Newborns’ Cranial Vault: Clinical Anatomy and Authors’ Perspective
Abstract
Cranial vault is the case surrounding the brain. Its structure differs in newborns than in adults in some aspects. Such differences should be clear for both health and family members. Moreover, the anatomy and embryology of fetal skull take a little attention in the previous literature and textbooks. Therefore, this short review aimed to clarify some aspects of anatomy and clinical importance of cranial vault features in newborns. The newborn vault is formed of multiple separate flat bones connected by fibrous tissues with wide soft gaps called fontanelles. Development of bones of skull vault is closely correlated with the expanding growth of the underlying brain. Such brain shouldn't be struggled by continuous tightening of the newborns' vault. Also, the newborn skull could be affected even by a fixed sleeping position.
Author Contributions
Academic Editor: Ismail Malkoc, Department of Anatomy, Faculty of Medicine, Ataturk University, Erzurum, Turkey.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2018 Abdelmonem A. Hegazy,et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
The neonatal skull is malleable; and could be affected or deformed by applying prolonged external forces. Abnormalities of the neonatal skull are common findings. Despite the recent advances in the global health care, the incidence of such anomalies is increasing with possible impact on the normal development and function of the underlying brain. The most encountered cranial anomalies are plagiocephaly and craniosynostosis 1. Both conditions are commonly associated with delays in the development of neonates. Plagiocephaly affects 13–48% of infants at the age of less than one year. Its increased incidence has been attributed to the prolonged supine positioning 2. In 1990s, the parents were advised by the American Academy of Pediatrics to keep their infants lying on their back during sleep with aiming to prevent or minimize the risk of sudden infant death syndrome 3. However, this position was noticed to be accompanied by positional skull deformity in many infants 4.
The research in the last decades clarified the process of brain development and the different factors influencing it and hence the human behavior 5.
Moreover, the previous literatures have mentioned the fetal skull with the impact of its anatomy on the subsequent development of the underlying brain. The most postnatal development of the brain occurs during the first year of life 6. At the same time, the skull vault increases rapidly during the first year of life, followed by a slow rate till the 7th year; a process is driven mainly by the expanding brain 7.
Any impediment to the normal development of the skull might result in bad sequelae to the person later on. Therefore, the current review aimed to highlight the anatomy of the neonatal skull and its clinical correlations.
Methods
The previous literature and textbooks were searched through the database including Google Scholar, PubMed, ScienceDirect, Ovid, SpringerLink, Scopus and Wiley Interscience using the keywords; skull vault, brain development, anatomy and newborns. The findings were recorded and discussed.
Results
Developmental Anatomy
The skull vault, also called calvarium or skull cap is the protective case of the brain. It is formed of flat bones forming the roof and sides of the skull. It constitutes with base of the skull the neurocranium. The neonatal vault is composed of pairs of the following separate bones: parietal, most of the two frontal and the squamous part of temporal bones as well as the squamous part of the single occipital bone 8. These bones are connected by narrow straight areas of fibrous tissues widened at the meetings of more than two bones to form large gaps closed by tough membranes and known as fontanelles (Figure 1).
Figure 1.Diagrams of neonatal skull showing bones, sutures and fontanelles of the vault: A) Lateral view; B) Superior view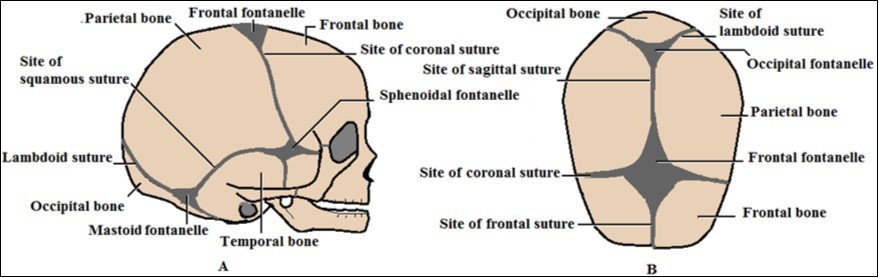
Ossification centers appear during the intrauterine life via intramembranous ossification within the outer layer of the condensed membrane surrounding the developing brain. These centers initiate the development of the individual bones forming the skull vault. The mesoderm in-between such individual bones forms fibrous tissues creating syndesmotic articulations called sutures 9.
The fontanelles are six in number including two singles: anterior "frontal" and posterior "occipital", and two pairs: anterolateral "sphenoidal" and posterolateral "mastoid". The parietal bone being situated centrally in the skull vault, it shares in the formation of all fontanelles 9. The more prominent fontanelle is the anterior rhomboidal one, situated between the two frontal and two parietal bones. The posterior fontanelle is triangular in shape lying at the junction of the two parietal bones with the occipital one. Other fontanelles are small 10. The fontanelles may reveal slight normal pulsation. The anterior fontanelle is the widest and the most clinical important one. It measures about 2.1 cm in size, and its median time to be closed is 13.8 months. On the other hand, the posterior fontanelle is about 0.5- 0.7 cm in size; and closes by the second month 11.
While bones of skull vault ossify through intramembranous ossification from bone centers appearing in the connective tissue membrane by the 8th week of intrauterine life, bones forming the skull base are formed by endochondral ossification 8,12. During infancy, the bony plates of skull vault resolve to form two tables 'outer and inner' of compact bones with intervening cancellous diploë. Also, within the first postnatal months, the fontanelles become ossified and disappear; and bones come close together forming sutures 13. The sutures appear as irregular lines resembling the surgical sutures or stitches; hence they might take their names.
Although the development of the brain is a continuous process throughout the life, the first years are the most important for healthy brain. Its development begins few weeks after conception and thought to be completed at the early adulthood 14. The developing brain is the primary inductor or initiative factor in appearance of the bones of the skull vault. Therefore, cases of anencephaly, in which there is failure of development of the cranial part of neural tube, are associated with absence of the skull vault. Moreover, failure of brain growth could lead to microcephaly 8. This is also marked in case of hydrocephalus occurring due to marked distension of the brain caused by increased CSF. Such increased CSF occurring after fusion of the sutures could compress the delicate brain tissue causing difficulties in the brain functions and even its damage 15.
The process of brain myelination begins after birth and includes all lobes of the cerebrum by the end of the first year. Such process is correlated to attaining certain cognitive abilities 6.
Clinical Correlations
The sites of sutures between the fetal bones give flexibility for the head to pass through the birth canal. These bones being separate entities override each other at birth to decrease the diameters of the delivered head 16. However, after birth the bones return away from each other to allow postnatal brain development. Later on, they become adherent together at more or less fixed "fibrous" joints called sutures. Mature closure of the sutures occurs by the age 12 years and completed at the third decade 12.
Variant sized skull vault of the neonates at birth should be investigated to determine its cause. Small sized vault might be due to brain cause including fetal alcohol syndrome and infections such as rubella, toxoplasmosis and herpes simplex. On the other hand, enlarged size might be caused by skeletal dysplasia or increased intracranial volume such as cases of hydrocephalus 17.
Margulies and Thibault concluded that the cranial sutures of the infant skull are weak; and have no the shock absorbing capabilities present in that of adults 18.
Despite the fontanelles are closed by strong membranes, they should be considered as potential weak points. The mothers and paramedical staffs should be oriented to such points. These fontanelles have been used in ultrasonography imaging of the neonatal cranial cavity and brain 19.
Palpation of the fontanelles at labor helps in diagnosis of the position of the head. Also, their inspection could assist in diagnosis of the state of growth of the surrounding bones; and to check the degree of dehydration and intracranial pressure. Cases of severe dehydration or malnutrition are commonly associated with depressed 'sunken' fontanelles; while increased intracranial tension causes their bulging 8. Abnormally large anterior fontanelle with an open posterior one is an early sign of the hypothyroidism. Also, these signs might denote other underlying causes such as increased intracranial pressure, achondroplasia, Down syndrome and rickets. On the other hand abnormal premature closure of the sutures might occur in craniosynostosis and abnormal brain development. Any abnormal size necessitates careful monitoring of the skull circumference to exclude pathologic causes 11.
Discussion
The neonatal skull vault is an important part to be thoroughly investigated for any anomalies in shape and size. It enables the physicians to check the newborn brain and general health. Development of the brain is the crucial factor in development and shaping of the overlying skull, especially before closure of the sutures. This is especially of particular importance in the first year, the age at which myelination of the brain takes place 6. Any interference on the skull could impede such development and expansion. Abnormal shape of the skull might occur by abnormal premature fusion of sutures. Examples include skull resembling a hump of camel called acrocephaly is caused by premature fusion of the coronal suture and scaphocephaly is the result of premature fusion of sagittal suture 8. Also, positional skull deformity could result from putting the infants on their back during sleep. This is manifested by flattening of the skull occiput accompanied with asymmetry of the bones of the face 20. Depending on this point, we suggest that continuous traction or tightening of the neonatal skull or any attempt to reduce its size might result not only in an interference with the normal development of the brain but also to abnormal shaping of the skull. Govaert reported that excessive displacements of the fetal bones might cause increased intracranial tension and even injury of the intracranial blood vessels 21. On the other hand, Bidhan et al reported that depression of the neonatal skull during labor has no intracranial involvement; and does not leave any impact on the developing brain but cosmetic and anxious for the parents 22. The depression disappears within six months of life; and managed conservative. This depression resolution might be attributed to growth and expansion of the underlying brain.
Through the process of fetal head molding the bones override each other at birth to decrease the size of the delivered head facilitating the parturition. However, excessive molding might cause severe complications through compression of the brain 16. Moreover, abnormal shape of the rapidly growing neonatal skull could result from continuous external pressure produced for example by keeping the same lying position of neonate for long time 2. Although cranial remolding of the cranium using helmet orthosis has been suggested for management of positional skull disfigurement, the procedure lacks deficient supporting data 23,24.
Conclusion
The general anatomical features of the newborns' skull vault are essential to be known not only for the health care members but also for mothers. It is formed of separate flat bones with soft pulsating spots "fontanelles". Continuous pressure or traction of the newborns' skull through fixed lying position or tight bandages should be avoided. This might result in bad sequelae regarding the normal skull shape and development and functions of the brain.
Acknowledgments
No external funding was provided.
References
- 1.Andrews B T, Fontana S C.Correlative vs. causative relationship between neonatal cranial head shape anomalies and early developmental delays. Front. Neurosci.2017;11: 708. DOI: 10.3389/fnins.2017.00708
- 2.W K Peitsch, Keefer C H, LaBrie R A, Mulliken J B. (2002) Incidence of cranial asymmetry in healthy newborns. Pediatrics. 110-72.
- 3.Binkiewicz-Glińska A, Mianowska A, Sokołów M, Reńska A, Ruckeman-Dziurdzińska K et al. (2016) Early diagnosis and treatment of children with skull deformities. Dev Period Med;XX,4:. 289-295.
- 4.Losee J E, Mason A C, Dudas J, Hua B S, Lan B et al. (2007) Nonsynostotic occipital plagiocephaly: Factors impacting onset, treatment, and outcomes. Plastic and Reconstructive Surgery;119(6):. 1866-1873.
- 5.Tierney A L, Nelson C A. (2009) Brain development and the role of experience in the early years.ZeroThree;30(2):. 9-13.
- 6.Paus T, Collins D L, Evans A C, Pike L B, Zijdenbos A. (2001) Maturation of white matter in the human brain: a review of magnetic resonance studies. , Brain Res Bull; 54(3), 255-266.
- 8.Hegazy A. (2014) Clinical Embryology for medical students and postgraduate doctors.LAPLambertAcademicPublishing. , Berlin
- 9.Patel M D, Swinford A E, Filly R A. (1994) Anatomic and sonographic features of the fetal skull. , J Ultrasound Med;13: 251, 257.
- 10.Shapiro R S, Robinson F. (1980) The embryogenesis of the human skull: an anatomic and radiographic atlas.HarvardUniversityPress. , Cambridge
- 12.Tubbs R S, Bosmia A N, Cohen-Gadol A A. (2011) The human calvaria: a review of embryology, anatomy, pathology, and molecular development. , Childs Nerv Syst 28(1), 23-31.
- 13.April E W. (1990) Anatomy (National Medicine Series for Independent Study),2nded.JohnWiley&SonsInc. , New York, Toronto
- 14.Tierney A L, Nelson C A. (2009) Brain development and the role of experience in the early years. , Zero Three 30(2), 9-13.
- 15.Pickard J D, Czosnyka M. (1993) Management of raised intracranial pressure. , J Neurol Neurosurg Psychiatry 56, 845-858.
- 16.PuF XuL, LiD LiS, SunL WangL, FanY. (2011) Effect of different labor forces on fetal skull molding. , Med Eng Phys 33(5), 620-625.
- 17.RBJ Glass, Fernbach S K, Norton K I, Choi P S, Naidich T P. (2004) The infant skull: A vault of information. , RadioGraphics 24, 507-522.
- 18.Margulies S S, Thibault K L. (2000) Infant Skull and Suture Properties: Measurements and Implications for Mechanisms of Pediatric Brain Injury. , J Biomech Eng 122, 364-371.
- 19.Di Salvo DN. (2001) A New View of the Neonatal Brain: Clinical Utility of Supplemental Neurologic US Imaging Windows. , RadioGraphics 21, 943-955.
- 20.Biggs W S. (2003) Diagnosis and management of positional head deformity. , Am Fam Physician 67(9), 1953-6.
- 21.Govaert P. (1993) Cranial haemorrhage in the term newborn infant.MacKeithPress,CambridgeUniversityPress. , London
- 22.Bidhan M S, LCS Shaw, Saravagi M G, Gupta B R. (2018) Congenital depression of neonatal skull. Available at:https://doi.org/10.1016/j.mjafi.2017.12.006 , Med J Armed Forces India
Cited by (12)
This article has been cited by 12 scholarly works according to:
Citing Articles:
Universal Journal of Public Health (2024) OpenAlex
Annals of Medicine & Surgery (2024) Crossref
Cureus (2023) OpenAlex
Tyler Morgan, John D. Ciubuc, D. Murray, M. Murray, Richard Murray - Cureus (2023) Semantic Scholar
Cureus (2023) Crossref
Annals of Medicine and Surgery (2023) OpenAlex
Immunological Reviews (2022) OpenAlex
William A Mills, M. Coburn, Ukpong B. Eyo - Immunological Reviews (2022) Semantic Scholar
Immunological Reviews (2022) Crossref
Frontiers in Cell and Developmental Biology (2021) OpenAlex
Connor Cross, R. Khonsari, Leila Galiay, Giovanna Patermoster, David Johnson et al. - Frontiers in Cell and Developmental Biology (2021) Semantic Scholar
Frontiers in Cell and Developmental Biology (2021) Crossref
Journal of Clinical Medical Research (2020) OpenAlex
L. Rana, D. Sood, N. Rana, Deepak Singh - (2020) Semantic Scholar
International Journal of Human Anatomy (2018) OpenAlex
