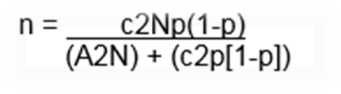Anatomical Variants of the Placenta in Sudanese and their Relation to the Neonatal Outcome
Abstract
Background
In recent years great attention has been focused on the structural and histological structures of the placenta and the umbilical cord due to their vital roles in fetal development and neonatal survival. While extensive studies have been documented in this area in the developed world, there is very little published information about the morphological variations that occur in human placenta in Sudan. Therefore, this study was designed to evaluate the structural variations in placental indices and its relation to neonatal outcome.
Methods
A prospective hospital - based study conducted in Wad Madani Maternity Teaching Hospital Department of Obstetrics and Gynecology between July 2014 and March 2018.
Results
Mean placental indices for weight, diameter and thickness were 515.51 g, 18.80 cm and 2.43 cm respectively. The mean neonatal indices were 2.95 kg, 33.19 cm and 44.42 cm for weight, head circumference and length respectively. Neonatal weight correlated significantly with placental weight, neonatal length and neonatal head circumference (P < 0.000). On the contrary, neonatal weight had no significant correlation with placental thickness and diameter
Conclusion
In this study, there was a strong relationship between the placenta and the fetus suggesting that the well-being of the fetus is highly dependent on the placenta since it serves as a link between the mother and the fetus.
Author Contributions
Academic Editor: Abdelmonem Awad Mustafa Hegazy, Professor and Former Chairman of Anatomy and Embryology Department, Faculty of Medicine, Zagazig University, Egypt.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2020 Motaz Ahmed Abdelrahman NuggedAlla, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
The placenta is a complex multifunctional organ of mainly foetal origin with pleiotropic roles during foetal growth. It has a portion derived from the developing embryo and a maternal portion formed by the modification of the uterine lining of the mother 1. Placenta with umbilical cord is a bridging organ between mother and developing fetus. The placenta is a fetal organ which provides the physiological link between a pregnant woman and the fetus. The placenta is a highly vascularized organ and its main functions are exchange of metabolic and gaseous products between maternal and fetal bloodstreams, and production of hormones 3. The placenta develops from the chorionic villi at the implantation site at about the fifth week of gestation and by the ninth or tenth week the diffuse granular echotexture of the placenta is clearly apparent at sonography 2. Placenta develops from two sources i.e. Foetal part from chorionic frondosum and the maternal part from decidua basalis 3. Development of placenta starts from the implantation. Blastocyst contains an outer unicellular layer called “Trophoblast” and an inner cell mass called “Embryoblast” 3. The trophoblast in the region of embryonic pole of blastocyst is called “Polar trophoblast” and the rest of the trophoblast is called “Mural trophoblast”. Placenta develops mainly from the polar trophoblast and primary mesoderm 3. This study aimed to assess the morphological study on placenta and its relation to the neonatal outcomes in Sudanese.
Materials and Methods
Study Design
This is a prospective hospital - based study conducted in Wad Madani Maternity Teaching Hospital Gezira State. It aimed to assess the morphological study on placenta and its relation to the neonatal outcomes.
Sample Size and Study Population
This study on the umbilical cord and placenta of 371 neonates who born in the Wad Madani Maternity Teaching Hospital during the period of the study during the period of the study (between July 2014 and March 2018), all these were included criteria. Sample size was calculated according to the following equation formula 4:
n= is the sample size required
N= is the whole target population in question
P= is the average proportion of records expected to meet the various criteria (1-p) is the average proportion of records not expected to meet the criteria
A= is the margin of error deemed to be acceptable
c= is a mathematical constant defined by the Confidence Interval chosen
Data Collection
Data has been classified into following three categories:
1. Maternal
2. Placenta
3. Neonate details
Maternal Details
Maternal details obtained from the case sheets of enrolled subjects, are as follows. Name (for file only), Hospital number, Age, last menstrual period (LMP), estimated date of delivery (EDD and term of gestation and Obstetric history.
Placenta
Placental details collected as below:
Technique
Morphological parameters measured as follows:
(A) Placental weight measured by sensitive balance.
(B) Placental diameters and lengths measured by plastic meter.
Placentae collected immediately after the delivery and examined within 30 minutes after delivery. Then weighted, measured immediately then washed well with running tap water to remove the blood clots from maternal surface of placenta. Gross morphological features, weight, diameter central thickness and attachment of umbilical cord were noted.
Neonate Details
The neonate details includes date of delivery (DOD), gender, APGAR Score at 1 minute and 5minutes, total length (Height), head circumference in centimeters, weight in Kilograms and anomalies if present were recorded from neonate’s file immediately followed by the delivery. All the neonates followed up for their hospital stay to rule out anomalies. Newborn’s birth weight < 2.5 Kg, 2.5 to 3.8 Kg and > 3.8 Kg has been considered as low birth weight, normal birth weight and large for gestational age (LGA) respectively. On the other hand gestational age (GA) less than 37 weeks has been considered as preterm, 37 to 42 weeks as term and more than 42 weeks was post term.
Data Analysis
Data was analyzed by statistical package for social science (SPSS). Data was displayed by tables and histogram.
Results
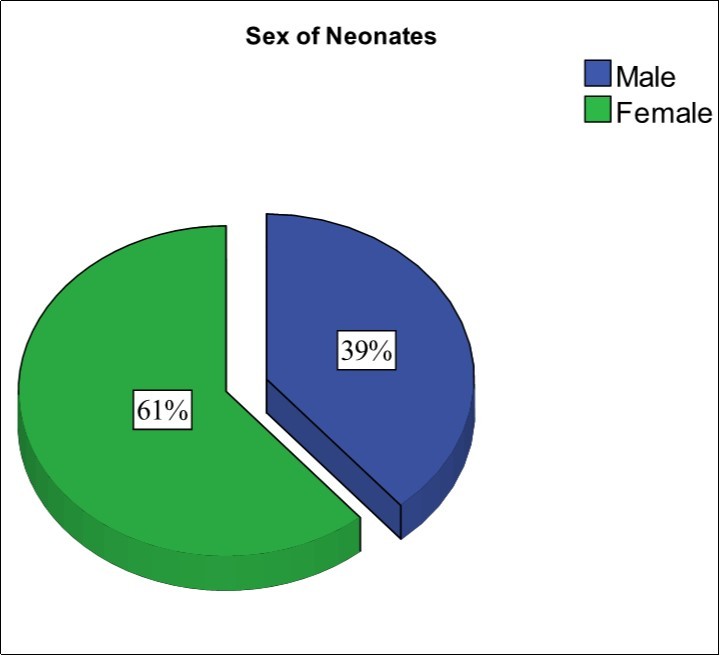
A total of 371 singletons uncomplicated pregnant women of mean age 31.06±5.6yrs with range 16 – 42 were enrolled in the study. Of all the deliveries 144 (39%) were Male babies and 227 (61%) were Female babies (Figure 1), (Table 1).
Table 1. Sex of Neonates| Frequency | Percent | Cumulative Percent | ||
| Valid | Male | 144 | 39 | 39 |
| Female | 227 | 61 | 100.0 | |
| Total | 371 | 100.0 | ||
Among 371 placentae 306 (82.4%) were Round (Figure 2), 65 (17.6%) were Oval (Figure 3). All the placentae were dark reddish-blue or crimson in colour without any palpable masses and nodosum.
Figure 2.Round shape placenta (Photographed)
Figure 3.Oval Shape placenta (Photographed)
The mean diameter of the 371 placenta was 18.8±1.5cm, thickness 2.42±.4 cm and the mean weight of placenta without umbilical cord was 515.5±65gms as Table 2.
Table 2. Descriptive statistics of placenta| Variables | Minimum | Maximum | Mean | Std. Deviation |
|---|---|---|---|---|
| Diameter of the placenta (cm) | 15.1 | 21.5 | 18.8396 | 1.48963 |
| Weight of placenta (gm) | 383 | 607 | 515.5121 | 65.02088 |
| Thickness of placenta (cm) | 1.5 | 3.1 | 2.4267 | .44050 |
The mean gestational age of 371 neonate s was 38 weeks ± 1.5 week. 138 (37.2%) were Preterm. APGAR score at 1 min and 5 min was 7, 8, 9 and 10 out of 10. Mean weight of 371 neonate s was 2.9 ±.5 kg with 33.1±1.5 cm head circumference and 44.4±7cm of total length of the neonates. (Table 3).
Table 3. Descriptive statistics of Newborn| Variables | Min | Max | Mean | Std. Dev |
|---|---|---|---|---|
| Gestational Age | 34 | 40 | 37.9704 | 1.50555 |
| APGAR Score | 7 | 10 | 8.9111 | .69761 |
| Weight of Neonates (Kg) | 1.8 | 3.7 | 2.9480 | .50667 |
| Head Circumference (cm) | 30 | 37 | 33.1887 | 1.50745 |
| Length of Neonates (cm) | 31 | 55 | 44.4488 | 7.35353 |
In measure of neonates there were 72 (19%) were Low Birth Weight neonates (LBW). The mean birth weight of 371 neonates was 2.95±.5 Kg. Figure 4.
Also when placentae parameters and neonatal outcomes were analyzed with Pearson’s correlation to find out the association between them, There was a significant strong positive correlation (r) between weight of placenta (M=515.5 SD=65gms) with APGAR Score (M=8.9 SD=.5) , (r= 0.42, p = 0.00) , weight of neonates (M=2.9 SD=.5 kg),(r= 0.75, p = 0.000) and significant negative correlation with length of neonates (M=44.4 SD=7cm), (r= - 0.25, p = 0.000). Large weight of placenta is associated with high APGAR score and large fetal weight but also associated with decreased fetal length.
Diameter of placenta (M=18.8 SD=1.5cm) had a positive correlation with APGAR score (M=8.9 SD=.5), (r= 0.42, p = 0.00), weight of neonates (M=2.9 SD=.5 kg), (r= 0.46, p = 0.000). Increased diameter of placenta is associated with high APGAR score and large fetal weight.
Also thickness of placenta (M=2.42 SD=.4 cm) had a positive correlation with weight of neonates (M=2.9 SD=.5 kg), (r= 0.39, p = 0.000) and significant negative correlation length of neonates (M=44.4 SD=7cm), (r= - 0.19, p = 0.000). As showed in (Table 3). That mean increased thickness of placenta is associated with large fetal weight and decreased fetal length. (Table 4)
Table 4. Correlation between placenta variables and neonatal outcome| AS | WN | HC | LN | |
| Pearson Correlation | .416** | .751** | .024 | -.248** |
| Sig. (2-tailed) | .000 | .000 | .644 | .000 |
| Pearson Correlation | .511** | .460** | -.037 | -.035 |
| Sig. (2-tailed) | .000 | .000 | .474 | .496 |
| Pearson Correlation | .087 | .385** | .037 | -.190** |
| Sig. (2-tailed) | .095 | .000 | .473 | .000 |
Discussion
The mean placental weight of 515.5 g in the present study was significantly lower than the sample means of 613, 630, 643 and 646.2 g reported in Israel by Barker et al 5 Eastern Nigeria by Adinma and Agbai, 6, Western Europe by Lurie et al., 7 and China by Lo et al., 8 respectively. Other investigators have reported normal placental weight to be within 400 - 600 g Sanin et al 9 Borton, 10.Other factors such as ethnicity, gestational diabetes, hypertension and hydrops foetalis have been reported to influence foetal and placental weight Asgharnia et al. 11 ,Leary et al., 12 and Van den Broek et al, 13. The findings of this study, placental weight was 515.5 g (SD = 65) with a range of 383-607 g suggest that the placenta is the single most appropriate sentinel for determining neonatal wellbeing. An effective and healthy placenta will provide a large surface area for transfer of substances between mother and fetus.
The mean placental diameter was 18.8 cm (SD=1.5) with a range of 15.1 to 21.5 cm. The diameter of the placenta did not correlate with neonatal head circumference or neonatal length but correlate significantly with weight of neonates (P < 0.46, r = 0.000). Increased diameter of placenta is associated with high APGAR score and large fetal weight.
The diameter of the placenta affects the amount of nutrients, oxygen and carbon dioxide that will pass from the mother to the child and vice versa. Borton 10 and Ohagwu et al. 14 reported a term placental diameter range of 15 cm to 25 cm whilst Yetter 15 reported a mean of about 22 cm. Comparing the mean placental diameter obtained in the present study with that of these investigators, the mean placental diameter is lower than that of Yetter 15 but falls within the range of Borton 10 and Ohagwu et al. 13.
The variations in the mean diameter may be due to racial, medical conditions or genetic since the study by Yetter, 15 involved European women while the study, of Borton 10 and Ohagwu et al. 13 in African populations.
The diameter of the placenta may give an indication of the size of the placenta which in turn may give indirect information about the foeto-placental ratio. The individual placentae with larger diameter have a large surface area for the exchange of substances and are most likely to result in increased neonatal weight.
In this study, the mean placental thickness was 2.42 cm (SD=0.4) with a range of 1.5 - 3.1 cm. Thickness of the placenta did not correlate significantly with APGAR score, neonatal head circumference or neonatal length. However, there was significant correlation between the placental thickness and the weight of the placenta (P < 0.000, r = 0.419), which mean that increased thickness of placenta is associated with large fetal weight and decreased fetal length.
Ohagwu et al. 13 reported an average placental thickness of 3.0 cm while Borton 10 reported a range of 2 cm to 4 cm. Yetter 15 gave the term placental thickness range of 2.0 cm to 2.5 cm. The average placental thickness for the present study is lower than that of Ohagwu et al. 13 but in agreement with the reports of Yetter 14 and Borton 10. However, the minimum placental thickness is lower than the minimum value of Yetter 14 and Borton 10, while the maximum value is above that of Yetter 14 but lower than that of Borton 10.
The significant correlation between the placental thickness and placental weight and placental weight with neonatal weight suggests that factors that affect the thickness of the placenta may have an indirect effect on the weight of the fetus and foeto-placental ratio. Placental thickness may give an indication of the amount of substances (nutrients, gases) that are exchanged between the fetus and the mother. Factors that affect placental thickness include nutrition, maternal genetics, maternal hemoglobin concentration gain and altitude.
Placentae less than 2.5 cm thick are associated with intrauterine growth retardation of the fetus while placentae more than 4 cm thick may be associated with maternal diabetes mellitus, foetal hydrops and intrauterine foetal infections 16.
Conclusion
In this study, there was a strong relationship between the placenta and the fetus suggesting that the well-being of the fetus is highly dependent on the placenta since it serves as a link between the mother and the developing fetus for nutritional support, excretory functions as well as immunological and hormonal support. Large placentae provide a large surface area for the exchange of substances from the mother to the fetus resulting in high fetal weight, length. The best indicator of fetal weight is gestational age. Critical examination of the placenta and umbilical cord immediately after delivery should be used to determine the well-being of the baby.
References
- 1.Sadler T W. (2004) Langman’s medical embryology. 9th edition. Baltimore,MD: Lippincott Williams and Wilkins. 11748.
- 2.Spirt B A, Gordon L P.Sonography of the placenta. In:Fleischer AC,Manning FA,Jeanty P,Romero R. (eds).Sonograpy in obstetrics and gynaecology: principles and practice. 5th edition. Connecticuit, USA. Appleton and Lange.1996: 173-2002.
- 3.Graham J Burton, Colin P Sibley, R M Eric, Jauniaux Ch.. (2013).1.Placental Anatomy andPhysiology.In:Steven G. Gabbe,Jennifer R. Niebyl,Joe Leigh Simpson et al.Obstetrics – Normal and Problem Pregnancies (6th Ed).Elsevier. New Delhi. 3 – 22.
- 4.Offredy M, Vickers P. (2010) . Developing a Healthcare Research Proposal (1sted.). Oxford: Wiley-Blackwell 131.
- 5.D J Barker, A R Bull, Osmond C, S J. (1990) Foetal and placental size and risk of hypertension in adult life. , British Medical Journal 301(6746), 259-262.
- 6.Adinma J I, Agbai A O. (1995) Foetal birth weight in Africa. , Journal of Obstetrics and Gynecology 15(5), 295-297.
- 7.Lurie S, Feinstein M, Mamet Y. (1999) Human Foetal-Placental Weight Ratio in normal Singleton Near-Term Pregnancies, Gynecologic and Obstetric Investigation. 48, 155-157.
- 8.Y F Lo, M J Jeng, Y S Lee, W J Soong, Hwang B. (2002) Placental weight and birth characteristics of healthy singleton newborns. , Acta Paediatr Taiwan 43(1), 21-25.
- 9.Sanin L H, Lopez S R, ETNO Olivares, Terrazas M C, MAR Silva et al. (2001) Relation between birth weight and placenta weight. , Neonatology 80(2), 113-117.
- 10.Borton C. (2011) Placenta and Placental problems. http://www.patient.co.uk/doctor/placenta-and-placental-problems,(accessed2017,July 3.at 22: 10 GMT).
- 11.Asgharnia M, Esmailpour N, Poorghorban M, Atrkar-Roshan Z. (2008) Placental Weight and its Association with Maternal and Neonatal Characteristics. Acta Media Iranica. 46(6), 467-472.
- 12.S D Leary, K M Godfrey, L J Greenaway, V A Davill, Fall C H D. (2003) Contribution of the umbilical cord and membranes to untrimmed placental weight. , Placenta 24(2), 276-278.
- 13.Broek N Van Den, Ntonya C, Kayira E, White S, J P Neilson. (2005) Preterm birth in rural Malawi: high incidence in ultrasound-dated population. , Human 20(11), 3235-3237.
- 14.Ohagwu C C, P O Abu, U O Ezeokeke, A C Ugwu. (2009) Relationship between Placental Thickness and Growth Parameters in Normal Nigerian Fetuses. , African Journal of Biotechnology 8(2), 133-138.
Cited by (2)
This article has been cited by 2 scholarly works according to:
Citing Articles:
Mohammadreza Dolikhani, Ghazaleh Shakibamaram, Delaram Sefidi, Dina Jalalvand, N. Faghih et al. - Journal of South Asian Federation of Obstetrics and Gynaecology (2025) Semantic Scholar
Journal of South Asian Federation of Obstetrics and Gynaecology (2025) Crossref
Journal of South Asian Federation of Obstetrics and Gynaecology (2025) OpenAlex