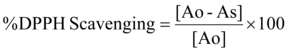Influence of Chemical Refining Processes on the Total Phenolics and Antioxidant Activity of Sunflower Oil
Abstract
The raw sunflower oil (SFO) has an undesirable flavour and odour. Therefore, to make it suitable for human consumption, the oil has to undergo a number of refining processes such as degumming, neutralization, bleaching and deodorization. During these refining processes, some of the phytochemicals present in SFO is lost. The aim of this study is to evaluate the loss in total phenolic, flavonoid and non-flavonoid contents and the antioxidant properties of the oil at the different chemical refining stages. The crude SFO oil has the highest total phenolic, flavonoid and non-flavonoid contents. The amount of phenolic compounds decreases as the oil undergoes different chemical refining processes. Results of this study indicated that the highest percentage loss of the phenolic compounds occurred during the deodorizing step.The statistical loss of the deodorized oil was 41.7, 63.9 and 27.6 % for total phenolic, flavonoid and non-flavonoid contents respectively. The free scavenging activity of sunflower oil was determined using DPPH and ABTS assays. The percentage scavenging activity ranged from 55.64 to 35.87 % for the DPPH assay while for ABTS assay the activity ranged from 59.46 to 31.43 % in a 50 mg/ml of SFO sample. This showed a decrease in antioxidant activity from crude to DNW to bleached and deodorized oil. The crude oil having the highest phenolic contents showed the highest antioxidant activity in both DPPH and ABTS assays.
Author Contributions
Academic Editor: LI Wang, University of Macau
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2015 Amanda Unanski Enright, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Free radicals are involved in various physiological reactions, causing serious cellular damage resulting in the complete inactivation of biomolecules (proteins, lipids and DNA) and cells, leading to a number of degenerative diseases 1. Antioxidants scavenge the free radicals thus helping in reducing the free-radical induced damage to the cells.
Oils contribute a major fraction of the human diet and are lipid materials derived from plants. Sunflower (Helianthus annuus L.) is one of the most important oilseed crop grown in the world and it contains unsaturated fats, protein, fiber and other important phytochemicals including phenolic compounds 2. SFO is widely used in nutrition and is a good source of polyphenols that can scavenge the free radicals produced in our body 3, 4. Crude SFO contains many unwanted compounds such as free fatty acids, odouriferous materials and others which are removed during chemical processing to yield an edible product 5. Chemical refining includes degumming, neutralizing, washing, bleaching, winterizing and deodorizing stages 6, 7. However, during these chemical processes many of the bioactive and nutritionally significant compounds are lost. Therefore, the present study was undertaken to assess the antioxidant potential together with the phenolic contents of crude and processed sunflower oil. The anti-oxidative properties of the oils were also correlated in terms of their phenolic, flavonoid and non-flavonoid contents.
Materials and Methods
Materials
Oil Samples
Sun flower oil (SFO) samples were collected in the month of November, 2012 from processing lines of Mauritius Oil Refineries (Port Louis, Mauritius). The samples were collected in plastic bottles (200 ml) at four different sampling stations during the manufacturing processes: Crude oil, Degummed/Neutralized/Washed oil (DNW oil), Bleached oil and Deodorized oil. The sample bottles were capped without any headspace and stored away from sunlight in refrigerator at 4 °C, immediately after sampling.
Chemicals
Ascorbic acid, gallic acid, 1,1-diphenyl-2-picrylhydrazyl (DPPH), 2,2′-azino-bis3 (ABTS) and trolox were purchased from Sigma Aldrich Chemical Co. (USA). Folin-Ciocalteu reagent was bought from BDH Company (UK). All the chemicals and reagents were analytical grade.
Preparation of Extracts
The oil (40.5 g) was extracted with 96 % ethanol in order to extract the phenolic components and the combined extracts were evaporated in a rotary evaporator (Buchi R-3000 Rotavapor) at 40 °C. The residue was dissolved in 5 ml ethanol and stored at 4 C for further analysis. The analysis was done in triplicates.
Determination of Total Phenolic Content (TPC)
The total phenolic content of the sunflower oil samples was determined using the Folin-Ciocalteu method 8. In 0.2 ml of the ethanolic oil sample, 0.5 ml of 0.5 N Folin-Ciocalteu reagent, followed by 2.5 ml of 20 % Na2CO3 and 0.8 ml of deionized water were added. The solution was incubated for 40 min at room temperature and the absorbance was read at 725 nm on a Biochrom Libra S22 UV/Vis spectrometer against a blank sample. Gallic acid was used as standard solution and the results were expressed as µg/ml equivalent of gallic acid per 40.5 g of oil extract.
Determination of Total Flavonoid Content (TFC)
The total flavonoid content in the oil samples was determined using the reported procedure 9. 0.5 ml of ethanolic solution of oil sample was mixed with 2 ml of distilled water and 0.15 ml of NaNO2 solution (15 %). 0.15 ml of AlCl3 solution (10 %) was added to the mixture and allowed to stand. 2 ml of NaOH solution (4 %) was then added and the total volume was brought to 5 ml. The absorbance was measured at 510 nm on a Biochrom Libra S22 UV/Vis spectrometer against a blank. Quercetin was used as the standard solution and the results were expressed as µg/ml equivalents quercetin per 40.5 g of oil extract
Determination of Total Non-Flavonoid Content (TNFC)
The total non-flavonoid content in the oil samples was determined according to reported method 10. 1 ml of hydrochloric acid (20 % v/v) and 0.5 ml formaldehyde (37 %) were added to 1 ml of ethanolic oil sample. The mixture was vortexed and left for 24 h and filtered. To 0.5 ml of the filtrate, 3.5 ml of distilled water and 0.25 ml Folin-Ciocaulteu reagent were added followed by 1 ml of Na2CO3 (20 %). The resulting mixture was vortexed and heated at 40 °C for 1 h and centrifuged. The absorbance of the solution was read at 685 nm on a Biochrom Libra S22 UV/Vis spectrometer against a blank. Gallic acid was used as the standard and the results were expressed µg/ml of gallic acid equivalents per 40.5 g of oil extract.
Antioxidant Activity
The antioxidant activity of the oil extracts was evaluated according to two assays.
DPPH Radical Scavenging Assay
The free radical activity of the oil samples of concentration ranging from 50000 to 24 mg/ml was assessed using DPPH (2,2’–diphenyl-1-picrylhydrazyl) assay according to the method described in literature 11. Absorbance was measured at wavelength 492 nm after 30 min using a labsystem multiskan Ms Eliza Reader. Ascorbic acid was used as positive control and methanol as blank. The percentage radical scavenging capacity was determined using the following formula:
Were is the absorbance of blank and is the absorbance of tested samples.
ABTS assay
The free radical scavenging activity of the different oil samples were assessed using ABTS (2,2'-azinobis-(3-ethylbenzothiazoline-6-sulfonic acid)assay with slight modifications 12. ABTS diammonium sulphate (0.054 g) and MnO2 (0.2 g) were dissolved in 100 ml of phosphate buffer and the solution was mixed using an orbital shaker for 15 min. The resulting bluish green solution was filtered. The freshly prepared ABTSradical cation was diluted with phosphate buffer solution to obtain an absorbance of 0.70 ± 0.05 at 734 nm. The SFO extract (0.5 g) was dissolved in 10 ml of chloroform. The solutions of concentration ranging 50000 to 3125 mg/ml were prepared through serial dilution. The ABTS activity was determined by mixing 0.25 ml of the diluted oil sample with 0.5 ml of the diluted ABTS. The decrease in absorbance was then measured at 734 nm using a Biochrom Libra S22 UV/Vis spectrometer. Trolox was used as the positive control. The percentage ABTS activity was determined using the following formula:
Where Ao is Absorbance of blank, As is Absorbance of tested sample
Result and Discussion
Phenolic compounds including flavonoids and non-flavanoids are important components present in SFO extract. The phenolic compounds constitute the main class of natural anti-oxidant present in SFO and they contribute mainly to its anti-oxidative potential. Raw SFO cannot be consumed directly due to its undesirable flavour and odour. It undergoes refining processes in order to make it suitable for consumption. During the chemical refining process the crude sun flower oil undergoes, (Degumming/Neutralization/Washing (DNW); Bleaching and Deodorization treatments 13,14,15,16. During these processes, unwanted components as well as some nutritive compounds including phenols are lost. The solubility of the phenolic compounds is affected by the polarity of the solvent used 17.The extraction efficiency of phenolic compounds was evaluated using different solvents including acetone (40 %), ethanol (96 %) and methanol (99.5 %). Ethanol was found to have the highest extraction efficiency and was used for further extraction in this study. The mass of the residue obtained after extraction were 1.11 ± 0.05, 0.57 ± 0.04, 0.53 ± 0.05 and 0.43 ± 0.06 g per 40.5 g of crude oil, DNW oil, Bleaching and Deodorized oil respectively. A decrease in the mass of the residues was observed for the different refining processes. The obtained residues were then dissolved in 5 ml of ethanol and were used for determining the phenolic, flavonoid and non-flavonoid contents.
Total phenolic (TPC), Flavonoid (TFC) and Non- Flavonoid Content (NFC)
Total phenolic, flavonoid and non-flavonoid contents of the ethanolic oil extracts and the percentage losses between the different oil samples weredeterminedandreported in Table 1. Crude oil possesses higher TPC, TFC and NFC as compared to the refined oil. Significant losses of the phenolic compounds including both the FC and NFC compounds were observed during the different chemical processes. The TPC decreases from 95.14 ± 1.20 to 41.18 ± 0.82 μg/ml, TFC from 37.42 ± 2.18 to 8.24 ± 0.34 μg/ml and the TNFC from 66.47 ± 0.53 to 30.44 ± 0.44 μg/ml. The highest percentage loss in PC and FC was observed during the deodorization step. In this process, the unwanted volatile components such as free fatty acids and the oxidation products of oil which are liable for producing unacceptable odour, colour, taste and flavour in the oil are removed by direct injection of heated steam at a temperature of 180–270 °C. Due to the vigorous conditions not only the undesirable components are stripped off but some important minor components including the phenolic compounds are also distilled off.
Table 1. TPC/TFC/TNFC of SFO during the chemical refining| Oil | TPC GAE/ μg /ml | % Loss | TFC Quercetin / μ g/ml | % Loss | TNFC GAE/ μg /ml | % Loss |
| Crude oil | 95.14 ± 1.20 | 37.42 ± 2.18 | 66.47 ± 0.53 | |||
| DNW oil | 82.99 ± 1.18 | 12.7 | 29.39 ± 1.93 | 21.4 | 51.52 ± 1.04 | 22.5 |
| Bleached oil | 70.69 ± 1.08 | 14.8 | 22.88 ± 0.35 | 22.1 | 42.09 ± 1.03 | 18.3 |
| Deodorized oil | 41.18 ± 0.82 | 41.7 | 8.24 ± 0.34 | 63.9 | 30.44 ± 0.44 | 27.6 |
Anti-oxidant Activity
The antioxidant activity is the ability to inhibit the process of oxidation. The DPPH and ABTS radicals are the two most commonly used and stable chromatogens to measure the radical scavenging activity of biologically active plant extracts. The different functional groups present in the bioactive compounds influence the capacity of the sample to react and quench different radicals. ABTS generally gives higher anti-oxidative activity compare to DPPH since ABTS radical reactions involves electron transfer and takes place at a much faster rate than DPPH radical 18.
DPPH Assay
The DPPH (2,2-diphenyl-1-picrylhydrazyl) assay is a spectrophotometric method to evaluate the free radical scavenging potential (antioxidant activity) of a sample. The scavenging ability of DPPH radical is its ability to accept a hydrogen atom or an electron from an antioxidant having RH group such as phenolic and flavonoid compounds. The methanolic-DPPH solution is reduced to the molecular (DPPH-H) by antioxidants 19. This reduction causes a colour change from purple to yellow. The degree of decolourisation indicates the radical scavenging potential of the antioxidant 20.
The antioxidant activity of the SFO oil samples was determined using the DPPH assay and the absorbances were read at 492 nm. The percentage radical scavenging activity of the ethanolic extracts of the different oil was determined with varying concentrations and is summarized in Table 2 and Figure 1. The free radical scavenging activities were significantly different for different oil samples. It was observed that crude oil has highest antioxidant activity of 55.64 % while the deodorized oil had lowest antioxidant activity of 35.87 % at a concentration of 50000 µg/ml of ethanolic solution.
Table 2. Average % DPPH scavenging activity of oil extracts| Conc µg/ml | Ascorbic Acid | Crude oil | DNW oil | Bleached oil | Deodorized oil |
| 50000 | 78.09 ± 0.59 | 55.64 ± 1.01 | 50.29 ± 0.25 | 43.92 ± 0.29 | 35.87 ± 1.01 |
| 25000 | 81.22 ± 0.89 | 53.00 ± 0.25 | 48.48 ± 0.66 | 40.46 ± 0.66 | 31.76 ± 2.25 |
| 12500 | 81.54 ± 0.74 | 51.13 ± 0.63 | 45.93 ± 0.95 | 31.48 ± 1.15 | 20.76 ± 1.56 |
| 6250 | 82.27 ± 0.59 | 47.70 ± 0.43 | 40.22 ± 0.80 | 20.87 ± 2.67 | 6.49 ± 2.25 |
| 3125 | 82.16 ± 1.33 | 41.48 ± 1.52 | 35.19 ± 0.29 | 11.75 ± 0.90 | 2.03 ± 1.50 |
| 1562 | 81.22 ± 0.00 | 35.35 ± 1.13 | 11.98 ± 3.99 | 7.05 ± 0.66 | 0.00 ± 0.00 |
| 781 | 82.86 ± 1.33 | 29.78 ± 0.66 | 4.92 ± 0.95 | 1.69 ± 1.30 | 0.00 ± 0.00 |
| 390 | 82.68 ±0.59 | 25.19 ± 0.25 | 1.61 ± 1.01 | 0.00 ± 0.00 | 0.00 ± 0.00 |
| 195 | 82.68 ± 0.00 | 13.53 ± 3.03 | 0.00 ± 0.00 | 0.00 ± 0.00 | 0.00 ± 0.00 |
| 97 | 82.68 ±0.59 | 1.00 ± 1.28 | 0.00 ± 0.00 | 0.00 ± 0.00 | 0.00 ± 0.00 |
| 48 | 82.05 ± 0.00 | 0.00 ± 0.00 | 0.00 ± 0.00 | 0.00 ± 0.00 | 0.00 ± 0.00 |
| 24 | 77.68 ± 2.01 | 0.00 ± 0.00 | 0.00 ± 0.00 | 0.00 ± 0.00 | 0.00 ± 0.00 |
Figure 1.Percentage of radical scavenging activity of sunflower oil using the DPPH Assay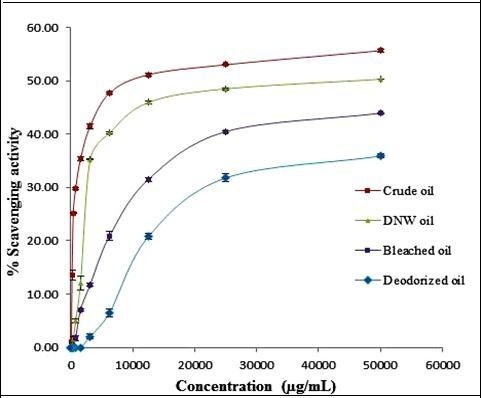
ABTS Assay
ABTS radical monocation is generated by the oxidation of ABTS with potassium persulphate. Similar to DPPH, the decolorization of ABTS radical reflects the capacity of an antioxidant species to donate electron or hydrogen atoms to inactivate this radical cation. The ABTS results were in good agreement with DPPH assay of the oil samples and decreases as the oil undergoes chemical processes. From the analysis of Table 3 and Figure 2, it can be concluded that the crude oil exhibited higher ABTS radical-scavenging activity (59.46 %) at a concentration of 50000 µg/ml of ethanolic solution.
Both assays showed comparable activities for the different oil extracts. However, DPPH radical being more stable and allows easy handling and manipulation.
Table 3. Average % scavenging activity of oil extracts using ABTS assay| Conc (mg/ml) | Crude oil | DNW oil | Bleached oil | Deodorized oil |
| 50000 | 59.46 ± 0.51 | 53.72 ± 0.43 | 38.17 ± 0.36 | 31.43 ± 0.51 |
| 25000 | 56.61 ± 0.57 | 50.92 ± 0.57 | 32.48 ± 0.43 | 26.84 ± 0.43 |
| 12500 | 53.67 ± 0.30 | 44.99 ± 0.59 | 24.66 ± 0.43 | 16.22 ± 0.43 |
| 6250 | 48.41 ± 0.57 | 42.25 ± 0.57 | 20.86 ± 0.67 | 10.91 ± 0.43 |
| 3125 | 45.00 ± 0.64 | 38.41 ± 0.57 | 15.6 ± 0.36 | 3.84 ± 0.38 |
Figure 2.Percentage of radical scavenging activity of oils using the ABTS Assay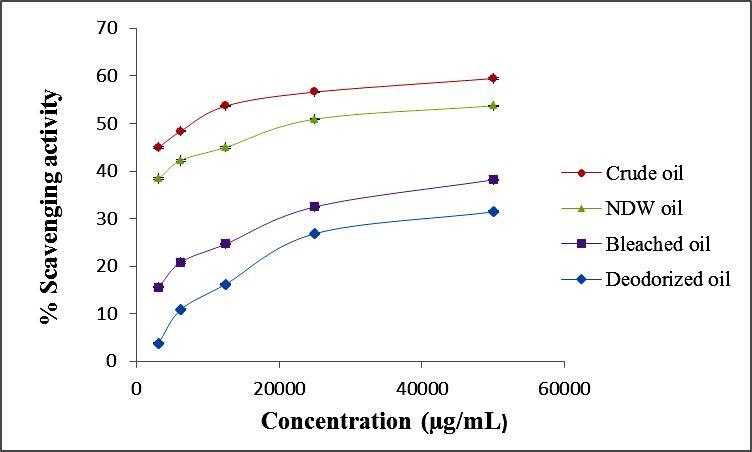
Figure 3.Correlation among phenolic compounds and antioxidant activity of oil samples measured according to DPPH assay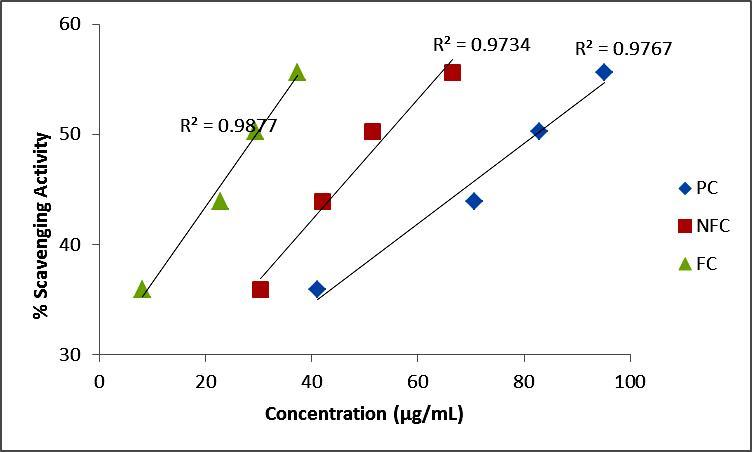
Relationship of Total Phenol and Antioxidant
Phenolic compounds are known to display anti-oxidant activity since they can either donates hydrogen or electrons and also form stable radical intermediates. The most important natural phenolics are flavonoids which are responsible for the free radical scavenging activities 21. There is a significant linear relationship between phenolic compounds and antioxidant activity 22, 23. Figure 3 highlights the strong positive correlation between PC, FC, NFC and antioxidant activity measured through the DPPH assay. Crude oil having more phenolic compounds showed higher antioxidant activity and vice-versa. A direct relationship was also found for ABTS assay and different phenolic compounds with R2 values ranging from 0.8877-0.9318.
Conclusions
There is a direct relationship between the phenolic compounds and the oil extracts obtained from the different stages of processing. It is clear from the results that the loss of phenolic compounds cause a decrease in radical scavenging activity. The present study indicated that sunflower oil can contribute to the dietary intake of antioxidants depending on the processing conditions. It is observed in the present work, that the unrefined oil possesses better activity which reduces as the oil is processed. This suggests the importance of optimizing and/or modifying the existing steps in the sunflower oil processing to retain the polyphenolic content in oil.
Acknowledgements
The authors are thankful to Mauritius Oil refineries for supplying the oil samples
References
- 2.B R Stefansson. (2007) Oilseed crops. The Canadian Encyclopedia (Historica Foundation. Available at:http://www.thecanadianencyclopedia.com , Toronto)
- 3.M, Muzquiz M, C Burbano Garcia-Vallejo, C, Ayet G et al. (2000) . , J. Sci. Food Agric 80, 459-464.
- 11.Jhaumeer-Laulloo S, M G Bhowon, Mungur S, M F, A H Subratty. (2012) . , Medicinal Chemistry 8(3), 409-414.
- 12.Re R, Pellegrini N, Proteggente A, Pannala A, Yang M et al. (1999) Free Radical Bio. , Med 26, 1231-1237.
- 14.Saba N, Syed T, Hussain S, N T Farah, Huseyin K et al. (2014) . , Pol. J. Food Nutr. Sci 64, 115-120.
- 15.Kady S A El, Gawwad A I El, A E Kassem, M 1 Hamed, M et al. (1994) Periodica Polytechnica Ser. , Chem. Eng 38(3), 147-152.
Cited by (19)
This article has been cited by 19 scholarly works according to:
Citing Articles:
Dumitrița Flaiș, Mariana Spinei, Mircea Oroian - Foods (2025) Semantic Scholar
Foods (2025) OpenAlex
Foods (2025) Crossref
Tepparit Samrit, Supawadee Osotprasit, Athit Chaiwichien, Phawiya Suksomboon, Supanan Chansap et al. - Pharmaceuticals (2024) Semantic Scholar
Pharmaceuticals (2024) OpenAlex
Pharmaceuticals (2024) Crossref
Journal of Agriculture and Food Research (2024) OpenAlex
Moussa Nid Ahmed, Aicha Eljid, Abderrahim Asbbane, Otmane Hallouch, Mohamed Ibourki et al. - Journal of Agriculture and Food Research (2024) Semantic Scholar
Journal of Agriculture and Food Research (2024) Crossref
O. Zufarov, K. Serkayev - Chemical Technology, Control and Management (2023) Semantic Scholar
Deleted Journal (2023) OpenAlex
P. Gumus, E. Decker, M. Maskan - Journal of the American Oil Chemists Society (2023) Semantic Scholar
Journal of the American Oil Chemists Society (2023) OpenAlex
Journal of the American Oil Chemists' Society (2023) Crossref
G. Sousa, M. I. Alves, M. Neves, C. Tecelão, S. Ferreira-Dias - Foods (2022) Semantic Scholar
Foods (2022) OpenAlex
Foods (2022) Crossref
O. K. Mosibo, Siwawoot Laopeng, G. Ferrentino, M. Scampicchio - Foods (2022) Semantic Scholar
G. Sousa, M. Trifunovska, M. Antunes, I. Miranda, Margarida Moldão et al. - Foods (2021) Semantic Scholar
Foods (2021) OpenAlex
Foods (2021) Crossref
M. G. Bhowon, S. Jhaumeer Laulloo, E. Hosten, Muzammil M. Khodabaccus, L. Rhyman et al. - Journal of Molecular Structure (2019) Semantic Scholar
Journal of Molecular Structure (2019) Crossref
Journal of Molecular Structure (2018) OpenAlex
International Journal Of Nutrition (2015) OpenAlex