Quercetin Attenuates Manganese-Induced Neurotoxicity in Mice Hippocampus by Regulation of Oxidative Stress and Tumour Necrosis factor Alpha Expression
Abstract
The hippocampus is involved in learning and memory processes, an integral component of cognitive function. The aim of this study is to assess the efficacy of quercetin on manganese-induced neurotoxicity in the hippocampus of the adult mice.
In this study, 40 adult mice of average weight of 18 –29g were randomly distributed into five groups of eight each. The brain was harvested and the region of the hippocampus was grossed for histological and immunohistochemical analysis.
The results revealed a significant increased level of oxidative stress markers of manganese treated mice when compared with the normal control and quercetin treated animals (p<0.05). Immunohistochemical analysis also showed a decrease expression of Tumour necrosis factor alpha (TNFα) with quercetin treated animals when compared with manganese treated animals indicating its neuroprotective function. In addition, quercetin treated animals all had an improved working spatial memory in Y-maze test. The histological results also revealed a degeneration of pyramidal cells with a characteristic pyknotic activities at the granular layer of the hippocampus leading to neuronal integrity damage following chronic exposure to manganese but normal architectural design was however maintained with quercetin.
Conclusively, exposure to manganese in excess may have adverse effect on extensive neuronal degeneration that could affect the learning, memory and possibly spatial navigation ability of miceand quercetin attenuates this induced neurotoxicity via inhibition of oxidative stress and reduction of TNF expression.
Author Contributions
Academic Editor: Manal ElSawaf, anta University, al-Gaish Street, Tanta, Gharbia, Egypt.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2020 Adeshina Oloruntoba Adekeye, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Neurotoxicity refers to a kind of toxicity by which different agents such as biological, chemical, or physical could cause an adverse effect on the anatomy of the central and peripheral neurons 1. It could be as a result of exposure to a substance known as neurotoxin to alter the normal structure of the nervous system and this might result into damage to nervous tissue which could perhaps destroy neurons 1. Exposure to other heavy metals and chemicals such as pesticides in the environment may lead to neurotoxicity 2. Neurodegeneration is the continuous process of losing the structure or function of neurons which can lead to death of neurons and these have a way to affect person’s movement and cognitive function 3. Neurodegenerative diseases are complex type of diseases that involves neurodegeneration in different areas of the brain. These diseases are characterized by a progressive degeneration of the both structure and function of the central nervous system. Most therapy adopted for neurodegenerative diseases such as Parkinson’s diseases and Alzheimer’s disease have been reported not to be completely efficient in the management of the disorders thereby leading into a state of progressive degeneration and death of neurons.
Manganese is an essential trace element present naturally in foods such soybeans, leafy vegetables, coffee and available as a dietary supplement and also a cofactor for many enzymes 4, 5. Evidence have shown that 1-100mcg/L of water contains small amounts of manganese 6. Evidence has not shown that high dietary manganese intakes could cause manganese toxicity 7. However, manganese toxicity has been reported in an individual working as welders and miners or those exposed to high amounts of manganese as result of chronic inhalation of manganese dust 8. Manganese toxicity is known to affects the central nervous system resulting into impaired fine motor and cognitive functions especially those related to memory and learning 8. However, the neural mechanism of alteration in hippocampus by manganese to affect cognition is still unclear, hence the reason for the study to understand the efficacy of quercetin on manganese-induced neurotoxicity.
Quercetin is grouped as one of the six subclasses of flavonoid compounds found in fruits and vegetables 9. Evidences revealed that quercetin have a therapeutic potential for the prevention of different diseases such as cardiovascular disease and neurodegenerative diseases 9, 10. However, quercetin has been shown to affect its antioxidant and anti-inflammatory properties in several areas of animal models as well as in humans by upregulating the signalling pathways via inhibition of oxidative stress markers and apoptotic factor expression involved 9, 10. For this present study, quercetin has been used on animal model to investigate its antioxidant activities on the cell death induced by excessive consumption of the manganese.
Materials and Methods
Experimental Animals
40 adult male BALB/c mice weighing 18-29g were purchased from Tolex Animal holding, Ekiti State, Nigeria. Approval was sought and obtained from the ABUAD Ethical committee on animal use. The animals were housed in well-ventilated cages and maintained under standard laboratory condition of temperature, humidity and light. The animals were allowed to acclimatize for two weeks and fed with rodent meal (ADEHEZ Rodent Feed®, Ilorin, Nigeria) and drinking water was made available ad libitum. After the acclimatization period, they were reweighed and randomly assigned to the five groups of eight (8) mice each. All animal experiment for the study complied with the ARRIVE guidelines 11 and was carried out in accordance National Institutes of Health guide for the care and use of Laboratory animals.
Other materials used for this study include:Quercetin were purchased from Medchem express, Manganese (II) chloride (Mncl2.4H2O) tetrahydrate, Sodium hydrogen Phospahte (Na2HPO4), Potassium chloride (KCL), Potassium dihydrogen phosphate (KH2PO4), Ketamine and all consumable used were purchased from Kermel laboratory (Colmar, France). The water used was double-distilled and other reagents were of analytical grade.
Preparation of Quercetin and Manganese
Quercetin was purchased from Medchem express while Manganese chloride was purchased from Kermel laboratory (Colmar, France). 10% DMSO was prepared by diluting 100ml of DMSO and 900ml of phosphate buffer solution (PBS). 400mg of Quercetin was then dissolved in 33.6ml of 10% dimethyl sulphur oxide (DMSO). The Manganese chloride was dissolved in distilled water;
Experimental Design
The mice were randomly divided into five (5) groups of 8 mice each. The first group represent the control group (C) that received normal saline for the duration of 14 days. The second group (Mn) received manganese (40mg/kg) for the 7days 12, 13. The third group (Q) received quercetin (50mg/kg) for 7 days. The fourth group (POST) was pre-treated with manganese (40mg/kg) followed by quercetin (50mg/kg) administration 14. The fifth group (CONC) received both manganese and quercetin concurrently for 7 days. (Mn: 40mg/kg; Q:50mg/kg). All administration was done by intraperitoneal route.
Animal Sacrifices
The mice that were set out for histological analysis were sacrificed by anaesthetized using ketamine followed by transcardially perfusion fixation (using 0.9% sterile saline followed by 4% paraformaldehyde) while the animals set out for biochemical analysis were sacrificed through cervical dislocation. The brain was harvested and the region of hippocampus was grossed for histological and immunohistochemical analysis.
Behavioural Analysis
The behavioural test was carried out with proper illumination and sound control in the behavioural room of Afe Babalola University. The behavioural test was conducted after the last administration. The behavioural test carried out were the Y-maze and the NOR (novel object recognition) test to assess learning and memory function, an integral component of cognitive behaviour in the experimental animalsandthe tests were recorded and properly documented.
Y-Maze Test
Y-maze test was carried out to investigate the spatial working memory of the animals based on the procedure used by Adeniyi et al., (15). The mice were placed in the middle of the Y-box and allowed their arm decision for 5 mins. The correct alternation was (ABC, ACB, BAC, BCA, CAB, CBA) between the arms (ABC) was video recorded and analysed later to determine the memory index which is the percentage of normal alternation. Animals with lower percentage alternation are likely to have lower spatial working memory index and vice versa.
Novel Object Recognition Test
The Novel object recognition test (NORt) was also carried out to evaluate cognition, particularly recognition memory and the test was done in a controlled arena. The objects used were similar in height and volume, but were different in appearance (colour). The frequency of times the animals recognized the new objects were video recorded with tally method.
Biochemical Analysis
Biochemical analysis was done using the whole brain tissue homogenate to assay the activities of malondialdehye and superoxide dismutase to quantitatively evaluate oxidative stress.
Photomicrography
Microscopy was conducted with a 5.1-megapixel MV550 research microscope connected to a computer and images captured and analysed.
Statistical Analysis
Statistical analysis was done using one-way analysis of variance (ANOVA) and differences between groups were evaluated using Newman-keuls for post hoc tests with the aid of GraphPad Prism V.5.0. The data statistical analysis was represented in graphs and bar charts with standard error of the mean (mean ± SEM) and significant level was set at *P< 0.05.
Results
Morphological Changes
There was a significant morphological change observed across the experimental groups. Quercetin (Q) and normal saline (C) treated mice revealed some level of positive changes in average body weight (26 to 32 g) but manganese treated mice experience a kind of decline in body weight (24 to 19g). The post treatment group (intervention) showed little or no changes in body weight. There has been no significant change in fur colour and colour of the eyes in all the groups.
Biochemical Assay
Lipid Peroxidation (MDA) and Superoxide Dismutase (SOD)
Statistical changes in the markers of oxidative stress in terms of Malondialdehyde (MDA) and SOD activities were observed (***P<0.001) from Figure 1 and Figure 2. Manganese-treated mice showed a significant increase of Malondialdehyde activities (MDA), which is an evidence of oxidative stress and this was invariably decrease with the quercetin administration as shown in Figure 1 when compared with the control group. Also, Activities of superoxide dismutase were significantly increased by quercetin-treated mice when compared with other experimental groups (Figure 2).
Figure 1.Concentration of MDA (µ/ml) in the brain tissue. There is a significant change observed when control and quercetin treated mice were compared with other experimental mice with manganese. Quercetin protects neurotoxicity by inhibition of markers of oxidative stress via reduction of lipid peroxidation. Control (C); Manganese (Mn); Quercetin (Q); Intervention (POST); Concurrent (CONC); Malondialdehyde (MDA) *P-value< 0.0001.
Figure 2.Concentration of SOD (µ/ml) in the brain tissue homogenate. Activities of superoxide dismutase were significantly increased by quercetin treated mice when compared with other experimental groups. Control (C); Manganese (Mn); Quercetin (Q); Intervention (POST); Concurrent (CONC); Superoxide Dismutase (SOD) **P<0.01, ***P< 0.001.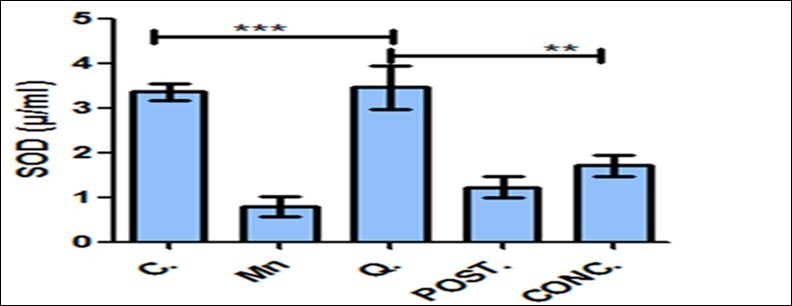
Behavioural Studies
Memory index was assessed in the experimental animals using NOR and Y-maze test.
Novel Object Recognition (NOR) Test and Y-Maze Test
These tests were carried out to assess the spatial working memory of the animals. The memory index for NORt was calculated using: Memory Index (%) = (T Novel) / (T Novel + T Familiar) x 100% (Figure 3). Quercetin improve spatial working memory with an evidence of statistical increase in memory index during novel object recognition test of all experimental groups that received quercetin when compared with the manganese-treated and normal control group in Figure 3 (*P<0.05, **P<0.01 and ***P<0.001). The percentage alternation as indices for memory index in Y-Maze test was calculated as: % Alternation = No. of right decisions / No. of total arm entries-2 ×100. This was indicated by higher alternation for the quercetin treated mice compared to control mice, although, manganese treated mice showed a decrease percentage alternation but no statistically significant changes were observed (Figure 4).
Figure 3.Memory index function with Novel object recognition test. Quercetin improve memory index when compared with other experimental groups. Control (C); Manganese (Mn); Quercetin (Q); Intervention (POST); Concurrent (CONC); *P<0.05, **P <0.001, ***P<0.0001.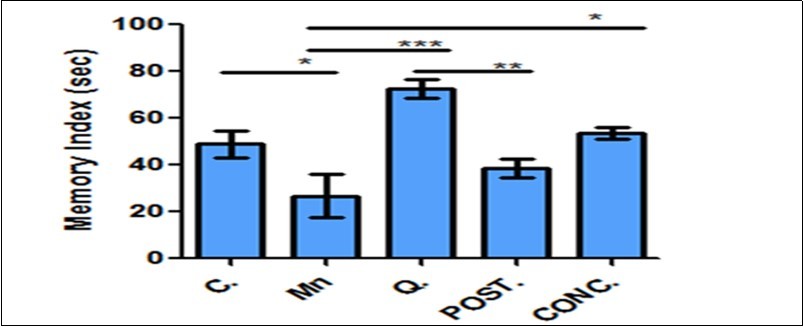
Figure 4.Percentage alternation as an indices of memory index in Y-maze test. Percentage alternation changes were observed across the groups but not significant based on the group comparison. Control (C); Manganese (Mn); Quercetin (Q); Intervention (POST); Concurrent (CONC); ns: not significant.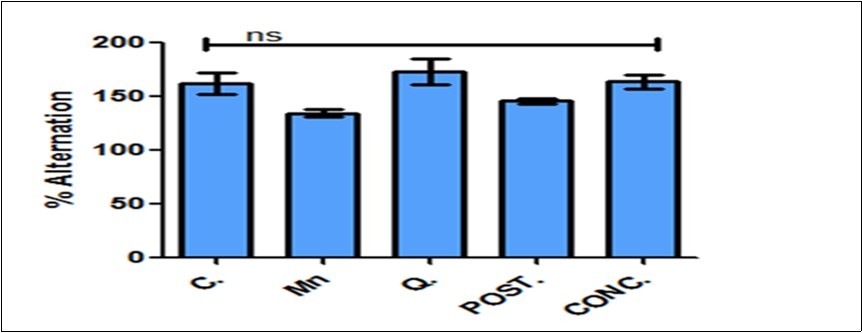
Histological Analysis
The hippocampus is divided structurally into hippocampus proper (cornu ammonis) and dentate gyrus with 3 cell layers and a remarkable distribution of densely packed pyramidal and other cells involved in encoding, organizing and storing memories. From the histological analysis in Figure 5, a distinct 3 marked layers of hippocampus with a normal distribution of pyramidal cells and a well stained nuclei were observed in normal control and quercetin treated group and the integrity of these neurons were also seen to be preserved around the dentate gyrus (DG) and cornu ammonis (CA1-CA3) with their principal cell layers known as granular and pyramidal layer respectively. As shown in Figure 5 with group that received Mn, the layers of the hippocampus there were shown to have histological alteration (Mn-treated mice). The pyramidal layer of the hippocampus was observed with extensive vacuolations of the cells and pyknotic appearance as compared to the control group, thus indicating neuronal cell death in the CA1 and CA2 region than CA3. The cells of the CA4 region, the hilus of DG and granular cell layer of DG, were least affected while the group that were received quercetin after manganese administration (intervention group) revealed mild neuronal death and co-treated animals (concurrent groups) revealed a very minimal vacuolation in normal appearance of the 3 distinct layers of hippocampus and neuronal distributions.
Figure 5.Photomicrograph of the dentate gyrus (DG) section in control (C) group, manganese (Mn) group, Quercetin (Q) group, post treatment (PT) group and concurrent (PT) group. Stained with H&E. Mg x800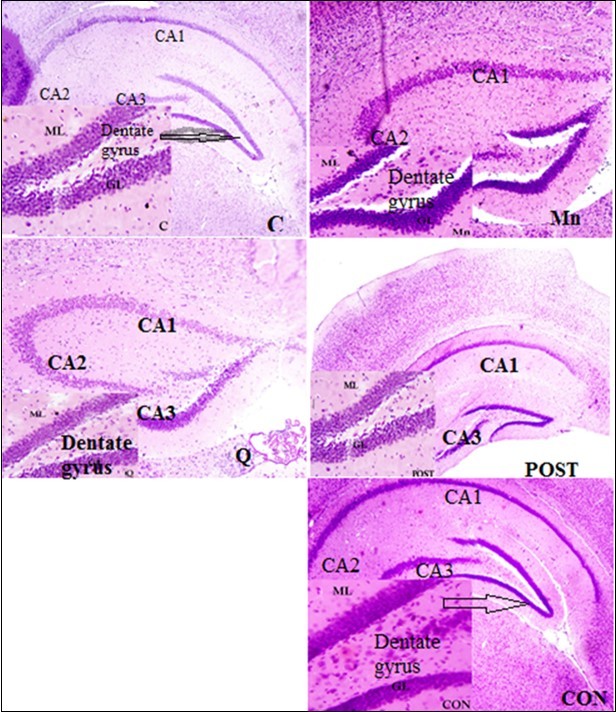
Immunohistochemistry (TNF-α)
Immunohistochemical results of hippocampal (dentate gyrus) section evaluating the ameliorative roles of quercetin following manganese-induced neurotoxicity. Normal control and quercetin treated mice revealed little or no expression of tumour necrosis factor alpha (TNF-α) in the dentate gyrus and cornu ammonis (CA1-CA3) but this was more pronounced in the Manganese-treated mice. However, other experimental animals that received quercetin recorded a minimal expression indicating the potential neuroprotective effect of quercetin during or after cell death. Figure 6.
Figure 6.TNF-α expression in there were no significant changes in fur and eye colour in all the groups throughout the experiment mice exposed to manganese chloride and quercetin. Control (C); Manganese (Mn); Quercetin (Q); Intervention (POST); Concurrent (CONC). The deep/dark brown precipitates indicate the expression of TNF-α. DG: dentate gyrus, CA: Cornu ammonis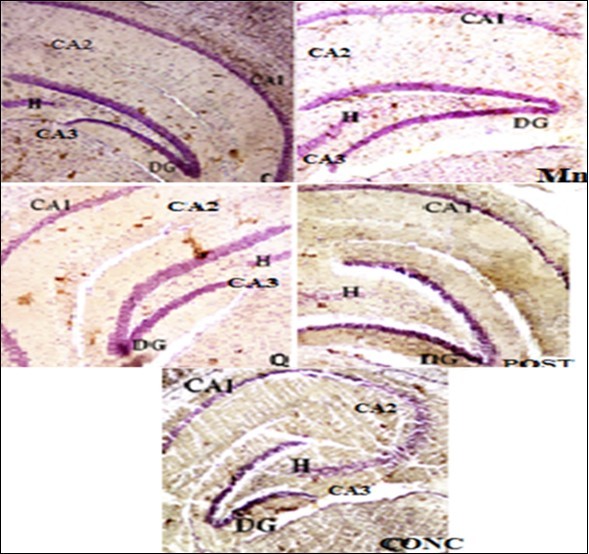
Discussion
The present study demonstrated the efficacy of quercetin on manganese-induced neurotoxicity in mice hippocampus. The weights of all the animals were taken into the consideration before grouping and experimentation. The normal control and quercetin treated mice showed an increase in weight gain but manganese-treated mice expressed a decline in size and weight indicating that quercetin enhances feeding efficiency. The post treated animals clearly showed a little or no changes in weight and size. However, there were no significant changes in fur and eye colour in all the groups throughout the experiment and this was supported by research done by Porwal et al. 5, 29. Oxidative stress increases the levels of reactive oxygen species (ROS) and give rise to lipid peroxidation that could have a deleterious effect on nervous tissues. An imbalance between oxidants and antioxidants form the underlying basis of oxidative stress leading to many pathophysiological conditions in the body, some of these include neurodegenerative diseases such as Parkinson's disease and Alzheimer's disease 6, 11, 12. Neurodegenerative diseases can also occur as result of a faulty metabolic process leading to a release of ROS and lipid peroxidation that affect cell membrane and other organelles to operate normally in terms of function 9. Also, excess manganese exposure in mice has been reported to induce neurotoxicity by generating reactive oxygen species 13, 14 and these free radicals cause oxidative stress that result in brain neuronal damage 10, 14. Malondialdehyde level is commonly known as a marker of oxidative stress. In the biochemical assay, malondialdehyde (MDA) concentration was done in this study to evaluate the level of lipid peroxidation as a marker of oxidative stress in cellular activities. Lipid peroxidation is considered as a major pathway of cell injuries in aerobic mechanism subjected to oxidative stress and the reaction sequences of lipid peroxidation increases the number of free radicals in the cells which leading to a cell death 10. Significant increase in oxidative stress in all the mice treated with manganese were observed and vice versa with mice treated with quercetin either with intoxicant or quercetin alone (Figure 1). This make us to confirmed indeed that oxidative stress via lipid peroxidation may cause cellular and molecular damage with increases reactive oxygen species (ROS) which further leads to tissue destruction where fragmentation of DNA material from the nuclei are evident 12, 13, 15.
Behavioural studies carried out at the end of drug administration using the novel object recognition and Y-maze test showed that manganese significantly affects the performance of mice and intervention of quercetin improves the spatial memory. Animals administered with manganese alone showed significantly reduction in memory index with longer duration to recognize the novel object compared to familial object which might as a result of hippocampal damage. This indicates poorer learning and memory capacity (Figure 4). This supports earlier evidence that showed that excess Manganese and other neurotoxin negatively affects learning and memory by damaging hippocampal formation 16. Results from Y-maze analysis (Figure 4) also showed that with the normal control animals being the reference point, the quercetin treated animals had an improved spatial working memory in terms of percentage alternation, a function of memory index when compared with manganese-induced animals. This result implies that quercetin improve memory and learning while exposure to high amount of manganese could affect cognitive function and this was supported by results obtained byShiva et al., Costa et al, and Jian et al., 17, 18, 19. The novel recognition and Y-maze are used to study the psychological and neural mechanisms of spatial learning and memory. The present behavioural studies have shown that Mn negatively affects learning and memory and this indicates an injury to the hippocampus which is involved in spatial memory but quercetin positively improves the spatial learning and memory 20. The hippocampus of mice is now known to have some pyramidal neurons that help the animals navigate better through their environment as the firing of these cells indicates a specific location in the environment of the mice. Extensive destruction of hippocampal pyramidal cells by excess Mn consumption would have affected the ability of exposed mice to correctly navigate in the NOR and y-maze tests and these could also have contributed to their poor cognitive performances. Mice with damaged cells of the dentate gyrus and cornu ammonis as a result of manganese treatment showed a decrease in spatial memory and cognitive function during neurobehavioural test with novel object recognition and y-maze test and this was significantly increase with quercetin (Figure 3 & Figure 4). Histological findings also showed that normal histoarchitectural design of the hippocampus were preserved in the control group with a normal distribution of neuronal cells seen in the pyramidal cell layer of the hippocampus proper (cornu ammonis;CA1-CA3) and granular cell layer of the dentate gyrus of the hippocampus which could indicates normal spatial memory of the mice in these group. However, the manganese-treated group (Mn-treated mice) revealed higher destruction of cells with pyknotic nuclei in the CA1 and CA2 regions than the CA3 region of the pyramidal cell layer. The cells of the hilus of dentate gyrus were least affected by the effect of excess manganese (Figure 5). Pyknosis refers to the condensation of nuclear materials as a result of a cell necrosis or apoptosis 22, 23. Excess manganese exposure become neurotoxin and these can extensively damage the hippocampal neurons with a characteristic pyknotic nuclei and intact densely dark nuclear fragments to influence learning, memory and possibly spatial navigation ability of mice 13, 23.Therefore,histological results of this study clearly support the reports that excess Mn exposure extensively destroys hippocampal neurons and induces neurotoxicity in the brain 21. Excess Mn consumption produces an extensive neuronal degeneration in the hippocampus leading to learning and memory loss 21. Figure 5 also showed an evidence of quercetin treated mice in adult neurogenesis where there was a regeneration and repair of cells of hippocampus from damage caused by oxidative stress of the induced-manganese mice.
Tumour necrosis factor alpha (TNF alpha) represent an inflammatory cytokine produced by macrophages during inflammation involving signalling events within cells leading to programmed cell death 24, 27. In the present study, the normal control and quercetin-treated mice according to Figure 6 showed no expression of TNF-α while the manganese-induced mice presented the highest expression of TNF-α which implies that high amount of manganese exposure in the brain may lead to neuro-inflammation and neurodegeneration that could be protected by the antioxidant effect of the quercetin via inhibition of pro-inflammatory cytokine expression 25, 26, 27.
Conclusion
In conclusion, the efficacy of quercetin on Mn-induced mice on the hippocampus in this present investigation showed an improvement of the endogenous antioxidant defence mechanism by way of inhibition of reactive oxygen species generation and suppression of inflammation as well as reduction of expression of tumour necrosis alpha. Hence, quercetin may have beneficial health effects in Mn-exposed individuals on Cognitive function.
References
- 1.Cunha-Oliveira T, Rego A C, Oliveira C R. (2008) Cellular and molecular mechanism involved in the neurotoxicity of opioid and psychostimulant drugs. , Brain research 58, 192-208.
- 2.Firestone M, Moya J, Cohen H E, Zartarian V. (2007) Identifying childhood age groups for exposure assessments and monitoring. , Risk Anal 27, 701-14.
- 3.Neubauer A C, Fink A. (2009) Intelligence and neural efficiency.Neuroscience &BiobehaviouralReviews;. 33(7), 1004-1023.
- 4.Tran T T, Chowanadisai W, Crinella F M, Chicz-DeMet A, Lönnerdal B. (2002) Effect of high dietary manganese intake of neonatal rats on tissue mineral accumulation, striatal dopamine levels, and neurodevelopmental status. NeuroToxicology. 23, 635-643.
- 5.Cabrera-vique C, Briones M, Muros J, Seiquer I, Sanchez J et al. (2015) A pilot duplicate diet study on manganese, selenium and chromium intake in institutionalized children and adolescents from Guatemala. British journal of nutrition. 114(10), 1604-1611.
- 6.Finley J W, Penland J G, Pettit R E, Davis C D. (2003) Dietary manganese intake and type of lipid do not affect clinical or neuropsychological measures in healthy young women. , J Nutr 133, 2849-56.
- 7.Yamada M, Asakura K, Sasaki S. (2014) Estimation of intakes of copper, zinc, and manganese in Japanese adults using 16-day semi-weighed diet records. Asia Pac J Clin Nutr. 23, 465-472.
- 9.Jomova K, Vondrakova D, Lawson M, Valko M. (2010) Metals, oxidative stress and neurodegenerative disorders. , Mol Cell Biochem.345(1-2): 91-104.
- 10.Khan A, Ali T, Rehman S U, Muhammad T Ikram, Saeed K et al.. Neuroprotective Effect of Quercetin Against the Detrimental Effects of LPS in the Adult Mouse Brain. Front. Pharmacol. 2018;https://doi.org/10.3389/fphar.2018.01383 .
- 11.Ostadmohammadi V, Milajerdi A, Ayati E, Kolahdooz F, Asemi Z. (2019) Effects of quercetin supplementation on glycemic control among patients with metabolic syndrome and related disorders: A systematic review and meta-analysis of randomized controlled trials. Phytother Res. 33(5), 1330-1340.
- 12.Percie du SertN, Hurst V, Ahluwalia A, Alam S, Avey M T et al.Clark A et al. The ARRIVE guidelines 2.0: Updated guidelines for reporting animal research.PlosBiology. 2020-10.
- 13.Fafure A A, Adekeye A O, L A Enye, A, M et al. (2018) Ficus Exasperata Vahl improves manganese-induced neurotoxicity and motor dysfunction in mice.Anatomy. , Journal of 7(2), 1206-1219.
- 14.Adekeye A O, Adumah C, Fafure A A, Ajao M, Sabiu S et al.. Adefule AK. (2018).Evaluation of the Effects ofGutenbergiaNigritanaLeaves Extract on Cerebellum of Adult Mice and Its Implication on Manganese Toxicity.World Journal of Pharmacy and PharmaceuticalSciences.7(3): 55-68.
- 15.Adeniyi P A, Omatsuli E P, Akinyemi A J, Ishola A O. (2016) Caffeine plus nicotine improves motor function, spatial and non-spatial working memory and functional indices in BALB/c male mice.Pathophysiology.
- 16.Broening H W, Morford L L, Inman-Wood S L, Fukumura M, Vorhees C V. (2001) . 3,4-Methylenedioxymethamphetamine (Ecstasy)-Induced Learning and Memory Impairments Depend on the Age of Exposure during 21(9), 3228-3235.
- 17.Samina S. (2017) Oxidative Stress and the Central Nervous System. , Journal of Pharmacology and Experimental Therapeutics 360(1), 201-205.
- 18.Meyle J, Chapple I. (2000) Molecular aspects of the pathogenesis of periodontitis. Periodontol. 69(1), 7-17.
- 19.Ebrahimpour S, Esmaeili A, Beheshti S.Effects of quercetin-conjugated superparamagnetic iron oxide nanoparticles on diabetes-induced learning and memory impairment in rats.Int. , J Nanomedicine2018 13, 6311-6324.
- 20.Kesner R P, Exans R B, Hunt M A. (1987) Further evidence in support of the neurological basis of an attribute model of memory: role of hippocampus. , Int. J. Neurosci 21(22), 184-196.
- 21.Nwoha P U, Ojo G B, Ajayi S A, Ofusori D A, Oluwayinka O P et al. (2007) Garcinia kola diet provides slight neuroprotection to mice hippocampal neurons against neurotoxin. , J. Environ. Neurosci 1(2), 125-136.
- 22.Costa L G, Garrick J M, Roque P J, Pellacani C. (2016) Mechanisms of neuroprotection by quercetin: Counteracting Oxidative Stress and More.Oxid Med CellLongev.
- 23.Jiang W, Luo T, Li S, Zhou Y, Shen X et al. (2016) Quercetin Protects against Okadaic Acid-induced injury via MAPK and P13K/Akt/GSK3β signaling Pathways in HT22 Hippocampal Neurons. Plos One. 11-4.
- 24.Kroemer G, Galluzzi L, Vandenabeele P, Abrams J, Alnemri E S et al. (2009) . Nomenclature Committee on Cell Death. Classification of cell death: recommendations of the Nomenclature Committee on Cell Death 2009. Cell Death Differ 16(1), 3-11.
- 25.Kumasaka M Y, Yajima I, Ohgami N, Naito H, Omata Y et al. (2014) Brain deposition and neurotoxicity of manganese in adult mice exposed via the drinking water. , Arch Toxicol 88(5), 1185-6.
- 26.Idriss H T, Naismith J H. (2000) TNF alpha and the TNF receptor superfamily: structure-function relationship(s). Microsc Res Tech. 50(3), 184-95.
- 27.Nair M P, Mahajan S, Reynolds J L, Aalinkeel R, Nair H et al. (2006) The Flavanoid Quercetin inhibits Proinflammatory Cytokine (Tumor Necrosis Factor Alpha) Gene Expression in Normal peripheral Blood Mononuclear cells via modulation of the NF-kβ System. Clin Vaccine Immunol. 13(3), 319-328.
Cited by (1)
This article has been cited by 1 scholarly work according to:
Citing Articles:
Food and Chemical Toxicology (2025) Crossref
Parinaz Javanbakht, Afshin Talebinasab, R. Asadi-Golshan, Maryam Shabani, I. Kashani et al. - Food and Chemical Toxicology (2024) Semantic Scholar
