Abstract
Aim:
The relation between inflammation and cancer has been known since the 19th century. However, investigations on the pathogenesis and pathophysiology of this relation have begun recently. It was demonstrated that increased neutrophil/lymphocyte ratio is a poor prognostic factor in some malignancies. The present study aimed to determine whether preoperative neutrophil/lymphocyte ratio has a prognostic value in larynx cancer.
Method:
Preoperative blood analyses of 139 patients, who underwent subtotal or total laryngectomy for larynx cancer between 2003 and 2013 at Marmara University School of Medicine, Department of ENT, were retrospectively evaluated. Neutrophil/lymphocyte ratio (NLR) was calculated dividing absolute neutrophil count by absolute lymphocyte count. Optimal cut-off value for NLR was determined by receiver operating characteristics (ROC) curve analysis. Statistical analyses were done using IBM SPSS statistic 22.0 (IBM SPSS, Turkey) and Med Calc 12.3.0 package programs.
Results:
The sensitivity of NLR in predicting advanced-stage (Stage 3 and 4) squamous-cell carcinoma of the larynx (LSSC), T4 LSSC and lymph node metastasis at different cut-off values were 66.2%, 83.9% and 73.8%, respectively and the specificity was 76.7%, 66.2% and 65.2%, respectively. Staging according to T classification revealed that NLR significantly increases with tumor stage (p<0.001). Statistically significant relation was determined between lymph node metastasis of tumor and neutrophil/lymphocyte ratio (p=0.003). Comparing overall survival (OS) and disease-free survival (DFS) between the cases with NLR <3.02 and the cases with NLR >3.02, it was demonstrated that OS and DFS are significantly lower in the cases with NLR<3.02 (p: 0.001 vs. p<0.05 for OS and p: 0.013 vs. p<0.05 for DFS)
Conclusion:
NLR increases with the stage of disease in LSSC. NLR is a simple, cheap, repeatable and valuable parameter that can be obtained from routine analyses, gives information about poor prognosis and survival, and is able to predict T4 LSSC, advanced-stage LSSC (stage 3-4) and lymph node metastasis.
Author Contributions
Academic Editor: Ioannis Chatzistefanou, 401 General Military Hospital of Athens
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2017 Muhammet fatih Topuz, et al
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Larynx cancer (LSSC) accounts for 2.4% of newly diagnosed malignancies each year in the world.1 It is the second leading head and neck cancer after the skin cancer and accounts for 25% of overall head and neck cancers.2 Almost all (95%) are squamous-cell carcinoma. LSSC is particularly more prevalent in the middle-aged males. The leading risk factors include smoking, tobacco use, alcohol consumption, air pollution and occupational factors. 3
TNM classification and histological grading system are the guides for us in identifying LSSC tumor behavior and designating treatment method. However, different outcomes despite the same treatment method implemented in the patients with similar characteristics suggest that these parameters are not adequate and accordingly direct us to investigate different prognostic factors. The aim of investigating different tumor markers in LSSC is to develop cancer-preventing treatments, to predict relapse and secondary cancers encountered in larynx cancer by enhancing knowledge on the cancer and biological behaviors of the host, and to provide more effective treatment planning.
The relation between inflammation and cancer is known since time immemorial 4. Inflammatory cells enhance genomic instability and angiogenesis with the cytokines and chemokines they produce in the early phases of neoplastic process, facilitate tumor cell migration, increases DNA injury, and accordingly they become strong tumor promoters.4 Recent studies as well demonstrated the relation of pretreatment peripheral leukocyte (neutrophil, lymphocyte, monocyte) count and thrombocyte count with prognosis in various types of cancer. It was demonstrated that preoperative NLR is a poor prognostic factor in the patients with non-small-cell lung cancer, small-cell lung cancer and renal-cell cancer.5 Again, NLR was shown to be associated with numerous malignancies including hepatocellular cancer6, gastric cancer7, epithelial ovarian cancer8, pancreatic cancer 9, and nasopharyngeal cancer 10. The present study aimed to expose the relation between NLR and LSSC.
Material and Method
Patients
A total of 139 patients, who admitted to our clinic between 2003 and 2013 and underwent subtotal or total laryngectomy for larynx cancer, were retrospectively analyzed. Patients’ medical histories, age and gender, history of smoking and alcohol consumption, results of preoperative blood analysis (lymphocyte, neutrophil, thrombocyte, etc.), localization of the lesion (supraglottic, glottic, subglottic and transglottic), and radiological imaging and pathological examination findings (tumor stage, lymph node metastasis, etc.) were retrieved from the patient files. After the surgery, the patients were followed every three months in the first year, every six months between 2 and 5 years, and every year after the fifth year. The last control was performed on 01. 07.2016.
Twenty eight patients, who had active infection or hematological disease or have been receiving hematological treatment prior to surgery, who had received preoperative radiotherapy or chemotherapy, had second primary cancer, chronic inflammatory or autoimmune disorder like SLE or missing data, were not included in the study.
Patients’ tumors were classified according to the TNM classification of The American Joint Committee on Cancer (AJCC), which was modified in 2002. Tumor stage and lymph node metastasis were evaluated taking histopathological examination as the basis.
The study was conducted after obtaining the approval of Marmara University Clinical Research Ethics Committee (date “07/03/2014”, number “09.2014.0027ˮ).
Amount of Peripheral Blood
Analysis was done in the blood samples, which have been routinely obtained from the patients one day before the surgery. NLR was calculated dividing absolute neutrophil count (neutrophil count/µL) by absolute lymphocyte count (lymphocyte count /µL) in the preoperative complete blood count. Blood samples were taken into the tubes containing ethylenediaminetetraacetic acid (EDTA) and analyzed using LH780 Beckman Coulter’s automated hematology analyzer in the Marmara University School of Medicine, Department of Biochemistry.
Statistical Analyses
Statistical analyses were done using Med Calc 12.3.0 and IBM SPSS statistics 22.0 (IBM SPSS, Turkey) package programs. Distributions were evaluated by Shapiro Wilk Test and Kolmogorov Smirnov Test. Independent two groups were compared using Student’s t-test or Mann Whitney U test depending on the distribution. Taking the distributions and variances into account, ANOVA or Kruskal Wallis test was used for the comparisons between more than two groups. In case Kruskal Wallis test was used, Post-hoc comparisons were done by Mann Whitney U test. In the event Post-hoc comparisons were done using Mann Whitney U test, the level of statistical significance was calculated by Bonferroni correction. Chi-square test was used to compare the categorical data. Predictive performance of NLR for the disease, stage and nodal metastasis was determined by drawing ROC curve. The sensitivity, specificity, and positive and negative predictive values were calculated after determining appropriate cut-off values. Overall survival was defined as the time from surgery to death or to the last control. Disease-free survival was defined as the time from surgery to the relapse, last control or death. Survival analysis was done using Kaplan-Meier test and the comparisons were done by Log-rank test. The level of statistical significance was predetermined to be p<0.05.
Results
A total of 111 patients, of whom 106 (95.5%) were male and 5 (4.5%) were female, were enrolled in the study. The mean age of the patients was 59.7 ±9.6 years. Demographic data of the patients and tumor-related factors are summarized in Table 1.
Table 1. Demographic data and tumor-related factors| Number of patients: 111 | |||
| Age at surgery (mean±SD) | 59.7 ±9.6 | ||
| Variables | Cases | NLR (min – max) | P |
| Gender | |||
| Male Female | 106(95.5%) 5(4.5%) | 3.19 (0.38 – 16.33) 3.03 (2.88 - 8.14) | |
| Primary localization: | |||
| Supraglottic: Glottic: Subglottic: Transglottic : | 33(29.7%) 23(20.7%) 8(7.2%) 47(42.3%) | 3.04 (1.38 - 16.33) 2.26 (1.15 - 8.14) 4.04 (2.88 – 8.80) 3.00 (0.38 – 13.80) | 0.027 |
| T class | |||
| T1 T2 T3 T4 | 24(21.6%) 32(28.8%) 24(21.6%) 31(27.9%) | 1.90 (1.15 – 5.40) 2.58 (0.78 – 16.33) 3.33 (0.38 – 5.41) 4.68 (1.75 – 11.30) | <0.001 |
| Lymph node metastasis | |||
| N0 N1 N2 | 69(62.2%) 19(17.1%) 23(20.7%) | 2.12 (0.38 – 5.77) 3.78 (2.18 – 8.43) 4.63 (3.38 – 16.33) | 0.003 |
| TNM stage | |||
| Stage 1 Stage 2 Stage 3 Stage 4 | 22 (19.8%) 21 (18.9%) 24(21.6%) 44(39.6%) | 2.08 (1.15 – 5.40) 2.38 (1.78 – 6.33) 3.63 (0.38 – 9.41) 4.98 (1.75 – 16.30) | <0.001 |
| Histopathological grade | |||
| Grade 1 Grade 2 Grade 3 | 12(10.8%) 73(65.8%) 26(23.4%) | 2.03 (0.38 – 5.49) 2.54 (2.08 – 9.46) 4.33 (3.40 – 16.33) | <0.001 |
The result of histopathological examination was reported as squamous-cell carcinoma in all patients. Primary tumor localization was supraglottic in 33 (29.7%), glottic in 23 (20.7%), subglottic in 8 (7.2%) and transglottic in 47 (42.3%) patients. According to the AJCC classification, 24 (21.6%) patients had T1, 32 (28.8%) patients had T2, 24 (21.6%) patients had T3, and 31 (27.9%) patients had T4 lesion. Of the patients, 22 (19.8%) were stage 1, 21 (18.9%) were stage 2, 24 (21.6%) were stage 3, and 44 (39.6%) were stage 4 larynx cancer. Histopathological grading of tumors revealed that 12 (10.8%) patients were grade 1, 73 (65.8%) patients were grade 2 and 26 (23.4%) patients were grade 3. Forty (39.5%) patients had lymph node metastasis.
Comparing the value of NLR according to the T classification, median (min-max) value of NLR in the patients with T1,T2,T3 and T4 larynx cancer were 1.90 (1.15-5.40), 2.58 (0.78-16.33), 3.33 (0.38 – 5.41), and 4.68 (1.75- 11.30), respectively (Table 1). Comparing the value of NLR between T1,T2,T3 and T4 groups, the difference between the groups was considered statistically significant (p<0.001)(Table 2). Paired comparisons revealed statistically significant difference between T1 and T3, T1 and T4, T2 and T4, and T3 and T4 groups.
Table 2. Between-group comparison of NLR according to T classification| T1 | T2 | T3 | T4 | |
| T1 | 1 | 0.029 | <0.001* | <0.001* |
| T2 | 0.029 | 1 | 0.118 | 0.004* |
| T3 | <0.001* | 0.118 | 1 | 0.004* |
| T4 | <0.001* | 0.004* | 0.004* | 1 |
Comparison of NLR according to the level of lymph node metastasis (N0, N1, N2) revealed statistically significant difference between the groups (p=0.003). While paired comparison revealed statistically significant difference between N0 and N1 (p=0.011) and between N0 and N2 (p=0.006), no statistically significant difference was determined between N1 and N2 (p=0.622).
The patients were compared in terms of the value of NLR according to the primary tumor localization; median (min-max.) value of NLR in the patients with supraglottic, glottic, subglottic and transglottic larynx cancer was 3.04 (1.38-16.33), 2.26 (1.15-8.14), 4.04 (2.88 – 8.80), and 3.00 (0.38- 13.8), respectively (Table 1). When the patients with transglottic larynx cancer were excluded because of unknown primary origin, the difference between the three groups in terms of median values of NLR was statistically significant (p=0.027). Comparing the groups between themselves, no statistically significant difference was determined between supraglottic and subglottic (P=0.130) and between supraglottic and glottic (p=0.077) localizations, whereas there was statistically significant difference between glottic and subglottic (p=0,014) localizations.
Optimal cut-off value for NLR
Cut-off value for optimum sensitivity and specificity was identified to determine the ability of NLR to predict advanced stage (stage 3 and 4) LSSC. When the cut-off value was taken as 3.02 based on the Receiver operating characteristic (ROC) curve, which was drawn to determine whether it is advanced stage (stage 3 and 4) LSSC, the sensitivity was 66.2% and the specificity was 76.7% (Graph 1) (Table 3)
Table 3. NLR’s ability to predict advanced stage (stage 3 - 4) LSSC, T4 LSSC and lymph node metastasis| Sensitivity | Specificity | AUC | 95% CI | PPV | NPV | |
| Stage 3 and 4 | 66.2% | 76.7% | 0.746 | 0.652-0.841 | 81.8% | 58.9% |
| T4 larynx cancer | 81% | 69.2% | 0.750 | 0.673-0.847 | 45% | 89.4% |
| Lymph node metastasis | 73.80% | 65.20% | 0.692 | 0.588-0.795 | 56.40% | 80.40% |
Graph 1. ROC curve demonstrating the ability of NLR to predict advanced stage LSSC 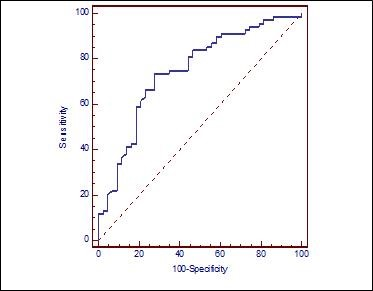
NLR was found 81% sensitive and 69.2% specific in predicting presence of T4 LSSC with a cut-off value taken as 3.02 based on the ROC curve (Graph 2) (Table 3)
Graph 2. ROC curve demonstrating the ability of NLR to predict T4 larynx cancer 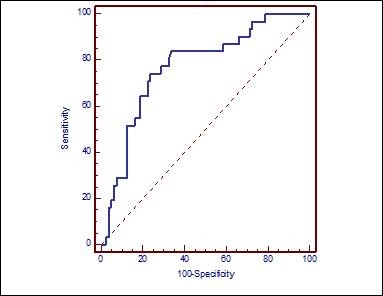
NLR was found 73.8% sensitive and 65.2% specific in predicting presence of lymph node metastasis with a cut-off value taken as 3.02 based on the ROC curve (Graph 3) (Table 3).
Graph 3. ROC curve demonstrating the ability of NLR to predict lymph node metastasis
OS and DFS according to NLR
One hundred and eleven participants were divided into two groups as those with NLR < 3.02 and those with NLR>3.02.
OS according to NLR
Seven (12.5%) of 56 cases with NLR<3.02 had died. The latest death occurred in the 7th year and the survival rate was 73.4% with standard error of 9.04%. The mean survival was 8.64±0.45 years and median survival was 10 years. One-, three-, five-, and 10-year cumulative survival rates were 98%, 95%, 79% and 73%, respectively. Twenty three (41.6%) of 55 cases with NLR>3.02 had died. The latest death occurred in the 6th year and survival rate was 45.7% with standard error of 8.1%. The mean survival was 6.29±0.55 years and median survival was 5.9 years. One-, three-, five-, and 10-year cumulative survival rates were 86%, 67%, 47% and 46%, respectively. Comparing the survival rates between the groups with NLR <3.02 and NLR>3.02 using Log Rank test, survival was found to be significantly lower in the group with NLR > 3.02 as compared to the group with NLR< 3.02 (p:0.001; p<0.05) (Graph 4).
DFS according to NLR
Relapse was seen in two (3.6%) of 56 cases with NLR<3.02. The latest relapse was seen in the 22nd month and survival rate was 95.6% with standard error of 3.1%. The mean survival was 9.62±0.27 years and median survival was 10 years. One-, three-, five-, and 10-year cumulative survival rate was 96%. Relapse was seen in 10 (18.2%) of 55 cases with NLR>3.02. The latest relapse was observed in the 20th month and survival rate was 78.6% with standard error of 6.2%. The mean survival was 8.06±0.55 years and median survival was 10 years. One-, three-, five-, and 10-year cumulative survival rate was 79%. Comparing the DFS between the groups with NLR <3.02 and NLR>3.02 using Log Rank test, DFS was found significantly lower in the group with NLR > 3.02 as compared to the group with NLR< 3.02 (p:0.013; p<0.05)(Graph 5).
Graph 5. DFS graph of LSSC patients after surgical resection 
Discussion
Today, many theorems have been proposed for the relation between NLR and cancer; however, it remains unclear. Nevertheless, it is suggested to be associated with the relation between cancer and chronic inflammation.22, 23 The inflammation around the tumor is considered to influence tumor growth, as well as tumor invasion of surrounding tissues and ability of metastasis.22, 23 Leukocytes -among which neutrophils and lymphocytes are the most effective-, dendritic cells, macrophages, eosinophils and mast cells in the environment play an important role in the entire process.
Lymphocytes inhibit proliferation and metastatic activities of tumor cells by causing cytotoxic cell death and cytokine production.12 Antitumor immune response of the host against tumor is a lymphocyte-dependent response 2, 11. It was demonstrated that neutrophils stimulate tumor growth by producing proangiogenic factors around the tumor such as vascular endothelial growth factor, chemokines and proteases 16, 17. Neutrophils and tumor-related macrophages are known to be the main sources of matrix metalloproteinase9. Proteinases lead to extracellular matrix destruction and accordingly facilitate infiltration of the cells into the tissues. Again, vascular endothelial growth factor synthesized by tumor-related macrophages causes mytogenesis in the endothelial cells and influences vascular permeability. 18, 19 Cytokines such as IL -6 and tumor necrosis factor alpha, which are encountered in cancer-related inflammation, and growth factors produced by the cancer cells induce the increase in neutrophil count. Cytokines and phagocytic mediators cause cellular DNA injury, inhibit apoptosis, and stimulates angiogenesis.20 For this reason, peripheral neutrophil count is accepted as a marker for cancer-related inflammation and tumor growth.21
NLR is an indicator of the balance between protumoral inflammatory status and antitumoral immune status. NLR increases due either to the increase in neutrophil count or to the decrease in lymphocyte count impairing the balance in favor of protumoral inflammatory status, whereas increase in lymphocyte count or decrease in neutrophil count reverses this balance in favor of antitumoral immune status.24 Therefore, while increased NLR is the indicator of poor prognosis, decreased NLR is the indicator of good prognosis. Earlier studies demonstrated that increased NLR is the indicator of poor prognosis in the ovarian cancer 25, colorectal cancer 26 and gastric cancer 27, whereas preoperative increased lymphocyte count, i.e. decreased NLR, is a good prognostic factor in pancreatic ductal adenocarcinoma 28.
Prognostic relationship between NLR and cancer depends on the type of cancer; therefore, different cut-off values of NLR need to be taken for each type of cancer. While the cut-off value was taken as 3.73 in a study suggesting that increased NLR is a poor prognostic factor in nasopharynx cancer, it was calculated to be 3.20 in another study investigating predictive value of NLR in T4 gastric cancer. 30 In the present study, we as well determined the cut-off value as 3.02 to demonstrate the predictive ability of NLR in LSSC. With this cut-off value, NLR has high sensitive and specific predictive ability for advanced stage larynx cancer, T classification and lymph node metastasis.
Evaluating OS and DFS in the cases with NLR < 3.02 and NLR > 3.02 using Log Rank test, it was determined that survival rate was significantly lower in those with NLR > 3.02 as compared to those with NLR < 3.02 (p:0.001; p<0.05, p:0.013; p<0.05). Kum et al. compared the NLR values in the differentiation between LSSC and benign and precancerous laryngeal lesions and found significant outcomes 31. The present study supports this study.
Conclusion
In the present study, we exposed the prognostic importance of NLR in LSSC. The surgeon’s knowledge about the stage, T class and lymph node metastasis of LSSC in the preoperative period is of critical importance in terms of treatment planning in the preoperative period. NLR may help us to predict LSSC T4, advanced stage LSSC (stage 3-4) and lymph node metastasis. Again, increased NLR is an independent poor prognostic factor for OS and DFS. Owing to these features, NLR is not a 100% diagnostic parameter, but is a non-invasive, cheap and valuable parameter, which could be obtained by simple and routine tests and used by the clinician while evaluating the patient.
