Abstract
Background:
Monitoring ultrasonic probe quality remains an important problem which impacts diagnostic accuracy. Here we present a quantitative method to assess probe quality primarily based on measuring probe maximum contrast (dB) and dynamic range.
Method:
Contrast relevant parameters of 26 transducer models manufactured by five manufacturers were measured with a novel Random Void Phantom (RVP) approach. 3D-data were acquired and analysed to determine image quality.
Results:
Acoustic contrast values ranging from 15dB to 36dB were observed.
Conclusion:
By examining artefact producing side lobes, the novel RVP approach presented here permits a quantitative assessment of ultrasound probe quality.
Author Contributions
Academic Editor: Yudong Zhang, Guest Professor, Kyushu Institute of Technology, Japan.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2019 Eckhart Fröhlich
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Like all devices, ultrasound probes, even those that are new and unused are subject to varying imaging quality. This results from minor variations in their manufacturing such as the imprecise nature of gluing the acoustic lens to the probe. Ageing and constant use introduce further variations which affect imaging quality and therefore diagnostic accuracy. The degradation of image quality is initially small, however progressively increases over time. These changes are barely perceptible to the naked eye compounding the problem.
Hypothesis 1: Initial probe imaging quality and the rate at which it degrades is not the same for all probe models from all manufacturers. There are clinically significant differences between different models 1, 2, 3, 4.
Hypothesis 2: The loss of quality is pronounced in the side coils suggesting that imaging quality loss may be detected and quantified by observing the side coils 1, 5.
Hypothesis 3: The novel Random Void Phantom (RVP) can be used to asses probe imaging quality in an objective test system. Reductions in grayscale resolution and maximum acoustic contrast of an ultrasound probe can be quantified. Criteria based on these measured parameters can be used to determine the suitability of a probe for clinical use. Probes deemed to be of inadequate imaging quality can be removed from further use in order to preserve diagnostic accuracy 6, 7.
Method
Measurement of 26 different transducer models from five manufacturers was performed using the RVP. The RVP for grayscale resolution consists of an artificial water filled foam sponge submersed in a water filled tank, Figure 1.
Figure 1. Random Void Phantom: Water-filled artificial foam immersed in a water tank. The 3D image shows the ultrasound image of the sponge acquired with a matrix probe.
Synthetic foam has an acoustic absorption similar to echo free cysts (voids) in living tissue. The voids are filled with pure water being free of reflective contaminants. The remaining background noise in the voids is caused only by the side and grating lobes. The tolerances of the foam inhomogeneities for maximum acoustic contrast are in the range +/- 1.5 dB 1, 6.
The transducers to be measured were motor-driven, or passed over the RVP manually. The main and side lobes were recorded. The ultrasound image sequences were documented in 3-sec loops as AVI files and subsequently processed as 3D-data blocks.
Maximum acoustic contrast and grayscale resolution of the various ultrasound probes was evaluated with software from Tissue Characterization Consulting (TCC), Figure 2. The higher and narrower the amplitude, the sharper the acoustic contrast 6, 8, 9.
Probe measurement was performed at four sites; Trnava, Bratislava, Ludwigsburg and Tübingen. Data analysis was performed by the author J.S. using the TCC software developed by J.S. (Medical Technology Lange, Rostock & SonoGalerie, 18211 Bargeshagen, Schliemannstraße 6).
Results
Acoustic contrast, using the RVP, was documented with linear arrays and matrix transducers. Low intensity contrast differences are indistinguishable and appear nearly black to the naked eye because the human eye can only discern 16 gray levels. These small differences in the low intensity range can however be visualized and evaluated when projected onto the 8-bit grayscale (256 shades of gray) of modern equipment. This is possible when the dynamic range (DR) is properly set and results in a smooth contrast graph, Figure 2.
Figure 2. measurement side lobes in different depths. A high and narrow amplitude indicates a sharp acoustic contrast. (azimuthal = lateral; elevational = perpendicular to lateral = transversal)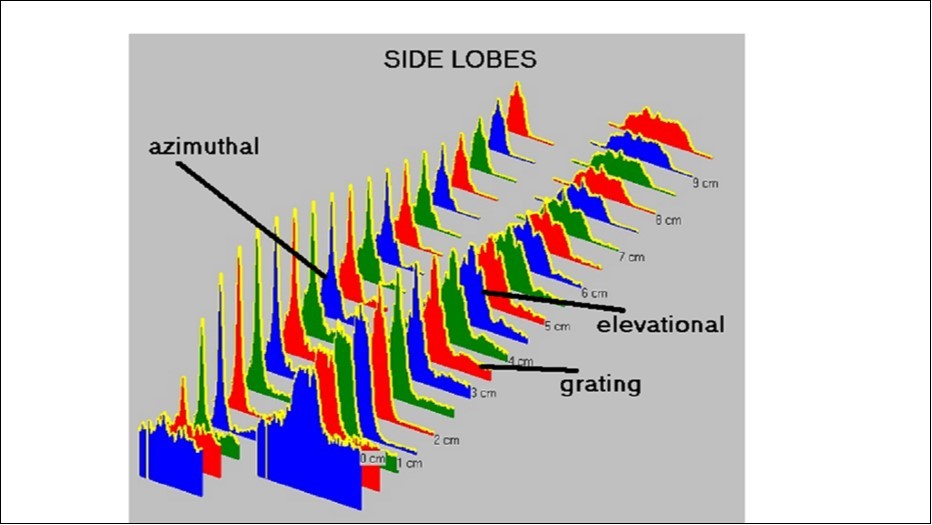
The maximum acoustic contrast and dynamic range of 26 transducers were measured. A surprisingly broad range of acoustic contrast values from 14.6 dB to 36.6 dB was observed, Table 1.
Table 1. Acoustic contrast values (dB) of 26 transducer models from five different companies| Company | Max. Contrast (dB) | Dynamic Range |
| > 25 dB Contrast: High Image Quality | ||
| Esaote My Lab 20 L523 | 37.25 | 100 |
| Philips IU22 ab L12-5 | 36.66 | 76 |
| Esaote My LabA533 | 36.08 | 100 |
| Philips IU22 L12-5 | 33.88 | 80 |
| Philips IE33 S5-1 | 32.71 | 60 |
| Philips IU22L17-5 | 32.63 | 80 |
| Philips IU22 dg L12-5 | 32.63 | 80 |
| ATL 350 C5-2 | 31.61 | 130 |
| Philips IU22 dg L17-5 | 29.67 | 85 |
| Esaote My Lab CA541 | 29.41 | 100 |
| ATL 350 L12.5 | 29.06 | 130 |
| Philips IE33 L9-3 | 28.55 | 70 |
| Philips IU22 C5-1 | 28.00 | 70 |
| Aloca SSD alpha C9130 | 26.73 | 96 |
| ATL 350 P4-2 | 26.51 | 130 |
| BK FlexFokus L8811 | 25.06 | 90 |
| < 25 dB Contrast: Average Image Quality | ||
| Philips IE33 ki55-1 | 24.71 | 60 |
| Philips IE33S5-1-3 | 24.71 | 60 |
| BK FlexFokus C8862 | 23.24 | 75 |
| Aloca SSD alpha L5548 | 22.61 | 93 |
| BK ProFokus L8811 | 22.24 | 90 |
| Aloca SSD alpha L5412 | 21.61 | 95 |
| Esaote My LabC PA240 23° | 20.00 | 100 |
| BK ProFokus C8820 | 17.65 | 90 |
| Philips EPIQ7c X5-1 | 15.53 | 60 |
| < 15 dB Contrast: Inadequate Image Quality | ||
| Aloca SSD alpha P52105 | 14.68 | 96 |
Probes with high acoustic contrast (32.63 dB to 36.66 dB) generated high quality images, Figure 3. Those with acoustic contrast values from 20 dB to 25 dB produced images of lower quality, Figure 4. A typical image produced by a damaged transducer measured as having an acoustic contrast of only 14.68 dB is shown below, Figure 5.
Figure 3. Random Void Phantom - transducer with high contrast resolution (36.66 dB, Philipps IU22.L12.5 MHz). Original 3D rendered image (left), with increased image contrast (right). Even voids within the RVP with weak contrast are discernible.
Figure 4. Random Void Phantom - medium contrast resolution (21.61 dB, Alloca SSD 5412). Original 3D rendered image (left), with increased image contrast (right).
Figure 5. Random Void Phantom) - damaged transducer with inadequate contrast resolution (14.68 dB, Alloca SSD P52105). Original 3D rendered image (left), with increased image contrast (right). Small voids within the RVP with weak contrast cannot be visualised.
Discussion
Acoustic Contrast in Living Tissue
The sonographic abdominal examination is regularly challenged by image contrast loss. Acoustic contrast is diminished by abdominal adipose and connective tissue. Additionally, small echo free objects are obscured by side lobe formation in fatty layers and other types of tissue. This predictably occurs with transducers having low acoustic contrast. Since the 1990´s, numerous investigators have shown the necessity for an acoustic contrast higher than 30 dB for all abdominal and chest sonographic examinations if diagnostic accuracy is to be maintained. When acoustic contrast falls below, echo free structures such as cysts in non-homogeneous tissues may not be visualised 10, 11.
We reviewed which transducers have high acoustic contrast and whether this information is well described by the technical specifications published by the manufacturer. It remains common practice to define the quality of probes and devices with technical parameters such as near field, focus zone, far field, and spatial resolution 12, 13.
These technical parameters remain stable, however provide no insight on how a particular transducer ages. Moreover they capture only a subset of clinically important technical parameters determining probe quality. Acoustic contrast is inappropriately ignored by this approach.
Detachment of the acoustic lens and changes in piezoelectric performance – caused by damage or wear – cause detrimental side lobes to multiply. Ultimately, transducer image contrast, image uniformity and image quality are reduced in a diagnostically significant manner.
Probe Quality Check
Previously, the quality of ultrasound probes was assessed with 2D phantom images displayed on the monitors of diagnostic ultrasound equipment. Image quality was determined by the ultrasonic wave front. All probes manufactured, independent of cost, are imperfect in that small and sometimes large side lobes are universally present. Surprisingly large variations in maximum acoustic contrast between brand new transducer models produced by different manufacturers have been observed. Minor deviations from design tolerances (e.g., acoustic apodization defects, irregularities in the adhesion of piezoceramics to the acoustic lens) likely explain the broad range of performance that we observed.
Our aim was to asses simple, appropriate and practical methods for ultrasound probe image quality monitoring. The RVP approach is a promising strategy. Side lobe detection requires reflectors in an echo-free environment. In practice, an echo free environment can only be simulated in water based phantoms 5, 8, 13.
If cysts (voids) are absolutely echo free, all side lobes being generated at the edges of the piezoelectric transmitter and the receiver will appear inside the cyst. Artificial foam in the form of a sponge submersed in water (RVP) is suitable for quantifying the grayscale resolution of an ultrasound probe. The sound attenuation in the foam behaves as in living tissue; the cavities (voids) are free of inclusions. The acoustic contrast thus largely depends on the side lobes.
The RVP model as described above is ideal for such a purpose with an attenuation that comes very close to echo free cysts (voids) in living tissue. When the voids are filled with pure water they are free of reflective impurities and only side and grating lobes can be the cause of apparent reflections within the voids. The number of side lobes ultimately determines image quality, or in other words the "acoustic contrast".
The 3D-acoustic contrast image is quantitative information about the quality of the probe which determines the limits of what can be done with optical image contrast enhancement.
The spectrum of grayscale values can be quantitatively expressed along three spatial axes; x, y & z. When the acoustic contrast is low, all three b-mode imaging planes appear black to the naked eye. When the dynamic range is properly set on the ultrasound equipment, diagnostically important small differences become perceptible 6.
Conclusions
This work highlights the importance of transducer maximum acoustic contrast for the imaging of non-homogeneous tissues. The best grayscale differentiation and highest acoustic contrast were achieved by matrix probes.
Based on our observations, applications demanding high grayscale differentiation as abdominal and breast examinations should only be performed with transducers measured as having a maximum acoustic contrast greater than 25 dB in order to optimally detect different tissue types. Ultrasound probes with an acoustic contrast between 15 dB and 25 dB are only appropriate for examinations where grayscale differentiation is not critical, such as is the case for orthopaedic examinations. Probes with a measured acoustic contrast of less than 15 dB should be retired to preserve diagnostic accuracy.
With the simple free hand scan transducer testing on RVP with 3D-data acquisition, quantitative determination of probe contrast quality and detection of small sound probe errors are possible.
Abbreviations and Definitions
• Design Parameters - The primary design of the probe, the hardware with defined near field, focus zone, far field and spatial resolution.
• dB - Decibel.
• DR: Dynamic Range; Ratio of the largest to the lowest signal amplitude; the lower limit is due to background noise, the upper limit is at the overdrive limit.
• Contrast (acoustic contrast): Quality differences between probes of the same design are generally associated with side lobes that increase in relation to the main lobe. The difference between major and minor lobes was therefore introduced as a new quality parameter, as acoustic contrast (or "contrast" for short).
• Quality Parameters - Probes of the same design can have different imaging quality due to production tolerances, or ageing processes. Thus, the lack uniformity of the piezo elements, or the acoustic lens can cause side lobes that acoustic contrast.
• RVP: Random Void Phantom - (Cyst Phantom)
• TCC: Tissue Characterization Consulting; http://www.3dultrasound.euweb.cz
• TMM: Tissue Mimicking Material
Acknowledgments
The authors thank the employees of General Electric for inspiring the idea of quantitatively measuring the quality differences between probes. We also thank the hospitals in Trnava and Bratislava in Slovakia and clinics in Ludwigsburg and Tübingen in Germany for their invaluable collaboration.
Funding Source
For statistics, we received financial support from the "Verein für Medizinische Weiterbildung Stuttgart"