Effect of Trigonella Foenum against Ethylene Diamine Tetra Acetic Acid induced Nephrotoxicity in Male Albino Rats
Abstract
Background
Nephrotoxicity is a complication due to the effect of some toxic chemicals on kidney. Current study planned to screen the effect of Trigonella foenumaqueous seeds extracts on EDTA induced nephrotoxicity. Trigonella foenum known for its various medicinal properties is also a natural antioxidant and a free radical scavenger with no documented evidence as a nephron-protective agent.
Objective
To investigate the protective effects of aqueous seed extracts of Trigonella foenum.
Material and Methods
The present study was used 40 male albino rats (Rattus albinus) with weight of (150 ± 10) g with divided into four groups: control gp; EDTA gp (95 mg/kg); Trigonella foenum gp (500 mg/kg) and EDTA + Trigonella f oenum gp by gastric tube daily for 4 weeks. Blood urea, creatinine, GFR, creatinine clearance, MDA and GPx analyses and microscopic examination of kidney were performed.
Results
In the present study, Blood samples were taken from all groups and concentration of serum urea, creatinine, GFR, Creatinine clearance, MDA and GPx were determined. Histopathological observations were observed in kidney tissue. Data were analyzed using analysis of variance (ANOVA). EDTA induced an increase in urea and creatinine as well as there was a decrease in the concentration of GFR and creatinine clearance. The level of MDA was increase while the concentration of GPx was decrease in the serum of EDTA group. The aqueous extracts of Trigonella seeds significantly prevented renal damage by normalizing increased levels of renal markers. The correction of oxidative stress biomarkers was consistent with amelioration of the histopathological changes induced by EDTA. Hence, it is suggested that ameliorative effect of aqueous extract of Trigonella foenumagainst EDTA induced nephrotoxicity.
Conclusion
The present data suggest that aqueous extract of Trigonella foenum exhibits reno-protective effect in EDTA induced renal damage.
Author Contributions
Academic Editor: Patricio De, Universidad de Chile.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2020 A I Barakat, et al
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
The salts of EDTA and EDTA are white crystalline or powder in structure shape. The effectiveness of EDTA as a chelator for a particular metal ion is given by its stability constant with the metal ion. The stability constants for metal-EDTA complexes vary considerably and any metal which is capable of forming a strong complex with EDTA will at least partially displace another metal with a weaker stability constant. The EDTA salts are soluble in water as well as have low sorption to soil and sediments, have no significant vapor pressure, and have a biodegradation half-life of weeks to months 1.EDTA has been used extensively in medicine as a chelating agent for the removal of toxic heavy metals and these minerals were bound to the EDTA. Trace amounts of various metals are necessary for the proper functioning of the body. The salt of EDTA is a common component in many eye drops and contact lens wetting and cleansing solutions and used in a number of personal care and hygiene products such as shampoos, liquid soaps, creams and lotions. Also used as a food additive in a range of products including canned shrimp and prawns, canned mushrooms and frozen French fries. It is added to salad dressings to prevent rancidity. EDTA is used in many industrial processes, in agriculture, in photochemical, pharmaceuticals, textiles and in galvanizing and paper manufacturing. The usage of EDTA in West Germany in 1986 by industry was: metal processing and galvanizing technology, 30% detergents, 20% photographic industry, 20% textiles, 10% paper and 5% miscellaneous (antioxidants in soaps and cosmetics, pharmaceuticals, and foodstuffs) 2. EDTA and its salts are eliminated from the body 95% via the kidneys and 5% by the bile, along with the metals and free ionic calcium which was bound in transit through the circulatory system. The binding of divalent and trivalent cations by EDTA can cause mineral deficiencies, which seem to be responsible for all of the known pharmacological effects. Sensitivity to the toxic effects of EDTA is related to the deficiency of zinc 3, 4. The long-term toxicity of EDTA is complicated by its ability to chelate essential and toxic metals. These toxicological studies that are available indicate that the apparent toxicological effects of EDTA have been due to zinc deficiency as a consequence of complication. EDTA is also used as a chelating agent in boiler and cooling water in nickel plating and in wood pulping processes. EDTA is also in detergents for household and textile use, industrial germicides, metal cutting fluids and as a bleaching agent in color film process. The EDTA salts are mild skin irritants but considered severe eye irritants. A report by the Scientific Committee on Toxicity, Ecotoxicity and the Environment 3. Diarrhea and lowered food consumption were reported by abnormal effects were seen in animals which were fed with mineral deficient diets. The symptoms were observed in male and female rats and have numerous toxicological studies including various acute, sub chronic, and chronic /carcinogenicity toxicity studies as well as mutagenicity studies. The exposure to EDTA arises directly from its use in food additives especially in human and medicines as well as personal care. Exposure to EDTA from drinking-water is probably very small in comparison with that from other sources. Once EDTA is present in the aquatic environment, its speciation will depend on the water quality and the presence of trace metals with which it will combine. The removal of EDTA from communal wastewater by biodegradation in sewage purification plants is very limited. There was a mild to moderate effect on the kidney tissue. The male rats which treated by EDTA for 14 days; all rats became lethargic; the kidneys were pale and swollen and there was moderate dilation of the bowel and subserosal haemorrhages. Histological observation of a number of sections showed lesions in the kidney tissue. All the experimental rats exhibited varying degrees of damage to the kidney 5.
Nephrotoxicity is characterized by functional changes in kidney tissue including inhibition of the protein synthesis, reduced glutathione antioxidant depletion, lipid peroxidation and mitochondrial damage. Oxidative damage is thought to be one of the main mechanisms involved in nearly all chronic renal pathologies 6. Exposure to chemical reagents like ethylene glycol, carbon tetra chloride, sodium oxalate and heavy metals like mercury, arsenic, lead and cadmium also induces nephrotoxicity 7. Oxidative damage in many tissues occurred by imbalance in the generation of reactive oxygen species 8. The free radical damage in tissues make by two mechanisms: Increased generation of reactive oxygen species (ROS), including hydroperoxides, singlet oxygen and hydrogen peroxides, and by causing direct depletion of antioxidant reserves 9.
Many plants have been used for the treatment of nephrotoxicity in traditional medicine. Several herbs are used for reducing renal damage and to avoid kidney failure complications 10. Medicinal plants are considered as a source for drug treatment and are playing an important role in drug programs. Numerous medicinal plants are rich by bioactive components which are mostly free of undesirable effects and have powerful pharmacological roles. Fenugreek (Trigonella foenum) is an annual crop belonging to Fabaceae family and native to an area spreading from Iran to Northern India and widely cultivated in China, India, Egypt, Ethiopia, Morocco, Ukraine, Greece and Turkey 11. In different parts of the world, leaves and seeds of fenugreek are often used as curries, dyes, medicines and vegetable 12. It has different active materials such as flavonoids, alkaloids, vitamins and very important types of amino acids 13. So, this study aimed to investigate the nephroprotective effects of aqueous seed extracts of Trigonella foenumon kidney in male rats against EDTA intoxication.
Material and Methods
The Experimental Animal
The present work was used male albino rats (Rattus albinus) with weight of (150 ± 10) gm. They were purchased from the Physiology Department, Faculty of Medicine, Alexandria University, Egypt. Rats were kept individually in wire cages; they were fed on standard commercial pellets diet 14 and water ad libitum acclimatization period (7 days) and also throughout all the experimental period (4weeks)
Preparation of Plant Extracts
Fenugreek seeds were obtained from local market, cleaned of extraneous matter and ground into a fine powder 200 ml boiling distilled water were added to 2.5 gm powder fenugreek seed, left it for 10 minutes and filtered. The filtrate was dried at 35-45C▫ in incubator.
Experimental Design
In all groups, animals were fed their corresponding diet daily for 4 successive weeks.
Rats were divided into 4 groups of 6 rats each as follows:
Group 1 (Normal control): rats were received distilled water by gastric tube daily.
Group 2 (EDTA group): rats were received 95 mg/kg by gastric tube daily 15.
Group 3 (Trigonella foenum group): rats were received 500 mg/kg by gastric tube daily 16.
Group 4 (EDTA+ Trigonella foenum group): rats received mixture 95 + 500 mg/kg by gastric tube daily.
Collection of Serum and Tissue Samples
At the end of the experimental period (4 weeks) and after overnight fasting, all rats were sacrificed and blood samples were collected from hepatic portal vein and portion of the blood was received into centrifuge tube and centrifuged at 1500 g for 15 min for obtaining serum while the other portion was received into heparinized tubes and centrifuged for obtaining plasma. Serum and plasma samples were stored at -20 °C for further assessment.
Detection of kidney Function
Biochemical Estimation of Blood Urea
The collected blood was centrifuged and plasma fraction was separated. Urea level of plasma was measured by commercially available standard Blood Urea Kit (Merck, Japan) using Semiautoanalyzer (Merck, Japan) by standard protocol for photometric determination of urea, according to the Urease GLDH method (kinetic UV test) 17.
Biochemical Estimation of Blood Creatinine, Glomerular Filtration Rate and Creatinine Clearance
The collected blood was centrifuged and plasma fraction was separated. The plasma creatinine level was measured by commercially available standard Blood creatinine Kit (Merck, Japan) using Semiautoanalyzer (Merck, Japan) by standard protocol for phtotometric determination of creatinine based on Jaffe kinetic method without de-proteinization. The calculation of creatinine clearance and glomerular filtration rate were estimated by using Urease Berthelot method kits 18.
Detection of Detection of Malondialdehyde (MDA) and glutathione peroxidase (GPx) were determined using commercial kits (Sigma–Aldrich, USA) according to 19, 20.
Histopathological Examination
Kidneys from the experimental rats were fixed in 10% buffered Bouins reagent and were processed for paraffin sectioning. Sections of about 5 mm thickness were stained with haematoxylin and eosin for photomicroscopic observations 21.
Statistical Analysis
All values were expressed as mean ± standard error (S.E). The differences were analyzed using one-way analysis of variance (ANOVA). The P-value <0.05 reflects significant differences.
Results
Effect on kidney Function
Serum urea levels, EDTA treatment for 4 weeks resulted in significant increase in serum urea level (44.0 ± 1.7 mg/ml) compared to control rats (Table 1). However, elevations in the blood urea were significantly (p<0.05) improved by aqueous seeds of Trigonella indicating their nephron-protective effects having lower values (25.6± 2.3 mg/ml) of urea level when compared to normal values.
Table 1. Effect of EDTA and Trigonella on urea, creatinine, GFR, Creatinine clearance, MDA and GPx (X ± S.E).| parameters | Control gp | EDTA gp | Trigonella gp | EDTA + Trigonella gp |
| Urea (mg/ml) | 20.3 ± 0.88 a | 44.0 ± 1.7 b | 22 ± 1.5 a | 25.6 ± 2.3 a |
| Creatinine (mg/ml) | 0.29 ±0.04 a | 0.62 ± 0.04 b | 0.27 ± 0.01 a | 0.30 ± 0.04 a |
| GFR (ml/min) | 91.6 ± 1.2 d | 42.5 ±1.2 a | 63.7 ± 1.8 b | 74.7 ± 1.4 c |
| C c (ml/min) | 125 ± 2.8 c | 80 ± 1.1 a | 92.3 ± 1.4 a | 106 ± 2 b |
| MDA (nmol/ml) | 9.1 ± 0.4 a | 44 ± 1.4 c | 10 ± 1.6 a | 20.6 ± 1.2 b |
| GPx (UlL) | 8 ± 0.2 c | 3.1 ± 0.4 a | 5.3 ± 0.1 b | 6.4 ± 0.2 b |
Serum creatinine was significantly increased (0.62 ± 0.04 mg/dl, p<0.05) in the group which treated with EDTA when compared to the control group (0.29 ±0.04 mg/dl) Table 1. However, group which treated with DETA and Trigonella had the numerically lowest value of serum creatinine (0.30± 0.04 mg/ml), indicating their improved nephroprotective activity. The concentrations of serum glomerular filtration rate and creatinine clearance were determined in the rats which induced by EDTA have decrease values (p<0.05). However, nephron-protective effects of Trigonella seeds were recorded in the group which treated with EDTA and Trigonella.
Effect of Trigonella Foenum on the Activity of Lipid Peroxidation and Antioxidant Enzymes in the Serum
The biochemical changes in lipid peroxidation and antioxidant enzymes were studied on the serum of rats (Table 1). Level of MDA was significantly increased (44 ± 1.4 nmol/ml) in the EDTA-treated group, when compared to the control ones (9.1 ± 0.4 nmol/ml). Higher values of MDA indicate the oxidative stress in EDTA treated group. After treatment the rats with Trigonella foenum showed significant decrease (p≤ 0.05) when compared to EDTA group. There was significant decrease (p<0.05) in the GPx activity (3.1 ± 0.4 U/L) in EDTA group when compared to control rats (8 ± 0.4 U/L). The activity of GPx enzyme showed significant increase (6.4 ± 0.2 U/L) in the group which treated by Trigonella foenumand EDTA when compared to control rats.
Histological Observations in kidney Tissues
The renal corpuscle is formed of the glomerular blood capillaries surrounded by capsular space and Bowman's capsule. The proximal convoluted tubules are lined with large cuboidal cells with deeply stained acidophilic cytoplasm with apical brush border and rounded vesicular nuclei. The distal convoluted tubules are lined by low cuboidal cells with distinct cell boundaries and less acidophilic cytoplasm. Displays that the glomerular capillaries showed congestion and dilatation in some renal corpuscles. The cells of the proximal convoluted tubules were swelled resulting narrowing or obliteration of the tubular lumen. The cells lining proximal convoluted tubules appeared with vacuolated cytoplasm and deeply stained nuclei. The brush borders of the proximal convoluted tubules cells were destructed. The distal convoluted tubules showed degenerative changes in the form of pyknotic nuclei and vacuolated cytoplasm shows mild cellular infiltration in the interstitial tissue. Some glomeruli showed degeneration with wide urinary space. The glomerular degeneration was thickening of Bowman's capsule and cell debris in tubular lumen. The renal corpuscles appeared nearly similar to the control. Most of kidney tubules exhibited acidophilic cytoplasm and rounded vesicular nuclei. Regarding the effect of studied treatments on kidney, it was found that CC administration increased significantly the serum level of creatinine and induced many histopathological changes in renal tissues Figure 1, Figure 2, Figure 3, Figure 4.
Figure 1.The effect of EDTA and Trigonella foenum on urea and creatinine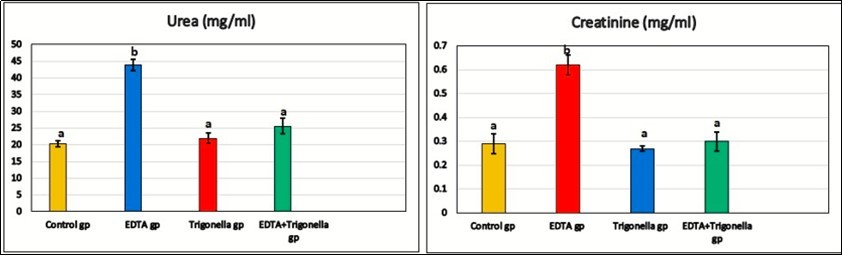
Figure 2.The effect of EDTA and Trigonella foenum on glomerular filtration rate (GFR) and creatinine clearance (Creat cl).
Figure 3.The effect of EDTA and Trigonella foenum on malondialdehyde (MDA) and glutathione peroxidase (GPx).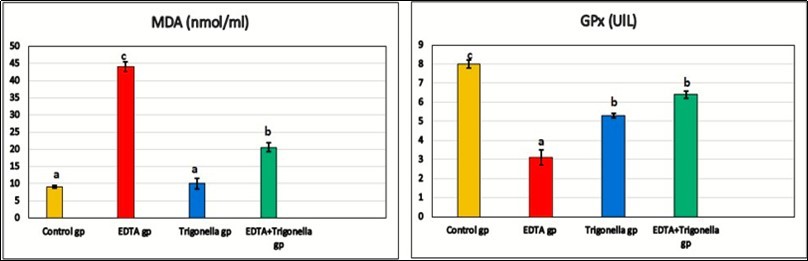
Figure 4.Photomicrographs of a section in the kidney tissue of the A) control group showing normal structure, B) EDTA treated group C) Trigonella foenum treated group D) EDTA + Trigonella foenum treated group (H&E, X. 400).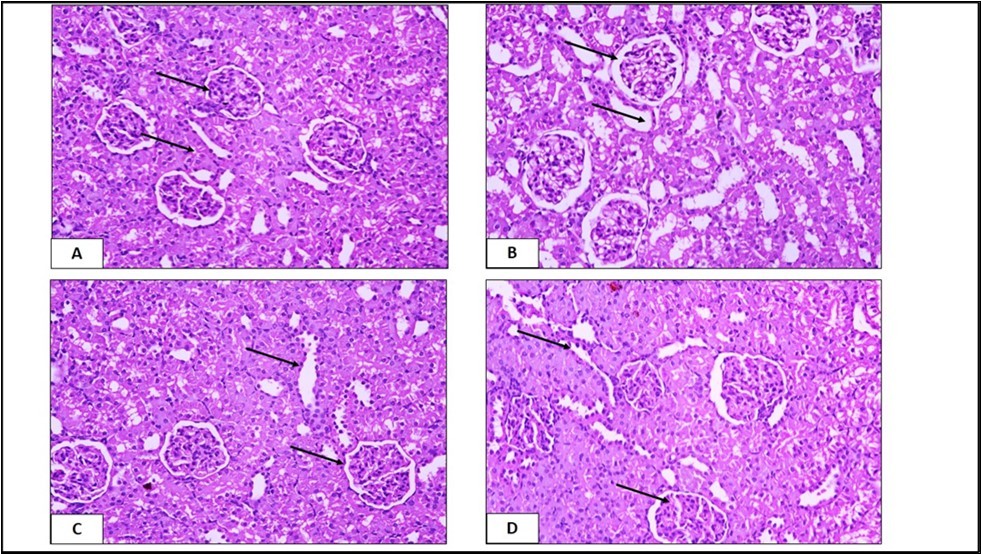
Discussion
The nephropathy is one of the important health hazards all over the world, in spite of age, race, environmental and geographical variations. The etiology behind this renal problem is widely ranging from substance induced to physiological and various metabolic complications, grading the nephropathy among the recent years leading to death of human. Medical literature has described an increase of nephrotoxic acute renal failure (ARF), by antibiotics drugs which are used, among which the aminoglycosides are most common. Elevation of the concentration of urea and creatinine levels in the serum of animal was considered as the indicator of nephrotoxicity. Majority of the medicinal plants have some organic compounds which provide many physiological functions; these bioactive substances include tannins, alkaloids, flavonoids, saponins, glycosides, carbohydrates, etc. The studies revealed that the bioactive constituents in Trigonella foenum22. The phytoconstituents detected in the plant tissue may be responsible for the nephroprotective activity 23. As well as these bioactive molecules also have antioxidant reaction. Several synthetic and natural compounds as Trigonella foenumhave been studied to protect the kidneys tissue against toxic material induced nephrotoxicity.
The current studies have shown that the fenugreek seeds exhibit anti diabetic action, hypolipidaemic effect, hypocholesterolemic effect, antitumor activity, antiulcer property, immunomodulatory effects and lactation induction. These seeds contains 45-60% carbohydrates, mainly mucilaginous fiber (galactomannans); 20-30 proteins high in lysine and tryptophan, 5-10%fixed oils (lipids), pyridine-type alkaloids mostly trigonelline (0.2-0.36%), choline (0.5%), gentianine, and carpaine, flavonoids, free amino acids, calcium and iron, saponins, glycosides, cholesterol and sitosterol, vitamins D, nicotinic acid and volatile oils 24, 25. In the current study, biochemical parameters such as blood urea and creatinine clearance have been estimated to investigate the nephroprotective effects of Trigonella foenumagainst EDTA induced nephrotoxicity in the treated animals. In addition, suppression of free radical defense mechanism has also been observed because selective accumulation of EDTA in kidney tissue can induce oxidative stress leading to lipid peroxidation as MDA. The generation of reactive oxygen species (ROS) mostly in mitochondria that can induce renal injuries. Morphologically, cellular necrosis, large lysosomes and myeloid bodies have been observed in the kidney sections. Even in the absence of major changes in the membrane permeability, the failure of plasma membrane pumps will cause potential changes in the cell. Regulating the ROS level in the kidney of rats, which might be ascribed to its ability to scavenge and prevent free radical generation 26.
Many studies have reported that the renal toxicity of EDTA. It is suggested that alter glomerular filtration because EDTA is actively transported into proximal tubules, here it accumulates and damages the tubular cells, hence; alters the renal circulation leading to reduced glomerular filtration rate (GFR). The major etiology behind this complication may be induction of oxidative stress which is the most common pathogenic inducer 27. We demonstrated that the administration of EDTA significantly reduced creatinine clearance as an index of GFR and enhanced plasma creatinine as well as urea-nitrogen concentrations. In addition, another possible mechanism regarding the action of EDTA is that EDTA may increase the production of hydrogen peroxide (H2O2), and it is well known that oxygen and H2O2 causes the contraction of mesangial cells, modify the filtration surface area and ultimately reduce the GFR 28.
The increase of concentration of EDTA in the kidney cells cause severe damage in renal tubules sections as well as decreasing in the number of the functional nephrons which weakens the reabsorption process and generate reactive oxygen species (ROS) which damage the kidney cells which leads to apoptosis process. These adverse effects on renal tubules function resulted in the elevation of nitrogen which containing compounds as urea, creatinine in the blood, proteinuria as well as reduction in creatinine clearance. The end product of protein catabolism is urea. The concentration of urea is elevated when GFR is markedly decreased in renal function. The nephrotoxic agents act on mesangial cells through contracting them and increasing cell proliferation and necrosis. All of which in turn make reduction in the filtration coefficient and lead to a decrease in GFR after treatment the animals with EDTA. The urea level begins to rise only after parenchymal tissue damage. Thus, serum urea concentration is considered a more reliable renal function indicator than serum creatinine. In the current study, urea concentration is significantly (P≤0.05) increased in EDTA group as compared to normal control rats. Trigonella-treatedgroup exhibited nephron-protective effects by significantly lowering in the serum urea level. The possible reason behind the serum urea accumulation may be an increase rate of serum urea production than the clearance rate. Creatinine derives from endogenous sources by tissue creatinine breakdown and its clearance enables a quite good estimation of the GFR. Plasma creatinine concentration is an important index than the urea concentration in the first phases of kidney disease. In the present study, mean serum concentration of creatinine increased significantly (P≤0.05) in EDTA treated group as compared to normal control group. All experimental rats which treated with Trigonella displayed nephron-protective action by lowering the serum creatinine level when compared to EDTA group. The increased level of blood urea and creatinine concentration in EDTA-toxicated rats suggests the inability of the kidney to excrete these products causing their increase in blood and decrease their excretion in urine. The enhancement in the total urinary protein excretion is due to decrease reabsorption of low molecular weight proteins by injured tubules 29. In kidney diseases, increased plasma urea is due to the higher rate of plasma urea production, which exceeds the rate of urea clearance 30. Tissue creatinine breakdown increases plasma creatinine level when nephrotoxicity occurs. Indicating chemical induced renal cellular toxicity as EDTA. The improvement in the blood urea nitrogen and creatinine levels in EDTA animals treated with fenugreek seeds indicates renal protective 31 nature of these extracts 32. Our results are in accordance with the finding of Pradeepa et al. 33.
Lipid peroxidation is the oxidative degradation of lipids which generates free radicals that cause damage in the cells. The end product of lipid peroxidation is malondialdehyde which is known as second messenger of free radicals. High concentration of MDA in kidney tissue indicates renal toxicity. In the present study, MDA level was significantly increased whereas GPx level was decreased noticeably in EDTA treated group as compared to control group. It has been proposed that oxidative stress may be responsible for tubular damage. Reduction in antioxidant enzymes in cells when there is disruption of cellular redox balance. Plasma levels of these enzymes are very sensitive markers of toxicity. When the endothelial cell layer of renal tubules is damaged, the enzymes are released into the blood stream and increase in level. It is well known that the production of ROS causes cell damage due to cytotoxic action of oxygen and nitrogen derived free radical species 34, 35, 36, 37, 38. Lipid peroxidation (LP) has a relationship with the release of lysosomal enzymes. Hence, LP activates the phospholipases and removes the peroxidized lipid from the membrane. The oxidation of unsaturated fatty acids in biological membranes by free radical leads to a decrease in membrane fluidity and disruption of membrane structure and function. The present results confirm the earlier findings, and concluded that EDTA administration caused severe damage to renal tissues most likely by ROS mediated mechanism as evident by decreased activities of antioxidant enzymes that led to increased lipid peroxidation 39, 35, 36, 37, 38. In addition, there is a highly significant increase of lipid peroxidation activity was reported in this study, these observations are in agreement with those reported earlier. Resulting in enhanced production of reactive oxygen species such as superoxide ions, hydroxyl radicals and hydrogen peroxide. These oxygen species resulted in lipid peroxidation 43. Production of ROS is followed by increased lipid peroxidation (LPO), increased activity of MDA and decreased activity of GPx which ultimately lead to cell death due to apoptosis. Lipid peroxidation is a common consequence of ROS formation and oxidative stress in the kidneys. In this study, changes in MDA 44, 35, 36, 37, 38 and alter the amount of tissue GPx in the EDTA group indicating the presence of oxidative stress and their reduced ability to cope with these changes. Induction of oxidative stress and deactivation of cellular defense mechanisms are also believed to be involved in EDTA-induced nephrotoxicity. Oxidative stress affects a vast range of intracellular targets as lipids, proteins and DNA and cellular defense mechanisms. Fenugreek aqueous extract exhibited antioxidant property 45. Enzymes involved in GPx metabolism were also measured in this study and it was found that there was a decrease in the activity of GPx in EDTA-rats, while the activity of these enzymes increased considerably in treated rats by Trigonella. GPx is a particularly important biomarker because of its ability to scavenge the effect of peroxides produced 46, 35, 36, 37, 38. Acute tubular necrosis was the most relevant histopathological changes observation. The histology of kidney control rats shows a normal morphology of renal parenchyma with Normal Tubular (NT) brush-borders and intact Normal Glomerulus (NG) and surrounding Bowman’s capsule. Glomerular damages were evident by glomerular bleeding and partial endothelial rupture in capsule. Proximal tubules were dilated with loss of cellular boundary and epithelial degeneration. We observed EDTA-induced tubule damage in the kidney. However, Trigonella foenumimproved this condition and attenuated EDTA-induced nephrotoxicity 47, 35, 36, 37, 38. The pathology of kidney appeared as dilatation and severe congestion of blood vessels, Majority of convoluted tubules cloudy swelling, contained hyaline cast in their lumen and vacuolar degenerative changes were observed in some renal tubules sections. Diffuse extravagations of red blood cells between the degenerated renal tubules. The glomerular tuft was congested, atrophied and some of them were swollen. In fenugreek treated EDTA significant decrease was noticed in urea and creatinine levels. An improvement in kidney structure occurred. The involvement of free radicals in EDTA nephrotoxicity may be normalize by fenugreek 48, 35, 36, 37, 38.
Abbreviations
Ethylene Diamine Tetra Acetic Acid (EDTA).
References
- 1. (2012) EPA (Environmental Protection Agency). Office of Pollution Prevention and Toxics.(2003). Structure Activity Team Report. Disodium EDTA (14025-21-9), isodium Zinc EDTA (73513-47-0),Disodium copper EDTA (73637-19-1),Disodium Manganese EDTA (15375-84-5),Disodium Manganese EDTA (73637-20-4),Calcium Disodium EDTA (62-33-9),Disodium Cupric EDTA (14025-15-1), Iron EDTA (17099-81-9),Sodium Iron EDTA (12389-75-2),Sodium EDTA(17421-79-3),Trisodium EDTA (150-38-9),Disodium EDTA (139-33-3),Potassium EDTA(7379-27-3),EDTA(60-00-4),Tetrasodium EDTA(67401-50-7).
- 2.Kalia K, S J Flora. (2005) Strategies for safe and effective therapeutic measures for chronic arsenic and lead poisoning. , Journal of occupational health 47, 1-21.
- 3. (2003) CSTEE (Scientific Committee on Toxicity, Ecotoxicity, and the Environment). (Opinionon the results of the Risk Assessment of:Tetrasodium EDTA and Edetic Acid (EDTA). Human Health Part. European Commission. , Brussels 1-7.
- 4.WHO. () Edetic acid (EDTA) in drinking-water. Background document for preparation of WHO Guidelines for drinking-water quality. Geneva, World Health Organization.
- 5.Frank R, Rau H. (1990) Photochemical transformation in aqueous solution and possible environmental fate of ethylenediaminetetraacetic acid (EDTA). Ecotoxicology and environmental safety. 19(1), 55-63.
- 6.Abd El-Ghany MA, Ramadan A M, Ghozy S F. (2012) Nutraceutical effects of curcuma, ginger, celery, yeast and honey on side effects of gentamicin induced nephrotoxicity in rats. , World Appl Sci J 16(5), 646-55.
- 7.Pydi R. (2012) Nephroprotective medicinal plants- A review. , Int J Univers Pharm Life Sci 1(2), 266-281.
- 8.N A Hamadouche, Slimani M, Merad-Boudia B, Zaoui C. (2009) Reproductive toxicity of lead acetate in adult male rats. , Am J Sci Res 3, 38-50.
- 10.J P Peesa. (2013) Nephroprotective Potential of Herbal Medicines: A Review. , Asian J Pharm Tech 3(3), 115-118.
- 11.Sadeghzadeh-Ahari D, Hassandokht M R, Kashi A K, Amri A, Alizadeh K H. (2010) Genetic variability of some agronomic traits in the Iranian fenugreek landraces under drought stress and non-stress conditions. , Afr J Plant Sci; 4(2), 12-20.
- 12.Acharya S, Srichamroen A, Basu S, Ooraikul B, Basu T. (2006) Improvement in the nutraceutical properties of fenugreek (Trigonella foenumgraecumL.). , Songklanakarin J Sci Technol; 28 (Suppl 1, 1-9.
- 13.Hasona N A, Ahmed M Q, Alghassab T A, Alghassab M A, Alghabban A A. (2016) Antihyperlipidemic effect of pomegranate peel and Iranian fenugreek extracts on cholesterol-rich diet induced hypercholesterolemia in Guinea pigs. , Merit Res J Med Sci; 4(4), 196-203.
- 14. (1995) National Research Council (NRC) Nutrient requirements of laboratory animals.4th revised edition.National Academy Press,Washington. , chapter 2, 11-79.
- 15.Kimmel C A. (1977) Effect of route of administration on the toxicity and teratogenicity of EDTA in the rat.Toxicology and applied pharmacology. 40, 299-306.
- 16.Radwan E H. (2018) Chapter of Soil Toxicology: Potential Approach on the Egyptain Agro-Environment. 76, 57-77.
- 17.C A Burtis, R A Edward. (1999) . Tietz Textbook of Clinical Chemistry. 3rdEdn., W.B , Philadelphia, PA.1917 .
- 18.Sabbagh M, Rick W, Schneide S. (1988) A kinetic method for the direct determination of creatinine in serum with 3, 5-dinitrobenzoic acid without deproteinization. , J. Clin. Chem. Clin. Biochem 26, 15-24.
- 19.Bernheim F, M L Bernheim, K M Wilbur. (1948) The reaction between thiobarbituric acid and the oxidation products of certain lipides. , J. Biol. Chem 1, 257-264.
- 20.Carlberg I, Mannervik B. (1975) Purification and characterization of the flavoenzyme glutathione reductase from rat liver. , JBC 14, 5475-5480.
- 21.Palani S, Raja S, R P Kumar, Jayakumar S, B S Kumar. (2009) Therapeutic efficacy ofPimpinellatirupatiensis(Apiaceae) on acetaminophen induced nephrotoxicity and oxidative stress in male albino rats. , Int. J. PharmTech Res 1, 925-934.
- 22.Schardein J L. (1981) Teratogenesis studies with EDTA and its salts in rats.Toxicology and applied pharmacology. 61, 423-428.
- 23.Albasha M O, El-Saied Azab A. (2014) Effect of cadmium on the liver and amelioration by aqueous extracts of fenugreek seeds, rosemary, and cinnamon in Guinea pigs: histological and biochemical study. , Cell Biol; 2(2), 7-17.
- 24.Hajimehdipoor H, Sadat-Ebrahimi S E, Amanzadeh Y, Izaddoost M, Givi E. (2010) Identification and Quantitative Determination of 4-Hydroxyisoleucine inTrigonella foenumgraecumL. from Iran. , J. Medicinal Plants; 9(6), 29-34.
- 25.Mehrafarin A, Qaderi A, Sh Rezazadeh, Naghdi Badi H, Gh Noormohammadi et al. (2010) . Bioengineering of Important Secondary Metabolites and Metabolic Pathways in Fenugreek (Trigonella foenumgraecumL.).J. of Medicinal Plants; 9(35), 1-18.
- 26.Flora G, Gupta D, Tiwari A.Toxicity of Lead: A Review with Recent Updates, Interdiscip Toxicol. 5(2), 47-58.
- 27.Nagi M N, Mansour M A. (2000) Protective effect of thymoquinone against doxorubicin-induced cardiotoxicity in rats, A possible mechanism of protection. , Pharmacological Research 41, 283-289.
- 28.Najafi H, Firouzifar M R, Shafaat O, Changizi-Ashtiyani S. (2014) Protective effects of Tribulus terrestris L. extract against acute kidney injury induced by ischemia/reperfusion in rats. , Iran. J. Kidney Dis.; 8, 292-298.
- 29.A I Morales, Vicente-Sanchez C, S J, Egido J, Mayoral P et al. (2006) Protective effect of quercetin on experimental chronic cadmium nephrotoxicity in rats is based on its antioxidant properties. , Food ChemToxicol 44, 2092-2100.
- 30.P D Mayne. (1994) The Kidneys and Renal Calculi. In:. Clinical Chemistry in Diagnosis and Treatment, Mayne, P.D. (Ed.). 6th Edn , Edward Arnold Publications, London, UK 2-24.
- 31.Ravikumar P, C V Anuradha. (1999) Effect of fenugreek seeds on blood lipid peroxidation and antioxidants in diabetic rats. , Phytother Res.May 13(3), 197-201.
- 32.Cuzzocrea S, Mazzon E, Dugo L, Serraino I, Paola R D et al. (2002) A role for superoxide in gentamicin-mediated nephropathy in rats. , European Journal of Pharmacology 450, 67-76.
- 33.Pradeepa S, Subramanian S, Kaviyarasan V. (2013) Biochemical evaluation of antidiabetic properties of Pithecellobium dulce fruits studied in streptozotocin induced experimental diabetic rats. , Int J Herb Med; 1(4), 21-28.
- 34.Hininger I, Waters R, Osman M, Garrel C, Fernholz K et al. (2005) Acute prooxidant effects of vitamin C in EDTA chelation therapy and long-term antioxidant benefits of therapy. Free Radic Biol Med;. 38(12), 1565-1570.
- 35.Radwan E H, Elghazaly M M, Hussein Hkh, KK Abdel Aziz, Barakat A I. (2020) The possible effects of sodium nitrite and sodium benzoate as food additives on the liver in male rats. , J. of Advances in biology V 13, 14-30.
- 36.Elghazaly M M, H K, KK Abdel Aziz. (2020) Adverse effect of mixture of food additives on some biochemical parameters in male albino rats. , A I Barakat, and E H Radwan 13, 1-13.
- 37.Radwan E H. (2015) Surveillance ecological study of cellular responses in three marine edible bivalve species to Cd present in their marine habitat, Mediterranean Sea in Alexandria Egypt. , J of Advances in biology, V 7(2), 1319-1337.
- 38.Radwan E H. (2016) Determination of total hydrocarbon and its relation to amino acid found in two bivalve edible species from Alexandria and Elismailia coast. , Egypt. J. of Advances in biology, V 9(5), 1834-1844.
- 39.Roussel A M, Hininger-Favier I, Waters R S, Osman M, Fernholz K et al. (2009) EDTA chelation therapy, without added vitamin C, decreases oxidative DNA damage and lipid peroxidation. , Altern Med Rev; 14(1), 56-61.
- 40.Elghazaly N A, E H Radwan, H, M.and Nour Eldein Allam (2019a). Interaction between Ator and Fennel remedies in the treatment of obesity in rats.J of obesity management. , V 1(3), 6-22.
- 41.Elghazaly N A, Radwan E H, Zaatout H H, Elghazaly M M.and Nour ElDein Allam (2019b). Beneficial effects of fennel (Foeniculum vulgare) in treating obesity in rats, (V),1. , issue 2, 16-33.
- 42.Elghazaly N A, Zaatout H H, Radwan E H, Elghazaly M M.and EA Elshikha (2019c). Trigonella foenum Graecum extract benefits on hematological, biochemical, and male reproductive system of a complementary thrapy with Glimepiride in treating streptozotocin induced diabetic rats. J of bioinformatics and diabetes. 1(3), 45-59.
- 43.Patrick L. (2002) Mercury toxicity and antioxidants: Part I: Role of glutathione and alpha-lipoic acid in the treatment of mercury toxicity. , Altern Med Rev 7, 456-471.
- 44.Ali S, Diwakar G, Pawa S, M R Siddiqui, M Z Abdin et al. (2001) Xanthine oxidase-derived reactive oxygen metabolites contribute to liver necrosis: protection by 4-hydroxypyrazolo [3,4-d] pyrimidine. , Biochim. Biophys. Acta 1, 21-30.
- 45.Thirunavukkarasu V, Anuradha C, Viswanathan P. (2003) Protective effect of fenugreek (TrigonellafoenumGraecum) seeds in experimental ethanol toxicity J. Ethnoph.3, APR.80(30): 237Toxicology and applied pharmacology. 61, 423-428.
- 46.Ali S, Pawa S, Naime M, Prasad R, Ahmad T et al. (2008) Role of mammalian cytosolic molybdenum Fe–S flavin hydroxylases in hepatic injury. , Life Sci 13, 780-788.
