1271.
- 7.Tey S L, Salleh N B, Henry J, Forde C G. (2017) Effects of aspartame-, monk fruit-, stevia- and sucrose-sweetened beverages on postprandial glucose, insulin and energy intake. DOI: 10.1038/ijo.2016.225. , Int J Obes (Lond) 41-3.
- 8.Tey S L, Salleh N B, Henry C J, Forde C G. (2017) Effects of non-nutritive (artificial vs. natural) sweeteners on 24-h glucose profiles. DOI: 10.1038/ejcn.2017.37. Eur J Clin Nutr 71-9.
- 9. (2021) Classification and Diagnosis of Diabetes:. Standards of Medical Care in Diabetes-2021. DOI: 10.2337/dc21-S002. Diabetes Care 1. 44(Suppl 1),S15-S33 , American Diabetes Association 2.
- 10.Flint A, Raben A, Blundell J E. (2000) Astrup A. DOI: 10.1038/sj.ijo.0801083. Int J Obes Relat Metab Disord 24(1).
- 11.Alison B Evert, Dennison Michelle, Christopher D Gardner, W Timothy Garvey. Ka Hei Karen Lau (2019) Nutrition Therapy for Adults With Diabetes or Prediabetes: A Consensus Report. , DOI:, Diabetes Care 42(5), 10-2337.
- 12.Sylvetsky A C, Rother K I. (2018) Non-nutritive Sweeteners. in Weight Management and Chronic Disease: A Review. DOI: 10.1002/oby.22139. Obesity 26-4.
- 13.Manavalan D, Shubrook C, Young C F. (2021) . Consumption of Non-nutritive Sweeteners and Risk for Type 2 Diabetes: What Do We Know, and Not? DOI: , Curr Diab Rep 21(12), 10-1007.
- 14.Fagherazzi G, Gusto G, Affret A, Mancini F, Dow C. (2017) Chronic consumption of artificial sweetener in packets or tablets and type 2 diabetes risk: evidence from the E3N-European prospective investigation into cancer and nutrition study. DOI: 10.1159/000458769. Annals of Nutrition and Metabolism 70-1.
- 15.Huang M, Quddus A, Stinson L, Shikany J, Howard B. (2017) Artificially sweetened beverages, sugar-sweetened beverages, plain water, and incident diabetes mellitus in post- menopausal women: the prospective Women’s Health Initiative observational study. DOI: 10.3945/ajcn.116.145391. , Am J Clin Nutr 106-2.
