Abstract
Head and neck cancers (HNCs) are the most prevalent and aggressive type of cancers. Genetic, epigenetic, environmental and viral risk-factors are associated with HNC carcinogenesis. Persistent infection of oncogenic human papillomaviruses (HR-HPVs) represent distinct biological, molecular and epigenetic entities in HNCs. There are three main epigenetic mechanisms that regulate transcription, these are DNA methylation, histone modifications and alteration in non-coding RNA networks, which can dissected to identify innovative and accurate epigenetic biomarkers for diagnosis and prognosis of HNC patients. Due to the lacunae of accurate distinctive biomarkers for the definite diagnosis of HNC, the identification of predictive epigenetic markers is necessary that might modify or increase HNC patient’s survival. In this mini review, we briefly summarize the current knowledge of different epigenetic biomarkers in HNC.
Author Contributions
Academic Editor: Xi Zhang, Co-founder & Scientist SinoScript LLC, USA.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2018 Shilpi Gupta, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Head and neck cancers (HNCs) are the 6th most common and heterogenous group of malignancies with about 780,000 new cases being diagnosed every year and more than 350,000 deaths reported annually 1.Head and neck carcinogenesis is mainly caused by chronic use of tobacco products, excessive alcohol intake and persistent infection with oncogenic human papillomaviruses (HR-HPVs). HPV-positive HNCs represent distinct biological, molecular entities in comparison to the HPV-negative and tobacco induced HNCs. HR-HPV infection significantly effect HNC prognosis. The HPV infected HNC patients are mainly non-tobacco smoking, younger age population with better prognosis than those with HPV-negative HNC patients 2, 3, 4, 5, 6. These strong evidences establish the distinct molecular and clinical role of HPV as a unique viral biomarker for HNC diagnosis and prognosis. These distinct entities can be due to epimutations inspiring further investigation on molecular and epigenetic modifications to modify HNC patient’s outcome.
It seems that HNC is as much a disease of misdirected epigenetic factors as that of genetic factors, which has emerged into one of the “hallmarks of cancers”. Various scientific studies have suggested that epigenetic alterations such as DNA methylation and the post translational modifications of histone proteins and/or mis-expression of non-coding RNAs (ncRNAs), are frequently associated with initiation, progression, differentiation and drug resistance in HNCs 7.Therefore, these epigenetic markers are emerging as important and reliable tools for early diagnosis and prognosis of HNC. In addition, dysregulation in any of these 3 distinct and mutually reinforcing epigenetic mechanisms lead to mis-regulation of gene expression, resulting in the development of HNCs 7, 8, 9.Epigenetic alterations causing aberrant transcriptional activity, provide selective advantage to the tumor through the silencing of tumor suppressor genes or activation of oncogenes and/or mutation effecting DNA repair genes. Epigenetic alterations are also involved in cellular plasticity during cancer progression, and further contribute to formation of cancer stem-like cells or cancer stem cells (CSCs) 10, 11.In this mini review, we explore different epigenetic mechanisms that are known to be altered during HNC carcinogenesis.
Methylation Specific Biomarkers
DNA Methylation
DNA methylationis the best studied epigenetic mechanism in HNCs. Alterations in epigenetic mechanisms controlling DNA methylation lead to changes in genome-wide methylation patterns as well as promoter-specific and/or CpG island specific DNA methylation which play a critical role in HNC growth and development 12.Aberrant changes in global DNA methylation profiles often accompany tumour development/carcinogenesis including HNC development. It has been reported that gene activation, due to hypomethylation of cytosine-phosphate-guanine (CpG)-dinucleotide rich promoter regions, was found in early grades of HNCs 13, 14.Esposti and co-workers showed, that differentially hypomethylated regions were mainly characterized by the loss of methylation at CpG island associated with HPV infection, which is independent of the HNC anatomic sub-sites. This hypomethylation has been related to the aberrant activation leading to overexpression of several oncogenes, thereby causing chromosomal instability. Further, analysis of DNA methylation and gene expression profiling in both HPV-positive and HPV-negative HNC cell lines, revealed that HPV-positive HNC cells show higher level of genic methylation along with over-expression of DNMT3A 15.Colacino et al analysed CpG methylation at 1505 CpG sites across 807 genes in 68 well-annotated HPV-positive and HPV-negative HNCs samples, they found that methylation in the CCNA1 promoter sites was higher in HPV-positive HNCs in comparison to HPV-negative counterparts indicating that epigenetic regulation may serve as novel targets for the development of virus-specific therapeutic regimens 16.Higher levels of salivary DNA methylation, in a panel of tumour-suppressor genes (RASSF1α, p16 INK4a, TIMP3 and PCQAP/MED15), was found in HPV-negative HNC patients as compared to HPV infected HNC patients. DNA hypermethylation of the anoactin-1 (ANO1) promoter at CpG’s was correlated with the higher expression of ANO1 in head and neck tumors 17.Recently, Misawa and co-workers demonstrated that Sal-like protein 1 (SALL) mRNA is downregulated in HNC patients due to DNA methylation which may serve as a methylation marker to identify HNC patients at high-risk for relapse 18.Thus, these studies are indicative of the fact that epigenetic alterations that aberrantly change transcriptional landscape, are crucial to the understanding of molecular mechanisms underlying development of HNC with respect to HPV infection.
Epigenetic Modifiers as Diagnostic Markers in HNC:
Writers (Histone Modifiers)
The structure of chromatin is dynamic and its fundamental unit is the nucleosome, which consists of 165bp of DNA strand wrapped around the histone octamer core, comprising of two copies of 4 globular histone proteins (H2A, H2B, H3, and H4). Histone proteins play an essential role in regulating the epigenetic state of the cell 19. The posttranslational modification (PTM) of histones and DNA is an important and frequently observed in HNCs. These histone alterations are closely related to DNA methylation, leading to carcinogenesis. Chromatin protein marks which cooperate with DNA methylation marks in maintaining gene expression, are altered in various cancer cells including HNCs. Expression of the genes coding for polycomb and trithorax group members (which frequently play roles during development to establish chromatin protein marks) are known to be overexpressed in HNCs as shown in Figure 1 (GEPIA). Over-expression of Polycomb complex member, enhancer of zeste homolog 2 (EZH2) in tongue cancer, correlates with reduced overall survival and was found to be of great prognostic importance 20. In another study, SUZ12 overexpression in HNCs correlated with cervical node metastasis along with reduced survival. It was further shown that the knockdown of SUZ12 inhibited cell proliferation and migration 21. In an independent study, mutations and copy number variations (CNVs) in MLL1, MLL2, MLL3, KDM6A and KDM6B were found to be a contributing factor in HNC progression 22. Number of missense, nonsense and frameshift mutations in MLL2 and EZH2 are associated with HNCs 23.Increased expression of histone deacetylase-2 (HDAC-2) associated with metastasis along with poor survival and further, served as a good prognostic marker for HNC patients 24. These studies suggest that treating HNC patients by targeting and modulating different epigenetic genes (responsible for control of cell survival, proliferation, migration, differentiation and apoptosis) may improve the efficacy of conventional HNC therapy.
Figure 1. Comparison of expression (transcripts per million) for Polycomb and trithorax group members in normal versus the HNC samples. Analysed using the data from GEPIA
Readers (Chromatin Remodelers)
ATP-dependent chromatin remodelers are responsible for opening and closing of chromatin. In the form of multimeric protein complexes, they catalyze the nucleosome sliding or repositioning, ejection and unwrapping. The chromatin remodeling enzymes are classified into different families on the basis of sequence similarities in the ATPase domain and differential combination of accessory protein domains. These are categorized in four types; (i) SWI/SNF (mating type SWItch/Sucrose Non-Fermenting) family, (ii) ISWI (Imitation SWItch) family, (iii) CHD (Chromodomain Helicase DNA-binding) family and (iv) INO80 (INOsitol requiring) family.
The chromatin remodelling enzymes consist of domains such as the bromodomain (recognition of acetylated lysines) 25, Chromodomain and PHD finger (recognition of methylated-lysines) 26, and HAND-SANT-SLIDE domains for DNA binding 27,Helicase SANT associated (HSA) domain, play an important role in recognition of various histone modification marks and catalyze nucleosome sliding or ejection. The HSA domain overlaps with the DNA binding domain known as DBINO (DNA binding domain of Ino80) 28, 29.
A number of genes coding for chromatin remodelling proteins show altered expression in various cancers. In Figure 2, meta-analysis using GEPIA for genes encoding for chromatin remodelling proteins, show majority of these genes over-expressed (BRG1, BAF47, BAF57, BAF53a, BAZ1A, CHRAC1, CHRAC17, CHD4, RUVBL1 AND RUVBL2) in HNCs. It is expected that over-expression of chromatin remodelling enzymes may lead to global changes in nucleosome occupancy profiles altering gene expression patterns. Altered expression of Remodelling and Spacing Factor-1 (RSF-1) has been analysed in several tumors including HNCs. Gain-of-function of RSF-1 leads to aggressive tumor behaviour, worst prognosis and treatment resistant in HNC patients 30.
Figure 2. Comparison of expression (transcripts per million) for Chromatin Remodelling family members in normal versus the head and neck cancer samples. Analyzed using the data from GEPIA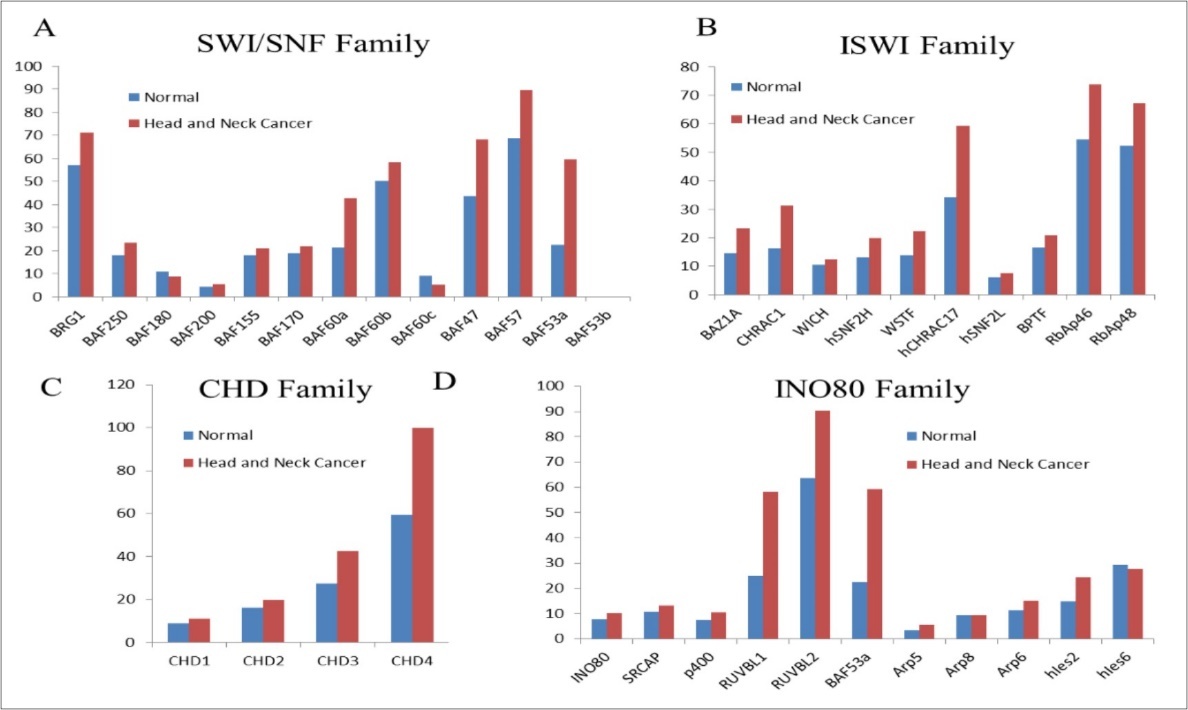
A number of mutations have been characterized in HNCs, that effect genes coding for chromatin remodelling enzymes such as CHD5 (mutated in 4% of HNSCCs), CHD8 (4.1%), SRCAP (4.1%), NSD1 (9.5%), PRMT5 (4.1%) 31.Altered nucleosome positioning around the transcription start- and the transcription termination sites can directly alter the transcriptional status of the target genes leading to disrupted transcription circuits thereby causing cancer phenotype. However, accurate data supporting the relevance of chromatin remodelers in different subtypes of HNCs have been limited. Thus, comprehensive understanding the role of chromatin remodelers associated with HNC carcinogenesis, may help in developing tumor-specific, safe and effective epigenetic drugs for HNC patients.
The Non-coding RNAs (ncRNAs)
Non-coding RNAs (ncRNAs) participate in complex networks of interactions with different nucleic acids and proteins and are routinely dysregulated in several cancers including HNCs. ncRNAs are not translated into proteins but have critical role in regulating diverse cellular processes including cell growth, development, gene expression, differentiation and disease 32, 33.ncRNAs can be classified into two main groups based on their transcript size; (i) small ncRNAs (∼19–200 nt) and (ii) long ncRNAs (>200 nt).
Small ncRNAs
sncRNAs include PIWI interacting RNAs (piRNAs), microRNAs (miRNAs), small nucleolar RNAs (snoRNAs) and the newly discovered transcription initiation RNAs (tiRNAs) and circular RNAs. Alterations effecting ncRNAs that regulate signaling networks, contribute to carcinogenesis and could be considered as potential biomarkers leading to enhanced biological and therapeutic relevance in cancers including HNCs 1.Most of the ncRNAs are involved in epigenetic regulations, mediating alterations in chromatin conformation by directly targeting promoter regions, and thus controlling transcription 34.
P-element-induced Wimpy Testis (PIWI)-Interacting RNAs (piRNAs)
piRNAsare the newest players in cancer biology and forms the largest group of single-stranded, small non-coding RNAs (sncRNAs) subfamily. piRNAs are about 26–31 nucleotides in length with over 20,000 unique piRNA genes in the human genome. PIWI proteins are functionally different from microRNAs and primarily described in germ line, but recent researches reveal that these proteins are expressed in a tissue-specific manner among several human somatic tissue types and have highly conserved roles across species including transposon silencing, epigenetic regulation, gene and protein regulation, genome rearrangement, spermatogenesis and stem cell maintenance in germline tissues 35. These researches highlight the potential role of piRNA-mediated epigenetic regulation in cancer-related processes in human somatic cancers including HNCs 36. Several studies have correlated piRNA expression to clinical parameters associated with HNCs 37.Dysregulation of piRNAs was found to be associated with overall survival and can be utilized as a signature in HNC patients 38.Since piRNAs are emerging players in HNCs, a comprehensive characterization of piRNAs can be used to develop effective therapeutic strategies for HNC.
MiRNAs
miRNAs are the class of small, single-stranded ncRNAs, of about 22 nucleotides in size39.miRNAsconstitute the largest class of non-coding RNA molecules that have either tumor suppressor or oncogenic effects and control various physiological and pathological processes such as cell growth, proliferation, apoptosis, and differentiation wherein each miRNA has the potential to target multiple mRNAs or gene targets. A number of studies have demonstrated that some miRNAs affect gene expression and dysregulate signaling networks involved in HNC pathogenesis 40, 41.Earlier, it was analysed that in majority of cases, 4 of the most important tumor suppressor miRNAs (miR-34b, miR-137, miR-193a, and miR- 203), which are loated in close proximity to the CpG islands, were silenced through hypermethylation 42.On the other hand, upregulation of oncogenic miR-21 was associated with increase nodal metastasis and invasiveness in HNCs 43, 44, 45.An early research revealed that polymorphisms in miR-146, miR-149, miR-196 and miR-499 were found to increase the risk of non-smoker HPV-induced HNC patients46.miR-146 polymorphism is associated with an increased risk of HNCs 47.Overexpression of miR-21, miR-181b, miR-184 and miR-345 has been associated with malignant transformation during HNC 48.Higher expression of miR-21, miR-34c, 184 and miR-155 is known to play a role in anti-apoptotic and proliferative processes leading to downregulation of SOX7 protein and overexpression of c-Myc, Cyclin D1 and Bcl-2 gene 49.DNA hypomethylation induced upregulation of miR-663, leads resistance to chemotherapy in breast cancer cells50and this has been exploited to serve as a promising non-coding biomarker for HNC. Comprehensive profiling of global miRNAs levels in HNCs is yet to be investigated. These studies are essential for stratification of HNC subtypes on the basis of HNC subsites, HPV status and mechanism along with detailed functions of miRNAs for development of effective therapeutic evaluations.
The Small Nucleolar RNAs (snoRNAs)
snoRNAs are a type of linear transcripts with 60– 300 nt size. They are primarily classified into two types of snoRNAs; (i) C/D box and (ii) H/ACA box 51, 52, 53, 54. snoRNAs are linked with a set of core proteins interacting partners known as sno-ribonucleoproteins (RNPs) to form stable and functional snoRNP particles 55.It has been shown that SNORD78 (Box C/D) is upregulated in lung and prostate cancer pateints 56, 57. Moreover, higher expression of SNORA42 (Box H/ACA) served as an essential prognostic marker for cancer patients 58. Chamorro-Petronacci and co-workers (2018) reported that 14 snoRNAs were downregulated and 1 snoRNA was overexpressed in HNC patients 59. These emerging findings highlight the potential role of snoRNAs in carcinogenesis and further point towards the possible role of snoRNAs as unique molecular markers and drug targets for the treatment of HNCs 60.
Transcription Initiation RNAs (tiRNAs)
tiRNAs are small RNAs (about 18nt in length) that are generated from sequences immediately downstream of RNA polymerase II-associated transcription start sites They are commonly associated with highly expressed genes and may participate in epigenetic regulation. Though, the molecular mechanisms connecting tiRNAs to epigenetic regulation in HNCs is as yet unknown it has been reported that the a subset of tiRNAs play an important role in gene regulation and are functional modulators of CCCTC-binding factor (CTCF) as they are enriched at CTCF binding sites. Therefore, understanding the role of tiRNA in malignant transformation and development of novel tiRNA-based therapeutic approaches will be the most exciting challenges in cancer research.
Circular RNAs
Circular RNAs is a class of RNAs regulation a number of molecular processes in eukaryotes. They are further reported to play prominent role in cancers. Expression of circPVT1 (a circular RNA) was checked in 115 HNSCC patients samples and was found to be high in comparison to non-cancerous tissues. CircPVT1 was particularly enriched in HNSCC patients with TP53 mutations thus establishing a significant correlation between circPVT1 expression and mutated p53. The malignant phenotype was further controlled by up- or down-regulation of circPVT1. circPVT1 controls the expression of genes involved in cell proliferation and further binds to and regulate miR-497-5p expression 61.
Long Non-coding RNAs (lncRNAs)
lncRNAs are regulatory noncoding RNAs that have been established to play essential roles in various biological and cellular processes such as developmental processes, stemness and cellular differentiation. Accumulating evidences have also demonstrated the potential role of lncRNAs in the development of various cancers including HNCs and are a focus of current cancer research. With several lncRNAs identified through high-throughput approaches, understanding their functions in cancer progression and pathogenesis will be of utmost importance in oncology research. lncRNAs have opened novel horizons in cancer research, diagnosis and potential anti-cancer therapeutic approaches 62.They may serve both, as oncogenic or tumor suppressor lncRNAs, by modulating cell growth, proliferation, differentiation and invasion in HNC cells 63. Metastasis-associated lung adenocarcinoma transcript 1 (MALAT1) was shown to be overexpressed in HNCs and have been further shown to play an essential role in maintaining epithelial-mesenchymal transition (EMT)-mediated migration in HNCs 64 hence serving as a unique prognostic biomarker. HOTAIR, a lncRNA act as a biomarker for aggressive HNCs as its overexpression is associated with bad prognosis in different subtypes of HNCs 65.Sincethe role of lncRNAs have been well-characterized in HNC carcinogenesis, they can be used as novel molecular markers for HNC diagnosis and further carry a potential therapeutic advantage for aggressive HNC
Concluding Remarks
Growing knowledge on epigenetic alterations such as DNA methylation, histone modifications, and various ncRNA signalling pathways in HNCs may help in the understanding of disease pathogenesis and offer to identify novel unique epigenetic biomarkers for disease diagnosis, prognosis and treatment response for highly heterogenous group of HNCs. Specific targeting of these epigenetic alterations will help in developing novel epigenetic therapeutic approaches to enhance chemo-radio sensitivity and efficacy in HNCs.