Survival Rate and Growth Performances on Pre-Grossing of the African Cyprinidae LabeobarbusBatesii (Boulenger, 1903) According to the Level of Dietary Protein
Abstract
In order to contribute to the domestication of Labeobabusbatesii, the survival and growth performances in pre-grossing according to the dietary protein level were studied between January and April 2020. A total of 400 fry (0.8 ± 0.02g), divided into four batches and distributed randomly in 8 happas (50 ind / m2), respectively received in duplicate the food at 25, 30, 35 or 40% crude protein (R25%, R30%, R35% and R40%). The results show that: survival rate and growth performances were significantly influenced (p≤0.05) by the level of dietary protein except for lengths (total, standard and length gain) whose highest values were obtained with the diet at 35% crude protein, the highest significant values of the other growth characteristics were obtained in the batch fed at 25% crude protein while the lowest were recorded on individuals of batches fed at 40% crude protein. Regardless of the level of food protein, the post-larvae presented an allometric minorant growth. So, 25% of dietary protein is therefore recommended for pre-grossing of this species.
Author Contributions
Academic Editor: Eman Hashem Radwan, Damanhour University, Egypt.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2020 Tonfack Achile Peguy, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Achieving the food security so coveted by the nations of the world in recent years requires the participation of a large number of activity sectors including aquaculture 1. This sector can be summed up in our regions to fish farming. In Cameroon, this sector contributes only less than 1% to meeting the fish needs of the populations 2. This is explained by the low fish production estimated at 1000 tons in 2003.
In addition, the promotion of fish farming requires mastering the technical route of breeding not only species of interest but also and above all endogenous species. Moreover, the development of fish farming in sub-Saharan Africa in general and in Cameroon in particular, has been the subject of many attempts from the 1950s and 1960s 3; 4. The majority of these attempts ended in failure, the major cause of which was the too short duration of the projects, which did not allow fish farmers to assimilate training on technological production in a still new sector 1. In addition, the species surveyed so far (Clariasgariepinus, Cyprinus carpio and Oreochromis niloticus) are mostly introduced species and therefore poorly adapted to our agro-ecological zones. Moreover, according to 5 the species introduced in Africa (common carp and Chinese carp) have not been able to stimulate the development of an aquaculture production sector. However, this continent is home to a diversity of freshwater fish species that is largely sufficient to support sustainable aquaculture development as in Asia. It is in this same vein that 6 adds by stating that the most harmful impact of the introduction of exotic species is probably to distract a certain number of countries from the attention paid to the aquaculture potential of native species. It is therefore clear facing all this facts that the domestication of indigenous species with high aquaculture potential would be a very promising alternative. Nowadays, the species of the Cyprinidae family are the highest in the world, accounting for 61% of production by volume 7; while almost no African cyprinid is currently used in fish farming 8. This production, based on the domestication of native species, is very important in Asia due to the mastery of breeding techniques by fish farmers 5. However, if the cyprinids have been very well studied in Eurasia and North America, this group is still very poorly known in Africa, where there are though nearly 500 species 9. Nevertheless, two genera of African Cyprinidae (Labeobarbus and Barbus), which are moreover the most privileged in research works, seem to have affinities with Eurasian species valuated by African producers. The work carried out by 10, 11, 12 and 13 on the bioecology of Labeobarbusbatesii (endogenous species in Cameroon) in a natural environment in the Mbô plain show that this species has a very high aquaculture potential. According to the same authors, this fish is omnivorous with an herbivorous tendency and presents an interesting growth potential. However, no study has yet been conducted to assess the nutritional requirements of this species in captivity. This work is part of a domestication program for the African carp Labeobarbusbatesii underway at the IRAD station in Foumban. It follows an evaluation of the effect of feed type on growth performance in post-larvae of this species 14. Its general objective is to contribute to the enhancement of the aquaculture potential of endogenous fishery resources in general and of Cyprinidae in particular. More specifically, this involves evaluating: The effect of the level of dietary protein on the survival and growth performances of post-larvae of Labeobarbusbatesii.
Material and Methods
Study Zone
The study took place between January and April 2020 at the station of the Institute of Agricultural Research for Development (IRAD) of Foumban, more specifically within his Koupa Matapit fish experimentation farm. This farm, located at 9 km of the town of Foumban (Highlands of West Cameroon) is located at 5 ° 45.826 'North latitude and 10 ° 48.516' East longitude with an altitude of 1147 m. The Sudano-Guinean climate includes a rainy season (March - October) and a dry season (November - February). The average temperature and rainfall are respectively 22º C and 1800 mm / year on average 15.
Animal Material
A total of 400 Labeobarbusbatesii fry with an average total length of 4.5 cm and an average weight of 0.8 g were collected in the nursery tanks of the IRAD station in Foumban. These fry had been collected a month before (in a post-larval state) in the natural environment from fishermen on the Nkam river and packaged in oxygenated plastic for transportation to the IRAD station in Foumban. These post-larvae had been fed post-larvae food for a month 14.
Breeding Structure
The test was carried out in 8 identical happas made of mosquito net of 3.375 m3(1.5 x 1.5 x 1.5 m), installed in a pond of 400 m2 previously cleaned and disinfected with quicklime (5 Kg/are) then dried for 7 days. The pond was equipped with a supply channel and overflow pipe. Each happas was equipped with a floating frame below which was placed a basin to collect food refusals.
Experimental Rations
Depending on the protein requirements of the Cyprinidae, four isocalorific experimental rations were formulated with various local ingredients and their bromatological compositions were analyzed (Table 1). The ingredients used were bought from local markets with fishmeal and soybean meal as the main sources of protein. The total energy of the ration was calculated from the following equivalences: 23.7; 39.5 and 17.2 kJ g-1 respectively for proteins, lipids and free nitrogen extracts.
Table 1. Experimental rations| Percentage and chemical composition | Type of compound feed d | |||
| R25% | R30% | R35% | R40% | |
| Ingredients | ||||
| Corn | 37 | 27.5 | 17.5 | 10 |
| Re-molding | 10 | 10 | 10 | 10 |
| Cotton cake | 2.3 | 2.3 | 2.3 | 2.3 |
| Soybean meal | 13 | 13 | 13 | 15 |
| Blood meal | 2.00 | 2.00 | 2.00 | 2.00 |
| Fishmeal | 18 | 27.50 | 37.50 | 45 |
| Shell | 0.10 | 0.10 | 0.10 | 0.10 |
| Bone meal | 0.10 | 0.10 | 0.10 | 0.10 |
| Palm oil | 15 | 15 | 15 | 13 |
| Iodized salt | 0.50 | 0.50 | 0.50 | 0.50 |
| Premix 2% | 2.00 | 2.00 | 2.00 | 2.00 |
| Total | 100.00 | 100.00 | 100.00 | 100.0 |
| Bromatological compositions analyzed dddd | ||||
| Dry matter(%) | 91.2 | 87.8 | 84 | 84 |
| CP (%) | 25.10 | 30.01 | 34.98 | 40.13 |
| GE (kcal / kg)(kcal / kg) | 3576.45 | 3548.87 | 3515.16 | 3447 |
| ME (kcal / kg) | 3220 | 3236 | 3267 | 3293 |
| Fat (%) | 6.54 | 6.24 | 5.65 | 5.24 |
| ash (%) | 2.50 | 2.67 | 2.71 | 2.78 |
| CP / GE | 0.01 | 0.01 | 0.01 | 0.01 |
Experimental Apparatus
The 400 fry were distributed randomly into four comparable lots (weight, size): R25% (feed composed of 25% crude protein), R30% (feed composed of 30% crude protein), R35% (feed composed of 35% crude protein) and R40% (food composed of 40% crude protein) with two repeations each. One of the 08 happas described above was randomly assigned to each of the duplicate batches described above, at a density of 20 fry / m2 (i.e 50 fry / happa) according to a completely randomized device.
Conduct of the Test
Throughout the trial, the fry were fed 4 times a day between 6 a.m. and 6 p.m. at 10% of the ichthyobiomass 16. A control fishery was carried out every month during which 20% of the size of each happa was taken at random using a dip net, for body measurements, just as at the start of the test. Thus, each fish was weighed individually using a precision 0.1 g electronic balance 17 and measured using a millimeter ichthyometer. The monthly weight assessment allowed us to readjust the amount of food to be distributed the following month. Food refusals were also collected every week, dried under the sun and then weighed, to estimate food consumption. The physico-chemical characteristics of the water (pH, dissolved oxygen, temperature, conductivity) were measured in situ and inside the pond, at the beginning of the trial, then each week between 6 and 8 a.m 15. At the end of the test, all of the individuals were fished, measured (weight and height) and counted.
Data Collection and Parameters Studied
The live weight, the lengths (total and standard) as well as the food refusals collected allowed us to evaluate the following growth parameters:
Food consumption (FC) = Quantity of food served - Refusal
Weight gain (WG) = Final weight - Initial weight
Average daily gain (ADG) = Poids final-Poids initial/Time(Number of days)
Specific growth rate (SGR) = (ln poids final-ln poids initialX100)/Time (Number of day)
ln = natural logarithm
Consumption index (CI) = (Quantity of food consumed)/(Final weight-Initial weight)
Condition factor K = 100(w/TL3) W = Weight (g) ; TL = Total length (cm)
Survival rate = (Initial number of fishes - Mortality)X100/(Initial number of fishes)
Statistical Analysis
The collected data was subjected to one-way analysis of variance. In case of significant differences, the Duncan's test at the 5% threshold was used to compare the means. The correlations were made in order to reveal the level of association between the characteristics studied. SPSS software version 21.0 was used to perform the analyzes.
Results and Discussion
Results
Effect of Dietary Protein Level on the Survival Rate of Labeobarbus Batesii fry in Pre-Magnification
The survival rate (Figure 1) in pre-magnification according to the level of protein in the compound food recorded the highest values (67%) in the R40% batches, corresponding to the fry fed with the compound feed containing 40% crude protein followed by those receiving R25%, and the lowest (57%) with the R35% treatment. At the end of the test, a significant difference (p <0.05) was observed between the individuals fed with the R25% feed and those of all the other batches. However, the fry receiving the R30% and R35% treatments were comparable.
Figure 1.Survival rate according to the dietary protein level in pre-magnification of Labeobarbus batesii fry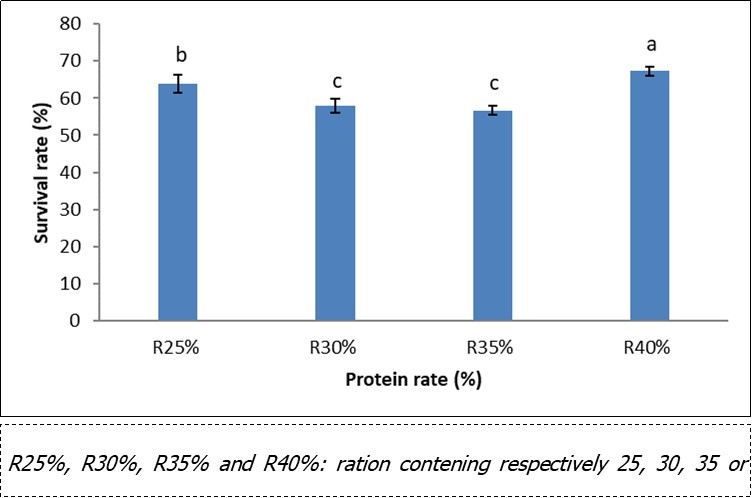
Effect of the Level of Dietary Protein on the Evolution of the Body Weight and the Total Length of the fry of Labeobarbus Batesii. Live weight
The live weight as illustrated in Figure 2 shows a comparable evolution throughout the test. Thus, whatever the period considered, the highest values were recorded in the batches fed with food containing 25% crude protein followed by compound food containing 30% protein. The lowest weight being obtained with the compound food containing 40% crude protein.
Figure 2.Evolution of body live weight of Labeobarbus batesii according to the level of protein in the compound food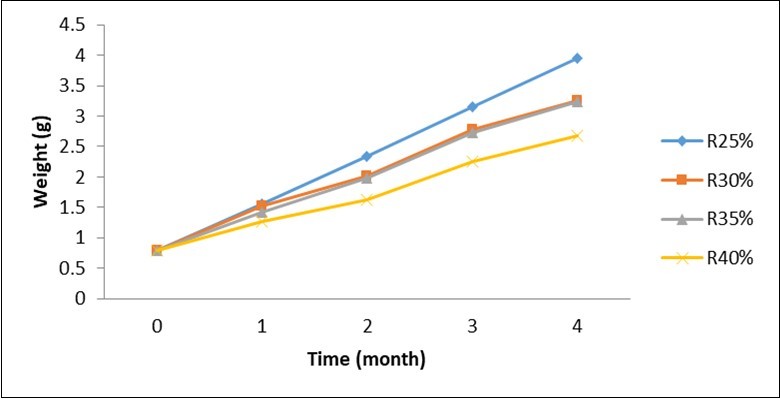
At the end of the test (Table 2), the fry fed on feed containing 30 and 35% protein, besides comparable, showed a significant difference (p <0.05) with the fishes receiving the composed feed of 25 and 40 % crude protein.
Table 2. Growth characteristics according to the protein level of the compound feed in Labeobarbus batesii| Growth characteristics | protein rate | |||
| R25% | R30% | R35% | R40% | |
| Live weight (g) | 3.958 ± 0.202a | 3.267 ± 0.299b | 3.300 ± 0.241b | 2.692 ± 0.318c |
| Weight gain (g) | 3.158 ± 0.202a | 2.467 ± 0.299b | 2.50 ± 0.241b | 1.892 ± 0.318c |
| Average daily gain (g/d) | 0.026 ± 0.002a | 0.021 ± 0.002b | 0.021 ± 0.002b | 0.016 ± 0.003c |
| Total length (cm) | 6.883 ± 0.212a | 6.866 ± 0.414a | 6.818 ± 0.306a | 6.400 ± 0.443b |
| Standard length (cm) | 5.725 ± 0.191a | 5.792 ± 0.380a | 5.800 ± 0.293a | 5.008 ± 1.318b |
| Length gain (Cm) | 2.383 ± 0.212a | 2.367 ± 0.414a | 2.418 ± 0.306a | 1.900 ± 0.443b |
| Food consumption (g) | 11.840 ± 0.812a | 9.171 ± 0.209b | 9.243 ± 1.238b | 7.924 ± 1.034c |
| Consumption index | 2.410 ± 0.102a | 1.850 ± 0.166b | 1.911 ± 0.379b | 1.632 ± 0.188c |
| Specific growth rate (%) | 1.332 ± 0.041a | 1.169 ± 0.077b | 1.179 ± 0.059b | 1.006 ± 0.101c |
| Condition factor K | 1.217 ± 0.089a | 1.017 ± 0.125b | 0.999 ± 0.072b | 1.044 ± 0.193b |
Total Length
The change in total length according to the protein level of the compound food (Figure 3) was comparable regardless of the period of the test. It was thus observed that the fishes receiving 25% crude protein in the food (R25%), presented the highest values whatever the period considered, followed by fry fed at R30% (containing 30% crude protein) and the lowest ones with the food at 40% crude protein.
Figure 3.Evolution of the total length of Labeobarbus batesii in pre-magnification according to the type of compound food
R25%, R30%, R35% and R40%: ration contening respectively 25, 30, 35 or 40%
At the end of the test, a significant difference (p <0.05) was observed between the individuals of the batches fed on composed feed of 25, 30, 35% of crude protein (otherwise comparable each other), and those receiving the food with the highest protein level (R40%).
Growth Performances of Labeobarbus Batesii fry According to the Dietary Protein Level
The growth performances of L. batesii fry according to the protein level of the compound feed is summarized in Table 2.
Food consumption and the consumption index as presented in Table 2 recorded the highest values in individuals fed with the food containing the lowest level of crude protein (R25%), followed by those receiving R35% (35% of protein) and the lowest values being obtained in individuals fed with the food containing 40% protein (R40%). There is a significant difference between individuals fed with the compound food containing 25% protein and those from all other batches, although individuals fed with R30% and R35% feed remained comparable (p˃0.05) between them.
The standard length and the gain in length of Labeobarbusbatesii in pre-magnification recorded the highest values with the feed composed of 35% crude protein followed by R30% (for the standard length) or R25% (for the gain length), while the lowest values were obtained with the compound feed containing 40% (R40%) crude protein. At the end of the trial, a significant difference was observed between the fry fed on a compound food containing 40% crude protein and the R25%, R30%, R35% treatments which were besides comparable (p˃0, 05) between them.
Weight gain, average daily gain, and specific growth rate in African carp fry were highest in subjects fed with 25% crude protein food, followed by those fed with 35% protein crude in the compound food and the lowest with R40%. A significant difference (p <0.05) was observed between the fry receiving the compound feed containing 30 and 35% crude protein, (besides comparable each other) and those of the batches fed at R25% and R40%.
The highest values of condition factor K were obtained with the compound food with the lowest protein level (R25%) followed by that of the highest level (R40%) and the lowest values with R35%. Moreover, the fry from the batches fed with the food containing 25% protein showed a significant difference (p <0.05) with those from the various other batches which were besides comparable each other.
Relative Growth of Labeobarbus Batesii fry According to the Protein Level of the Compound Feed
The effect of the protein level of the compound feed on the characteristics of the weight-length relationship is summarized in Table 3. It appears that the weight of Labeobarbusbatesii fry is strongly associated with lengths (total and standard). This relationship follows a power-type equation and the coefficients of determination (R²) are very high with the compound food containing 35% crude protein, medium with the lots receiving R30% and low in the fry fed on compound food containing 25 and 40% crude protein. Furthermore, the allometric coefficient (b) varies from 0.60 (R40% with the standard length) to 1.37 (R35% with the total length). In short, the growth is of the allometric type minorant whatever the protein level of the compound feed and of length considered.
Table 3. Length-to-weight relationship of Labeobarbus batesii fry according to the protein level of the compound feed| Protein rate | Relationships | ||||
| Equations | R2 | a | b | Growth type | |
| R25% | PV = 0.54LT1.03 | 0.53 | 0.054 | 1.03 | Minorant |
| PV = 0.63LS1.05 | 0.51 | 0.63 | 1.05 | Minorant | |
| R30% | PV = 0.27LT1.29 | 0.75 | 0.27 | 1.29 | Minorant |
| PV = 0.40LS1.19 | 0.74 | 0.40 | 1.19 | Minorant | |
| R35% | PV = 0.23LT1.37 | 0.93 | 0.23 | 1.37 | Minorant |
| PV = 0.37LS1.24 | 0.94 | 0, 37 | 1.24 | Minorant | |
| R40% | PV = 0.67LT0.75 | 0.54 | 0.67 | 0.75 | Minorant |
| PV = 0.97LS0.60 | 0.41 | 0.97 | 0.60 | Minorant | |
Discussion
During this test, the values of total length (TL) and length gain (LG) were respectively between 6.4-6.9 cm and 1.9-2.42 cm. They have tended to decrease with increasing protein levels. These values are comparable to the 3-10 cm reported by 18 in Clariidae as well as those observed by 2 (8-10 cm) in juvenile Cirrhiusmrigala (after 2 to 3 months). These observations contradict those of 19 who in 60 days of breeding shows that increasing the rate of dietary protein from 28 to 32% increases the total length from 13.74 cm to 20.69 cm in Osteochilusvittatus. It is the same for those of 20 with the fry of Poecilia reticulata, for which the increase in the level of crude protein in the ration of 18 to 43% leads to an increase in TL from 21.8 to 36.03 cm after 07 months of observations. Although the direction of evolution is the opposite, these values remain higher than those reported by 21 with Puntius vittatus with an increase in total length from 3.31 to 4.52cm when the level of dietary protein increases from 15% to 35% after 75 days of aging. These results would suggest medium growth in length in this species at this stage of development. The differences noted between them are essentially due to the genetic determinism of the species.
The values obtained during this work between 2.9-3.95 g; 1.89-3.16 g and 0.016-0.03 g / D respectively for body weight, weight gain and average daily gain were very low compared to that reported by 22 with Cyprinus carpio fingerlings and that WG increased from 7.68 to 13.41 g as much as ADG (0.17 - 0.30) after 45 days of breeding with crude protein levels between 30 and 54.7%. The same is true with the observations of 19 for whom increasing the protein level from 28 to 32% increases the GW from 13.74 to 20.69 g and the ADG from 2.56 to 3.11 g in juveniles of Osteochilusvittatus. The work of 23 in the Aspikutum hybrid go in the same direction in showing an increase in GW from 5.19 to 8.62g and ADG from 0.28 to 0.46 g / J with an increase in protein level from 30 to 35%. These results are nevertheless higher than those reported by 21 showing a variation in GP between 0.31g and 0.84g when the level of dietary protein goes from 15 to 35% after 75 days of observation in an ornamental Puntius vittatus fish. Beyond the species, the relatively low values of the growth characteristics could be explained by the poor adaptation of these animals because at this stage of domestication they would hardly adapt to a compound food regularly served earlier than the diversity food found in the natural wild milieu.
The specific growth rate between 1.01 and 1.33% obtained during this work is relatively comparable to that by 20 whose work revealed that the SGR varies between 1.24 to 0.9% when the protein level drops from 43 to 18% in Poecilia reticulata. The same is true of the 1.4-2.04% reported by 24with the post-larvae of the Keurelian fish Tor tamba. The same trend was observed by 25 with the post-larvae of Pethiareval (1.3-1.49%). However, this rate remains low compared to the values of 3.33 and 4.72% obtained by 14 with Labeobarbusbatesii post-larvae. These differences could well be explained by the poor adaptation of this new species to artificial food. The values of the condition factor K included during this test between 0.99 and 1.22 were comparable to the values (0.89-1.096) obtained by 10 in a natural environment as well as those (0.9-1.5) obtained by 14 in captivity with post-larvae of the same species, and those of 0.93-1.23 reported by 26 with post-larvae of Cyprinus carpio. This value remains relatively high compared to that (0.73-0.77) obtained in the hybrid Aspikutum by 23. They also remain very low compared to the values of 0.87-3.14 obtained by 27 with Garraruffa. The values of K, comparable during this test to other values already obtained with this species, both in the wild and in captivity, would suggest that it is a question of the genetic determinism of the species. In short, regardless of the level of dietary protein, the fish showed good nutritional and health status since all the values of K remained greater than 1 (K≥1).
The food consumption values during this test ranged from 7.92 to 11.84; 1.63 and 2.41 respectively. Moreover, the values of the consumption index increased with the increasing rate of protein, which is contradictory to the observations of 19 with Osteochilusvittatus fingerlings who report that increasing dietary protein level from 28 to 32% increases dietary consumption from 17.46 to 19.03 g. The same is true of the work of 23 carried out on juveniles of Aspikutum (hybrid resulting from the crossing Leuciscus aspirus ♀ x ♂ Rutilus frisii) which revealed that the increase in the protein level from 30 to 35% contributed to a change in food consumption of 14.58g to 19.57g in 60 days. This contradiction in the direction of evolution although the values remained comparable could be explained by the too high mage of variation of the protein level during this test which ranged from 25 to 40%. However, the optimal rate seems to be reached from the lower end (25%) of this margin. The lowering of the consumption index during this test with the increasing level of protein corroborates the observations of 22 whereby with common carp fingerlings, the CI drops from 3.77 to 2.16 as the crude protein level increases from 30% to 54%. It is the same for the works of 19 where the CI drops from 1.28 to 0.92 with fingerlings of Osteochilusvittatus when the dietary protein level drops from 28 to 32%. However the work of 20 demonstrated the contrary by revealing that the CI increases from 1.70 to 3, when the protein level drops from 43 to 18%. This variation could be linked to genetic determinism in relation to the requirements of the species.
The values of this parameter during the test varied significantly between 57 and 67%. This result is similar to that observed with Cirrhiusmrigala by 2, which reports survival rates ranging from 60-70% during 2-3 months of pre-magnification in this species. In addition, our rates are higher than those obtained by 14 with Labeobarbusbatesii post-larvae (32-56%). However, it remains very low compared to those (79-92% and 78-93%) obtained with the fry of Poecilia reticulata and Pethiareval respectively by 20 and 25. This variation is explained by the fragility and therefore the difficulty for wild fry to feed on a single food although it is complete.
As for the relative growth coefficient b, it allows to indicate the type of growth. Indeed, when b is equal to 3, the growth is said to be isometric, and when it is different from 3 the growth is said to be allometric ; hence the notions of negative or minorant allometry if b <3 and positive or upper allometry for b> 3 28. In our study, b ranges from 0.6 to 1.37, indicating a minorqnt allometry. This result is lower than the observations of 29, who, comparing 03 strains of Nile Tilapia in nursery, obtained values of b varying between 2.28 and 3. As well as those of 10 and 14 who report values of this parameter between 2.785 and 3.088 then 2.029-2.88 respectively in adults and post-larvae of Labeobarbusbatesii.
Conclusion
At the end of this study on the effect of the level of dietary protein on the survival and growth performances of Labeobarbusbatesii in captivity, it appears that the level of protein significantly influenced both the survival rate and all of the growth characteristics. The best growth performances and survival rate were obtained with the lots fed at the lowest protein level as opposed to the lots fed at the highest level. The use of 25% crude protein is therefore recommended for pre-fattening diets in Labeobarbusbatesii. However, for better control of the nutritional needs of this species, it would be necessary not only to continue this work until adulthood, but also to assess its energy needs.
References
- 1.Efolé E T. (2011) Optimisation biotechnique de la pisciculture en étang dans le cadre du développement durable des Exploitations Familiales Agricoles au Cameroun. Institut Supérieur des Sciences Agronomiques,Agro-Alimentaire, Hrticoles et du Paysage.2011-28(H-84).
- 2.FAO. (2009) The state of Food unsecurity in the wold:Economiccrisis-impacts and lessonslearned. , Rome
- 3.Kouao B. (1979) Cameroon. Rapport de stage sur les activités du Cameroun et de Centrafriquedans le domaine de la Pêche et de la pisciculture.
- 4.Lazard J, Lecomte Y, Y Stomal B ET Weigel J. (1991) Pisciculture en Afrique subsaharienne: situation et projets dans les pays francophones; proposition d’action. Ministère de la coopération et du Développement: Paris. 155.
- 5.C Lazard J et Levêque. (2009) Introductions et transferts d’espèces de poissons d’eau douce. In Lazard J. et Lésel R. pisciculture: poisson de demain.CahAgric. 18(2), 157-173.
- 6.Lazard J. (2006) Introduction /Domestication d’espèces de poissons : quelques éléments de réflexion. Unité propre de recherche « aquaculture et gestion des ressources aquatiques ». Département envt. CIRAD. Avenue Agropolis, TA 30/01,34398 Montpellier Cedex 5, France8 .
- 7.FAO. (2018) La situation mondiale des pêches et de l’aquaculture 2018. Atteindre les objectifs de développement durable. , Rome. Licence: CC BY-NC-SA 3, IGO..
- 8.J F Guégan, Paugy D.et Lévêque C.(1993).L’étude desBarbusdans le cadre d’un programme PICADO. Cahiers d’éthologie. 13(2), 183-184.
- 9.CLOFFA. (1984) Check-list of the freshwater fishes of Afirica. Daget J,Gosse JP. and Thys Van Der Audenaerde D.F.E. (eds scientifiques), ORSTOM Paris. , MRAC Tervuren, Tome 1, 410.
- 10.M Nguenga D et Tchoumboué J T. (2010) de morphologie générale et de croissance du Cyprinidae africainLabeobarbusbatesiidans la plaine inondable des. Mbô, Cameroun. International journal of Biology and Chemestrics sciences 4(6), 1988-2000.
- 11.T C, Minette T E M, Tchoumboué J. (2013) Reproductive Strategy ofLabeobarbusbatesii(Boulenger. , International Journal of Zoology 2013, 8.
- 12.T C, Tomedi E T M, Nguenga D, Tekou G, Tchoumboué J. (2014) Habits of the African CarpLabeobarbusbatesii(Pisces: Cyprinidae) from the Mbô Floodplain Rivers. Advances in Research. AIR.2014.12.006 1(12), 9.
- 13.Tomedi E T M, T C, E T, Kenfack M, Tchoumboué J. (1903) 2014.Population Structure, Condition Factor, Length-weight and Length-age Relationships ofLabeobarbusbatesii(Boulenger. , International Journal of Aquaculture 19(4).
- 14.A P Tonfack, N A Kpoumie, Ngoula F, Djikengoue K p L, T C et al. (2020) rate and growth performances of post-larvae of the African CyprinidaeLabeobarbusbatesii(Boulenger. , International Journal of Fisheries and Aquatic Studies 8(3), 128-134.
- 15.D. (2006) Contribution à l’amélioration de la production d’alevins au Cameroun: essais de reproduction et d’élevage de nurserie avecClariasgariepinuset deux autres espèces. Compte rendu de mission. 64, p..
- 16.E T. (2009) Ecloserie artisanale familiale de silure africain (ClariasgariepinusBurshell. 1822) et de carpe commune (Cyprinuscarpio) de la région Ouest-CamerounEC FP7 Project, SARNISSA: 1-23.
- 17.Tomedi-Eyango M, O Tchoumboue J et Jamtade. (2008) Effet du poids de mise en charge sur la survie et la croissance des fingerlings du poisson chat africain (ClariasgariepinusBurchell, 1822).Science AgronomiqueDeveloppement. 4(1), 41-47.
- 18.Viveen W J A R, Richter C J J, Oordt P G W J V, Janssen J A L, E A Huisman. (1985) Manuel pratique de pisciculture du poisson-chat africain (Clariasgariepinus). Département de Pisciculture et de Pêche de l'Université Agronomique de Wageningen. 91, p.
- 19.Niagara Muhammad A S, Mia S, H. (2018) Influence of different protein levels and protein to energy ratios on growth, feed efficiency and survival of bonylipbarb(OsteochilusvittatusCyprinidae) fingerlings. , Pakistan Journal of Nutrition 17(5), 228-235.
- 20.Paliitha K H M, Prakash S, Syeddian Z S G. (2010) Growth and reproductive performance of female guppy,Poeciliareticulata(Peters) fed diets with different nutrient levels. , Indian Journal of fisheries 57(1), 65-71.
- 21.A R, Jayaprakas V.(2014).Effect of dietary protein on the growth and reproductive performance of the indigenous ornamental fish,Puntiusvittatus(day).Journal of Aquatic Biology & Fisheries. 2(1), 62-67.
- 22.C O, MiresanV Cocan D I, R D Vâtu, C M Raducu, Coroian A. (2015) Growth performance of common carp (CyprinuscarpioL.) fingerlings fed with various protein levels. , Aquaculture Aquarium, Conservation and Legislation International Journal of the bioflux society 6(8), 11.
- 23.Haghparast P, Falahatkar B, Khoshkholgh M R &Meknatkhah, B. (2016) Influence of dietary protein/lipid ratio on growth performance and body composition of Aspikutum, a new hybrid ofLeuciscusaspius♀. , Rutilusfrisii♂ (Teleostei: Cyprinidae). Iranian Journal of Ichthyology 3(4), 304-315.
- 24.Muchlisin Z A, Murda T, Yulvizar C, Dewiyanti I, Fadi N et al. (2017) Growth performance and feedutilization of keurelingfishTortambra (Cyprinidae) fed formulated diet supplemented with enhanced probiotic. F1000 Research 2017: 137 (doi: 10 ,12688/f1000 research.10693f)
- 25.Rathnayake R M L W, Shirantha R R A R, Rupuka G R H, Kithsiri H M P, Nayananjali W A D.2016.Evaluation of growth and breeding performances of Pethiareval (red fin barb), with different feeds under aquarium condition. , International Journal of Scientific and Research 10(6), 191-195.
- 26.Djikengoue k P L, T F Mambe, N A Kpoumie, A P Tonfack. (2020) Effects of the level and frequency of fertilization with hen droppings on zooplanktonic density and growth performance of common carp post-larvae (Cyprinuscarpio). International journal of aquaculture research and development,2691-6622:. 13-21.
- 27.Abedi M, A H Shiva, Mohammadi H.and Malekpour R.(2010).Reproductive biology and age determination of Garrarufa Heckel, 1843 (Actinopterygii: Cyprinidae) in central Iran. , Turk. J. Zool 34, 1-7.
