A Triple-Blind, Placebo-Controlled Randomized Trial of the Effect of Bilateral Alternating Somatosensory Stimulation on Reducing Stress-Related Cortisol and Anxiety During and After the Trier Social Stress Test
Abstract
The aim of this clinical study was to determine the efficacy of bilateral alternating somatosensory stimulation for the management of stress and anxiety during and after the Trier Social Stress Test (TSST), a laboratory procedure for reliably inducing stress in human subjects. For this, a randomized, placebo-controlled, triple-blinded clinical trial of 80 qualified subjects was conducted. Subjects were randomized into two groups, a treatment group (n=40) and a control (placebo) group (n=40). Metrics of emotional stress assessed were a subjective rating of the level of emotional stress and salivary cortisol levels, both obtained at 3 timepoints: before treatment (baseline), immediately following completion of the TSST, and after 20 minutes of rest following completion of the TSST. Results showed that the treatment group had a statistically greater decrease in the subjective rating of stress relative to the control group both immediately following the TSST and 20 minutes after the TSST. Salivary cortisol levels in the treatment group were also lower than the control group at those same time points. These results suggest that bilateral alternating somatosensory stimulation may be effective in reducing subjective levels of stress and anxiety. It also may actively attenuate stress-related cortisol levels, which may reflect a mechanism for reducing cortisol-induced inflammation back to baseline after exposure to stressful situations.
Author Contributions
Academic Editor: Hammad Afzal, SZABIST, Karachi, Pakistan.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2019 Ernesto Cesar Pinto Leal-Junior, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
This study was funded by the company who invented, manufactures and sells the device (TouchPointsTM) used to produce the therapeutic effect seen in this study. However, this company played no role in the study design, data collecting, or analysis.
Citation:
Introduction
Stress and stress-related disorders are responsible for a significant amount of disability worldwide. Although the term “stress” is used in a wide variety of contexts, it has consistently been demonstrated that individuals with high levels of stress experience impaired physical and mental functioning with more work days lost and a greater utilization of health care services 1. The total estimated number of people living with anxiety disorders in the world is 264 million 2. The Agency for Healthcare Research and Quality found that, in 2006, $57.5 billion was spent on mental health care in the Unites States 2. Excessive stress is a common complaint in modernized countries, which negatively affects quality of life. Chronic stress has a negative effect on multiple brain functions, causing impairment of many executive skills, including working memory, decision making and attentional control 3. Excessive stress can also cause physical symptoms, including headaches, insomnia, reduced immune function, and pain. Previous work has demonstrated that repeated social and physical stressors are associated with changes in the amygdala that often results in clear social avoidance 4. In the clinical setting, the TSST is a valid and reliable method for inducing stress in a human subject 5, 6, 7. It induces stress by requiring participants to make an interview-style presentation, followed by a surprise mental arithmetic test, in front of an interview panel who do not provide feedback or encouragement 5. The TSST was employed in this study to induce a stress response to a socially evaluative situation. The period of induced stress lasted approximately 15 minutes and was divided into 5-minute components.
There are many and varied treatments for stress and anxiety, which can be divided into pharmacologic and non-pharmacologic methods. Pharmacotherapies for stress and anxiety, such as selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs), have the most evidence to support their usage 8, but are plagued with side effects such as sexual dysfunction and sleep disturbance 9. Techniques for reducing stress and anxiety by non-pharmacological methods can potentially make a great impact on quality of life and stress-related morbidity. Multiple previous studies have shown that techniques utilizing biofeedback, neurofeedback and noninvasive brain stimulation are effective in mitigating stress and anxiety. One non-pharmacologic approach for altering how the brain manages stress and anxiety is through a non-invasive somatosensory-based therapy called eye movement desensitization and reprocessing (EMDR). EMDR is a psychotherapy treatment originally designed to alleviate the distress associated with traumatic memories 10. Bilateral stimulation is a core element of EMDR and involves a stimulus (visual, auditory or tactile) which occur in a rhythmic, alternating left-right pattern, such as watching a hand or moving light alternating from left to right or listening to tones that alternate between the left and right ears.
Multiple previous studies have demonstrated a positive therapeutic benefit of EMDR on individuals with high or pathological levels of anxiety or stress 11. Several clinical trials have also shown positive effects of EMDR in adults with autism spectrum disorder 12, depression 13, 14, and post-traumatic stress disorder 15, 16. In addition, techniques, such as EMDR and bilateral alternating somatosensory stimulation, do not have adverse drug interactions or side effects 17. In this study, bilateral alternating somatosensory stimulation was administered using TouchPoints™ 18. TouchPoints™ are wearable, non-invasive EMDR-based devices to relieve stress using a patent-pending technology called BLAST (bilateral alternating stimulation in tactile form) and have been shown to have a clinical effect on the human stress response. The therapeutic effect of TouchPoints™ is thought to involve reduction of electrical activity in the salience network, which can be measured on EEG 18, 19, 20. Previous work has demonstrated a statistically significant reduction in subjective stress levels in as little as 30 seconds using Touch Points™ 18. However, these studies were not based on controlled experimental conditions and were not blinded.
The purpose of this randomized placebo-controlled triple-blind study is to more precisely and quantitatively evaluate the effects of BLAST, applied using TouchPoints™, on subjects with stress and anxiety induced by the TSST, a clinically validated technique for reliably inducing stress in human subject 5, 6, 7. The level of stress and anxiety was assessed both subjectively and objectively through the subject’s subjective rating of emotional stress and the salivary cortisol levels, respectively, sampled at multiple time points before and after the TSST.
Materials and Methods
Human Subjects and Informed Consent
A triple-blinded placebo-controlled randomized clinical trial was performed with approval from the institutional research board/research ethics committee (IRB process number 2366732). The IRB approval was conducted through the UNINOVE University (Brazil). Informed consent was obtained (signed form) from all subjects prior to enrollment in this study. The study was conducted at the Laboratory of Phototherapy and Innovative Technologies in Health, between January and June of 2018. 80 subjects participated in this study in 2 groups of 40 subjects each. Human subject recruitment had the following inclusion and exclusion criteria:
Inclusion Criteria
To be Eligible for Study Participation, Each Subject had to Satisfy each of the Following Criteria
male (to avoid the effect of menstrual cycle and oral contraceptive use on cortisol levels),
between the ages of 18-35 years old,
fluent in Portuguese,
healthy, with no illness, injury or disease for the past 30 days.
a self-reported subjective baseline stress rating of 5 or more (on a 0-10 scale with 0 being no stress and 10 being the worst stress of the subject’s life),
rank “speaking in public” as 4/10 or greater on a Pain/Fear Catastrophizing Scale (PCS) (ensures subjects will experience some anxiety during the test)
a Generalized Anxiety Disorder-7 (GAD-7) score of 9 or less (e.g., diagnosis of generalized anxiety disorder (GAD) is unlikely).
not currently taking prescription medications for the management of stress/anxiety,
not currently using any recreational drugs (i.e., marijuana),
be able to be present for the experimental procedure at a specific time of the day (minimizes inter-subject circadian variability in cortisol levels).
Exclusion Criteria
Subjects who Satisfied any of the Following criteria were excluded from participation in this study:
previously hospitalized for a mental health condition.
history of traumatic brain injury or migraines.
diagnosed with post-traumatic stress disorder (PTSD) or with dissociative identity disorder.
diagnosed with a chronic pain disease, including chronic fatigue syndrome, fibromyalgia, endometriosis, inflammatory bowel disease, interstitial cystitis or diabetic neuropathic pain.
diagnosed with a serious mental health illness, such as dementia or schizophrenia,
any psychiatric hospitalization in the past two years,
diagnosed with a developmental disability or cognitive impairment,
participated in a clinical study or other type of clinical research in the past 30 days.
Blinding and Group Randomization Process
Subjects were randomized into two groups: a treatment group and a control (placebo) group with 40 subjects per group. Subjects were allocated to one of the two groups via variable block randomization with varying block sizes of two and four used at random to minimize the likelihood of predicting the next treatment group assignment. Randomization was performed using an automated computerized sequence methodology, which insures that the methodology and the sequence were concealed from the investigator and the subjects. Blinding of investigator to subject/group was insured by the following steps:
Each computer-generated randomization sequence was unique and was, therefore, not be able to be replicated.
Randomization occurred to either ‘Procedure Group A’ or ‘Procedure Group B’ instead of treatment or placebo group to conceal their identity.
Only the study Sponsor knew which assignment (Procedure Group A or B) corresponded to the TouchPoints™ devices and which one corresponded to the mock placebo device. The Sponsor did not reveal this information to any source (investigators, subjects, study monitors, or study analysts) until the final data analysis was completed.
Both the TouchPoints™ devices and the mock (placebo) devices were visually identical in appearance with the only difference being the lack of tactile response in the placebo devices.
Time Points and Metrics Measured
Two metrics of stress were measured in each subject: a subjective rating of the level of emotional stress, and the level of salivary cortisol, a hormone whose release is associated with psychosocial stress 21, 22. Measurement for both metrics were taken at three time points for each subject: baseline (pre-treatment), immediately following completion of the TSST, and after 20 minutes of rest following the completion of the TSST. A 20-minute rest period was chosen for one of the timepoints because previous studies looking at the effect of meditation or relaxation techniques on stress hormones showed that the greatest drop in cortisol levels occurred after 20 minutes 23, 24, 25. Moreover, since cortisol is stable in saliva, it can be collected at discrete time points to look for a time-dependent response 21.
Salivary Cortisol Assay
Salivary cortisol samples were all collected daily between 11AM and 1PM to minimize Circadian variations 22. The amount of salivary cortisol in a sample was measured using a competitive immunoassay. The cortisol in the saliva competes with antibody binding sites on a test strip with cortisol conjugated with horseradish peroxidase. After incubation, the unbound cortisol is washed away, and a developing solution is added which reacts with horseradish peroxidase to produce a visual color. The amount of salivary cortisol is can be then calculated from a spectrophotometer reading of the developed test strip and is inversely proportional to the amount of cortisol conjugate present (optical density).
Subjective Rating of Emotional Stress
For the subjective stress rating, subjects were asked to rate their stress and anxiety on a scale ranging from 0 to 10, with 0 being “no anxiety” and 10 being “the worst anxiety ever.” All measurements were collected by an assessor who was not aware of the group assignment of each subject.
Group Specific Interventions
For each subject randomly divided into two groups (control versus treatment), according to the method described above, the following group-specific interventions were performed:
Treatment Group
Subjects randomized to the treatment group performed the TSST with the active Touch Points™ devices. The active devices are worn on the Volmer aspect of each wrist and administer a bilateral alternating pattern of vibration, as well as an audible “buzzing”
Control Group
Subjects randomized to the control group performed the TSST with a mock (placebo) device. The mock (placebo) Touch Points™ devices are worn in the same way and have the same physical appearance as the actual device, including the appearance of any visible light output. However, the bilateral somatosensory vibrations were disabled prior to the study via a Bluetooth activation device. The investigator enabled both the active and placebo devices from a distance using a Bluetooth-enabled tablet using the same series of activation steps.
For both groups, immediately following the completion of the TSST, salivary cortisol levels and subjective stress rating were measured for each subject. This was followed by 20 minutes of rest, after which these measurements were repeated.
Statistical Analysis
All statistical analysis was performed using the IBM Statistical Package for the Social Sciences (SPSS) 25.0 26. The intention-to-treat analysis was performed a priori. The researcher who performed the statistical analyses was blinded to the results of the randomization of subjects to the two groups. Data were first assessed for a normal (Gaussian) distribution using the Shapiro-Wilk test. Group results (i.e., subjective stress ratings, salivary cortisol levels) were expressed as a mean and standard deviation. A two-way ANOVA test was then used, followed by a Bonferroni post hoc test, to assess the statistical significance of these group results. The significance threshold was set at p < 0.05. Results of the two-way ANOVA were presented as mean and standard error of the mean (SEM).
Results
Eighty healthy, male subjects were recruited and completed all procedures with no dropouts. The average age of 26.21 years ( 5.38 years). Table 1 lists the group mean and standard deviation (SD) for each of the chosen metrics (subjective stress rating (0-10 scale); salivary cortisol level) evaluated at each of the three time points in this study: baseline (prior to start of TSST); immediately following completion of TSST; after 20 minutes of rest following completion of TSST. There were no statistically significant group differences in the baseline (initial) measurements for either metric.
Table 1. Outcomes in absolute values.| Subjective Stress Ratings | |||||||
| Treatment | Recruitment | Baseline | After initial rest | After presentation preparation | After presentation | After math challenge | After final rest |
| M | 8,47 | 6,32 | 1,56 | 2,61** | 3,79**** | 7,18* | 5,15*** |
| SD | 1,68 | 3,17 | 1,59 | 2,23 | 3,15 | 1,96 | 3,25 |
| Placebo | |||||||
| M | 8,83 | 7,17 | 2,27 | 4,41 | 6,30 | 8,49 | 6,99 |
| SD | 1,15 | 2,14 | 1,69 | 1,31 | 1,90 | 1,04 | 1,92 |
| Cortisol | |||||||
| Treatment | Baseline | After TSST | After final rest | ||||
| M | 1,99 | 1,94 | 1,74 | ||||
| SD | 0,91 | 0,93 | 0,97 | ||||
| Placebo | |||||||
| M | 1,79 | 1,94 | 2,05 | ||||
| SD | 0,93 | 1,03 | 0,94 | ||||
| GAD7 | |||||||
| Treatment | Recruitment | Baseline | |||||
| M | 5,79 | 5,41 | |||||
| SD | 6,03 | 5,70 | |||||
| Placebo | |||||||
| M | 6,37 | 5,55 | |||||
| SD | 6,47 | 5,51 | |||||
| Catastrophizing Scale | |||||||
| Treatment | Recruitment | Baseline | |||||
| M | 2,67 | 3,20 | |||||
| SD | 0,65 | 1,45 | |||||
| Placebo | |||||||
| M | 2,85 | 3,19 | |||||
| SD | 0,62 | 1,01 |
However, the results showed that the application of BLAST using TouchPoints™ significantly decreased the subject’s subjective stress level, when evaluated immediately following the completion of the TSST and after 20 minutes of rest following the completion of the TSST (Figure 1).
Figure 1.Comparison of the subjective stress ratings between the treatment and control groups.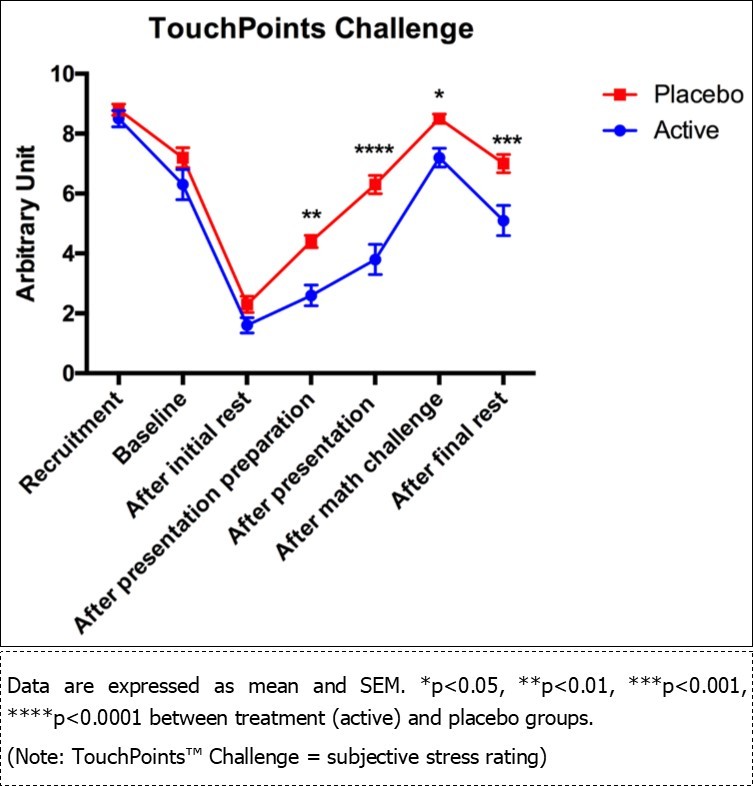
Results of the salivary cortisol levels did not reveal any statistically significant difference between the two groups (p > 0.05) at the initial time point. However, analysis of the change in salivary cortisol levels revealed that the subjects in the treatment group had a statistically significant difference in salivary cortisol levels immediately following the completion of the TSST, and at 20 minutes following the completion of the TSST, as compared to placebo (Figure 2).
Figure 2.Change in cortisol levels between time points for the treatment (active) group and the control (placebo) group.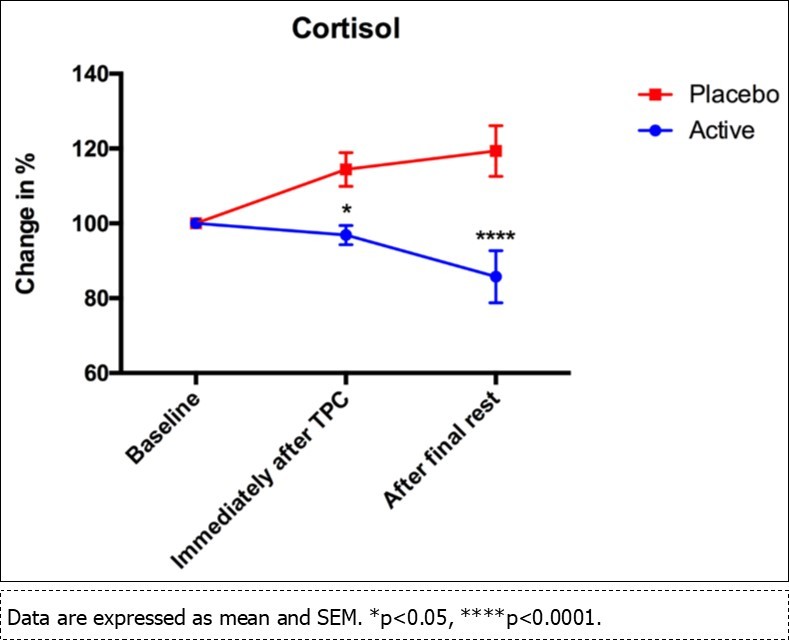
Discussion
In this study, our results showed that treatment with bilateral alternating somatosensory stimulation resulted in a statistically significant reduction in both subjective and quantitative metrics of stress and anxiety, as compared to placebo, assessed in subjects undergoing the TSST both immediately following the completion of the test and after 20 minutes of rest following the completion of the test. This suggests that this technique was effectively reducing stress during the active portion of the TSST and that this effect persisted following the TSST. This agrees with previous work which supports a role for bilateral alternating somatosensory information and EMDR in attenuating the stress response and returning stress induced reactions back to baseline. The statistically significant reduction in subjective stress ratings following treatment with BLAST agrees with a previous study by our group showing significant reductions in subjective ratings of both physical and psychological stress following 30 seconds of treatment with BLAST as compared to their baseline 18. It also supports several studies which have shown a beneficial effect of EMDR-based treatment on patients with high levels of anxiety 13, 15, 27. The significant reduction in salivary cortisol levels in the treatment group as compared to the control group also is consistent with these results but, as a more quantitative and objective metric of the stress response 22, provides a novel and compelling additional support for the benefit of BLAST in attenuating the stress response. As with the subjective stress ratings in these subjects, there were no statistically significant differences between the two groups at the initial assessment. However, there was a statistically significant (p < 0.05) difference seen immediately following the TSST and after a twenty-minute rest following the TSST, when compared to the placebo group (p < 0.0001). A graph comparing the time course of the salivary cortisol levels between the two groups revealed a noticeable downward trending of the salivary cortisol levels in the treatment group that was not present in the controls. This may indicate a beneficial effect of bilateral alternating somatosensory stimulation in stabilizing cortisol levels and returning subjects to a normal baseline after a stress-induced response 22.
Limitations
Although the current study provided more rigorous experimental conditions and statistical power than our group’s previous retrospective study showing the effect of BLAST on attenuating the stress response, there still were limitations. Specifically, this study included only males which, although it avoided the effects of the menstrual cycle and oral contraceptives on serum cortisol levels, does not allow us to assess for any sex differences in the efficacy of BLAST and limits the degree to which we can extend the findings in this study sample to the population 28. In addition, only two time points were chosen at which to calculate the two metrics of the stress response. This limited sampling perhaps fails to fully characterize the time dependent response of BLAST on the stress response induced by the TSST. Increasing the frequency of datapoints and extending the time period may reveal more precisely how rapidly the stress response (as estimated by salivary cortisol levels) is attenuated and for how long this effect lasts.
Conclusion
The application of BLAST using TouchPoints™ was effective in reducing subjective levels of emotional stress, as well as physiological stress as evaluated by salivary cortisol levels in subjects undergoing the TSST as compared to controls. This suggests that bilateral alternating somatosensory stimulation may provide a non-invasive, non-pharmacologic means of managing stress in real time situations when other treatments may not be available or practical. Further research is needed to more completely evaluate these effects and to validate them against current conventional methods for reducing stress.
References
- 1.Kalia M. (2002) Assessing the economic impact of stress--the modern day hidden epidemic. , Metabolism, Suppl1 51(6), 49-53.
- 2.Health World.Organization. Depression and Other Common Mental Disorders Global Health Estimates. n.d
- 3.Subhani A R, Kamel N, Mohamad Saad MN, Nandagopal N, Kang K et al. (2018) Mitigation of stress: new treatment alternatives. , Cogn Neurodyn 12(1), 1-20.
- 4.Patel D, Anilkumar S, Chattarji S, Buwalda B. (2018) Repeated social stress leads to contrasting patterns of structural plasticity in the amygdala and hippocampus. , Behav Brain Res 347, 314-24.
- 5.Kirschbaum C, Pirke K-M, Hellhammer D H. (1993) The ‘Trier Social Stress Test’ – A Tool for Investigating Psychobiological Stress Responses in a Laboratory Setting. , Neuropsychobiology 28(12), 76-81.
- 6.Birkett M A. (2011) The Trier Social Stress Test Protocol for Inducing Psychological Stress. , J Vis Exp 56, 3238-10.
- 7.Allen A P, Kennedy P J, Dockray S, Cryan J F, Dinan T G et al. (2017) The Trier Social Stress Test: Principles and practice. , Neurobiol Stress 6, 113-26.
- 8.Charney M E, Hellberg S N, Bui E, Simon N M. (2018) Evidenced-Based Treatment of Posttraumatic Stress Disorder. , Harv Rev Psychiatry 26(3), 99-115.
- 9.Ferguson J M. (2001) SSRI Antidepressant Medications: Adverse Effects and Tolerability. , Prim Care Companion J Clin Psychiatry 3(1), 22-7.
- 10.Shapiro F. (1989) Eye movement desensitization: A new treatment for post-traumatic stress disorder. , J Behav Ther Exp Psych 20, 211-217.
- 11.Davidson P R. (2001) Parker KC. Eye movement desensitization and reprocessing (EMDR): a meta-analysis. , J Consult Clin Psychol 69(2), 305-16.
- 12.E Lobregt-van Buuren, Sizoo B, Mevissen L, A de Jongh. (2019) Eye Movement Desensitization and Reprocessing (EMDR) Therapy as a Feasible and Potential Effective Treatment for Adults with Autism Spectrum Disorder (ASD) and a History of Adverse Events. , J Autism Dev Disord 49(1), 151-64.
- 13.Chen Y R, Hung K W, Tsai J C. (2014) Efficacy of eye-movement desensitization and reprocessing for patients with posttraumatic-stress disorder: A meta-analysis of randomized controlled trials. , PLoS One 1(17), 9-8.
- 14.Banerjee S, Argaez C. (2017) Eye Movement Desensitization and Reprocessing for Depression, Anxiety, and Post-Traumatic Stress Disorder: A Review of Clinical Effectiveness. Canadian Agency for Drugs and Technologies in Health. , Ottawa (ON);
- 15.Acarturk C, Konuk E, Cetinkaya M. (2016) The efficacy of eye movement desensitization and reprocessing for post-traumatic stress disorder and depression among Syrian refugees: results of a randomized controlled trial. , Psychol Med 46(12), 2583-93.
- 16.BA van der Kolk, Spinazzola J, Blaustein M E. (2007) A randomized clinical trial of eye movement desensitization and reprocessing (EMDR), fluoxetine, and pill placebo in the treatment of posttraumatic stress disorder: treatment effects and long-term maintenance. , J Clin Psychiatry 68(1), 37-46.
- 17.Shapiro F, Solomon R M. (1995) Eye Movement Desensitivation and Reprocessing. , New York:JohnWiley&Sons
- 18.Serin A, Hageman N S, Kade E. (2018) The Therapeutic Effect of Bilateral Alternating Stimulation Tactile Form Technology on the Stress Response. , J Biotechnol Biomed Sci 1(2), 42-7.
- 19.Harper M L, Rasolkhani-Kalhorn T, Drozd J F. (2009) On the neural basis of EMDR therapy: Insights from qEEG studies. , Traumatology (Tallahass Fla) 15(2), 81-95.
- 20.Pagani M, G Di Lorenzo, Verardo A R. (2012) . , Neurobiological Correlates of EMDR Monitoring – An EEG Study. PLoS One 1(12), 7-9.
- 21.Racic M, Ivkovic N. (2014) Salivary Cortisol Levels as a Biological Marker of Stress Reaction Značaj molekularne biologije u dijagnostici i terapiji tumora pluća View project Competency based Curriculum Reform. in Nursing and Caring in Western Balkan Universities-CCNURCA TEMPUS View.doi: .
- 22.Dedovic K, Duchesne A, Andrews J, Engert V, Pruessner J C. (2009) The brain and the stress axis: The neural correlates of cortisol regulation in response to stress. , Neuroimage 47(3), 864-71.
- 23.Klatt M D, Buckworth J, Malarkey W B. (2009) Effects of Low-Dose Mindfulness-Based Stress Reduction (MBSR-ld) on Working Adults. Heal Educ Behav. 36(3), 601-14.
- 24.Barnes V A, Treiber F A, Turner J R, Davis H, Strong W B. (1999) Acute effects of transcendental meditation on hemodynamic functioning in middle-aged adults. Psychosom Med. 61(4), 525-31.
- 25.Mohan A, Sharma R, Bijlani R L. (2011) Effect of Meditation on Stress-Induced Changes in Cognitive Functions. , J Altern Complement Med 17(3), 207-12.
Cited by (2)
This article has been cited by 2 scholarly works according to:
Citing Articles:
Occupational Therapy In Health Care (2021) OpenAlex
K. McGhee, Emily Kidney, Kaelah Pou, Heather Pruyn Bouley, S. Reynolds - Occupational Therapy in Health Care (2021) Semantic Scholar
Occupational Therapy In Health Care (2021) Crossref
