Abstract
The aim of this review is to compile our understanding of microRNA (miRNA) and its significance in Hypertension (HTN) pathophysiology. The wide spectrum of health disparity is one of the reasons for the dominance of HTN in humans for decades. We are striving hard to understand these variations, and we know to some extent that genetic susceptibilities do exist in HTN. Understanding miRNA will add to the current understanding of the disease process. In later parts, we discussed possible clinical implications of miRNAs in HTN as a biomarker of disease expression and its potential in prognostic and therapeutic applications in HTN.
Author Contributions
Academic Editor: Sandeep Kumar, Emory University and Georgia Institute of Technology
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2015 Sawan Jalnapurkar, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Over 70 million American adults (29%) suffer from Hypertension (HTN) 1. If this trend continues to rise, there will be additional 9.9% adults with HTN by 2030 2. HTN is a well-known risk factor for development of cardiovascular diseases, stroke, and End Stage Renal Disease. Even with advances in medicine and novel therapeutic agents, HTN was responsible for 1000 deaths/day in 2013 in the United States alone 3. One possible explanation of the higher mortality rate from cardiovascular diseases could be variability in disease presentation and diverse responses to therapy. This is further augmented due to ethnic diversity in the United States; for instance, HTN occurs earlier in onset and more prevalently in African American populations, suggesting that a gap exists in our understanding of HTN mechanics and its inconsistent presentation. Growing evidence in the field of miRNA could add to our current understanding of disease variation, and it could be a potential tool for diagnosis, prognosis, and therapy in HTN 2.
Hypertension Epidemiology and its Impact on Health Care Economics
HTN is one of the major risk factors for cardiovascular disease (CVD) and stroke 4. CVD risk is multiplied by factor of 2 for each BP increment of 20/10 mm of Hg 4. HTN is a major burden within the global population as well, with 972 million adults suffering from HTN worldwide 5. There are sizeable variations in the prevalence of HTN in the USA, with 43% and 45.7% of African American men and women respectively as compared to 33.9% and 31.3 Caucasian men and women 3. In a 45 year old healthy African American, the 40 year risk of developing HTN is 92.7%, while in Hispanic populations it is 92.4%. On the other hand there is relatively low risk of developing HTN among Caucasians (up to 86%), and an even lower risk in Asian populations (84.1%) 6. Furthermore, total life expectancy was found to be 5.1 more years for men with normal blood pressure and 4.9 more years for a healthy women as compared to their own gender at 50 years of age with HTN 7. The etiology of this great diversity lies in environmental factors and socio-economic conditions; it also adds significant financial burden on society. Healthcare costs, both direct and indirect (due to loss of productivity), attributed to hypertension alone were $73.5 billion in 2010 and are projected to be over $200 billion by 2030 2.
MiRNA Biology
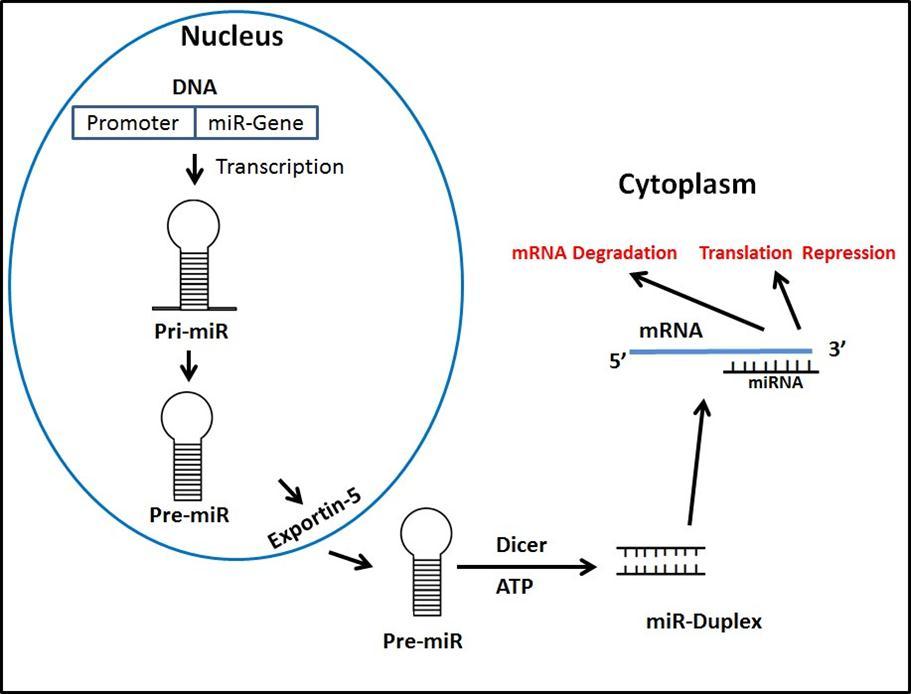
microRNAs are approximately 22-25 nucleotide long regulators of posttranslational activity of messenger RNA (mRNA). miRNAs play an important role in cell proliferation, differentiation, migration, and apoptosis during both normal development and disease progression 8. Two decades ago in 1993, Ambros, Ruykun, and their colleagues discovered a new unexpected cellular regulatory mechanism involving a non-protein coding transcript by using a C. elegans model 9, 10. It has been predicted that miRNAs regulate approximately 30% of the human protein-coding genome 11, and it was soon discovered that they exist across all forms of life. miRNA was first studied in malignancies where it was found to act as a tumor stimulant or tumor suppressor. Thus, controlling miRNA activity is crucial. miRNAs are first developed as longer molecules by RNA polymerase II called Pri-miRNAs. They are maturated by ribonuclease Drosha into hairpin structures called Pre-miRNAs and are transported into the cytoplasm where the loop is cut by another ribonuclease, Dicer, which forms a double stranded RNA molecule (Figure 1). One strand of this double stranded RNA is degraded, and then the remaining strand is incorporated into a RNA induced slicing complex (RISC) which further carries out its action 8. miRNAs act by binding to their target mRNAs at 3’-UTRs, using a partial base-pairing mechanism. miRNA at the 5’end has a sequence 7-8 nucleotides complimentary to the target mRNA which is called the “seed” region 12. The number of binding sites, overall degree of complementarity, and accessibility of the mRNA determines the inhibition or expression of miRNA that could lead to the translation or degradation of target mRNA 13, 14. Uncontrolled hypertension is one of the risk factors which leads to atherosclerosis and subsequently CVD. miRNA plays a crucial role in development of HTN, and there is potential for targeting these miRNAs as preventive and reparative therapeutics for HTN. We will now focus on the in vivo contribution of miRNAs in the etiology and pathogenesis in developing HTN.
Pathophysiology of Hypertension
Adequate perfusion to tissues depends on blood pressure, and this is determined by the interactions of cardiac, renal, vascular, and neurohormonal mechanisms, accompanied with altered angiogenesis and impaired platelet function 15. Any aberrancy in any of these mechanisms due to environmental or genetic susceptibilities 16, 17 results in developing HTN. Hypertension is a key risk factor for developing CVD, cerebrovascular accidents (CVAs), and target organ damage (TOD) 15.
miRNA in Vascular Mechanism:
Vascular Smooth Muscle Cell ( VSMC)
Vascular smooth cells (VSMC) constitute the medial layer of arteries and veins. VSMC contractility is important in vascular tone, which will determine vascular resistance, perfusion to tissues, and blood pressure. miRNAs have proved to play an integral role in the transcriptional regulation of VSMC development, phenotype, and function during vascular pathology. Lack of miRNA in embryonic or adult VSMCs results in embryonic lethality or loss of VSMC function respectively, suggesting that miRNA expression is necessary for the development and maintenance of VSMCs 18, 19.
microRNAs in Endothelial Dysfunction.
Vascular endothelial function controls vascular tone by synthesizing and releasing nitric oxide (NO), a potent vasodilator. Increased oxidative stress leads to reduced NO, resulting in impaired vasodilatation causing endothelial dysfunction - a potential mechanism mediating the adverse effects of hypertension. Reduced availability of NO makes vascular endothelium more prone for atherosclerosis and increases the stiffness of the arteries 20. Endothelial nitric oxide synthase (eNOS) is responsible for producing nitric oxide within the endothelium. miR-155 binds to the 3’ end of its target mRNA to control the expression eNOS, thus contributing to bioavailability of NO responsible for maintaining homeostasis 21. Sun et al. carried out an experiment in human internal mammary arteries that showed increased expression of miR-155 is inversely related to eNOS expression and NO 21. In addition, NO also functions as an anti-inflammatory substance, and it inhibits leukocyte adhesion, which in turn prevents vascular inflammation which leads to impaired vasodilation and hypertension 22. L-arginine is the precursor and key component for the synthesis of NO 23. SLC7A1 is a transporter gene required for normal transportation of Larginine and normal NO metabolism 24. Polymorphisms in the 3'UTR of SLCA1 may cause a decrease in gene expression 25. The 3’UTR of SLCA1 has binding sites for miR-122, and polymorphism in this site results in lower levels of SLC7A1. This reduced expression of SLC7A1 is responsible for deficiency of NO which results in endothelial dysfunction as seen in hypertensive subjects 26. Another study revealed that miR-9 and miR-126 are closely related to essential hypertension in humans as they show a distinct expression profile in hypertensive patients relative to healthy individuals. These miRNAs are associated with clinical prognostic indices of hypertensive target-organ damage in hypertensive patients 27.
microRNAs in Vascular Integrity and Impaired Angiogenesis
Endothelial cells constitute the innermost lining of vascular structures and play a pivotal role in maintaining vascular integrity, angiogenesis, and wound repair 28. Lack of balance between proangiogenic and antiangiogenic factor is found in hypertensive patients 29. Two factors, FGFs and VEGFs, are required for angiogenesis as they bind to receptors in order to activate MAP kinase activity, thus promoting angiogenesis. miR-126 is shown to control the activity of VEGF, subsequently controlling angiogenic signaling 28.
Wang et al. demonstrated that target deletion of miR-126 in mice results in antiangiogensis in vivo. miR-126 controls the expression of Spred-1, a negative regulator of MAP kinase signaling which promotes angiogenesis. The pro-angiogenic actions of miR-126 correlate with its repression of its target Spred-1. miR-126 deletion in mice resulted in enhanced expression of Spred-1, and corresponding reduced angiogenic signaling of VEFG and FGF 30. Nicoli et al. proved that klf2a, a mechanically sensitive transcription factor, stimulates the expression of miR-126, activating VEGF signaling. This further emphasizes the role of miR-126 in angiogenesis 31. Reduced angiogenesis within the level of microvessels called rarefaction results in target organ damage, a major pathogenic feature and complication of hypertension 32.
In another study, levels of miR-126 and miR-9 were analyzed in 60 patients with uncontrolled HTN and 29 healthy subjects 27. Hypertensive patients showed significantly lower miR-9 and miR-126. In order to identify target organ damage, subjects underwent 2-D echocardiography and 24 hour mean pulse pressure tests. The researchers found that LV mass index was positively correlated with miR-9, similar to what was shown in previous animal studies stating that miR-9 is a negative regulator of cardiac hypertrophy 33. miR-126 and miR-9 were positively related to the 24 hour mean pulse pressure, which can predict advanced target organ damage in HTN34. In addition, one study compared healthy male subjects between 30 years to 60 years of age with hypertensive subjects without other comorbid conditions. Increased expression of hsa-miR-505 was discovered in hypertensive subjects, which was reproducible in vitro 35. FGF18 was found be to be a direct target of hsa-miR-505 and is a pro-angiogenic factor responsible for antiagiogenic effects of has-miR-505.
Role of SNPs (Single Nucleotide Polymorphism) in HTN
Most miRNA binding sites are present in the 3’UTRs of RAS (Renin Angiotensin System) component of mRNAs. Probability of SNPs increase in occurrence at miRNA target sites. SNPs can modify, create, or destroy the efficiency of miRNA binding to mRNA if SNPs occur at miRNA (i.e. seed region) 36, 37, 38. Therefore, these miSNPs can either enhance the function of miRNA (reducing protein expression of target mRNA) or diminish the function of miRNA, causing overexpression of that particular mRNA. For example, if a loss-of-function miRSNP occurred in the AGTR1, ACE, REN, or ATP6AP2 gene, an increased incidence of hypertension, cardiac/vascular remodeling, and atherosclerosis would be observed. In contrast, if a loss-of-function miRSNP occurred in the AGTR2, ACE2, MAS1, or LNPEP gene, a decreased incidence of hypertension, cardiac/vascular remodeling, and atherosclerosis would be expected. To support this hypothesis, Yang et al. studied epistasis networks in eight SNP-SNP interaction models. AGT, ACE, and AT1R genes had overall effects of susceptibility to hypertension, and the SNPs of ACE had a hypertension-associated effect to show the interacting effects of the SNPs of AGT and AT1R genes 39.
miRNA in Renin-Angiotensin-Aldosterone-System (RAAS)
RAAS plays a vital role in controlling blood pressure via its biologically active component Angiotensin II (AngII). Its mechanisms include maintenance and response to hemodynamic changes, aldosterone production, renal function, thirst responses, sympathetic nervous system responses, and maintenance of appropriate peripheral vascular resistance. There are several miRNAs studied that influence RAAS (Table 1). Jackson el at (2013) studied the role of miR-181a in RAAS signaling and its contribution to HTN 40. Authors used genetically hypertensive mice from increased SNS tone and found elevated levels of RAAS during the hypertensive period correlated with higher Ren1 mRNA expression and reduced expression of renal specific miR-181a 40. It is a negative regulator of Ren1 mRNA expression proven in previous studies where transfection with miR181a in mice showed reduced levels of Ren1 and subsequently lower blood pressure 41. Marques et al. showed similar results in their experiments with human subjects in addition to miR 181a regulating renin expression via Ren mRNA. They also found miR-663 which controls renin expression via Ren and ApoE genes 41; however, sample size in the study was low and larger prospective studies are required to understand Renin regulation via microRNA. Kemp et al. showed that the miR-483-3p can possibly be a target in RAAS. Results showed that miR-483-3p is involved in regulation of AT2R, ACE and ACE2, all key components of RAAS 42. In another experiment, associations between SNPs in RAAS related genes were demonstrated, and the investigators genotyped a case control study on myocardial infarction Leiden (SMILE) for 10 SNPs in potential miRNA binding sites in 8 RAAS related genes, discovering 9 SNPs. Of these 9 SNPs, 4 were located in Arginine vasopressin 1A receptors (AVPR1A), bradykinin 2 receptors (BDKRB2), and thromboxane A2 receptors (TBXA2R). These genes were associated with blood pressure regulation. The rare allele of the AVPR1A SNP rs11174811 was associated with increased blood pressure, whereas the rare alleles of the two linked BDKRB2 SNPs rs5225 and rs2069591 as well as the TBXA2R SNP rs13306046 were associated with decreased blood pressure 43. Another interesting correlation from this study was in individuals with Down’s syndrome (DS) comparatively had lower systolic and diastolic pressures resulting in reduced risk of vascular anomalies including cardiovascular diseases. Analysis of fibroblasts in monozygotic twins with DS showed overexpression of miR-155. It is hypothesized that overexpression of miR-155 in DS reduces levels of the protein hAT1R (Human Angiotensin Type 1 Receptor) in fibroblasts and VSMCs. Reduced hAT1R also attenuates angiotensin II induced signaling in fibroblasts and VSMCs, resulting in lower systolic and diastolic blood pressures. 44, 45, 46, 47, 48, 49, 50, 51, 52 .
Table 1. MicroRNAs involved in pathophysiology of HTN and their target gene/protein and Functions.| Pathophysiologic process of HTN | Microrna / MiR Involved | Target mRNA protein | Function | References |
| Endothelial Dysfunction | ||||
| Polymorphism MiR 122 | ↓SLCA1Expression | ↓NO level | 26 24 | |
| MiR-155 Overexpression | ↓eNOS Expression | ↓NO level | 21 | |
| Vascular Integrity and Angiogenesis | ||||
| MiR126deletion | ↓Angiogenic signalling via VEGF and FGF | ↓Angiogenesis | 28, 30 | |
| MiR-505 | ↓FGF18 | ↓Angiogenesis | 35 | |
| RAAS | ||||
| MIR-155 overexpression | ↓hATR1 and ↓Ang II signaling | ↓ BP | 44 45 46 47 48 49 50 51 52 | |
| MiR-181a Overexpression | ↓Ren1 expression | ↓BP | 40 | |
| MiR 438-3pOverexpression | ↓ACE, ACE2, AT2R | ↓BP | 42 | |
| End Organ Damage | ||||
| Mir-9 and MiR126 | Thrap1 & Myostatin2 | ↑Hypertrophy | 27 |
Micrornas in Pulmonary Hypertension :
Pulmonary hypertension (PHT) is the result of proliferation of pulmonary artery smooth muscle cells (PASMC) and endothelial cells (PAECs) responsible for remodeling of pulmonary vessels and subsequently right heart failure 53. Pulmonary vascular homeostasis is regulated by the bone morphogenic protein receptor type II (BMPR2) signaling pathway and dysfunction in this pathway results in PHT 54. Brock et al. demonstrated that activation of STAT3 via IL-6 results in increased expression of miR-17-92 in PAECs. Specific microRNAs miR-17-5p and miR-20a have targets at BMPR2 resulting in its downregulation and is a possible mechanism of developing PAH 54. Hypoxia is another pathophysiologic process involved in developing PHT. Mizuno et al. showed that P53-miR34a clusters play crucial role in hypoxia induced PHT 55. miR 145 on the other hand is potentially an important entity in prevention of PHT induced by hypoxia. In wild type hypoxic mice, miR-145 found to have elevated expression and also in patients suffering from PHT
56. As described, miRNAs play vital roles in development of PHT. Its role as a therapeutic agent has potential, but it has yet to be studied extensively in humans to understand their targets and networks.
Summary
As previously discussed, miRNA appears to be involved in multiple pathophysiologic processes in the development of HTN. Although HTN has been identified for decades, it continues to be serious risk factor for cardiovascular disease and stroke. Understanding its disease variability and pathogenesis at the molecular level is still not completely understood. Development of hypertension is multifactorial, and the addition of miRNA machinery in its development is a promising and novel approach to close gap in understanding of HTN.
The field of miRNA is rapidly growing; its research is no longer limited to oncology. miRNA has been studied to identify its role in various disease processes. There are multiple methods that have been developed to control its expression. A single miRNA controls the expression of more than one gene, and multiple miRNAs are involved in controlling the expression of one gene, making the process of understanding miRNA mechanisms more complicated. Hence, diminishing or augmenting expression of one miRNA can have unwanted or unanticipated effects. Several ways exist to suppress the expression of miRNA, which include anti-miRNAs, miRNA inhibitors, miRNA sponges, and its mimic. The path to develop new therapeutics can be quite protracted and difficult. Recently, inhibition of miR-122 entered the clinical phase to treat hepatitis C virus infections. Multiple other miRNA inhibitors are soon going to be transitioning from the preclinical phase to the clinical phase. miRNAs are readily detected and are easily accessible in extracellular fluids such as blood and urine, and they are currently explored as biomarkers in a wide range of cardiac conditions. In order to efficiently use miRNAs in clinical practice and replace the current standard of care, rigorous research is needed to understand its molecular mechanisms, the extent of its effects, and its signaling pathways. More than 1800 miRNAs have been identified so far in humans. The miRBase (www.mirbase.org) database is a searchable database of published miRNA sequences and annotations, with more than 1800 miRNAs having been identified so far in humans 57. With many new miRNAs still being discovered, we will continue to increase our knowledge base about field of miRNA. It is certain that we have identified a controlling mechanism at the terminal stage of gene expression via miRNAs, and the discovery has impacts on various systems including the cardiovascular system. Understanding miRNA in detail also has the potential to close the gap in our lack of understanding of diversity in expression of disease profiles and variable responses to identical managements.