Abstract
The present study aims at integrating the characterization studies conducted on the Cuban germplasm collection of Xanthosoma sagittifolium (Araceae) preserved in the Research Institute of Tropical Roots and Tubers Crops (INIVIT). Differentiation of accessions was done by combining qualitative and quantitative descriptors to clarify the relationships between six well-defined groups. The results may be used to establish a core collection for improved management of Xanthosoma spp. germplasm.
Author Contributions
Academic Editor: Neelam Rani Yadav, Department of Molecular Biology, Biotechnology and Bioinformatics.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2018 Marilys D. Milián Jiménez, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Cocoyam (Xanthosomasagittifolium(L.) Schott, Araceae) is cultivated throughout the tropical areas and it plays a major role for food security in developing countries 1. It is an important food for 400 million people and has overtaken taro (Colocasia esculenta (L.) Schott.) as the main edible aroid in many tropical areas 2, 3 especially in Africa. This South American species was probably domesticated on the northern side of the Amazon basin. It was introduced by Portuguese slavers to Sao Tomé and Principe islands and from there to Central and West Africa. They were looking for high yielding crops that could feed the slaves before their transport to the West Indies. Its introduction in Asia and Oceania occurred in the 19th century and it was the result of missionaries’ intervention 4.
There are about 40 species of the Xanthosomagenus. The taxonomic position of the cultivated Xanthosomaspp. is unclear and there is a tendency to call all cultivated Xanthosomaspp., X. sagittifolium5, 6. The genus includes three important food species: X. sagittifolium, X. atrovirens and X. violaceum. Another species, X. brasiliense is grown for its edible leaves. Cultivars of X. saggitifolium are differentiated mainly by leaf pigmentation, plant size, cormel number and shape, cormel buds shape and pigmentation, spatial arrangement of cormels and cormel flesh pigmentation. Young leaves of some cultivars can be used as vegetable. The total number of collected accessions of Xanthosomaspp.is about 1000 1. Collections were made during the 1980s in Cameroon, Equatorial Guinea, Gabon, Ghana and Togo. Over 300 accessions were evaluated for yield and incidence of Pythium infection 7, 2. In Ghana, the collection of 70 X. sagittifolium accessions was thought to present sufficient diversity to interest plant breeders but ploidy levels were not determined 8. Cocoyam improvement has been done in Cameroon with some crosses producing seeds but not others, perhaps due to ploidy differences 9, 7, 2. DNA analysis of cocoyam collection in Florida showed very little genetic variation 10.
In Cuba, cocoyam (called malanga) is a very important crop with a fairly stable annual production around 220 000 tons. The average yields are about 16 tons per hectare in twelve months for the traditional Cuban cropping system 11. There is an evident need to identify accessions with superior traits that could be used as selected parents to initiate a breeding program. The Research Institute of Tropical Roots and Tuber Crops (INIVIT) has maintained a collection of Xanthosoma spp. since 1967 which represents one of the most diverse germplasm of cocoyam in the world. Some work has already been done, X.violaceum, X. atrovirensand X. caracu accessions have been morphologically described 12. Over the years, this collection has been described with morpho-agronomic descriptors, ploidy levels have been determined and isozyme markers have been used to clarify the genetic relationships among accessions 13 and to attempt to select cultivars best suited and resistant to the attack of pests and diseases. The present study aims at analyzing the existing diversity within the Xanthosomaspp. collection in Cuba by combining cytological, morphological and isozyme characterization of accessions.
Materials and Methods
The work was conducted in the Cuban germplasm collection of Araceae species at the Research Institute of Tropical Root and Tuber Crops (INIVIT) located in Santo Domingo municipality, Villa Clara Province, Cuba. Since its foundation in 1967, this institution assembled cocoyam (Malanga: Xanthosoma spp.) germplasm through collections, donations and introductions. The present collection includes 71 accessions (Table 1) preserved ex situ in field conditions and in vitro; 56 of them are Cuban and 15 are foreign introductions 13.
Table 1. Accessions studied from the Cuban national collection of Xanthosoma genus| No.* | Accessions | Cormel flesh color | Origin | No.* | Accessions | Cormel flesh color | Origin | |
| 1 | ‘Inivit-84’ | Light purple | Cuba | 37. | ‘Mexico-8’ | pink | Mexico | |
| 2 | ‘Morada Cabaiguán’ | Light purple | Cuba | 38. | ‘Morada De Mexico’ | pink | Mexico | |
| 3 | ‘Amarilla Riza’ | Yellow | Cuba | 39. | ‘Cuarentena’ | Light purple | SaoTomé and Príncipe | |
| 4 | ‘Amarilla Trinidad’ | Orange yellow | Trinidad and Tobago | 40. | ‘Cuarentena-1’ | Light pink | Sao Tomé and Príncipe | |
| 5 | ‘Riza’ | white | Cuba | 41. | ‘Batabala Blanca’ | Creamy white | Sao Tome and Principe | |
| 6 | ‘Blanca’ | white | Cuba | 42. | ‘Morada-2’ | purple | Cuba | |
| 7 | ‘Amarilla Criolla’ | yellow | Cuba | 43. | ‘Blanca-1’ | Creamy white | Cuba | |
| 8 | ‘Morada’ | Light purple | Cuba | 44. | ‘Tricolor’ | white | Cuba | |
| 9 | ‘Japonesa’ | pink | Indonesia | 45. | ‘Ceniza’ | purple | Cuba | |
| 10 | ‘Rosada’ | Light pink | Cuba | 46. | ‘Blanca-2’ | white | Cuba | |
| 11 | ‘Jordín’ | Light pink | Cuba | 47. | ‘Morada-3’ | purple | Cuba | |
| 12 | ‘Macal’ | purple | Mexico | 48. | ‘Blanca-3’ | white | Cuba | |
| 13 | ‘Amarilla Especial’ | yellow | Cuba | 49. | ‘Picante’ | Creamy white | Cuba | |
| 14 | ‘Javanica’ | Creamy white | Cuba | 50. | ‘Amarilla-1’ | Orange yellow | Cuba | |
| 15 | ‘Macal Sport’ | White | Cuba | 51. | ‘Blanca-4’ | Creamy white | Cuba | |
| 16 | ‘Blanca Venegas’ | white | Cuba | 52. | ‘Sergio Cuarentena’ | pink | Cuba | |
| 17 | ‘Criolla -1’ | Light pink | Cuba | 53. | ‘Morada-4’ | purple | Cuba | |
| 18 | ‘Amarilla’ | yellow | Cuba | 54. | ‘Blanca-5’ | white | Cuba | |
| 19 | ‘Stoupan’ | white | Guade loupe | 55. | ’Morada Ceniza’ | Light pink | Cuba | |
| 20 | ‘Belembe’ | Light yellow | Guadel oupe | 56. | ‘Amarilla-2’ | yellow | Cuba | |
| 21 | ‘Viequera’ | Creamy white | Puerto Rico | 57. | ‘Encintada’ | yellow | Cuba | |
| 22 | ‘Blanca Mutación’ | white | Cuba | 58. | ‘Morada-5’ | pink | Cuba | |
| 23 | ‘Blanca P. Del Rio’ | white | Cuba | 59. | ‘Blanca-6’ | Creamy white | Cuba | |
| 24 | ‘Morada 1727’ | pink | Cuba | 60. | ‘Blanca Morada’ | Light pink | Cuba | |
| 25 | ‘Mexico-1’ | Light pink | Mexico | 61. | ‘De Seda’ | yellow | Cuba | |
| 26 | ‘Mexico -27’ | pink | Mexico | 62. | ‘Americana’ | white | Cuba | |
| 27 | ‘Mexico-16’ | pink | Mexico | 63. | ‘Chopo Amarillo’ | Orange yellow | Cuba | |
| 28 | ‘Mexico-2’ | pink | Mexico | 64. | ‘Morada-6’ | Light purple | Cuba | |
| 29 | ‘Mexico-3’ | Light purple | Mexico | 65. | ‘Blanca-7’ | white | Cuba | |
| 30 | ‘Amarilla Ceniza’ | yellow | Cuba | 66. | ‘Morada-7’ | purple | Cuba | |
| 31 | ‘Blanqui-Morada’ | Light pink | Cuba | 67. | ‘Blanca-8’ | white | Cuba | |
| 32 | ‘Morada-1’ | purple | Cuba | 68. | ‘Blanca-9’ | Creamy white | Cuba | |
| 33 | ‘Morada-1726’ | Light purple | Cuba | 69. | ‘Morada Jibacoa’ | Light purple | Cuba | |
| 34 | ‘Blanca Selección‘ | white | Cuba | 70. | ‘Blanca-10’ | white | Cuba | |
| 35 | ‘Amarilla Espec. -4’ | yellow | Cuba | 71. | ‘Morada-8’ | pink | Cuba | |
| 36 | ‘Blanca Baracoa’ | white | Cuba | * Order Number Per Accession | ||||
Chromosome Counts
Counts were conducted on root tips. Five accessions of each accession were planted in polyethylene bags filled up with potting mix, according to the protocol developed 13.
Morpho-Agronomical Descriptors
Soil preparation, plantation and cultivation techniques were those standards for the cultivation of cocoyam in Cuba 14, 15 and so it was the methodology for the establishment of the germplasm collection 16. The germplasm collection was replanted every year in April and harvested 11 months later for eight consecutive years from 2006 to 2014. Morphological descriptions were conducted every year. A set of descriptors based on the list published by the International Board for Plant Genetic Resources 17 and the List of descriptors 13 was used, 27 descriptors were selected for the identification of accessions.
Isozymes Studies
Numerous enzyme systems were assayed using a variety of buffer systems but three enzyme systems were selected: Esterase (EC.3.1.1), Peroxidase (EC.1.11.1.7), Polyphenol-oxidase (EC1.10.3.1) 18.
For esterase, the staining solution was made of 0.3 g of Fast blue RR Salt, 2 ml of α- naftil acetate dissolved in 5 ml acetone and 10 ml of distilled water. The stain was made up to 100 ml of phosphate buffer at pH 6.4. The gel was kept in the dark in this solution until the bands appeared. For peroxidases, staining solution was made of 2 g hydrochloric bencidine dissolved in 14 ml glacial acetic acid and made up to 100 ml with distilled water. A solution of hydrogenate peroxide at 1% was added and the gel was submerged during 1 to 3 minutes until the bands appeared. For polyphenol-oxidases, the staining solution was made up of 0.1 g of dihidroxifenil alumina (L-DOPA) and L-proline in 100 ml of phosphate buffer at 0.1 molar, pH= 6.5. The gel was kept in the dark in this solution until the bands appeared.
Once stained, the gels were washed with distilled water and kept in a solution of glacial acetic acid at 10%, until zymograms were scored. Position of each band was measured in cm starting from the migration point. Because the genetic interpretation of the zymograms was excessively complex, the electromorphs were used as isozymic descriptors. Each electromorph was considered as a character with presence scored as 1 and absence scored as 0, being the only two modalities. A total of twenty-two distinct electromorphs were used as isozymic descriptors, and if two accessions were different for at least one electromorph, they were considered to exhibit two different zymotypes. For esterases, six electromorphs were scored and five zymotypes were identified. For peroxidases, nine electromorphs were scored and nine zymotypes were identified. For polyphenol-oxidases, seven electromorphs were scored and eleven zymotypes were identified.
Data Analysis
A Categorical Principal Component Analysis (CATPCA) 19 was conducted using the qualitative morpho-agronomic variables and the number of chromosomes (2n= 24 or 26). With the resulting dimensions and quantitative variables of the principal components (PCA), successive attempts were made to concentrate the analysis with less dimensions. Scores of the accessions were used in each component in order to compare the formed groups. Variables presenting poor contribution were eliminated from the analysis 20 in order to achieve acumulative variance of more than 70% 21 in the first four components. For a list of descriptors, minimum averages between the major and minor of the eigenvectors values were taken and descriptors were selected whose eigenvectors were average above; also the correlations of the original variables with components and their coefficients of determination in the same were taken into account 22.
The conglomerate analysis (CA) of all the accessions was carried out with these descriptors from the agglomeration UPGMA hierarchical method (Unweighted Pair-Group Method using Arithmetic Averages), and the Euclidean distance as a measure of dissimilarity 23. The accuracy of the classification of the PCA was integrated via discriminant analysis (DA) data and assessed the contribution of studied isoenzymatic systems, classification of accessions. Diversity groups were created through a binary system. Processing of data in all cases was made with the statistical package SPSS® 15 24 and the results of the CA were represented in a graph with the use of the options provided by the STATISTICA® program 6 25.
Integrated analysis of cytogenetic, morpho-agronomic and isoenzymatic results A similar procedure to the one described in the Data analysis section was followed with the integration of the morpho-agronomic data and the number of chromosomes from the selected minimum descriptors and information from the three isoenzymatic systems (nominal variables). The accuracy classification of the PCA was verified through DA and the contribution of the studied isoenzymatic systems to the classification of the accessions were evaluated. Data processing was also performed with the SPSS® 15 statistical package 24. AC graphics were shown through the STATISTICA® 6 program, 25.
Results and Discussion
Chromosome Counts
All accessions with white- or yellow- fleshed cormels presented a chromosome number of 2n = 26 (Figure 1 A) which represents 59.2% of the total number of accessions. All accessions with purple-fleshed cormels presented 2n = 24 chromosomes corresponding to the remaining 48.8% (Figure 1 B) (Table 1).Previous studies have observed for X. sagittifolium, a chromosome number of 2n = 26 26 confirmed that all Xanthosoma species are diploid with 26 somatic chromosomes. More recently, 27, designates a chromosome number of 2n = 26 for accessions producing white fleshed cormels in Cameroon. However, some autors 28, 29 revealed the existence of 2n = 24 chromosomes for X. nigrum with purple fleshed cormels confirming the first results 30. 31, proposed a basic number x = 13 chromosomes for Xanthosoma spp. and recognized diploid accessions with 2n = 2 x = 26 chromosomes and the formation of 13 bivalents in the diakinesis and accessions of tetraploid 2n = 4 x = 52 chromosomes.bivalents in the diakinesis and accessions of tetraploid 2n = 4 x = 52 chromosomes.
Figure 1. Cells of cocoyam accessions of the Xanthosoma genus in mitotic metaphase (100X): A) 2n = 26 chromosomes. B) 2n = 24 chromosomes.
Accessions with purple fleshed cormels with 2n = 24 chromosomes must have formed during the process of evolution of this genus by the loss of two chromosomes which, according to the classification 32, 33 would be aneuploids of the type nullisomic (loss of a pair) or double monosomic (loss of a chromosome from different pairs).
In the case of the monosomics and nullisomics, the loss of genetic material can lead to the infeasibility of individuals, if it is diploid organisms, unless the affected chromosomes are carriers of unimportant genetic information 32. Nullisomy is caused by non-disjunction during meiosis that causes two of the gametes to have no chromosomal material, leaving the other two gametes to have double the amount of chromosomal material. Due to the lack of genetic information, the nullisomic gametes are rendered unviable for fertilization. In addition to, these mutations are more likely to perpetuate in asexually propagated species like Xanthosoma spp. It is however, more likely that the accessions with 2n = 24 chromosomes are double monosomics. Another hypothesis that could explain the existence of accessions with different chromosome numbers is the fact that the numerical and structural changes of chromosomes are cytogenetic differentiation and speciation mechanisms, so within a same genus, species with different chromosome numbers could exist and contribute to the evolutionary process. The accessions of white- and yellow-fleshed cormels would be x = 13 and the purple-fleshed x = 12. This would confirm previous statements found in the literature recognizing X. sagittifolium as 2n = 2 x = 26 chromosomes and X. nigrum as 2n = 2 x = 24 chromosomes 34.
Interestingly, the relationship between the color of the cormel flesh and the number of chromosomes indicate that this character must be determined by a gene with two alleles, and purple dominates over yellow, but there must also be an epistatic gene whose dominant allele prevents the manifestation of color by which the accessions of white phenotype which are more abundant in the collection due to this relationship are not allelic. As the flesh color presents variation, it is possible that modifier genes acting on the flesh colour and the same allelic combination determine the phenotype. For accessions with 2n = 24 chromosomes, they may have lost the chromosomes carrying (n) information of the epistatic gene for which 100% of individuals manifest purple or yellow colors in their cormel flesh.
Morpho–Agronomic Descriptors
Analysis of successive categorical main components carried out with the qualitative variables (nominal and ordinal) including the number of chromosomes considered as a nominal variable, allowed to concentrate the greater variability in three dimensions with an accumulated total of 74.10%. From measurements obtained in the PCA and the quantitative variables in the second analysis, the PCA revealed a group of accessions in correspondence with the classification according to the coloration of the flesh cormel 35, who explained that this character cannot be used to identify accessions or groups of these, nor to distinguish species. The first four major components were taken explaining the 77.818% of the variability according to Cliff criteria in 1987 21, who indicated that components whose own values explain 70% or more of the total variance must be accepted. All variables showed a high contribution.
Two groups (I and II) of accessions are presented in Figure 2 in which the white flesh accessions and the flesh residues overlap. In group IV, the yellow flesh accessions (2n = 26 chromosomes) are located in a well-defined group which in addition coincides with this character, present green leave blades with green petioles, yellow buds, low height (between 30 and 50 cm), high content of dry matter in the primary and secondary cormels and abundant tillering. The accessions 'Amarilla Trinidad' (4), 'Belembe' (20), 'Jardín' (11) and 'Picante' (49) are spatially separated from each other and from the other accessions.
Figure 2. Spatial distribution of the accessions generated by principal component analysis using the morpho-agronomic variables and the number of chromosomes. The number of each accession is shown in Table 1.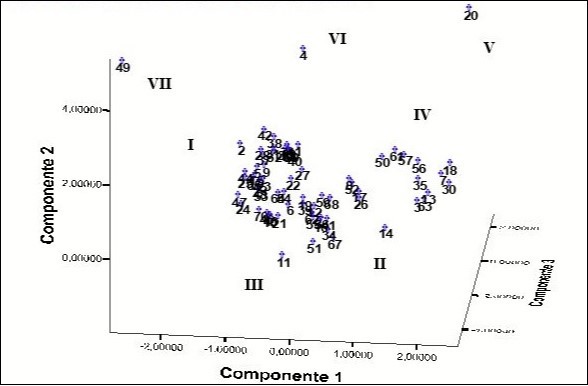
The PCA allowed to develop a list of 17 minimum qualitative and seven quantitative descriptors (Table 2) that showed the largest contribution to the variance in the first three components in PCA and to test their capacity to discriminate accessions.
In this sense, as far as possible, some high heritability descriptors should be used so that the variability is sufficiently discriminated 36.
Table 2. Selected minimum descriptors (24) for the study of the accessions of the Cuban Xanthosoma collection| Qualitative characters | Quantitative characters |
| Chromosome Number | Plant Height |
| Growth habit | Petiole length |
| Leaf blade orientation Leaf Blade Edges | Petiole length / leaf blade width |
| Leaf Blade Shape | Sucker number |
| Venation color | Dry matter percentage in the leaf |
| Venation color under side | (blade, petiole and sheath) |
| Petiole color | Dry matter percentage in primary cormels |
| Color of the basal part petiole | Dry matter percentage in secondary cormels |
| Color of the petiole edge | |
| Presence of wax on the petiole | |
| Color of the leaf blade | |
| External color of primary and secondary cormels | |
| Color of the primary and secondary cormels buds | |
| Color of the flesh of primary and secondary cormels | |
| Shape of secondary cormels | |
| Inflorescence emergence |
The analysis of conglomerates (CA) based on the coordinates of the main components of greater weight allowed the formation of seven groups of variability (Figure 3), taking into account the criteria of 22, which corresponds to those obtained in the PCA. The use of the qualifications of the main components in the formation of the conglomerates increases its effectiveness and helps to validate its results 37.
Figure 3. Dendrogram showing the grouping of the accessions of the Cuban collection of the Xanthosoma genus from the minimum descriptors selected according to the morpho-agronomic data and the number of chromosomes. The UPGMA method and the Euclidean distance were used.
Accessions with white flesh cormels that share the group I with the purple ones which have the peculiarity of having clearer shades within the range of purple colors, possess green foliage and in the petiole, the sheath and in the blade presents some pink shades.
White flesh accessions that share group II with the purple ones which have darker shades, present a more intense purple coloration of the vegetative structures mentioned above.
According to the interpretation in chromosomes counts section, accessions with cormels white flesh (2n = 26) could have the genetic information for the manifestation of the purple color, but the presence of epistatic genes prevent its expression which may explain the presence of white flesh and purple flesh accessions in the same group and with morpho-agronomic traits coincident. This would support the hypothesis that purple accessions (2n = 24) are aneuploid because if they had different basic numbers, their location in different groups should be expected because of the genetic differences they would present.
Different ranges of purple color shown in the cormels flesh of the accessions mentioned above may be related to the action of modifying genes which modulate the color expression.
This reveals the existence of a single group with two subgroups for these accessions which basically present as distinctive trait, the color of the flesh of cormels and the number of chromosomes.
'Jardín' accession is located in group III which has distinctive features among the other accessions with purple cormels, it has 2n = 24 chromosomes and the leaf blade is deep green with purple tones, venations on the underside are purple, the same as the petiole and its edge and sheaths; they have abundant wax along the petiole and this one is white-reddish at the base of its sheath. 'Jardín' is the only entry of purple flesh that shows dark purple color in the petiole and its edge, as well as in the venation of the leaf blade on the underside.
All accessions with yellow flesh cormels (2n = 26) appear in group IV. This group of accessions has low yields which limit their dispersion and determine a greater use of accessions of other groups in practice, despite having a high nutritional value mainly due to its higher content of proteins, essential amino acids, phosphorus and carotene than the white fleshed and purple fleshed accessions 38.
Although the number of chromosomes is the same that in the case of the white fleshed accessions, according to the above, the yellow fleshed accessions must be those where the colour and the epistatic genes are recessive, therefore the yellow color is expressed and different ranges of this color appears in the flesh of the cormels of these accessions due to the action of the modifying genes.
'Amarilla Trinidad', 'Belembe' and 'Picante' accessions show a well-defined separation, the same in this analysis as in the PCA. Each one of them has many different characteristics that propitiate this distance observed in the dendrogram.
It is observed that there are no duplicates in the collection base of cocoyam which is preserved in Cuba as a result of this study since all accessions are distant from each other.
It is possible to assume that two accessions are morphologically identical if the distance between them is equal to 0 39. In this sense, the multivariate analysis allowed to discriminate which are the most important variables (minimum descriptors) in the identification of possible duplicates, and what additional information they provide for future evaluation. The same author adds that in the case of large collections, the identification of these possible duplicates contribute significantly to the conformation of core collections.
In the Discriminant Analysis (DA), 98.6% of accessions were correctly classified since just one accession of group II was relocated in group I for a 96.4% correct classification. The 'Jardín' accession is correctly located as the only member of group III in correspondence with its particular traits that distinguish it from other accessions. Three other accessions were located in different groups, 'Picante' in group V, 'Belembe' in group VI and 'Amarilla Trinidad' in group VII because they have many phenotype characteristics.
Isozymes Studies
Esterases presented 22 bands with 100% of polymorphism. In this system 59 accessions are identical, but with 14 combinations of zymotypes corresponding to 28 electrophoretic patterns. The largest number of bands is located in the lower Anodic migration area. Four unique bands appear, two of them (3.3 and 5.7 units) identify the accession 'Jardín' (11); one corresponds to the 'Picante' accession (49) (4.5 units) and the other for 'Amarilla Trinidad' (4) (6.9 units).
Similar results with the use of this and other isozyme systems have been published in the genus Musa 40, in the genus Colocasia41 and in Manihot esculenta Crantz 42 whose information has been useful in several genetic studies. Although the functional characterization of esterases of plants has been limited 43, these enzymes are very abundant in plants, they play an important role in photosynthesis and its high polymorphism makes them widely used in studies of characterization 44.
For the peroxidase system there is a total of 27 bands with 100% polymorphism. The band of 0.9 units is common to all the genotypes, except for 'Picante' accession which confirms the results analyzed so far where there has been a marked tendency to separate from the rest of the accessions by its particular characteristics. Different banding patterns suggest a high polymorphism of electrophoretic patterns obtained in the isoenzymatic peroxidase system with 46 accessions showing equal zimotipo within eight combinations in terms of the number and position of the bands. Two unique bands for the accession 'Picante' (2.7 and 6.1 units) and two for the accession 'Amarilla Trinidad' (3.2 and 4.0 units) were observed.
The obtained results in this system confirm its wide use as a genetic marker in plants 40. According to the quantitative activity on this isoenzymatic system in accessions of Xanthosoma, 27, points out that peroxidase activity is constitutively in the accessions with Cormels of yellow flesh compared with the white flesh and the purple flesh 45.
Some authors 46 confirmed that peroxidases are hemeproteins widely distributed in plants and linked to numerous processes of cell physiology and its content and composition vary before physiological changes caused by biotic and abiotic stress.
Results for polyphenoloxidase isoenzymes also show 100% polymorphism compared with a total of 21 isoenzymatic bands.
These results are evidence of the existence of a high polymorphism in this isoenzymatic system; many of the accessions with own zymotypes coincide with the results of the analysis of the two previous isoenzymatic systems reaffirming the 'Picante' accession with an own electrophoretic pattern in the three systems studied, like 'Belembe'.
The polyphenoloxidases are widespread in plants and their catalytic action is linked to the appearance of undesirable brown color, discoloration and darkening, as well as the loss of the taste of storage food, fruits and vegetables which imply a loss of nutritional value 47. The presence of polymorphic patterns of this isozyme has been informed 48, 40 in other crops of economic importance. In general, it has been determined that polyphenoloxidases as well as the esterases are among the most polymorphic systems in plants, 49 and results match those found in the genus Xanthosoma.
Peroxidase system is very well extended, biochemical genetic studies by the important role in the biosynthesis of wall components, as well as the regulation of growth, cell differentiation and resistance to biotic and abiotic factors 44. The polyphenoloxidases are also widely used in studies of plants which can be found in chloroplasts and are associated to biotic and abiotic stress for their ability in the immobilization of enzymes and oxidation of some donor phenols 50. Moreover, as already reported, esterasesplay a very important role in photosynthesis.
The results show that isoenzymatic systems have 100% variability, allowing a better analysis of the genotypes of malanga of the Xanthosoma genus included in this study, according to the number and position of the revealed bands. 71 accessions were analyzed, 26 have identical isoenzymatic for these isozymes also reflected in the analysis. One of them is the 'Picante' accession.
The variability in patterns of bands of the three studied systems found in accessions in the collection of the Xanthosoma genus, allowed to distinguish four groups within the collection in correspondence with the color of the flesh in the primary and secondary cormels. Group I concentrated on the accessions of purple flesh; while the ones of white flesh are located in Group II, and in IV are those of yellow flesh. The 'Picante' accession is singly separated in Group III which adds new elements on its particular characteristics, away from the rest of the genotypes of the collection.
Integrated Analysis
The morpho-agronomic evaluation of the accessions may be integrated with direct studies of the genome through the cytogenetic analysis, electrophoresis of enzyme, protein and DNA 51.
The procedure for the integrated analysis of the 71 accessions from the selected minimum descriptors and taking into account the analysis of the quantitative and qualitative morpho-agronomic traits (nominal and ordinal), the number of chromosomes and the information provided by the considered nominal variables, isozyme systems consisted of a CATPCA allowing to concentrate the variability in three dimensions.
These dimensions were used in the PCA, the results indicate a total acumulative variance to the third component of 74.14%, so it was also considered adequate, according to Cliff’s criteria in 1987 21 shows that quantitative variables provided the biggest contribution to the variability in component 1, except the relationship between the length of the petiole and leaf blade width and foliage dry matter percentage that they made in component 2.
As for dimensions, where the isozymes information is included, there is a greater and also positive contribution of dimension 1, this time in component 1. Three dimensions showed their largest negative contribution to component 2 and 2 dimensions contributed most to the variability in component 3. This indicates that the contribution of the information generated by the three isozyme systems distributed was mostly in the first three components.
Distribution in three-dimensional space of the accessions of Xanthosoma taking into account the integrated results of the morpho-agronomic, cytogenetic and isozymes characterization are shown in Figure 4.
Figure 4. Spatial distribution of the 71 accessions in three components. The distribution was generated by the PCA of the cytogenetic, isoenzymatic and morpho-agronomic data analyzed in an integrated way.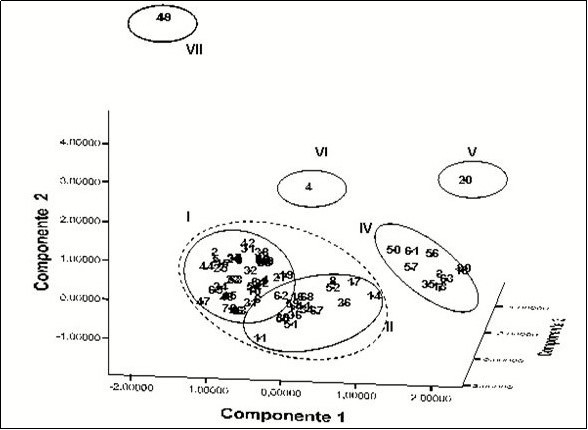
It was more evident the separation and grouping of accessions in well-defined groups as groups III, IV, V, VI and VII, while accessions in groups I and II are masked with each other forming a greater grouping.
The dendrogram in Figure 5 shows the formation of six groups. Seen as the genetic component provided by the analysis of the three isozyme systems, it seems to be crucial in not separating the 'Jardin' accession in a different group which presents many common characteristics with the accessions of purple flesh and have, like these, 2n = 24 chromosomes.
Figure 5. Dendrogram generated by cluster analysis of the integrated data of cytogenetic, morphoagronomic and isoenzymatic markers in the Cuban collection of genus Xanthosoma with the UPGMA algorithm and the Euclidean distance . Note the formation of six variability groups
The CA showed differences among all accessions indicating the absence of duplicates when descriptors data are analyzed in an integrated manner.
The DA proved the classification made with the PCA with 100% of the cases grouped correctly and 95.8% of clustered cases validated by crossed-validation. If we compare these results with those shown by the DA without the integration of the data, it could be considered that the use of the information from different types of descriptors 52, 53, is important to achieve a better classification of the germplasm studied because it obtained a greater accuracy in the classification in this case which offers a more complete assessment of the diversity of germplasm and it can also detect the absence of duplicates in the collection.
By integrating cytogenetic, morpho-agronomic and isozymes data, we studied groups of variability for accessions of the Xanthosoma genus (71 accessions from the Cuban collection, which provides the basis for the creation of the core collection as a starting point for the genetic improvement of the crop. This has direct implications for taxonomic studies and breeding, and allowed to rule out the presence of duplicates in the collection of the studied accessions.
According to the results obtained in the present study, we propose the following groups in the Cuban collection of the genus Xanthosoma:
Group I: 31 accessions: seven that has white flesh of its primary and secondary cormels, and a chromosome number of 2n = 26 and 24 accessions of purple flesh with 2n = 24 chromosomes. These accessions had common characteristics like the height of the plants, the green color of the leaves and the sucker number that made possible its location in the same group.
Group II: 26 accessions: 17 with cormels of white flesh of its primary and secondary cormels with a chromosome number of 2n = 26 and nine with purple flesh presented 2n = 24 chromosomes. Note that it is included in this group the Jardín which had been separated in the analysis not integrated accession, but has its particular characteristics with 2n = 24 chromosomes and cormels with purple flesh, but it is the only entrance with these characteristics, presenting the dark purple color in the petiole and its ridge, as well as through the ribs of the blade on the underside. This group has also common characteristics as the green color of leaves, plant heightand number of suckers.
Because they appear within a same group of accessions of white flesh and accessions of purple flesh, it implies the presence of aneuploid nulisomics or double monosomic and this assumption must be tested.
Group III: 11 accessions with cormels of yellow flesh and a chromosome number of 2n = 26. These accessions from the morphoagronomic point of view are distinguished by the presence of smaller plants and a higher yield than the rhizomes of white mass and purple mass; the leaves have the sagittal lamina of light green color, not shiny, with wax in the lamina and also in the petiole. The rhizomes have yellow buds and the color of the flesh varies from light yellow to orange-yellow.
Group IV. 'Belembe', with cormels of yellow flesh and 2n = 26 chromosomes. It was introduced in Cuba from the island of Guadeloupe, grows in tillers from a meter high, but it usually has about 50 cm. high, with multiple stems arising from a small cormel of yellowish flesh that is not used as food. The petioles are long and thin, from 20 to 40 cm. in length; the blade is hastate arranged in a horizontal plane in cup with two basal lobes often asymmetrical, triangular shape, and a basal acute apex. More tender cooked leaves are consumed to remove oxalate crystals and latex. These features make that 'Belembe' present their own identity within the collection.
Group V.: 'Amarilla Trinidad', also with cormels of yellow flesh and a chromosome of the 2n number = 26, is an accession introduced from the island of Trinidad and Tobago, presents yellow pigmentation in the flesh of their cormels, but it is the unique accession with this feature which has the leaf blade hastate of intense green color with purple edge, petioles in purple with red base and the edge deep purple.
Group VI: 'Picante', with cormels of creamy white flesh and 2n = 26 chromosomes; It shares features with the accessions of the Cuban collection of the genus Xanthosoma, as not peltate sagittate shape of its leaves, the presence of a white creamy of the flesh of their cormels and a chromosome number of 2n = 26, basically. However, morpho-agronomic characterization, as well as, the analysis of the three systems carried out in this work, it has been shown that cannot be assured that 'Picante' belongs to this group. It has leaves with almost vertical growth with the apex directed upwards, ribs are prominent and does not produce secondary cormels. In addition to, the petiole at the base has a white color and you can distinguish a greenish white color with purple pigmentation which extends to the ribs on the underside. The edge is dark purple, blades blade presented a glossy deep green color and the ribs by the beam are dark green with an abundant emission of inflorescences. This description confirms its genetic distance from the rest of the accessions preserved in the Cuban collection of the genus Xanthosoma. Its characteristics make it closer to the Alocasia specimens described 54, 55.
Morpho-agronomic characterization, as well as the analysis of the three isozyme systems in this work shows the separation of the accessions 'Belembe', 'Amarilla Trinidad ', 'Picante'.
Integrated data analysis revealed that all the accessions in the collection are different. The cluster analysis showed a high polymorphism ruling out the presence of duplicates in the studied collection.
Despite the variability found in the Cuban collection of the genus Xanthosoma, the results demonstrate the need to broaden the genetic base taking into account the groups of accessions with a limited representativeness.
Compliance with Ethical Standards
Acknowledgements
The authors would like to thank the agronomist field technicians Miguel Angel Lago and Juan Carlos Hernández for their contribution in the characterization and evaluation of germplasm.