Abstract
The growth characteristics of Conraua goliath (Boulenger, 1906), an endemic and endangered species in Cameroon, were evaluated in its natural habitat between October 2021 and November 2022 in order to contribute to its domestication and conservation. For this purpose, 139 specimens (55 males, 55 females, and 29 individuals of undifferentiated sex) were collected by fishermen from 3 sites (Loum, Penja, and Nlonako) in the Department of Moungo, Coastal Region of Cameroon. The frogs were measured, and their growth parameters were determined after dissection and macroscopic and microscopic observations. The maximum size observed is 33.5 cm (SVL) and 2529 g body weight (BW), and the minimum size is 5.3 cm (SVL) and 12 g body weight (BW). This species is more abundant in the rainy season, in frogs between 18 and 24 cm, and has a Caucasian and uni-modal size structure. Independent of sex and season, this species has a significant and high regression coefficient between length, weight, and isometric growth pattern. However, this growth varies according to sex. It ranges from isometric in males (b = 2.82) and undifferentiated sexes (b = 2.85) to negative allometry in females (b = 2.21). The condition factor K ranges from 3.28 to 14.24 (mean: 7.80 ± 1.95) and fluctuates according to the sex and size of the frog. According to our results, this species has a large size and a high growth rate and could therefore be an excellent candidate for domestication to ensure its conservation. However, studies on reproductive characteristics are needed to complete this work.
Author Contributions
Academic Editor: Eman Hashem Radwan, Damanhour University
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2023 Liliane Fleurette Moumegni Monthe, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
The world is facing a massive decline in amphibian biodiversity 1 due to overexploitation and fragmentation of their habitats 2, 3.According to the IUCN 4, approximately 220 amphibian species are used for food. In 2001, the Food and Agriculture Organization of the United Nations estimated the annual global consumption of frog legs at 4,716 tonnes 5. Furthermore, livestock farms are a very small part of this market and more than 85% of the amphibians consumed would come from the wild, at the expense of the species concerned 6, 7. In Africa, the majority of over-exploited amphibians are edible frogs8, 9. In countries such as Republic of Nigeria, Benin, Burkina Faso, Republic of Ghana and Madagascar, frogs are widely used as a source of animal protein, leading to a thriving trade 7, 9, 10, 11.
In Cameroon, the consumption of goliath frog meat is mainly observed among some populations in the coastal and southern regions 12, 13, 14. Its growing market demand could be one of the main factors that significantly contributed to the decline of its natural population 12, 13, 15, as it is classified as an endangered species by the IUCN 1. Despite its commercial importance and potential use as an aquaculture species, studies interested in its bio ecology are quite rare and limited 16. Previous studies on the African giant frog have mainly focused on its distribution 15 and phylogeny 17, 18. Some investigations have provided information on different aspects of the life cycle, including parental care, parasites and diet12, 13, 19, 20, but no studies have been done to determining growth characteristics. However, the development of production of this species implies the control of its biological cycle. For that purpose, knowledge of growth and reproduction in the natural environment is essential. In order to contribute to a better understanding of the life cycle of Conraua goliath in its habitat and to assess its potential for aquaculture, this study investigated several aspects of growth, such as population structure, length-weight relationships and condition factor (K), which are crucial for the domestication of this species to ensure its conservation and enhancement.
Material and methods
Study area and period
The study was carried out in the Department of Moungo, the coastal region of Cameroon (Figure 1), more specifically in the districts of Loum (LN: 4o41 'and LE: 009o43'), Penja (LN: 4o45 'and LE: 009o 45') and Nlonako (LN: 4o40' and LE: 009o43') with an average altitude of 665 m. The study area is located in the Littoral Region of Cameroon. This area is characterized by an average temperature of 22.4°C, an average annual rainfall of 2700 mm 21, 22, and a relative humidity of 75%. The hydrographic network is dense and includes three main rivers, namely the Dibombé, the Mabombé and the Nkam, as well as a large number of rivers and streams. These rivers are perennial and their flow varies with the seasons.
Figure 1.Location of the study area and hydrographic network.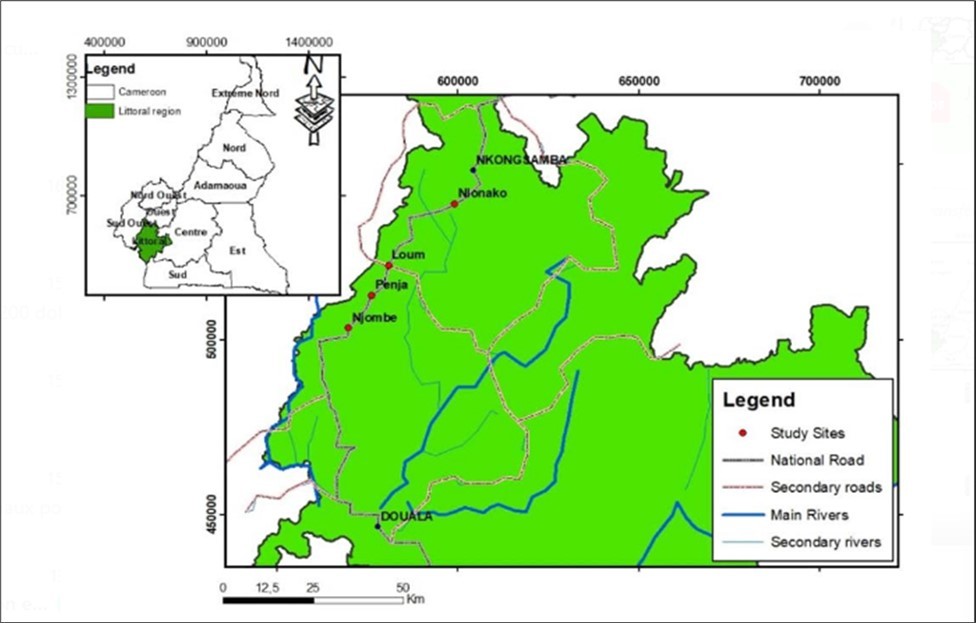
Animal Material and Essay Conduct
A total of 139 specimens of Conraua goliath, 55 females, 55 males and 29 juveniles were collected between October 2021 and November 2022 at the sites of Loum, Penja, and Nlonako. The frogs were captured at night or before sunset using traps and hawks, then placed in plastic containers with air holes filled with river water at a depth of about 5 cm. The next morning, the frogs were individually weighed using a 1g precision portable scale, anaesthetized with chloroform and then sacrificed by cutting the nerve cord behind the brain 23. Each specimen was marked according to the date of capture, packed in a glacier and examined within 12 hours at the Laboratory of Animal Biology and Physiology of the University of Yaoundé 1.
For each animal, the total length (TL); the snout – vent length (SVL) was measured using a 1 mm precision tape measure and an electronic Vernier caliper (F.F. The resolution is 0.01 mm / 0.0005 inc). Body weight (BW) and eviscerated weight (EW) were obtained using an electronic scale balance (Sartorius Competence) of 0.01g precision.
A longitudinal incision was made in the abdomen to carefully, remove the liver and gonads, which were weighed using a 0.01g precision electronic balance.
The sex of the animals was determined after dissection by the presence of eggs in females and testicles in males. Maturity was determined by visual inspection of the gonads. On the basis of this examination, individuals whose sex could not be determined were considered immature (sex undetermined).
Studied Parameters
Population structure and weight-length relationship
The size structure of the populations was determined according to the snout - vent length (SVL) 24.
The total length / Snout – vent length and body weight – eviscerated weight relationships were determined by linear regression according to the least squares method, and the equations were as follows:
TL = a + b SVL and BW = a+ b EW
Where a is the ordonate of the origin and b the slope of the regression line. TL = total length, BW = Body weight, EW= eviscerated weight.
The length-weight relationship was determined according to the method defined by Peig and Green 25 in the form BW = a SVLb,
where a is the regression constant, b is the regression coefficient or allometric coefficient, SVL = Snout – vent length (cm) and BW = Body weight (g).
Condition Factor K
The condition factor K, was calculated using the allometric method from the following formula:
K = 100 X BW/SVL b ,
Where BW is the Body weight and SVL is the size of the individuals. To estimate the value of the coefficient b, the weight-length equation was established (BW = a SVLb) and applied to all the individuals collected 23, 25, 26, 27.
Statistical analysis
Descriptive analysis (means, standard deviations, percentages) was used. The means were compared using the ANOVA I test, followed by the Duncan test to separate the means. The statistical significance of the coefficient of determination R2 was estimated and the student’s t-test was used to check whether the values of b were significantly different from the isometric value of b = 2.50 at the 5% threshold.
Size classes were defined according to Sturge's rule 28.
Number of classes (NC) = 1 + 3.3 log10 (n)
Where n = total number of individuals. The class interval (I) was determined by the following report:
I = (SVL max - SVL min) / NC, where SVL= Snout –vent length.
All these analyses were carried out using SPSS version 21.0 with a probability threshold of 5%. The Microsoft Excel 2019 spreadsheet was used to highlight graphs.
Results
Morphometric characteristics of Conraua goliath in the natural environment according to season, maturity stage and sex
The morphometric characteristics of Conraua goliath in the natural environment are shown in Table 1. It appears that the weight of the 139 specimens collected varied between 13 and 2529 g, with significantly higher values (P<0.05) in the dry season (mean: 860.30 ± 672.13 g, n = 65), in adults (mean: 963.78 ± 472.39 g, n = 110) and in males (mean: 1082.332 ± 576.91 g, n = 55). Similarly, their size varied between 5.3 and 33.5 cm, with comparable values (P > 0.05) depending on the seasons, and significantly lower in juveniles (mean: 10.05 ± 2.38 cm, n = 29).
The snout – vent length (SVL) varied between 12 and 33.5 cm in males (mean: 22.35 ± 4.60 cm, n = 55), between 12 and 31 cm in females (mean: 21.11 ± 3.09 cm, n = 55), and between 5.3 and 14.3 cm in juveniles (mean: 10.05 ± 2.38 cm, n = 29).
Table 1. Morphometric characteristics of Conraua goliath in their habitat according to season, maturity stage and sex.| Factor | N | BW (g) | SVL (cm) |
|---|---|---|---|
| Mean ± Standard deviation | Mean ± Standard deviation | ||
| Season | |||
| Dry season | 65 | 860.30 ± 672.13b | 19.28 ± 6.63a |
| Rainy season | 74 | 686.67 ± 394.74a | 19.30 ± 5.46a |
| Maturity stage | |||
| Adults | 78 | 963.78 ± 472.39 b | 21.86 ± 3.93 b |
| Juveniles | 29 | 87.54 ± 50.91 a | 10.05 ± 2.38 a |
| Sex | |||
| Male | 55 | 1082.33 ± 576.91 c | 22.35 ± 4.60 c |
| Female | 55 | 812.12 ± 285.57 b | 21.11 ± 3.09 b |
| undetermined sex | 29 | 87.54 ± 50.91 a | 10.05 ± 2.38 a |
Structure by size of Conraua goliath in the natural environment according to season
The size frequency distribution of this species' populations (Figure 2a, Figure 2b, and Figure 2c) is Caucasian and unimodal (20 cm), with sizes between 18 and 24 cm being most prevalent, and large sizes (29 cm) being less common. When looking at the seasons, the frequency peaks are practically identical. In the dry season, the frequency is bimodal (22 and 23 cm) and unimodal in the wet season (20 cm).
Figure 2.Size frequency distribution of Conraua goliath depending on the: (a) total population; (b) dry season, and (c) rainy season in its natural environment: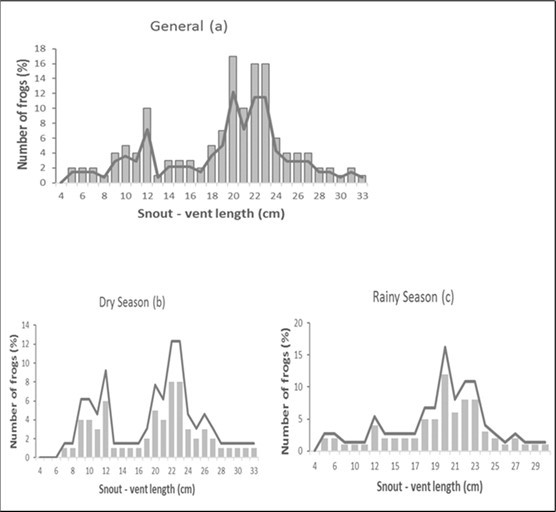
Body weight / eviscerated weight and Total length / Snout – vent length relationships
Body weight has a strong positive and significant correlation (P < 0.05) with eviscerated weight (r = 0.992). The regression equation was PT = 0.846 Pev + 6.380 (R2 = 0.986, N = 139), (Figure 3a).
Total length is strongly and significantly (P 0.001) correlated with snout-vent length (r= 0.984). (Figure 3b) The regression equation was TL = 2.245 SVL+ 1.791 (R2= 0.969, N = 139).
Figure 3. Total weight-eviscerated weight relationships (a), Total length- snout vent length relationship (b) of Conraua goliath in its natural environment.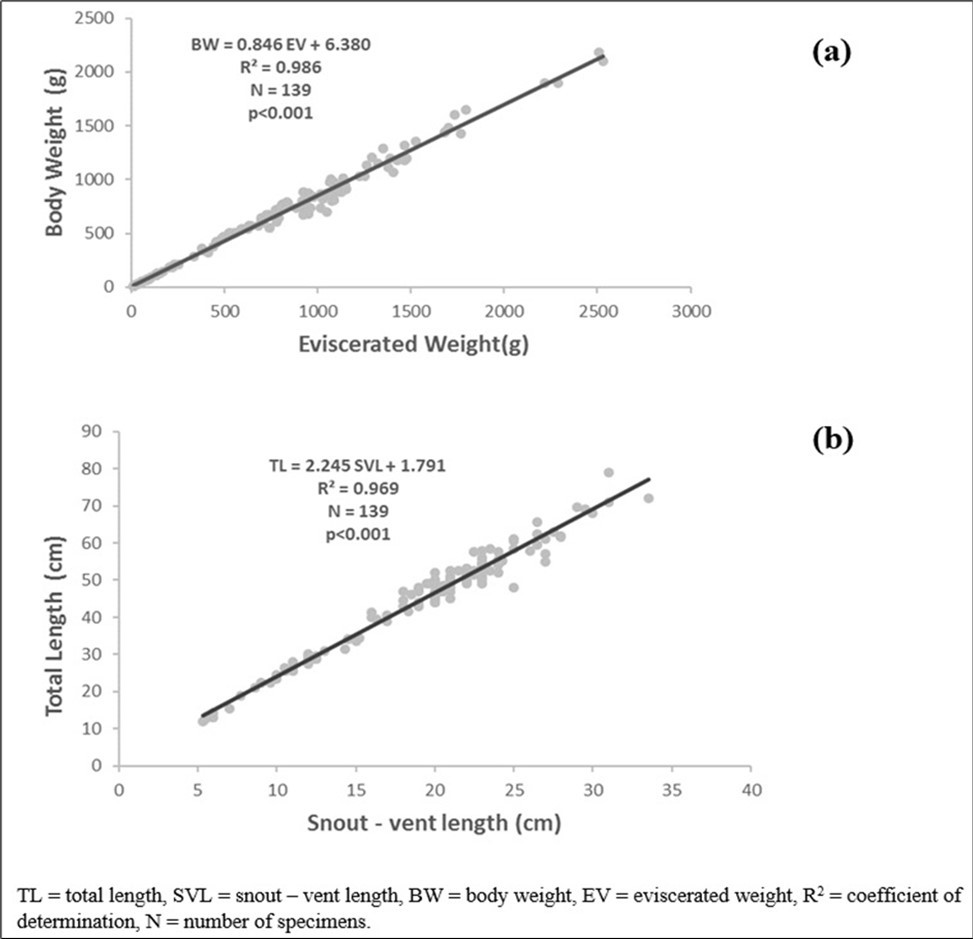
BodyWeight / Snout – vent length relashionship of Conraua goliath
Snout-vent length is strongly and positively correlated (P <0.05) with body weight (R2=0.975). The general equation was BW = 0.074 SVL3.033 (Figure 4). In addition, the value of b was significantly higher than 2.50 (P<0.05). Regarding the sex, equations were BW = 0.136 SVL2.853 (R2 = 0.867, N = 55), BW = 0.898 SVL2.213 (R2 = 0.613, N = 55), and BW = 0.096 SVL2.889 (R2 = 0.953, N = 29) respectively for males, females, and undetermined (Figure 4). b was less than 2. 50 in females and greater than 2.50 in males and undetermined sex (P < 0.05), indicating two types of growth respectively: negative allometric growth pattern (b=2.213) and isometric growth pattern (b=2.853 and b=2.889).
Figure 4.Body weight – snout – vent length Relationship of Conraua goliath depending on sex in its natural environment 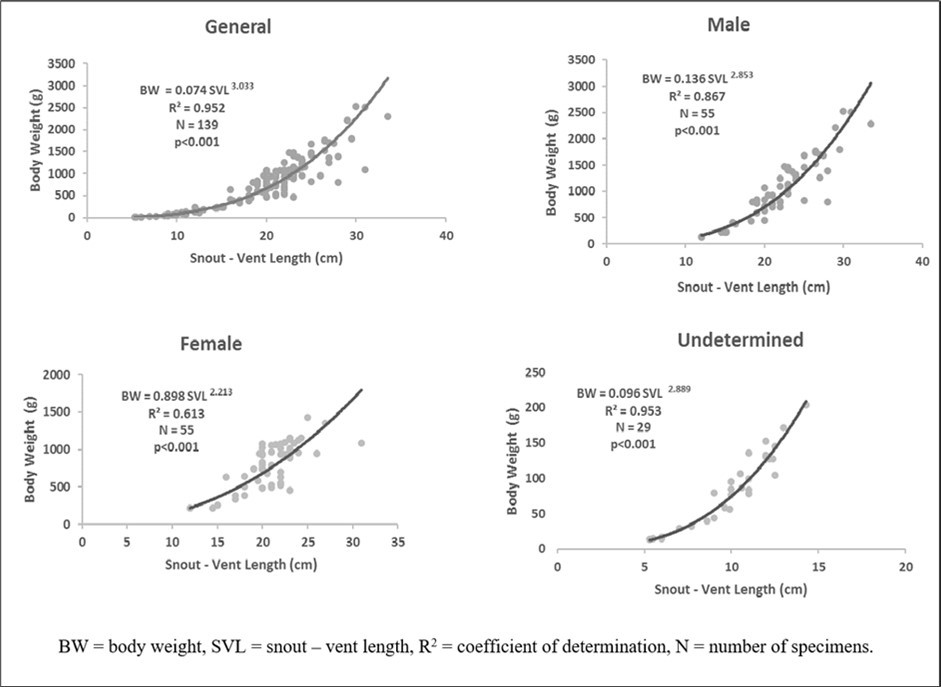
Body Weight / Snout – vent length relationship, growthpattern, and condition factor K depending on season
Depending on the season, snout-vent length is positively and significantly correlated (P < 0.05) with body weight (dry season: r =0.988; rainy season: r =0.964). The allometric coefficient b, was larger than 2.50 (P 0.05), indicating isometric growth (dry season: b = 3.24; rainy season: b = 2.84). Table 2 summarizes the condition factor k values, which show that they were comparable (P > 0.05). The highest readings, however, were recorded during the dry season (7.91 1.61).
Table 2. Body Weight – Snout Vent Length Relationship, growth type and condition factor K of Conraua goliath according to season.9906009264015N = number of specimens; BW= Body weight; SVL = Snout Vent Length; R² = Coefficient of determination; r = Coefficient of correlation; a = significance threshold; b = allometric coefficient, Mean ± Sandard Deviation, ***high significant correlation (p < 0.001). 00N = number of specimens; BW= Body weight; SVL = Snout Vent Length; R² = Coefficient of determination; r = Coefficient of correlation; a = significance threshold; b = allometric coefficient, Mean ± Sandard Deviation, ***high significant correlation (p < 0.001). 990600784161500| Season | Body weight – Snout - Sent Length Relationship Parameters | Type of growth | Condition factor K | |||||
| N | Equation BW = a SVL b | R 2 | r | a | b | |||
| Dry | 65 | BW = 0.042 SVL 3.241 | 0.978 | 0.988*** | 0.042 | 3.24 | isometric | 7.91 ± 1.61a |
| Rainy | 74 | BW = 0.128 SVL2.840 | 0.930 | 0.964*** | 0.128 | 2.84 | isometric | 7.69 ± 2.21 a |
| Total | 139 | BW =0.074 SVL 3.033 | 0.952 | 0.975*** | 0.074 | 3.03 | Isometric | 7.80 ± 1.95 |
Condition factor K depending on sex and size classes
Figure 5a and Figure 5b illustrate how condition factor K varies with sex and total length class. In general, the values of this parameter ranged from 3.28 to 14.24 (mean: 7.80 ± 1.95). When considering the sexes, the highest values were recorded for males. In fact, the condition factor varied from 3.28 to 12.14 (mean = 8.13 ± 1.87) for males, from 3.63 to 13.28 (mean = 7.84 ± 2.23) for females, and from 4.95 to 10.14 (mean = 7.08 ± 1.26) for undifferentiated sexes. However, no significant differences in sex were found for this parameter (P>0.05).
The condition factor K had statistically significant low values (P<0.05) (mean: 5.45 ± 2.15) in frogs having total lengths greater than 70 cm compared to values obtained in frogs with total lengths between 40 and 45 cm (mean: 9.29 ± 2.61).
Figure 5. Condition factor K depending on the sex (a) and size (b) in Conraua goliath 
Discussion
Size structure of the population
The maximum size (SVL) observed in Conraua goliath was 33.5, corresponding weight of 2529g. This is the world largest frog species 29. This biological characteristic is a good indicator of zootechnical performance that can be expressed in livestock environments. In this study, large-sized subjects are much more common in males than females, which contradicts the general patterns of body size variation in anurans that females are larger than males. 30, 31, 32, 33, 34. Studies conducted on parental care behavior in Conraua goliath by Schäfer et al. 12 report that, the atypical nest construction could explain the gigantism observed in this species. In fact, according to the research carried out by these authors, the males took care of their offspring by constructing nests with rocks that weighed up to 2 kg, or two-thirds of their own weight. According to the same authors, it is not because these frogs are large that they build such nests, but it is because they initially decided to build these nests that they became bigger over time. However, some studies corroborate our findings that size sexual dimorphism may be in favor of males 23, 35, due to the beneficial effect of competing for food resources 36.
The distribution frequency of the sizes is bimodal in the dry season (22 and 23 cm) and unimodal in the rainy season (20 cm), and the largest number of individuals collected is concentrated between 18 and 24 cm. These differences may be related to different environmental conditions and a high segregation in microhabitats in terms of maturity stages 15. On the other hand, these modes represented on the histogram in the dry season would correspond to the breeders who gave birth to individuals who became juvenile in the rainy season. Our observations also revealed that C. goliath is relatively abundant during the rainy season. This result is consistent with that of the study of Rhinella yanachaga in Peru 37 which shows that this species is abundant during the rainy season. Indeed, the breeding season in the Goliath frog is between October and March, which could explain the abundance of C. goliath during this season since the genitors and juveniles derived from the metamorphoses of the calves dense the effectiveness of the population. Several studies have shown that anurans are more abundant during the rainy season, as higher rainfall promotes their development and reproduction 38, 39, 40, 41, 42. For example, in Venezuela, during the heavy rainy season, Hyalinobatrachium duranti showed high abundance in a cloudy forest 43. In fact, the constant presence of fog and clouds leads to a reduction in solar radiation, which has as a result a decrease in temperature and an increase in relative humidity that favor the activity of amphibians throughout the year 44.
Weight - length relashionship
A high correlation have been observed between Body weight and Snout vent length of Conraua goliath, confirming the results obtained by Nguessu 13 in studies conducted on the same species in the Moungo and Nkam.
According to Vazzoler 45, populations with a regression coefficient b between 2.50 and 4.00 have an isometric growth pattern. The regression coefficient (b = 3.03) obtained in C. goliath shows that the shape of the body has not changed with growth, which is called isometric growth pattern. These results are comparable to those obtained in L. macrosternum from Brazil (b = 3.012) 27 and L. latrans from Argentina (b = 3.13) 23. In this study, males are significantly larger than females. It is also shown that the sex of the frogs influences the relationship between Snout – vent length and body weight. This disparity in the weight-length relationship could be explained by sexual dimorphism, which leads in distinct body structures between the sexes and thus dramatically different b regression coefficients. Thus, growth range from isometric in males (b = 2.82) and undifferentiated sexes (b = 2.85) to negative allometry in females (b = 2.21). This difference in growth between males and females could be explained by the breeding season, during which females preparing to lay eggs stop feeding, whereas males do not, as they are occupied with preparing to lay eggs and therefore expend more energy and must compensate for this by eating more. The value of the regression coefficient b observed in C. goliath ranged from 2.89 in the rainy season to 3.24 in the dry season. This result contradicts the hypothesis of Brown et al. 46 that during the rainy season, frogs may exhibit greater size, higher growth rates, accumulation of energy reserves and increased gonadal development. However, the difference in regression coefficients between the two seasons could be explained by the fact that b depends on food availability, habitat type and even breeding season.
Condition factor K
The condition factor K is an index that indicates the effect of environmental factors on animal comfort 27. In this study, Conraua goliath recorded a lower value (7.80) than value observed (8.64) in Sclerophrys maculate 47. The highest K values were obtained in the dry season (7.91) compared to those recorded in the rainy season (7.69). In addition, this parameter oscillated between the different sizes, so we registered low and high values in both juveniles and adults. This variation can be attributed to the influence of the reproductive cycle and to an annual or seasonal variation in the abundance of food in the habitat, confirming the studies carried out by Brodeur et al. 23 on L. latrans. Energy reserves and body condition of amphibians are closely linked to their reproductive cycle, which in turn is highly dependent on climate. According to Zhelev et al. 48, the variation in this parameter may also be related to the presence of pesticides and fertilisers used in agriculture, which are found in the frog ecosystem. The low values recorded in females (7.84) indicate that C. goliath uses its energy reserves in muscle and fat during the reproductive period. In fact, during egg maturation, females significantly increase the use of gonadal adipose tissue and its decrease can be observed during this period 46. Studies conducted by Chaves et al. 27 on the species L. macrosternum showed a significant correlation between the condition factor k values and the gonado-somatic ratio, which reflected greater energy expenditure for gametogenesis. On the other hand, higher K values in males (8.13) may be associated not only with a higher intensity of the food activity of the species Conraua goliath, with subsequent accumulation of body fat, but also with the fact that males have the burden of preparing for the ponte and therefore expend more energy and, therefore, must compensate for these effort by feeding more. It is interesting to note that a lower value of K was recorded in juveniles (7,08). This result corroborates that obtained by Brodeur et al. 23 with L. latrans. This could be related to their sexual maturity. In fact, juvenile frogs have not yet started to accumulate energy reserves and gonadal tissue for reproduction. However, literature contains very little information about the body condition and energy reserves of juvenile frogs to further explain or describe this phenomenon.
Conclusion
At the end of this study on the structure size, weight-length relationship, and condition factor K of Conraua goliath in the natural environment, the main conclusions are as follows:
Analysis of the size structure of Conraua goliath reveals a significant difference in size between the sexes. Independently of season, the maximum size was observed in males, while the minimum size was recorded in undetermined, followed by females. The frequency of size distribution varies according to the season. This species is more abundant during the rainy season.
The frogs captured in this habitat showed an isometric growth pattern. However, this growth varied according to sex. It ranged from isometric in males and undetermined sexes to negative allometry in females.
The condition factor K was greater than 1 regardless of season or sex, which indicates good physical condition in the habitat. The values of this parameter were significantly different between the length classes, but comparable between the sexes.
Conraua goliath can be considered as a large frog, a biologically characteristic species that is used to indicate optimal zootechnical performance. However, to better understand its life cycle, its growth characteristics need to be studied.