Correlation of Modified Computed Tomography Severity Index with Biochemical Markers in Acute Pancreatitis
Abstract
Introduction
Multislice computed tomography examinations performed 3 to 4 days after the onset of symptoms is the imaging procedure of choice in acute pancreatitis, which is mainly done for evaluating the complications. Due to radiation exposure and cost, alternative scoring systems like Ranson’s score and Glasgow’s score were devised but were cumbersome. The serum amylase and lipase were found to increase with progression of disease. The objective of this study was to evaluate the association of acute pancreatitis by modified CT scoring system with the serum levels of amylase and lipase.
Material and Methods
In hundred patients presenting with acute pancreatitis, the severity of pancreatitis was recorded using the modified CT severity index. The serum amylase and lipase levels were recorded and correlated with modified CT severity index score.
Results
Among 100 patients studied, 46%, 40% and 14% had mild, moderate and severe grades of pancreatitis respectively by modified CTSI score. No significant correlation was found between the levels of serum amylase (r = -0.104, p = 0.301) and lipase (r = -0.177, p = 0.079) with the modified CT severity index in patients with acute pancreatitis.
Conclusion
The serum level of amylase and lipase had non-significant correlation with the modified CT severity index score and thus did not indicate severity of pancreatitis.
Author Contributions
Academic Editor: Qiuqin Tang, Obstetrics and Gynecology Hospital Affiliated to Nanjing Medical University, China.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2021 Joshi BR, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Acute pancreatitis is an acute inflammation of pancreas which varies in severity, from mild self-limiting pancreatic inflammation to pancreatic necrosis with life-threatening sequelae. Biochemical markers like serum amylase and lipase have been used for diagnosis of acute pancreatitis(AP). Amylase was first used as a laboratory test for pancreatic disorders in 1916.1Because of its short half-life, the serum amylase level decreased sooner than other pancreatic enzymes and returned to normal despite persistence of inflammation2and also had a low specificity for AP. Serum Lipase was another commonly used biochemical marker for diagnosis of AP.
Contrast Enhanced CT (CECT) is considered to be the gold standard imaging modality in the evaluation of patients with AP.3The role of imaging is not only to diagnose AP but to demonstrate the presence and extent of pancreatic necrosis and the complications of AP. Ideally, doing CECT after 48-72 hours of onset of an acute attack, increases the chances of picking the necrotising pancreatitis.4The CT imaging features of acute pancreatitis include focal or diffuse enlargement of the pancreas, peripancreatic fat stranding, peripancreatic fascial thickening and fluid collections.5 CT has an overall accuracy of 87% and sensitivity and specificity of 100% in the detection of pancreatic necrosis.6,7 Modified CT severity index MCTSI is the most frequently used scoring system used for assessing the severity of acute pancreatitis.
The purpose of this study was to evaluate the association of AP by MCTSI with the serum levels of amylase. This can help patients not to undergo hazardous radiological exposure if biochemical parameters can estimate same findings with a certain level of confidence.
Methods
After approval from the Institutional Review Committee, this prospective study was done in the Department of Radiodiagnosis and Imaging in the tertiary hospital for a period of 12 months from October 2018 to October 2019. Hundred patients admitted with clinical impression of acute pancreatitis who underwent contrast enhanced MDCT during study period within three days of admission were included in the study. Informed consent was received before the study.
All scans were done using Seimens 128 - slice MDCT with 120 KVp and 300 mAs with 5 mm slice thickness. Bolus tracking method was used for post contrast scan with the tracker placed in the descending aorta at the level of dome of diaphragm. 70-80 ml of 350mg/ml non ionic iodinated contrast (IOHEXOL) was injected using pressure injector at the rate of 3-4ml/sec. Threshold set at 150 Hounsfield units(HU) and delay of 3 seconds was given after the attainment of threshold for arterial phase. Venous phase acquired after a delay 60 seconds from the time of contrast injection. Scanning was done in cranio-caudal direction in arterial and venous phases; from the level of diaphragm to aortic bifurcation in the arterial phase and from the level of diaphragm to the level of pubic symphysis in the venous phase. Images were retro reconstructed with 1.25 mm slice thickness and reformatted in sagittal and coronal planes for analysis.
The severity of pancreatitis was scored using modified CT severity index and classified into three categories (mild, moderate and severe). The modified index is a 10 point scoring system derived by assessing the degree of pancreatic inflammation (0 to 4 points) pancreatic necrosis (0 to 4 points) and extrapancreatic complications (0 or 2 points). Clinical outcome parameters included the length of hospital stay, the need for surgical intervention and the occurrence of infection, organ failure and death. All patients were graded into mild (score 0-2), moderate (score 4-6) or severe (score 8-10) pancreatitis.
Collected data were analyzed by Chi square test to assess the statistical significance. P-values <0.05 was considered statistically significant.
Results
Out of the 100 patients studied, 62 (62%) were male and 38 (38%) were females. The age range was from 21 to 79 with a mean age of 40 years. Maximum number of patients was seen in the age group of <35 years of age group which consisted of 29 (29%) patients. Most common presentation was pain abdomen (seen in 100% of cases), followed by fever (58%), vomiting (24%) and jaundice (17%).
In 54 (54%) patients, ultrasound examination of the abdomen was normal. Forty six (46%) patients had abnormal ultrasound findings such as bulky pancreas with altered echogenecity, peripancreatic fat stranding, fluid collection, ascites or pleural effusion.
In our study, pancreas was normal in 1 (1%) patient. Fifty (50%) patients had intrinsic pancreatic abnormalities to whom 2 points were assigned. Remaining 49 (49%) patients had peripancreatic inflammation, to whom 4 points were assigned.
A total of 75 (75%) patients had no evidence of pancreatic necrosis on CT scan. Twenty five (25%) patients had less than 30% necrosis to which 2 points were assigned. Twenty-nine (29%) patients had one or more extra pancreatic complications such as pleural effusion, ascites, vascular complications, parenchymal complications, or gastrointestinal tract involvement.
Modified CT Severity index was calculated by adding points assigned to each parameter. The severity of pancreatitis is classified into three categories: mild (0-2 points), moderate (4-6 points) and severe (8-10 points). According to the Modified CT Severity Index, the patients were graded into mild (n=40), moderate (n=46) and severe (n=14) i.e. 40% patients had mild, 46% patients had moderate and 14% had severe pancreatitis (Figure 1, Figure 2)
Figure 1.Percentage of patient with scoring based on pancreatic inflammation, necrosis and extra pancreatic complications.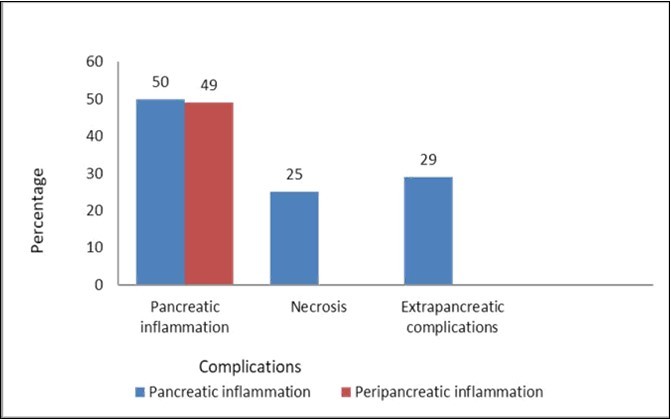
Figure 2.Number and percentage of patients with grades of severity assessment based on MCTSI.
The mean CTSI score ± S.D. was 4.04 ± 2.10. The mean serum amylase and serum lipase levels were 906.98 ± 660.629 IU/L and 354.79 ± 295.144 IU/L respectively. Mild negative correlation was seen between the serum amylase levels and the modified CTSI score, however the correlation was not significant (r= -0.104, p value=0.30). Similarly mild negative correlation was seen between the serum lipase levels and the modified CTSI score, however the correlation was not significant (r= -0.177, p value= 0.07). (Figure 3,Figure 4) (Table 1, Table 2 )
Figure 3.Scatter plot of the diagram showing correlation between serum amylase and MCTSI score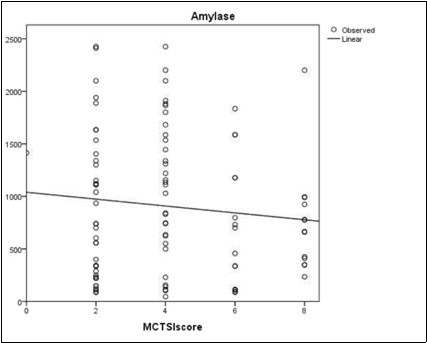
Figure 4.Scatter plot of the diagram showing correlation between serum lipase and MCTSI score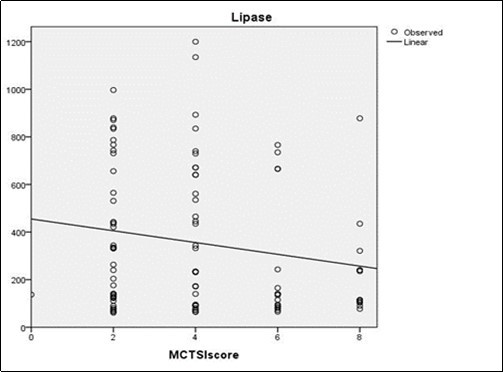
| Mean | Std. Deviation | N | |
| MCTSI score | 4.04 | 2.108 | 100 |
| Amylase | 906.98 | 660.629 | 100 |
| Lipase | 354.79 | 295.144 | 100 |
| Amylase | Lipase | ||
| MCTSI score | Pearson Correlation | -.104 | -.177 |
| Sig. (2-tailed) | .301 | .079 | |
| N | 100 | 100 |
Discussion
Initial clinical assessment of the progression of AP alone has been inadequate in identifying patients who develop a severe disease. Identifying severe cases are important and can play a significant role in management decision and in reducing the morbidity and mortality associated with severe AP. Ranson score, Glasgow score, Marshall and SOFA (Sepsis-related organ failure assessment) scoring system are different severity scoring system studied in the past and proven to be indicators of the clinical severity. However, most of the above systems are cumbersome to calculate and none of the above scoring system are proven to be the precise indicators of the adverse clinical outcome. Biochemical tests such as serum amylase, lipase, c-reactive protein(CRP) and trypsinogen levels have been variably studied by different studies to see any correlation with the severity of AP and variable results have been observed.
During the last two decades, management of severe AP has changed from a more aggressive surgical intervention towards a more conservative approach, except when infected necrosis has been confirmed. Hence it is very important from the treatment aspect to assess the severity of AP and the presence of necrosis by CECT. The CT severity assessment scoring system of AP has changed over a period of time. The Balthazar grading system did not incorporate the presence of organ failure8, extra pancreatic parenchymal complications 9,10 or peripancreatic vascular complications and their correlation with the final outcome. Secondly, as documented in some studies, inter-observer agreement for was only moderate, with a reported agreement of approximately 75%. 9, In view of these limitations, MCTSI was proposed by Mortele et al., which is easier to calculate and correlates more closely with the patient outcome measures like the occurrence of infections, organ failure, the need for surgical or percutaneous intervention, the length of hospital stay, and death than the CT Severity Index. In this regard, we also used the MCTSI for severity assessment in our study.
The mean serum amylase and serum lipase levels were 906.98 ± 660.629 IU/L and 354.79 ± 295.144 IU/L respectively. The mean CTSI score ± S.D. was 4.04±2.108. Mild negative correlation was seen between the serum amylase levels and the modified CTSI score, however the correlation was not significant (r= -0.104, p value=0.301). Similarly mild negative correlation was seen between the serum lipase levels and the modified CTSI score, however the correlation was not significant (r= - 0.177, p value= 0.079).
Serum amylase was elevated in 92% patients and lipase was elevated in 100% patients at presentation. In a study done by HS Batra et al 11, the findings were similar and 84% patients had raised amylase (79).
Similarly, Chamara Basnayake et al 12found that the serum concentrations of amylase and lipase elevated within hours of the pancreatic injury. A threshold concentration 2–4 times the upper limit of normal was recommended for diagnosis. Serum lipase was the preferred test due to its improved sensitivity, particularly in alcohol-induced pancreatitis. Its prolonged elevation created a wider diagnostic window than amylase. However, they concluded that neither enzyme was useful in monitoring or predicting the severity of an episode of pancreatitis in adults.
In a study done by Coffey MJ et al 13to evaluate the diagnostic yield and concordance for serum pancreatic enzymes and imaging in children with AP, overall, lipase, amylase, ultrasound US and CT were consistent with an AP diagnosis in 93% (93/100), 54% (43/80), 27% (21/77) and 67% (28/42) of cases respectively. The diagnostic yield for combinations of blood(s) and imaging(s) tests was higher than any single test and blood tests alone. The observed agreement between bloods 'lipase or amylase' and imaging 'US or CT,' was 40%. In 55% of cases, enzymes were positive whilst imaging was negative and the converse was evident in 5% of cases. There was no agreement between the various diagnostic tests, except between amylase and US, which had fair agreement. They concluded that the elevations in serum lipase contributed to the diagnosis more often than other tests. Combinations of blood and imaging tests had an increased diagnostic yield. At least 5% of cases of AP may be missed if imaging was not performed.
Sarfaraz Jasdanwala et al 14 found no advantage of testing both lipase and amylase, as well as no advantage in serially trending them for monitoring the clinical progress of the patient. Individual studies done by Ranson et al15, Clavien et al16, Winslet et al17and Pezzilli et alfound that serum amylase and lipase levels were not correlated to the severity of the disease.
The limitations of the study were small sample size and only two serum markers were taken into consideration. Future studies with other serum markers such as trypsinogen and CRP levels are recommended.
Conclusion
Among 100 patients 46% and 14% had moderate and severe grading of severity by MCTSI score respectively. The serum level of amylase and lipase had non-significant correlation with the MCTSI score and thus did not indicate severity of pancreatitis.
References
- 1.Lessard F, Dion R. (1989) for alpha-amylase isoenzymes by automated isoelectric focusing. Clinical chemistry. 35(10), 2116-8.
- 2.Malbergier A.Oliveira Júnior HPd. Sertraline and acute pancreatitis: a case- report. , Brazilian Journal of Psychiatry 26(1), 39-40.
- 3.Banks P A, Freeman M L. (2006) Practice guidelines in acute pancreatitis. , The American journal of gastroenterology.; 101(10), 1-23.
- 4.Isenmann R, BÜChler M, Uhl W, Malfertheiner P, Martini M et al. (1993) Pancreatic necrosis: an early finding in severe acute pancreatitis. Pancreas. 8(3), 358-61.
- 6.Alhajeri A, Erwin S. (2008) Acute pancreatitis: value and impact of CT severity index. Abdominal imaging. 33(1), 18-20.
- 7.Basterra G, Alvarez M, Marcaide A, Delgado E, R de Otazu Díaz et al. (1999) Acute pancreatitis: evaluation of the prognostic criteria of the latest Balthazar tomographic classification. Revista espanola de enfermedades digestivas: organo oficial de la Sociedad Espanola de Patologia Digestiva. 91(6), 433-8.
- 8.Balthazar E J, Freeny P C, vanSonnenberg E. (1994) Imaging and intervention in acute pancreatitis. , Radiology 193(2), 297-306.
- 9.Mortele K, Mergo P, Taylor H, Ernst M, Ros P. (2000) Renal and perirenal space involvement in acute pancreatitis: spiral CT findings. , Abdominal imaging 25(3), 272-8.
- 10.Wiesner W, Studler U, Kocher T, Degen L, Buitrago-Tellez C et al. (2003) Colonic involvement in non-necrotizing acute pancreatitis: correlation of CT findings with the clinical course of affected patients. European radiology. 13(4), 897-902.
- 11.Batra H, Kumar A, Saha T, Misra P, Ambade V. (2015) Comparative study of serum amylase and lipase in acute pancreatitis patients. , Indian Journal of Clinical Biochemistry 30(2), 230-3.
- 12.Basnayake C, Ratnam D. (2015) Abnormal laboratory results: blood tests for acute pancreatitis. Australian prescriber. 38(4), 128.
- 13.Coffey M J, Nightingale S, Ooi C Y. (2014) Diagnosing acute pancreatitis in children: what is the diagnostic yield and concordance for serum pancreatic enzymes and imaging within 96 h of presentation?. , Pancreatology 14(4), 251-6.
- 14.Ranson J. (1982) Etiological and prognostic factors in human acute pancreatitis. , Am J Gastroenterol 77, 633-8.
- 15.Clavien P-A, Robert J, Meyer P, Borst F, Hauser H et al. (1989) Acute pancreatitis and normoamylasemia. Not an uncommon combination. Annals of surgery. 210(5), 614.
