Abstract
The purpose of this study was to investigate the effect of cremophor RH-40 and polysorbate 80 with hydroxypropyl methylcellulose (HPMC) F4M on the development of formulations of intranasal erythropoietin with low sialic acid content (Neuro-EPO) as a neuroprotective agent. Parameters such as pH, osmolality, apparent viscosity, and protein concentration were controlled for minimizing the differences between formulations. All Neuro-EPO formulations showed similar behaviour in the physicochemistry quality control. However significant differences between formulations were observed in the permanent unilateral ischemia model. The formulations and the vehicles containing cremophor RH-40 showed higher neurotoxicity levels than those containing polysorbate 80 as a nonionic surfactant. Formulations containing HPMC F4M at 0.6% as a bioadhesive polymer showed higher levels of survival and better neurological status than those without the polymer. The formulations with polysorbate 80 and HPMC F4M showed a higher index of survival, smaller incidence of clinical signs of stroke, and similar behavior in the learning and the memory to the false injured animals used as control. These findings suggest that the intranasal pathway constitutes a safe and alternative route of access of the Neuro-EPO to the brain.
Author Contributions
Academic Editor: Satheesh Podaralla, Research Scientist: Formulation R&D SRI International Menlo Park, CA
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2014 A. Muñoz-Cernada, et al
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
The neuroprotective effect of recombinant human erythropoietin (rHu-EPO) administered intravenously (IV) in many preclinical studies showed reduction of injury caused by ischemic stroke 1, 2, 3. Similar effect occurs in humans 4, 5. However, clinical and experimental results show a high risk of cardiovascular complications by induction of erythropoiesis 6. When sialic acid content is less than 9 sialic acid molecules per molecule protein, is considered a low sialic acid erythropoietin, and is identified like Neuro-EPO, is normally eliminated, since it is not biologically active by the systemic route7, 8. Surprisingly, it has been found that intranasal (IN) administration of Neuro-EPO has higher therapeutic values than the traditional rHu-EPO 9, 10.
The non-hematopoietic derivatives of rHu-EPO and the intranasal route are the most promising perspectives for the treatment of acute and chronic neurological diseases 11, 12. The rate and capacity of drug transport from the nasal cavity to the brain depends primarily on its physicochemical properties. For high-molecular-weight drugs such as rHu-EPO, IN pharmacokinetics exhibit relatively low bioavailability (less of 1%) and relatively high variability compared to IV 13, 14. This can be ameliorated by use of permeation enhancers such as surfactants and mucoadhesive agents which enhance bioavailability and reduce variability 15.
Nonionic surfactants like cremophor RH-40 and polysorbate 80 protect proteins against agitation-induced damage via hydrophobic interactions and tend to be widely regarded as safe. Nonionic surfactants generally have low chronic toxicity and have shown to enhance the flux of materials permeating through biological membranes 16, 17, 18. Neutral mucoadhesive agents like HPMC are expected to substantially increase residence time of the formulation in the nasal cavity, but this result does not always improve absorption of drugs 19, 20.
Permanent unilateral occlusion of the common carotid artery in Mongolian gerbils was the biomodel used to establish therapeutic effectiveness of intranasal formulation of Neuro-EPO. The purpose of this study was to determine the effect of cremophor RH-40 and polysorbate 80 with HPMC on the development of formulations of Neuro-EPO as a neuroprotector agent, by using permanent unilateral ischemia model.
Materials and Methods
Materials
Drug: Low sialic acid rHu-EPO (Neuro-EPO) (not a commercial product, publication number: Patent WO/2007/009404, international application No.PCT/CU2006/000007)21 was supplied by the Center of Molecular Immunology (CIM, Havana, Cuba) with protein concentration of 2mg/mL (L-C0702). The preparation was buffered with anhydrous monobasic sodium phosphate (BDH, England) and anhydrous dibasic sodium phosphate (Merck, Germany). Nonionic surfactant: Polyoxyethylene 20 sorbitan monooleate (Polysorbate 80) was purchased from Riedel-de Häen (Germany) and Polyoxyl 40 hydrogenated castor oil (Cremophor RH 40) was purchased from Panreac (Spain). Isotonic agent sodium chloride was supplied from ESCO (Spain). Benzalkonium chloride (BAC), purchased from Merck (Germany). Bioadhesive polymer HPMC F4M was obtained from Blanver, Brazil. Trifluoracetic acid and acetonitrile of chromatographic grade were purchased from Merck, Germany. All other chemicals were of analytical grade.
Viscosity Study
Universal viscometer RV-20-M5 with coaxial cylinder sensors (HAAKE, Germany) was used for measurements of polymer viscosity. A computer controls the shear rate and the test temperature through a Rheocontroller RC20 which is connected to the rotovisco RV20 and to a HAAKE circulator. Sensor NV one software was used to control the rheometer and to capture the data.
The polymer was initially analysed using concentrations at 0.3, 0.6, 0.9, and 1.2%w/v in water for injection (WFI). All determinations were done with 9mL of polymer solution with the sensor M5/NV over a shear rate range of 1–250 s−1for the rheological behaviour and 100 s−1 for the apparent viscosity. The sample stage was held at 25°C by using the Rheocontroller RC20. This temperature was chosen to reflect normal room temperature when using a nasal device.
Formulation Preparations
For in vivo study four different formulations at 1 mg/mL of Neuro-EPO were employed together with the vehicle as detailed below. BAC was used as an antimicrobial preservative.
(F1) Neuro-EPO in isotonic saline-phosphate buffer with cremophor RH-40.
(F2) Neuro-EPO in isotonic saline-phosphate buffer with cremophor RH-40 and HPMC F4M.
(F3) Neuro-EPO in isotonic saline-phosphate buffer with Polysorbate 80.
(F4) Neuro-EPO in isotonic saline-phosphate buffer with Polysorbate 80 and HPMC F4M.
The vehicles were constituted by the same excipients of the formulations but without protein. They were identified like V1, V2, V3 and V4.
All formulations and vehicles were adjusted at pH 6.2 ± 0.2 with phosphate buffer (anhydrous monobasic sodium phosphate and anhydrous dibasic sodium phosphate). pH measurements were performed with a Radiometer PHM 61 (Denmark). The concentration of HPMC F4M was selected on the basis of the results obtained in the viscosity study and evaluated formulations. Cremophor RH-40 and Polysorbate 80 had saturable binding to Neuro-EPO with a molar binding stoichiometry of 6 : 1 and 0.25 : 1 (Surfactant: protein), respectively. The effective critical micelle concentration (CMC) of surfactant solutions containing Neuro-EPO was determined using a Kruss K8 surface tensiometer (Germany). The measure conditions were: temperature 25◦C, accuracy ±0.1mN/m, resolution 0.05 mN/m, ring Ptlr 20, and welded 20mm diameter. Osmolality measurements were performed using a Cryoscopic Osmometer Osmomat 030 (Germany). The formulations were filled into glass vials with its chlorobutyl rubber plug and aluminium overcap.
Method of Formulation Preparation with HPMC F4M
Polymer was dispersed in WFI in a volume of 15% of the total volume of the formulation at a temperature of 85–95◦C. Water for injection was added up to 30% of the total volume. Finally the volume was completed until 100% with a previous stock solution (twice the concentration in formulations final). Stock solution was prepared containing 2mg/mL of Neuro-EPO, Surfactant (polysorbate 80 or cremophor RH-40), monobasic sodium phosphate, dibasic sodium phosphate, sodium chloride and preservative BAC and filtered through a 0.2 µm filter. Osmolality and pH were tested to control physic parameters.
Chemical Analysis
Protein Identification. Identification of Neuro-EPO was carried out by sodium dodecyl phosphate-polyacrylamide gel electrophoresis (SDS-PAGE) in 13% polyacrylamide gels under denaturing conditions 22. The gel was stained with Coomassie Brilliant Blue R-250 by using standard protocols and Low Molecular Weight Marker (LMWM, Pharmacia) was used to determine the relative molecular weight of Neuro-EPO.
Protein Concentration. The total protein concentrations in formulations by Reverse phase—High Performance Liquid Chromatography (RP-HPLC) were performed using a KNAUER HPLC system connected to a Discovery BIO Wide Pore C5 column (25 cm × 4.6mm,5 μm) and a KNAUER UV detector adjusted at 214 nm. The chromatography was carried out with a lineal gradient from 2-70 % of eluent (AcN). The mobile phases were: H2O containing 0.1% Trifluoracetic acid (TFA) (A) and a mixture of H2O/Acetonitrile (AcN) (30:70, v/v) containing 0.08% TFA (B) which were filtered through 0.45 μm filter (Millipore). The flow rate was 1 mL/min and the column was maintained at ambient temperature. The injection volumes were 20 μL.
Method Validation
The method was subsequently validated by following the validation protocols for analytical methods in the International Conference for Harmonisation (ICH) Guidelines23. The method´s specificity, linearity, precision, and accuracy were determined. The specificity of RP-HPLC method was assessed using the forced degradation conditions for proteins stability like overnight vigorous shaking and formulation exposed at 40 ˚C for 4 hours. Chromatogram of formulation, with the best neurological behavior was selected. The linearity of test solution was prepared at seven concentration levels covering from 40-120 % of Neuro-EPO in the formulations. The method reproducibility was evaluated by determining precision of five repeated injection by two different analysts. The accuracy of the method was checked for four known concentration levels (60 %, 80 %, 100 %, 120 %) making by different dilutions, and peak areas was recorded. The mean recovery and the recoveries obtained for each level are described in detail in Table 1.
Table 1. Statistics values for the validation parameters| Parameter | Test | Results | Acceptation criteria |
|---|---|---|---|
| Linearity | Correlation and determination coefficients of standard curve | r = 0.9995r2= 0.9991 | r ≥ 0,999r2 ≥ 0,98 |
| Statistical signification of slope variance | Sbrel = 0.854 % | Sbrel ≤ 2 % | |
| Variation coefficient for response factors | VCf= 1.77 % | VCf ≤ 5 % | |
| Proportionality test | -4.12 ≤ a ≤ 69.65 | Interval (a) includes 0 | |
| Accuracy | Mean recovery | R = 99.97 % | 98 % ≥ R ≥ 102 % |
| Repeatability | Variation coefficient of means obtained for one analyst | CV = 0.56 % | CV ≤ 2,0 % |
| Intermediate precision | Fisher test (comparison of precisions for two different analysts) | Fexp = 5.28Ftab (4; 4; 0,05) = 6.39 | Fexp ≤ Ftab |
| Student test (comparison of means for two different analysts) | t exp = 1.419ttab ( 8 ; 0,05) = 2.306 | texp ≤ ttab | |
| Total variation coefficient | CVtotal = 1.04 % | CVtotal ≤ 3,0 % |
Experimental In Vivo Preclinical Study.
Animals: 108 male Mongolian gerbils (12–15 weeks; 70–90 g) were provided by the National Center for Laboratory Animal Breeding (CENPALAB, Havana, Cuba) and adapted to experimental conditions for 7 days. The animals were maintained in controlled environmental rooms at 22 ± 2◦C, relative humidity 55–60%; light-dark cycle was 12 h/12 h and there were 15–20 room air changes per hour. Food and water was provided ad libitum. Protocols were discussed and approved by the Institutional Ethics Committee, considering the international standards established by ICLAS 24.
The animals were distributed according to a completely randomized design in three groups of 12 animals each: Group I: control group. (Sham operated gerbils), n =12; Group II: unilateral permanent ischemia with nasal vehicle (placebo formulation), n = 12 for each vehicle; Group III: unilateral permanent ischemia coupled with IN Neuro-EPO treatment, n = 12 for each formulation. It was incorporated a diagram with animal study plan and time points (see Figure 1).
Figure 1. Diagram of animal study plans and time points.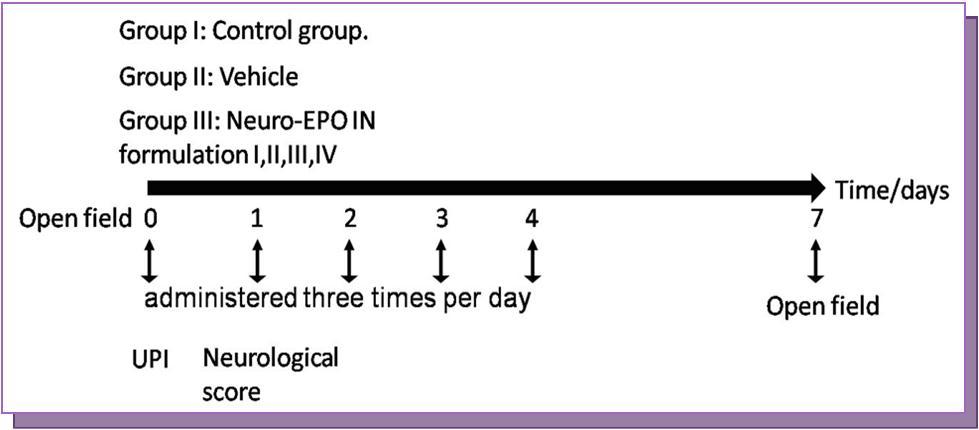
Gerbils were anesthetized with ketamine-atropine-diazepam (47, 0.02, and 5 mg/kg, resp.). Lesions were performed according to Butterfield and McGraw’s method 25. Briefly, the right common carotid artery (CCA) was isolated, double ligated using silk 5-0 suture, and sectioned.
In control operated animals, the artery was only isolated. IN administration was performed essentially as described previously 9, taking into account the established Guide to the Care and Use of Experimental Animals 24. Immediately after surgery, the gerbils were placed on their backs and a total of 10 μg of Neuro-EPO or a corresponding volume of vehicle solution per gerbil was given in nose drops (5 μL/drop) over a 1- to 2-min period, alternating props. between the left and right nares. The mouth and the opposite naris were closed during the administration so the drops could be naturally inhaled high into the nasal cavity. Neuro-EPO IN and the corresponding volume of vehicle IN, were administered three times per day during 4 days, beginning immediately after surgery.
Twenty-four hours after unilateral permanent ischemia, the appearances of the following clinical signs of infarction were assessed: palpebral ptosis, bristling, loss of tone and reflexes in the four limbs, postural asymmetry, rolling or circling, and death. Each sign was scored separately. The sum of scores creates a general neurological score for each animal 26. The clinical signs of brain infarction were assessed as previously established 9. In brief, each animal was evaluated to determine its neurological state according to a scale (with a maximum of 30 and a minimum of 0), grasping strength, and disturbances in posture and gait. Gerbils were placed in the centre of a round open field (30 cm diameter and 25 cm high). “Rearing” was considered as standing straight up on the hind limbs and tail, until the animal returned any forelimb to the floor or touched the open field wall with any forelimb. Exploratory activity was determined by the rearing counted at 3, 6, and 9 min in the open field. An average of the total rearing counts was calculated for each group, rendering three dots per trial. Dots were plotted, and the line obtained by the minimal square method was calculated and considered as the habituation curve. The slope of the habituation curve was used to characterize the animal state.
Statistical analysis was performed by GraphPad Prism Version 5 (GraphPad Software, San Diego, California, USA). All data were represented as mean ± SD, n = 12. Chi-square for viability and U-Mann Whitney Test for neurological state and spontaneous exploratory activity were using. Statistical significance was considered with P < 0.05.
The linearity of test solution was prepared at seven concentration levels covering from 40-120 % of Neuro-EPO in the formulations. The value of 100% is 200µg/ml (dilution of the sample 1mg/ml/5).The method reproducibility was evaluated by determining precision of five repeated injection by two different analysts.
Results
Viscosity Study.
The apparent viscosity of a formulation is an important parameter related with the administration route and clearance from the nasal cavity. Table 2 shows apparent viscosity values for HPMC F4M at different concentrations in WFI. A shear rate-time profile was programmed at constant temperature 25˚C, every shear rate defined the resulting shear stress was measured and used to calculate the rheological behaviour. For the polymer concentrations of 0.3, 0.6, and 0.9% the solutions behaved in a newtonian manner, shear rate had no effect on the apparent viscosity. With at the highest polymer concentration, the solutions exhibited pseudoplastic shear thinning profiles, the apparent viscosity decreased with increasing shear rate.
Table 2. Apparent viscosity of HPMC F4M solutions and rheological behavior. All data were represented as mean ± S.D, n=3.| HPMC F4M | Apparent viscosity(mPa.s) at 100 s−1 | Rheological Behavior(1–250 s−1) |
| 0.3% | 4.5 ± 1.2 | Newtonian flow |
| 0.6% | 40.2 ± 2.0 | Newtonian flow |
| 0.9% | 89.5 ± 6.0 | Newtonian flow |
| 1.2% | 237.0 ± 18.2 | Pseudoplastic flow |
Formulation Preparations.
Based on the results obtained in viscosity study (Table 2), the HPMC F4M concentrations of 0.3, 0.6, and 0.9% were selected for the evaluation in combination with the other ingredients. With concentrations of 0.3 and 0.6% precipitate-free translucent solutions were obtained. However, at higher concentrations, HPMC F4M was unstable due to turbidity. At concentrations of 0.3 and 0.6% of HPMC F4M, with the rest of ingredients, the values of viscosity were lower than those obtained with WFI, for both surfactants (data not shown). Only formulations at 0.6% HPMC showed apparent viscosities in the range suitable for nasal application (4.0–60.0mPa.s) 27, 28.
Physicochemical Quality Control.
The physicochemical determination of Neuro-EPO in the preformulation experiments showed that all variants were colorless, precipitate-free solutions, with concentrations of Neuro-EPO inside the range expected approximately 1 mg/mL (see Table 3). SDS-PAGE analysis showed a unique band corresponding to the relative molecular weight of 29–33 kDa as described for rhEPO (see Figure 2).
Table 3. Physicochemical quality control of the formulation variants. All data were represented as mean ± SD, n=3.| Formulation | pH | Osmolality(mOsmol/kg) | Protein Concentration(mg/mL) |
| 1 | 6.1 ± 0.06 | 288 ± 1.5 | 0.981 ± 0.032 |
| 2 | 6.1 ± 0.08 | 294 ± 1.0 | 1.06 ± 0.055 |
| 3 | 6.2 ± 0.03 | 292 ± 1.0 | 1.03 ± 0.050 |
| 4 | 6.2 ± 0.05 | 302 ± 0.5 | 1.04 ± 0.060 |
Figure 2. SDS PAGE of Neuro-EPO run under denaturing conditions. Samples consists of (Lane 1) Formulation 1, (Lane 2) Formulation 2, (Lane 3) Formulation 3, (Lane 4) Formulation 4 and (Lane 5) molecular weight standards.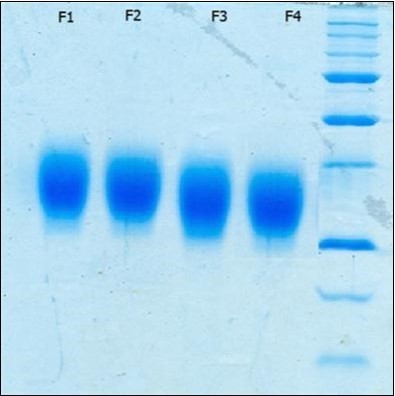
Method Validation
Validation procedure assessed the methods ability in four parameters: linearity, accuracy, precision, and specificity. The statistic values of all tests performed to evaluate these parameters are within the acceptation criteria (see Table 1). Likewise, the method displayed an acceptable degree of linearity (r2= 0.9995) in a concentration range of 80- 320 µg/mL of NeuroEPO. The percent recovery of the method was between 98.41 and 101.53 % for the concentration range of 120- 240 µg/mL and the mean recovery value was 99.9 % ± 1.56, indicating that the accuracy of the method is acceptable for quantitative porpoises. In the repeatability study was obtained a coefficient of variation value less than 2%, which is the maximum limit allowed for chromatographic methods. For intermediate precision is demonstrated that there are no statistically significant differences neither between the means obtained by different analysts according to the Student test (t-test) for a 95% probability and 8 degrees of freedom, nor between the precisions achieved by both analysts for a 95% probability according to Fisher's F test. According to these results, the method is precise and can be found values with a level of significance of 5% in the range between 100.6 and 101.6%.
The visual inspections of the well shaped peaks in the chromatograms (see Figure 3) verify that the proposed method demonstrates satisfactory specificity.
This method complied with international standards for the validation of analytical techniques, and therefore guarantees a procedure linear, precise and exact for the protein quantification, within the range of 100.6 and 101.6%.
Figure 3. Chromatograms for the specificity of RP-HPLC method.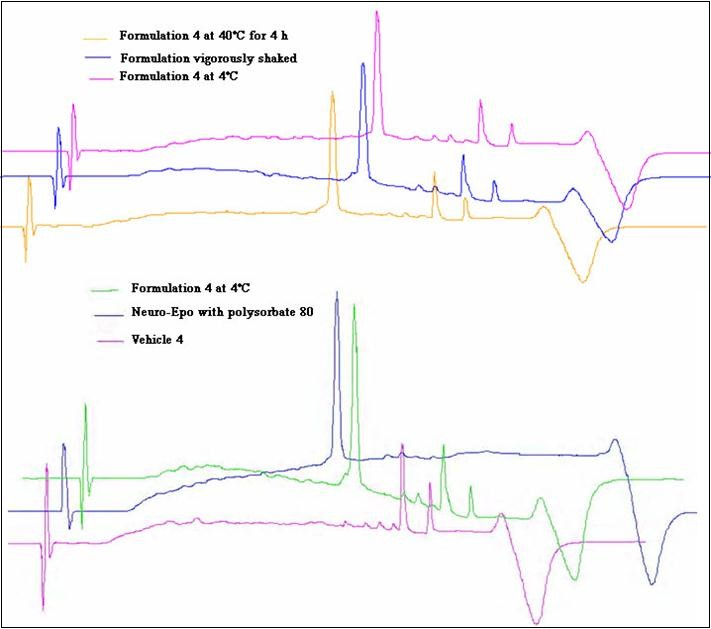
Experimental In Vivo Preclinical Study.
During the experiments described with nasal administration of Neuro-EPO, a higher survival percentage was observed with formulations 3 and 4 during the 7 days subsequent to surgery as evidenced by the analysis of the proportions (Figure 4). However, only formulations 2 and 4 were significantly different (P < 0.05) to their vehicle. Twenty-four hours after the unilateral ligature, animals showed affectations in the neurological status, revealed by means of the test described, and expressed by the score (Figure 5). Clinical signs of stroke were significantly smaller with formulation 4 and showed significant differences (P < 0.01) to their vehicle, but there was no difference in the rest of the formulations.
Figure 4. Effect on viability, 7 days after the injury. Chi-square, which enabled us to compare the vehicle-treated variant with the corresponding Neuro-EPO-treated formulation. (*p<0.05).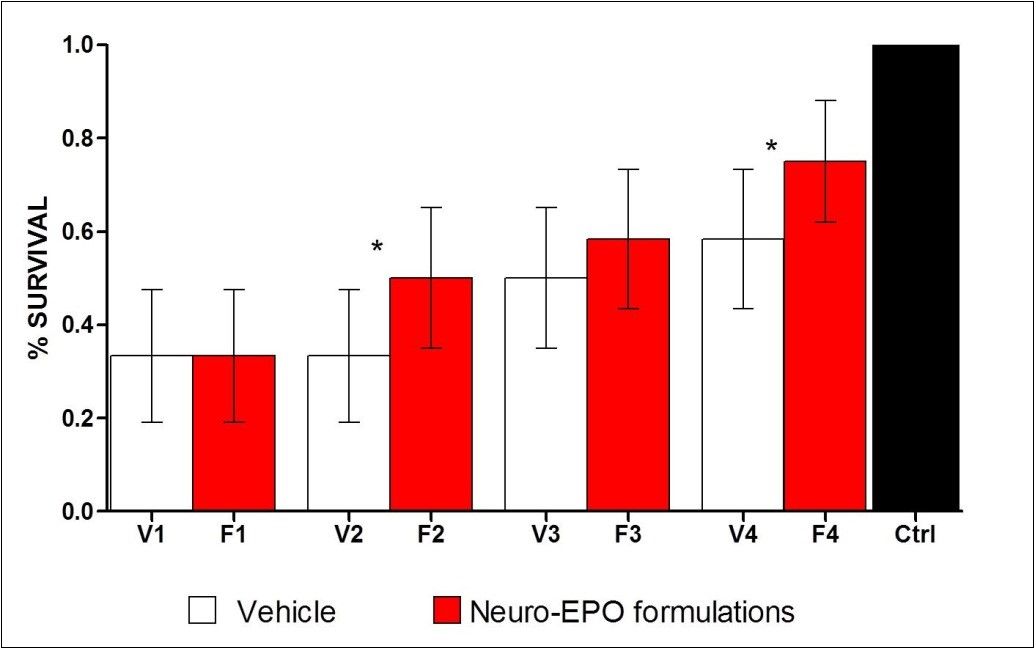
Figure 5. Neurological state score of the animals, 24 hours after the injury were evaluated by the two-tailed Mann-Whitney U test, which enabled us to compare the vehicle-treated variant with the corresponding Neuro-EPO-treated formulation. ** p < 0.01. 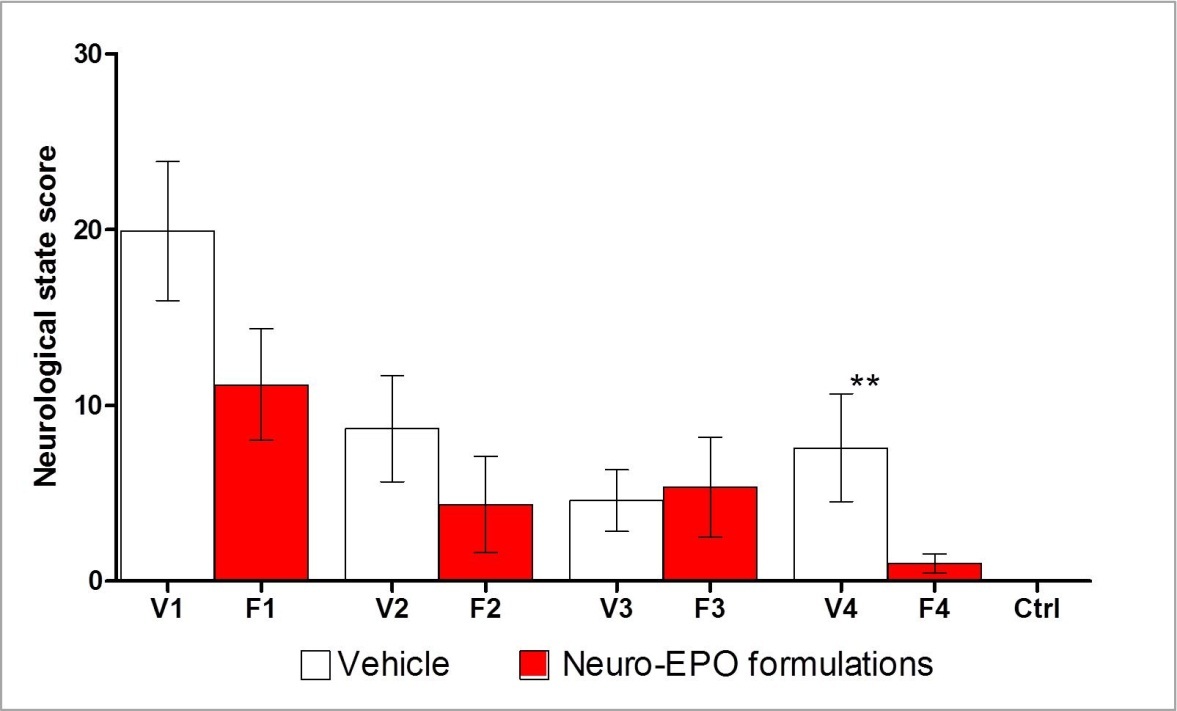
In spontaneous exploratory activity model, slopes of the habituation curves in the open field test were analysed (Figure 6). It could be established that formulations 1, 2, and 3 exhibited significant differences between the habituation curves 7 days after carotid occlusion and control group. The motor-functional exploratory activity appeared depressed with the same formulations (Figure 6), while the animals treated with formulation 4 showed similar activity to the control group.
Figure 6. Slope values before and 7 days after the injury were evaluated by the two-tailed Mann-Whitney U test, which enabled us to compare the control group with the Neuro-EPO-treated formulations 7 days after injury. * p < 0.05, ** p < 0.01, *** p < 0.001.
Discussion
Viscosity Study.
A solution should have sufficiently low viscosity to flow through the respiratory and olfactory epithelium, but must be sufficiently viscous to retain the protein in the nasal cavity 27, 29. The concentrations of 0.3 and 0.6% of HPMC in WFI had an apparent viscosity in the range suitable for nasal application (4.0–60.0mPa.s) 25, 26. With increasing concentration of the polymer at 0.9 and 1.2%, the solution exhibited a greater apparent viscosity.
Formulation Preparations.
The apparent viscosity of the formulations may occasionally result considerably lower than expected. This phenomenon can be caused by the interaction of polymer with one or more ingredients of the formulation; this effect usually depends on the concentration of the interacting materials and on the presence of other ingredients such as: electrolytes, salts, or preservatives. In general, as the ion concentrations in a polymer solution increases, polymer hydration or solubility decreases. The amount of water available to hydrate the polymer is reduced because more water molecules are required to keep the ions in solution. Changes in the hydration state of a polymer in solution are manifested primarily by changes in solution viscosity, turbidity, or cloud point 30.
Physicochemistry Quality Control
Increasing evidence suggests that pH, osmolality, molecular weight, and additives, can all influence drug deposition in the nasal cavity and the pathway a drug follows to the central nervous system (CNS) after intranasal administration 31. The effects of additives on nasal mucociliary functioning is an important issue, adverse effects on the ciliated epithelium may limit “patient” acceptance of formulation and, thus, the use in sub chronic nasal drug delivery 32. For physiological reasons, aqueous nasal preparations are usually in the range of pH 4.0–7.4 and formulated products are isotonic 33.
One of the most challenging tasks in the development of protein pharmaceuticals is to deal with physical and chemical instabilities of proteins 34. Protein can be adsorbed by many surfaces and interfaces, such as container surface, and air/water interfaces. Nonionic surfactants are generally preferred in protein stabilization 35. Low concentrations of nonionic surfactants are often sufficient to prevent or reduce protein surface adsorption or aggregation due to their relatively low CMC 36. These excipients generally act via one or a combination of mechanisms: increasing membrane fluidity, and solubilizing or stabilizing the protein.
Erythropoietin is often stable in a narrow pH range (6.0-7.0) in isotonic solution with phosphate buffer. Formulations of rHu-EPO contain polysorbate 80 as stabilizer with an approximated concentration of 0.03%. This value represents twenty times the critical micelle concentration (CMC) of this non-ionic surfactant 37. There are no reports which relate to the use of cremophor RH-40 and rHu-EPO. The CMC of polysorbate 80 is 0.00157% 38 and cremophor RH-40 is 0.039% 39. Those concentrations are not toxic on the human nasal epithelium 17, 40.
Multidose formulations must contain antimicrobial agents to protect them from microbial contamination during multiple dosage withdrawals from vials. The antimicrobial preservative BAC showed absorption enhancement effect at 0.01% concentration. This could be attributed to its surface active properties. BAC is a positively charged surface active agent which may interact with negatively charged cell membrane via electrostatic binding to surface proteins, may cause structural destabilization of the epithelium, and could thus increase membrane permeability 41.
Increasing the residence time of the dose on the nasal epithelium may have an effect on the absorption of drugs across the nasal epithelium although this has not been proven incontrovertibly 42. Pennington, et al. found that an increase in the viscosity of a nasal solution with the bioadhesive material HPMC resulted in a prolonged clearance rate from the nasal cavity 43. Hence, with progressive increase in concentration of HPMC and thereby also viscosity (0.6%, 0.9%, and 1.25%) an increased halftime of nasal clearance (1.0, 1.7, and 2.2 h, resp.) was found in human volunteers. Polymers may also inhibit protein aggregation through one or more of these properties: surface activity, preferential exclusion, steric hindrance of protein–protein interactions, and increased viscosity, limiting protein structural movement 35.
Our hypothesis is that the increase of the residence time of Neuro-EPO by using a mucoadhesive neutral polymer like HPMC F4M, in combination with nonionic surfactants and absorption enhancement excipients, may have a positive effect on controlled release of the drug to the brain tissues. The pH, osmolality, apparent viscosity, and protein concentration of the formulations were evaluated to ensure physiological acceptability.
All formulations showed adequate physicochemical properties for nasal administration. All were formulated inside the range of pH 6.0 to 7.0 and having the same osmotic pressure as that of 0.9%w/vNaCl solution. The identification and concentrations of Neuro-EPO were the same in all case, indicating the absence of adsorption of protein by container surface. SDS-PAGE resulted in only heavy chains of Neuro-EPO with expected molecular weight that could indicate adecuate stabilization of both surfactants. Neuro-EPO formulations showed similar behaviour in the physicochemistry quality control.
Experimental In Vivo Preclinical Study.
Permanent unilateral occlusion of the common carotid artery has been used for studying the physiopathology of the brain ischemia and for the evaluation of putative neuroprotective agents. In this model, a mortality rate from 25 to 50% has been reported 44, 45.
When comparing the results of the formulation and vehicle of the variants 1 and 2 containing cremophor RH-40 as nonionic surfactant with the formulation and vehicle of the variants 3 and 4 containing polysorbate 80, smaller levels of survival and a more deteriorated neurological state are clearly observed with the first ones. This is evidence of a higher level of damage in the cases that were formulated with cremophor RH-40.
There is evidence that suggests that the derived polyoxyl hydrogenated castor oil like Cremophor EL and Cremophor RH40 could be related with neurotoxicity 46, 47. Our results are in agreement with them, more deteriorated neurological state in formulation and vehicles with this surfactant. Although significant differences between V2 and F2 were observed attributable to bioadhesive polymer. This effect didn't have realistic importance because toxic effect of cremophor RH-40 had bigger impact. It was evident that vehicle V3 group showed similar % survival rate that formulation F2 (Figure 4).
Only Formulation 4 with polysorbate 80 and HPMC F4M polymer showed higher indexes of survival, smaller clinical signs of stroke, and, in general, better neurological status than those similar with vehicle. These results suggest that the bioadhesive polymer HPMC F4M increase the residence time of Neuro-EPO in the nasal cavity due to the effect of mucus adhesion and/or prolonged clearance rate from the nasal cavity.
Animals treated with formulations 1, 2, and 3 did not remember the environment explored 7 days after having explored it for the first time, thus they continue zooming with the same frequency in the three intervals of time. Moreover, a greater functional integrity at day 7 in formulation 4 with Neuro-EPO treated gerbils was noted, when compared with other variants. Motor and exploratory activities expressed by rearing counts in a new recipient, were preserved in the treated animals.
The formulations with polysorbate 80 and HPMC F4M showed a higher index of survival, smaller incidence of clinical signs of stroke, and similar behaviour in the learning and the memory to the false injured animals used as control.
These results constitute an indirect evidence that formulation 4 reached the brain and exerted a neuroprotective effect in a model of ischemia, reflected in a significant improvement of the functional variables.
Other investigations with the same formulation have demonstrated neuroprotective effects in different animal models 48, 49, 50. On the other hand it showed absences of hematological side effects in acute and subacute nasal toxicological evaluation 51. Therefore, the results using formulation 4 suggest that the intranasal administration is effective in the treatment of acute brain ischemia in animal models.
Conclusions
The concentration at 0.6% HPMC F4M was selected as the most suitable in the evaluated formulations. The four Neuro-EPO formulations showed similar behaviour in the physicochemistry quality control. In the permanent unilateral ischemia model, significant differences between the four formulations were observed. The formulations and the vehicles containing cremophor RH-40 showed higher neurotoxicity levels from those containing polysorbate 80 as nonionic surfactant. The formulation containing 0.6% HPMC F4M as bioadhesive polymer showed a higher level of survival and better neurological status than the others without polymer. The formulation 4 showed a higher index of survival, smaller incidence of clinical signs of stroke for 24 h, and a similar behaviour in the learning and the memory to the false injured animals used as control.
These findings suggest that the intranasal pathway could be an alternative route of access of the Neuro-EPO to the brain. However, the final evidence needs to be scientifically established with clinical trials in stroke patients.