Abstract
Many lay people along with some so called “key opinion leaders” have a common slogan “There's no answer for cancer”. Again, mistake delays proper treatment and make situation worse, more often. Compliance is crucial to obtain optimal health outcomes, such as cure or improvement in QoL. Patients may delay treatment or fail to seek care because of high out-of- pocket expenditures. Despite phenomenal development, conventional therapy falls short in cancer management. There are two major hurdles in anticancer drug development: dose-limiting toxic side effects that reduce either drug effectiveness or the QoL of patients and complicated drug development processes that are costly and time consuming. Cancer patients are increasingly seeking out alternative medicine and might be reluctant to disclose its use to their oncology treatment physicians. But there is limited available information on patterns of utilization and efficacy of alternative medicine for patients with cancer. As adjuvant therapy, many traditional medicines shown efficacy against brain, head and neck, skin, breast, liver, pancreas, kidney, bladder, prostate, colon and blood cancers. The literature reviews non-pharmacological interventions used against cancer, published trials, systematic reviews and meta-analyses.
Author Contributions
Academic Editor: Wei Wu, Nanjing Medical University, China
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2019 Abdul Kader Mohiuddin
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
In 2019, 1.8 million new cancer cases and 0.6 cancer deaths are projected to occur in the USA 3. Globally, cancer responsible for at least 20% of all mortality 4, 18.1 million new cancer (9.5 million cases were in men and 8.6 million in women, according to AICR), 9.5 million death in 2018 5, 6, 5- year prevalence 43.8 million (a nearly 67% of total cancer patients) 7, 8, is predicted to rise by 61.4% to 27.5 million in 2040 9. Cancer is the second most common cause of death in the USA and rest of the world 10, 11. It is estimated that there will be 18 million new cases of cancer and 9.6 million cancer deaths in 2018 (GLOBOCAN 2018). Mortality rates in LMICs were 2-fold higher for cervical cancer and 40% higher for male lung and liver cancers during 2012-2016 3, with around 70% of deaths from cancer reported in LMICs 12, 13. Asia, Africa, and Latin America are home of 50% of cancer patients collectively; with more than half of global cancer-associated mortalities occurring in Asia alone 13. Lung cancer is the leading cause of cancer death (nearly 1 in 5 of all cancer death) 14, stomach cancer is the 3rd15 and pancreatic cancer is the 7th16 leading cause of cancer-related deaths worldwide. Prostate cancer is the second most frequent cancer diagnosis made in men and the fifth leading cause of cancer deaths for males 17. Asia and Europe are the home of more than 60% prostate cancer patients 18. Breast cancer is prevalent in 12% of women in the USA more than 2.5 million new cases of breast cancer were diagnosed in 2017 19. The rate for breast cancer declined by 35% in last 3 decades but number of deaths remain same 20. The 3 most prevalent cancers in 2019 are prostate, colorectal and skin melanoma among males, and breast, uterine corpus, and colorectal among females 21. Overall cancer death rates declined faster in blacks than whites in US, although rates for cancers of the breast, uterine corpus, and pancreas are increasing in black people 22. Also, black men have a 70% higher prostate cancer and a more than 2-fold higher mortality rate compared with white men 23. The cost of delivering cancer treatment is estimated to rise globally with a projected total spending of $458 billion by 2030 24. However, the financial burden stems from employment loss, cost of care even when patients don't require chemotherapy, out of pocket costs' opportunity costs of informal care time and can continue long after the death of the patient 25, 26. Studies say 46 billion in productivity lost in major emerging economies due to cancer 27 and economic costs of tobacco-related cancers exceed USD 200 billion each year 28. Also, cancer causes 2.6 times more likely to file for bankruptcy than the non-cancer people 29. Cancer trends in young adults, reflect recent changes in carcinogenic exposures, which could foreshadow the future overall disease burden 30. Cancer cachexia (anorexia, weight loss, loss of adipose tissue and skeletal muscle) is reported in 30%-80% cancer patients and causes 20% of all cancer deaths 31. Worldwide, some 60%-80% people depend on alternative medicines 32, 33, 34, which is also true for nearly 40% to 70% European 35, 36, 50% Italian, 40% Korean, 30% British 37 and up to 87% of Australian cancer patients 38. Use of unapproved/unlabeled/wrong herbal treatment is not uncommon 39, 40 and also drug interactions reported phyto-therapeutics in oncology 41. So, Proper and up-to-date knowledge is necessary in using alternative treatment options as patients who received alternative medicines had a 2.5 greater risk of dying compared to those who received conventional cancer treatment 42.
Reasons Behind Choosing Alternative Care
Pain is affects approximately 66% cancer patients 43, distressing or intolerable in more than one‐third of patients 44 and chronic pain is associated with primary cancer itself or metastases or its treatment (chronic post-cancer treatment pain) 43, 44, 45, 46. Although, WHO described opioids as essential medicines for pain control but distribution shows substantial inequity, a less than 20% of the world’s population consuming more than 90% of the world’s supply 46. Also, some 85% of PCPs perceived their training in pain management to be inadequate in a Pan-European survey 47. Along with these, fear of dependence, prescription diversion, regulatory scrutiny, withdrawal symptoms, opioid-related adverse events and deaths limit its use 46, 48, 49, 50, 51, 52. There is a lack of high-quality evidence regarding the analgesic efficacy of NSAIDs in cancer; contradiction and inconsistent findings also reported 53, 54, although advocated as a useful adjunct for management of cancer pain 55, 56. In addition, long-term use of NSAIDs is often associated with many serious cardiovascular, gastrointestinal, renal, and other side effects 54, 57. Some other studies also reveal association of NSAIDs with certain cancer types 58, 59. Several studies support use of cannabis/marijuana in cancer pain management 60, 61, 62, 63, 64, 65, 66, 67, 68, 69, 70. Its social acceptability is gradually increasing around the world 71, but many studies oppose it’s use or at least demand further investigation of benefit risk ratio 72, 73, 74, 75, 76, 77, 78, 79, 80. Chemotherapy and radiotherapy are still commonly conventional approaches for treatment of patients harboring advanced cancer 81. Traditional chemotherapy also associated with neuropathic pain 82, fatigue and sleep disturbance 83, anxiety and depression 84, mouth sores, nausea and vomiting, early satiety 85, alopecia 86, bone and muscle wasting 31, 87. Futile medication use in management of terminally ill cancer patients has also been reported, one-fifth of cancer patients at the end of their life took futile medications (statins and antidementia drugs in nearly 100% cases, antihypertensives and bisphosphonates in nearly 30% cases) 88. The goal of cancer palliative care is to prevent or treat, the symptoms and side effects of the cancer type and its treatment, caregiving to any related physical, emotional, social, and spiritual aspects 89, 90, 91. Some alternative therapies, like acupuncture, physical therapy, aromatherapy, CBT are widely recommended along with mind-body interventions like yoga, tai chi, meditation and mindfulness, that keep people fit and energetic as they undergo treatment 1, 2.
Herbal and Non-Herbal Plant Derivatives
Medicinal plants are a rich source of secondary metabolites with interesting biological and pharmacological activities 92. Kuruppu et.al, 2019 reported that there are 3000 plants possess some anticancer properties and nearly 75% cancer drugs are derived from natural sources, 40% of them are FDA approved 93, 94, 95, 96, 97. Only a small number of natural anti-tumor products including vinblastine, vincristine, podophyllotoxin, paclitaxel (Taxol) and camptothecin have been tested clinically, while vinflunine ditartrate, anhydrovinblastine, NK-611, tafluposide, paclitaxel poliglumex, combretastatins, salvicine, curcumin, indirubin, triptolide, homoharringtonine are still on trial 98. In addition, there are 195,000 pharmacologically active compounds for which the interactions are quantitatively known 99. According to an estimate, more than 300,000 secondary metabolites exist in nature 100. Glycosides 92, alkaloids, polyphenols, saponins, tannins and terpenoids 101 have shown promising results in cancer research. Chinese herbal medicines (CHM) also have been demonstrated to exert synergistic effects with other anticancer drugs, improved efficacy and reduced side effects 81, 102, 103, 104, 105, 106, 107, 108, 109, 110. It is an independent medical profession in Hong Kong and mainland China 108. Cancer patients used CHM to improve their physical and emotional well-beings and to reduce cancer therapy-induced toxicities 111. Nutrition and foods are related to about 30% of all the cancers cases 112. Omega-3s from fish pack a stronger punch than other oils when it comes to cancer prevention 113, 114, 115. Seaweeds are specifically used to treat tumors in CHM 116. Several studies revealed that active metabolites among the terpenoids, including carotenoids, polyphenols and alkaloids that can obtained from marine source 117, 118, 119, 120, 121, 122, 123, 124, 125. Compounds from natural sources with anti-proliferative activity represent an important and novel alternative to treat several types of cancer.
Lung Cancer
American Cancer Society estimated that in 2018 lung and bronchus cancers would be responsible for 234,030 new cases which represent 14% of all new cancer cases and 154,050 deaths 126. Non-small-cell lung cancer (NSCLC) is the most common type of lung cancer, accounting for about 80%-85% of all cases 127, 128. More than half of the NSCLC cases are diagnosed at an advanced stage (stages III and IV) 129. Smoking causes at least 80% of lung cancer deaths 130. Lin et.al, 2019 concluded association between lung cancer incidence and increased reliance on coal for energy generation 131. Other possible reasons are exposure to indoor and outdoor air pollution, exposure to radiation, and occupational exposure to agents such as asbestos, nickel, chromium, and arsenic 132. Cannabidiol (a non-psychoactive compound from Cannabis sativa), significantly inhibits the recruitment of tumor-associated macrophages (TAM) in primary tumor stroma and secondary lung metastases 133. Table 1.
Table 1. Medicinal Plants Used in Lung Cancer| Plant | Plant Parts Used | Important Constituents | Mode of Action/ Pathway Modulation/ Study Results | |
| Allium sativum L | Bulbs | Organo-sulfur compounds (OSCs) like allicin (Figure 2a), allixin (Figure 2b), diallyl sulfide, diallyl disulfide, etc. | OSCs are antioxidant, detoxify carcinogen & have antiproliferative properties. Raw garlic intake, <2 times per week, ≥2 times per week was inversely associated with lung cancer 134, 135, 136. | |
| Curcuma longa | Rhizome | Curcumin (Figure 2c) | Inhibition of telomerase activity, dose-dependent cytotoxic effect on A549 lung cancer cell line 137. | |
| Ferula assa-foetida | Resin | Conferone (Figure 2d), a sesquiterpene-coumarin | Cytotoxic effect on A549 lung cancer cell line 138. | |
| Astragalus cytosus | Root | Polysaccharide (and flavonoids of other Astragalus spp.) | Inhibit the proliferation and delay the tumor growth xenograft of human NSCLC in vivo and in vitro through the down-regulation of NF-κB activity 139. Astragalus polysaccharide injection integrated with vinorelbine and cisplatin offered an improved QOL for patients with advanced NSCLC 140. Astragalus-based CHM may increase effectiveness of platinum-based chemotherapy 141. | |
| FritillariaeThunbergii(Bei-Mu) (Figure 3a) | Plant extract | Alkaloids | MDR reversal activity on human lung adenocarcinoma parental cells A549 with dosage dependence and the apoptosis rate was increased over time 142. | |
| Platycodon grandiflorum(Jie-Geng or balloon flower) (Figure 3b) | Roots | Platycodin D (Figure 2e) (Saponin) | Blocks reduction of AKT expression by small interfering RNA (siRNA) and enhance apoptotic effect 143, Modulation of the AMPK/mTOR/AKT, MAPK signaling pathways in A549 cells 144, 145, reduces the protein level of PD-L1 in lung cancer cells 146 and enhances autophagic cell death. | |
| Catharanthus roseus | Root, stem, bark and flower | Vinca alkaloids, vinblastine, vinorelbine (Figure 2f) and vincristine | Vinorelbine plus cisplatin is used for non-small-cell lung cancer. vinorelbine and cisplatin combination in patients with stage III A and stage III B non-small-cell lung exhibited positive results 147. Resistance to chemotherapy of Vinca alkaloids (Microtubule-targeting agents) has been a major obstacle to the treatment of lung cancer 148. New generation agents that have the potential to overcome the mechanisms of resistance to the available drugs may provide new therapeutic opportunities 149. High vinorelbine blood levels were associated with severe toxicity 150. | |
| Selaginellatamariscina(Spike Moss) (Figure 3d) | Whole plant | Amentoflavone (biflavonoid) | S. tamariscina ethanolic extract (STE) potently inhibited human AKR1B10 and synergistically increased the doxorubicin anti-proliferative effect in A549 and NCI-H460 human lung cancer cells 151. STE decreased expressions of matrix metalloproteinase (MMP)-2, -9 and urokinase plasminogen activator (u-PA) reported in an older study 152. Oral administration of STE could not prevent the tumor formation but provided strong inhibition of tumor growth 153. | |
| Crocus sativus L. (Saffron) | Dried stigmas | Crocetin (natural apocarotenoid dicarboxylic acid) | In vivo protective effect 154, induction of apoptosis 155, suppression of pulmonary tumor promotion 156 and also chemosensitizer for vincristine via p53-dependent and independent pathway 157, 158. | |
| Ophiopogonis Decoction(Mài-Mén- Dōng-Tāng) (Figure 3c) | Tuber and Root | Ginsenosides, lignans, steroidal saponins, and homoisoflavanones. | Modulatory effects on apoptosis, autophagy, cell cycle progression, and cell proliferation 159. | |
| Sesbania grandiflora (Humming-bird tree) (Figure 3e) | Dried leaves | Saponosides | Methanolic fraction of S. grandiflora exerted potent antiproliferative effects in the human lung cancer cell line, A549, involve a pathway that prevents NFκB activation, induction of apoptosis with high levels of ROS intermediates 160. | |
| Toona sinensis | Leaves | Terpenoids, phenylpropanoids, gallic acid (Figure 2g) and flavonoids | Inhibited H441 xenograft tumor growth in vivo and in vitro, induction of apoptosis in vitro 161. | |
| Phyllanthus emblica and Terminalia bellerica | Fruits | Pyrogallol and gallic acid (tannin) | With doxorubicin or cisplatin resulted in a synergistic effect and the possibility of reducing the doses of the chemotherapeutic drugs 162. Antiproliferative and antitumor properties on lung cancer cells and lung adenocarcinoma xenografts due to presence of tannins (P. emblica) 163. T. belerica induced apoptosis in lung cancer through regulation of Bax/Bcl-2 is also reported 164. | |
| Cinnamomum subavenium | Leaves and bark | Subamolide A (butanolide) | Induced lung cancer cell death by ROS generation, which triggers mitotic catastrophe followed by apoptosis 165. | |
| Camellia sinensis(Tea) | Leaves | Catechins (Polyphenol) | (−)-Epigallocatechin-3-gallate (catechin), the major polyphenol in green tea induces ROS and oxidative DNA damages as well as apoptosis in vivo and in vitro 166. | |
| Panax ginseng | Roots | Saponins | Ginsenoside Rh2 mediates changes in the microRNA expression, related to angiogenesis, apoptosis, chromatic modification, cell proliferation and differentiation. Another important mechanism is the inhibition of tumor cells angiogenesis that inhibits VEGF expression in LLC. Rg3 has been found to be a potent inhibitor of invasion of several tumor cell lines H1650, H520 and H1963 in SCLC 167. | |
| Taxus brevifolia | Bark | Paclitaxel (Figure 2h)(Taxol®) (taxane dipertene) | The paclitaxel-based formulation Abraxane® is the only nanomedicine approved by EMA and the FDA in combination with carboplatin for the first-line treatment of advanced NSCLC in adult patients who are not candidates for potentially curative surgery and/or radiation therapy 168. Inhaled submicron particle paclitaxel (NanoPac®) demonstrated substantial deposition and retention of paclitaxel in sampled lung tissue in rodent model 169. | |
| Curcuma longa | Rhizomes | Curcumin(yellow polyphenol compound) | Curcumin (4 mg/kg every 2 days for a total of 7 injections) exhibits a better treatment efficacy of doxorubicin (0.4 mg/kg) in cancer due to its efflux inhibitory effect of curcumin 170. | |
Figure 1. Alternative Treatment Options 12. One third cancer patients use alternative medicine-are not well regulated and may interact with conventional treatments like chemotherapy and radiation. Some alternative therapies, like acupuncture, physical therapy, aromatherapy, CBT are widely recommended by oncologists for cancer pain management. Mind-body interventions like yoga, tai chi, meditation and mindfulness, which were each used by less than 10% of patients, can keep people fit and energetic as they undergo treatment, reduce the side effects of traditional therapies and improve patients’ sleep, stress and mental health. Many hospitals even have alternative medicine centers that offer these programs.
Figure 2. Plant derived biomolecules studied in lung cancer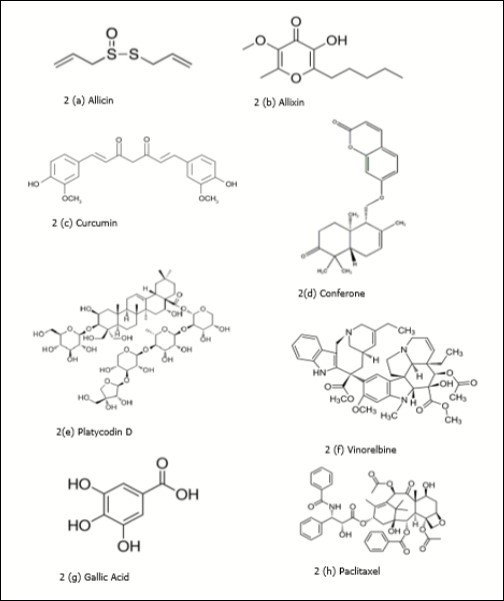
Chemotherapy remains the indispensable choice for the vast majority of patients with advanced NSCLC, including primary tumors and lung metastases 168, 171. Use of the pulmonary route is a promising way to decrease the severe systemic toxicities associated with chemotherapy. Inhalation allows the administration of high drug doses directly to lung tumors without prior distribution in the organism 172, 173. However, Bei-Mu, Jie-Geng, and Mai-Men-Dong-Tang are important CHMs that have improved the survival rate 174. Euphorbia mauritanica and Kedrostishirtella extracts may play a role in inducing cell death in lung cancer cells 175. Several 2019 reviews reveal fucoidans (sulfated polysaccharide mainly derived from brown seaweed) in lung cancer management. Brown algae like Fucusvesiculosus, Turbinariaconoides, Laminaria japonica (Figure 3g) are reported in inhibition of tumor migration and invasion, apoptosis induction and inhibition lung cancer cell progression respectively 176. Fucusevanescens, Sargassum sp., Saccharina Japonica was reported to inhibit proliferation and metastasis, and inducing apoptosis in vitro 177. Undaria pinnatifida acted on ERK1/2 MAPK and p38, PI3K/Akt signaling, F. evanescens increased metastatic activity of cyclophosphamide and showed cytolytic activity of natural killer cells in 2 different studies and F. vesiculosus (Figure 3f) decreased NF-κB in LLC 178. U. pinnatifida (Figure 3h) was found to show average antitumor and superior efficacy against LLC in review of Misra et.al, 2019 179. Sponge alkaloids from Aaptos showed potential in human lung adenocarcinoma A549, from Fascaplysinopsis exerted an anti-proliferative and pro-apoptotic effect in lung cancer, from blue sponge Xestospongia showed apoptosis as well as stimulate anoikis in H460 lung cancer cells in review by Ercolano et.al, 2019 180. Polyphyllin D from Paris polyphylla is well known for its induction of endoplasmic reticulum (ER) stress and mitochondria-mediated apoptotic pathways against lung cancer 181. High fish consumption was significantly associated with a decreased risk of lung cancer 182, 183. Possible mechanism could be changes in formation of PGE2 and PGE3 and alteration of Akt phosphorylation 184. However, other studies reveal healthy dietary intake like high fruit, vegetable, soy protein, poultry (white meat), low-CHO, fish oil-containing diets, together with exercise also decline risks of lung cancer among non-smokers 185, 186, 187. Conversely, long-term use of high doses of some supplements, such as retinol, β-carotene, B vitamins, and vitamin E, increase lung cancer risk in current and former smokers 188. Smokers should continue to avoid β-carotene supplementation 189, 190. When a person stops smoking before the age of 40, they reduce their chances of dying from smoking-related disease by 90% 191. Telephone counseling reduce cost of nicotine replacement therapy (NRT). Again, cessation with intensive telephone counseling and NRT could be over 20% 186. Also, lung cancer mortality can be reduced by 20% via low dose CT lung cancer screening and treatment of early-stage disease 192.
Figure 3. Plants studied in lung cancer (3a). Fritillariae Thunbergii (Bei-Mu) (Source: Inner Path)
Figure 3(b). Platycodon grandiflorum (Jie-Geng) (Source: Wikipedia)
Figure 3(c). Ophiopogonis Decoction (Mài-Mén- Dōng-Tāng)
Figure 3(d). Selaginella tamariscina (Spike Moss)
Figure 3(f). Fucus vesiculosus L. (Source: Seaweed Site of M.D. Guiry)
Figure 3(g). Sesbania grandiflora (Humming-bird tree)
Figure 3(h). Undaria pinnatifida (Source: The Marine Life Information Network)
Blood and Bone Marrow Cancer (Leukemia)
Hematopoietic cancers constitute a diverse group of diseases including leukemias, lymphomas, plasma cell tumors, myelodysplastic syndromes, and mastocytosis. They arise primarily from two categories of immunological cell types, myeloid and lymphoid cells 193. AML is the most common form of acute leukemia in adults, accounting for over 80% of all diagnosed acute leukemias 194, 195. Globally, between 1990 to 2018, the number of leukemia cases markedly increased from 297,000 to 437, 033 196, accounting for close to 250,000 annual deaths due to AML worldwide 197. Optimization of post-remission therapies to maintain complete remission and prevent relapse is a major challenge in treating patients with AML 198. Children with Down syndrome have a 150-fold increased risk of developing AML and 20-fold increased risk of developing ALL 199. The incidence of ALL is about 3.3 cases per 100,000 children 200. Outcomes for patients with CML have substantially improved due to advances in drug development and rational treatment intervention strategies 201. Allowed costs for leukemia patients averaged almost $157,000 in the year after diagnosis, with costs for AML almost tripling that amount, according to a new report from the Leukemia & Lymphoma Society (LLS) 202. Table 2.
Table 2. Traditional Plants Used in Leukemia| Plant | Plant Parts Used | Important Constituents | Mode of Action/ Pathway Modulation/ Study Results |
| Zingiber officinale | Rhizome | 6-gingerol (Figure 4a), 6-shogaol (Figure 4b), and 6-paradol. | Combined with MTX showed synergistic effects on CCRF-CEM, Nalm-6 and ALL primary cells 203. 8-shogaol, originated from ginger, elevated the level of ROS, c-caspase-3, -9, c-PARP, c-DFF-45, and decreased the level of glutathione, MMP, caspase-8 and Bid 204. Enhanced cell growth inhibition while combined with Nerium oleander and imatinib 205. |
| Paris polyphylla | Tubers | Polyphyllin D (Figure 4c) (Steroids) | Polyphyllin D induces apoptosis and differentiation in K562/A02 cells through G2/M phase arrest 206, 207. |
| Withaniasomnifera(Ashwagandha/'Indian ginseng') | Root or the whole plant | Alkaloids, flavonoids, steroids, and terpenoids | Significant cytotoxic and cytostatic potential human T-lymphoblastoid cell line, and induces ICD. Its proapoptotic mechanism involves intracellular Ca2+ accumulation and the generation of ROS 208. Withaferin- A (Withanolides, steroids) from the root induced oxidative stress in human leukemia HL-60 cells 209, 210. |
| Cephalotaxusharringtonia(Korean plum) | Leaves | Homoharringtonine (Figure 4d) (non-proprietory name omacetaxine mepesuccinate, alkaloid) | Suppression of the SP1/TET1/5hmC/FLT3/MYC signaling pathways in AML 211. Elevated ROS generation in *etoposide-treated AML cells and exhibited synergistic cytotoxicity 212. Combined treatment with HSP90 inhibitor provides an alternative way for the treatment of FLT3-ITD positive AML 213. FDA approved for treatment of patients with CML resistant or intolerant to tyrosine kinase inhibitors (TKI) 214. Also reported synergism with Ibrutinib (BTK) 215 and arsenic trioxide on AML stem cells by KG-1 (CD34+/CD96+/CD38+/-) and Kasumi-1 (CD34+/CD38-) cells 216 and by suppressing Mcl-1 through glycogen synthase kinase-3β (GSK3β) 217. Deregulates MYC transcriptional expression by directly binding NF-κB repressing factor and potentiates the therapeutic efficacy of anthracycline/cytarabine induction regimens 218. |
| Ancistrocladus cochinchinensis | Leaves | Naphthalene derivatives and isoquinoline alkaloids | Expresses cytotoxicity against HL-60 cancer cells 219. |
| Chondrodendron platyphyllum | Root barks | Curine (4e)(Bisbenzyl-isoquinoline alkaloid) | Disrupts MMP and curine presented a cytotoxic effect and induced apoptosis in HL-60 cells 220. |
| Alpinia intermedia (Hardy Wild Ginger) | Seeds | Intermedin A (calcitonin family peptide, labdane diterpene) | Increases cleaved (c)-PARP and c-caspase-3 levels, thus inducing apoptosis in HL-60 cells at a dose of 30 µg/mL 221. |
| Thalictrum cultratum(Meadow-rue) | Root | Thalicultratine C (Aporphinoid Alkaloids) | Downregulates MMP and induces apoptosis 222 |
| Artemisia annua L. (sweet wormwood) | Leaves | Artemisinin (Figure 4f) (sesquiterpene lactone) | Initiation of apoptotic cell death through ROS dependent and independent mechanisms, inhibition of cancer proliferation, metastasis and angiogenesis, and modulation of the cell signal transduction pathway and cause lysosomal disruption 223, 224. Combination of artesunate with lenalinomide, commonly used for the treatment of Multiple Myeloma 225. Hybrid 25 which proved even more potent than clinically used doxorubicin against CEM/ADR5000 cells 226. Significantly enhanced NK-92MI cell (natural killer cells) cytotoxicity against K562 cell line 227. |
| Clausenalansium(Wampee) | Stems | Coumarins (8-geranyloxypsolaren) and 2-methoxy-1-(3-methyl-buten-1-yl)-9H-carbazole-3-carbaldehyde | Cytotoxicity against K562 cell line 228. |
| Hericiumerinaceus(Yamabushitake, mushroom) | Whole plant | polysaccharide | Activation of mitochondria-mediated caspase-3 and caspase-9, induce apoptosis through down-regulation of anti-apoptotic proteins (Bcl-2, Bcl-xL(S), XIAP, and cIAPs) in U937 human monocytic leukemia cells 229. |
| Inonotus obliquus(Chaga mushroom) | Whole plant | Inonotodiol and inonotsuoxides (lanostan-type triterpenoids) | Inotodiol inhibits cell proliferation through apoptosis induction by activating caspase-3 230. |
| Allium sativum | Bulbs | Ajoene (Figure 4g) (OSC) | Ajoene induced apoptosis in human leukemic cells via stimulation of peroxide production, activation of caspase-3-like and caspase-8 activity 122, 231. The MAPK family member ERK1/2 was also activated by bisPMB (Ajoene Analogue) 232. |
| Securinega suffruticosa | Callus | Virosecurinine (Figure 4h) (alkaloid) | Inhibits proliferation and induce apoptosis in THP-1 cells by exerting an inhibitory effect on the activation of PI3K/AKT/mTOR signaling pathways 233. Inhibited the growth and proliferation of the K562 cell lines and induced apoptosis in K562 cells by affecting the expression of mTOR, SHIP2, BCR/ABL and PTEN 234. |
| Cynanchum atratum | Root | BW18 (a C-21 steroidal glycoside) | Potential alternative for CML patients. Regulating MAPK pathway leading to S phase cell cycle arrest and apoptosis, inhibited cell viability and proliferation of K562 cells 235. |
| Curcuma longa | Rhizomes | Curcumin(yellow polyphenol compound) | Curcumin targets multiple enzymes (eg. NRF2 targeted NQO1) involved in the ROS metabolic pathway to suppress tumor cell growth, increases ROS levels in CML-derived human leukemic cell 236, 237, 238. Potentiates the efficacy of vincristine and imatinib, significantly increased the apoptosis degree, decreased the activation of NF-κB and the expression of its regulated genes 239. |
| Camellia sinensis(Tea) | Leaves | Epigallocatechin gallate (EGCG) (Catechin) | Inhibits growth of human myeloid leukemia cells through the regulation of pRb synthesis and formation of pRb-E2F complexes 240. Causes caspase-independent necrosis-like cell death in CML 241. In early-stage CLL, the use of 2g of EGCG from the green tea extract twice a day was able to reduce the absolute leukocyte count 242, 243, 244, 245, 246. EGCG was also shown to inhibit DNA replication in leukemia cell lines and to modulate vascular endothelial growth factor leading to apoptosis in leukemic cells 247, 248. |
| Vitis vinifera(Grape) | Seed | Proanthocyanidins, Resveratrol (Figure 4i) (Phenols) | Involves sustained JNK activation and Cip1/p21 up-regulation, culminating in caspase activation 249, 250, apoptotic cell death and cell growth arrest in human promyelocytic leukemia HL-60 cells 251. Effective against the proliferation of both types of acute leukemic lymphocytes of AML and ALL patients 252. Polydatin, a natural precursor of resveratrol (RSV) induces cell cycle arrest and apoptosis in MOLT-4 leukemia cells 253. Combination of chloroquine with RSV and 2 other stilbenes induced significant cell death and toxicity in on RCH-ACV and 697 ALL cells 254. RSV is a molecule without known severe toxicities 255. Proanthocyanidin extract induced mitochondria-associated apoptosis in human AML 14.3D10 cells 256. |
| Punicagranatum(Pomegranate) | Fruit | Gallic acid, ellagic acid, caffeic acid, chlorogenic acid, cyanidin, delphinidin, pelargonidin, gallotannins and ellagitannins | Induced apoptosis and preferentially alters the cell cycle in leukemia cell lines compared with nontumor control cells 257. Flavonoid-rich fractions had proportional inhibitory effects on HL-60 cell proliferation 258. Peel extract promotes growth inhibition of K562 cells mainly via G2/M phase arrest while still conserving apoptosis induction, but at a lower rate 259. Juice extract significantly induced apoptosis in all leukemic cell lines and also induced cell cycle arrest in vitro 260. The polysaccharide PSP001, isolated from the rind of pomegranate fruit exhibited anti-oxidant activity in addition to growth inhibitory effect on leukemic cell lines 261. |
| Panax ginseng | Root | Ginsenoside | Ginsenoside Rh1 showed a suppressive effect on the MAPK signaling pathway, resulting inhibition of invasion and migration of THP-1 acute monocytic cells 262. A metabolite of ginseng saponin, compound K induced apoptosis in human leukemia cells and also induced the activation of caspase-3, -8, and -9, and modulation of Bcl-2 families 263. In addition to cell growth inhibition, compound K suppresses cell DNA synthesis and induces cell cycle arrest at G1 phase in pediatric AML 264. It might have antileukemia activity through CD11c+ cell-mediated antitumor immunity 265. Down-regulated the expression of human telomerase reverse transcriptase, with inhibiting the expression of c-Myc in a concentration-dependent manner 266. |
| Podophyllum emodi(Indian Origin), Podophyllum peltatum(American origin) | Roots and rhizomes | Podophyllotoxins | Podophyllotoxin derivatives showed promising cytotoxicities against a set of human cancer cell lines HL-60 267. Teniposide and etoposide have been reported as inhibitors of MYB transcription factor 268. GMZ-1 suppresses growth and induces apoptosis in adriamycin-resistant K562/A02 cells through modulation of MDR1 expression 269. A398was cytotoxic to the HT-29, MCF-7, MOLT-4 and HL-60 tumor cell lines 270. |
| Coleus forskohlii | Root | Forskolin (Diterpene) | Forskolin as a GSKJ4 sensitizer/adjuvant in vivo, sensitizes Human AML Cells to H3K27me2/3 eemethylases GSKJ4 Inhibitor via Protein Kinase A 271. |
| Andrographis paniculate(Green Chirata) | Leaves | Andrographolide (Figure 4j) (Diterpene) | Pretreatment of U937 with andrographolide (AGP) followed by low doses of topotecan showed an enhancement in inducing apoptosis 272. Inhibits growth of human T-ALL Jurkat cells by downregulation of PI3K/AKT and upregulation of p38 MAPK pathways 273. AGP was most potent to induce cytotoxicity in NALM-6 cells, effectively induced apoptosis by arresting cell cycle progression and increased the nuclear break down in NALM- 6 leukemic cells 274. Cell cycle arrest and mitochondrial-mediated apoptosis in human leukemic HL-60 cells also reported 275. Along with potent derivative NCTU-322, downregulated Bcr-Abl against imatinib-resistant CML cells 276. Inhibits growth of acute promyelocytic leukemia cells by inducing retinoic acid receptor-independent cell differentiation and apoptosis 277. inhibits MV4-11 cell proliferation and reduces drug resistance by blocking FLT3 signaling in AML 278. |
Pterostilbene (Figure 4k) (phytoalexin) isolated from grapevine leaves and blueberries, showing no toxicity in humans up to a dose of 250 mg/day 279, increases Fas expression in T-lymphoblastic leukemia cell lines 280. Blueberry extracts exerted anti-AML efficacy and specifically provoked Erk and Akt regulation within the leukemia stem cell subpopulation 281. Quercetin (Figure 4l) is a polyphenol partially responsible for the anti-AML efficacy of blueberry extracts. It can augment and focus the anti-AML efficacy of nano-liposomal ceramide (Lip-C6) and other ceramide-based therapeutics 282. Plant Homeodomain Finger 6 (PHF6) is frequently mutated in T-cell ALL (T-ALL), or AML 283, are present in about 20% of T-ALL that causes self-renewal and hematopoietic recovery after chemotherapy 284. PHF6 mutations have a significant role in leukemia stem cell activity in the pathogenesis of T-ALL 285. PHF2 low expression was significantly associated with leukemia cell proliferation and several poor prognostic indicators in adult ALL patients. By restoring IKAROS function (zinc finger transcription factor encoded by the IKZF1 gene), PHF2 can be promoted through histone modification 286. Sarkar et.al, 2019 depicted plants of West Bengal (bark of Flacourtiaindica, leaves of Madhucalongifolia (Figure 5b) and Prosopis cineraria) (Figure 5c) showed better cytotoxicity in both AML and CML cell lines (HL-60 and K562) 287. Danışman et.al, 2019 reported combination of flavonoids (apigenin, luteolin, 5-desmethyl sinensetin) and imatinib mesylate were able to enhance the cytotoxic effect on K562 cells in CML 288. Fucoidan (complex polysaccharide from brown seaweeds) inhibited proliferation of the SKM-1 AML cell line via the activation of apoptotic pathways and production of ROS 289 and also induced apoptosis in U937 Cells through activation of p38 MAPK and modulation of Bcl-2 Family 290. In another studies, in vitro and in vivo growth suppression 291 and enhancement of therapeutic potential of arsenic trioxide and all-trans retinoic acid 292 in acute promyelocytic leukemia cells also reported. Fucoxanthin (Figure 4m) induced apoptosis in human promyelocytic leukemia HL-60 cell 293, inhibited growth of leukemia cell lines by ROS generation 294, inhibited phosphorylation of ERK1/2 and histone H3, which are direct downstream signaling targets of lymphokine-activated killer T-cell-originated protein kinase (TOPK) 295, increased cytotoxicity against K562 cells and decreased cell proliferation of K562 and TK6 cells in vitro in imatinib and doxorubicin combination 296. Phlorotannins (algal polyphenols) showed antiproliferative activity in vitro against human leukemia THP-1 and U-937 cells 297. Heteronemin (Figure 4n) (a marine sesterterpenoid) effectively down-regulated cytarabine-induced activation of MAPK, AP-1, NF-κB and c-Myc, the down-stream targets of Ras signaling 298. Cichoriumintybus, Rheum ribes, Alhagipseudalhagi and Glycyrrihza glabra (Figure 5a) (Iranian Traditional plants) also showed notable effects on the leukemia cell lines 299. Juniperus sp. (Figure 5d) can be considered as an alternative source of podophyllotoxin (Figure 4o) and deoxypodophyllotoxin 300.
Figure 4. Plant derived biomolecules studied in leukemias
Figure 5. Plants studied in leukemias (a). Glycyrrhiza glabra (Licorice)
Figure 5(b). Madhuca longifolia (Mahua/Madhuca) (Source: Useful Tropical Plants - Ken Fern)
Figure 5(c). Prosopis cineraria (Khejri/Shami tree) (Source: Greensouq.ae)
Figure 5(d). Juniperus communis (Common Juniper) (Source: IUCN Red List)
Breast Cell Carcinoma
The most common breast cancer type is the invasive ductal carcinoma accounting for 70-80% of all breast cancers diagnosed 301. It starts in a milk passage (a duct), breaks through the wall of the duct and invades the tissue of the breast 302. In US, 232,000 new cases of breast cancer were diagnosed 303 and claimed the lives of 40,290 women 304 in 2015. First-degree relatives of patients with breast cancer have a 2-fold to 3-fold excess risk for development of the disease 305. BRCA1 and BRCA2 are the 2 most important genes responsible for increased breast cancer susceptibility 306. Early breast cancer detection programs depend for effectiveness on the participation rate, which is affected by risk factor awareness 307. Since 1990, between 384,000 and 614,500 breast cancer deaths have been averted due to increased mammography screening and improved treatment 308. However, more than 25% breast cancer is projected to be increased by 2020 309. Women with breast cancer had a higher risk of developing new comorbidities than women without cancer 306. stressful life 310, urban living, mastectomy 311, lower socioeconomic status 309, 312, genetic predisposition, African-American origin, not having children or breastfeeding, early menstruation/late menopause, obesity, alcohol abuse, HRT after menopause, benign breast conditions or having breast proliferation, using contraceptives and exposure to diethylstilbestrol 313, age between 40-60, late age first pregnancy, smoking 314, abortion history 315 are the associated factors. Distressingly, the 5-year cumulative mortality remains unacceptably high at 50%, primarily due to a late-stage presentation 316. Wearing bra is not associated with breast cancer risk 317 but wearing (tight) bras for many hours and having breast implants 315, 318 may have associations. Around 60% of breast cancer mortality occurs in LMICs 319. The prevalence costs of breast cancer care in the US in 2010 was $16.5 billion 320, 321, and exceeded $39 billion before 2017 322. Table 3.
Table 3. Traditional Plants Used in Breast Cell Carcinoma| Plant | Plant Parts Used | Important Constituents | Mode of Action/ Pathway Modulation/ Study Results |
| Podophyllum peltatum | Roots and rhizomes | Etoposide (Figure 6e) and teniposide (semisynthetic derivatives of podophyllotoxin) | Target topoisomerase II and forms a complex with topoisomerase II and DNA. The complex induces breaks in double-stranded DNA and prevents repair by topoisomerase II binding 93. Etoposide alters the balance between CDC25 splice variants in human breast cancer cell lines both at the mRNA and protein levels 323. |
| Juniperuscommunis(Juniper Berry) | Leaves | Deoxypodo-phyllotoxin(aryltetralin lignan or cyclolignan) | Induced apoptosis in malignant MB231 breast cancer cells and inhibited MAPK/ERK and NFκB signaling pathways within hours of treatment 324. Antitumor effect of DPT on MDA-MB-231 human breast cancer xenografts in vivo 325 and in vitro 326. Better efficacy to MDR breast cancer than paclitaxel via avoiding efflux transport 327. Crude aqueous extract of J. communis L. significantly decreased the growth of MCF-7/AZ breast cancer cells 328. |
| Catharanthus roseus | Root, stem, bark and flower | Vinorelbine, vindesine, vincristine and vinblastine (Vinca alkaloids, microtubule inhibitors) | Vinca alkaloids showed Golgi-disrupting activity in 3 different human breast cancer cell lines, BSY-1, MDA-MB-231 and MCF-7 329. Possible involvement of miR-222-3p expression in breast cancer cell apoptosis 330. Quercetin and vincristine are both active against ER breast cancers and exhibit synergism in vitro 331. Vinorelbine-phospholipid complex reduced injection irritation and maintain an antitumor effect in breast cancer in mouse models 332. |
| Taxus brevifoliaand Taxus baccata | Trunk bark | Paclitaxel and docetaxel (Taxanes) | Taxanes are among the most active chemotherapy agents in the management of metastatic breast cancer, associated less nausea and vomiting compared to non-taxane-containing regimens 333, 334, disrupt the equilibrium between polymerized and depolymerized forms of microtubules, the cellular structures required for cell division 335. Paclitaxel+ bevacizumab exhibits synergetic effects and anti-tumor efficacy 336. Paclitaxel/cyclophosphamide better tolerated adjuvant regimen for elderly patients than docetaxel combination in elderly patients 337. Intensive paclitaxel NCT has the lowest incidence rate of neutropenia among other available NCTs 338. Docetaxel combined with trastuzumab and Pertuzumab is the standard first-line therapy for HER2-positive metastatic breast cancer 339, regimens containing docetaxel were associated with lower CIPN severity than paclitaxel 340. Adding taxane to an anthracycline-based regimen improves survival in node-positive breast cancer patients 341. Taxane/cyclophosphamide was more effective than taxane/anthracycline in HER2-negative, breast cancer 342. |
| Syzygiumaromaticum(cloves) | Stems and flowers | β-caryophyllene (Figure 6a) (Sesquiterpene), Eugenol (Essential oil) | Dried flower buds of cloves lowered VEGF-A expressions at high dose and decreased MDA in vivo, induce adoptosis, decrease in CD24 and EpCAM expression in mammary cells 343. β-caryophyllene potentiated the anticancer activity of paclitaxel by facilitating the passage of paclitaxel through the plasma membrane 344. β-caryophyllene induced ionstitutive activation of PI3K/AKT/mTOR/S6K1 signaling and activation of ERK, JNK, and p38 MAPK in tumor cells 345, suppressed constitutive STAT3 activation in breast cancer cell lines and found cytotoxic to MDA-MB-231 346. Eugenol inhibits the cell proliferation and induces the apoptosis in human MCF-7 breast cancer cells 347. |
| Angelica archangelica | Leaves | β-phellandrene (Figure 6b) (cyclic monoterpene) | Moderate antiproliferative activity against Crl mouse mammary carcinoma cells in vitro 348. |
| Nigella sativa | Seed | Thymoquinone (Figure 6c) | Sustained inhibition of breast cancer cell proliferation with long-term treatment potential 349. |
| Viscum Album(European mistletoe, a hemi-parasitic shrub) | Leaves | Mistletoe lectin | Cytotoxic effect breast cancer cells (MFM-223, HCC–1937, KPL-1, MCF-7) 350. Addition to targeted therapy (with mAbs, TKIs, ICIs) significantly reduced adverse effect induced treatment discontinuation 351. Devoid of herb-drug interaction and interference with cytostatic effects of trastuzumab on SK-BR-3 cells in vitro 352. Decreased cell proliferation, increased apoptosis and necrosis reported with human ductal breast carcinoma cell line BT474 353. An increase in the number of neutrophils and the activation of the phagocytic cells also reported with V. Album354. Apoptosis was induced by the activation of its mitochondrial pathway 355. |
| Camptotheca acuminate(xǐ shù or happy tree) | Leaves | Camptothecin (Figure 6d)(Insoluble pentacyclic mono-terpene alkaloid) | Low dosage camptothecin combined with oncolytic measles virus found to elicit the same therapeutic effect as high doses of camptothecin in breast adenocarcinoma 356. |
Figure 6. Plant derived biomolecules studied in breast cancer
Ethanolic extract of Juniperus turbinate was more potent cytotoxic than cisplatin in human breast adenocarcinoma MDA-MB-231 cell lines 357. Juniperusoxycedrus ethanolic extract from needles and berries showed potent cytotoxic effects against two breast cancer cell lines (MDA-MB-468 and MCF-7), with no cytotoxicity towards normal cells (PBMCs) 358. Saturejakhuzistanica (Figure 7a) (Lamiaceae), Casearia Sylvestris (Figure 7b) (Salicaceae), Cedrelopsisgrevei (Rutaceae), Solaniumspirale Roxb. (Solanaceae), carbazole alkaloids, Helichrysumgymnocephalum (Asteraceae), Pituranthostortuosus (Apiaceae), Melaleuca armillaris (Myrtaceae), Rosmarinus officinalis (Lamiaceae), Schinus molle L. and Schinus terebinthifolius Raddi(Anacardiaceae), Erigeron acris L.(Asteraceae), Aquilaria sinensis (Thymelaeaceae), Thymus vulgaris L. (Lamiaceae), Schefflera heptaphylla L. (Araliaceae) showed antiproliferative actions on human MCF-7 breast cancer cells 359. XWL-1-48, a potent orally podophyllotoxin derivative suppress Topo II, induce DNA damage and apoptosis, blocks PI3K/AKT/Mdm2 pathway 360. Alteration of Chk-2 signaling in MCF-7 cells reported with 4'-Demethyl-deoxypodophyllotoxin glucoside isolated from Podophyllum hexandrum(Figure 7c)361. Genistein (most abundant and active isoflavone in soy), binds to the FIH-1 binding site of HIF-1α protein and downregulates HIF-1α in breast cancer cell line 362. Individuals with the habit of green tea (due to presence of EGCG) were found to have a negative association with the risk of future breast cancer (significantly increases circulating estradiol) 363, 364, 365, 366, 367, 368, 369, 370, 371, 372, 373. Compared with the US and EU, some Asian countries like China and Japan have lower breast cancer 364, where dietary consumption of soy products is much higher than US and EU 374. Brown seaweed fucoidan inhibited human breast cancer progression by upregulating microRNA (miR)-29c and downregulating miR-17-5p, thereby suppressing their target genes 375. Lophocladia sp (Lophocladines), Fucus sp (fucoidan), Sargassum muticum (7f) (polyphenol), Porphyra dentata (sterol fraction), Cymopoliabarbata (Figure 7e) (CYP1 inhibitors), Gracilariatermistipitata was found to be effective in breast cancer studies 376. High Urokinase-type plasminogen activator receptor (uPAR) expression predicts for more aggressive disease in several cancer types 377, dietary seaweed may help lowering breast cancer incidence by diminishing levels of uPAR 378. The tropical edible red seaweed Eucheuma cottonii L. (Figure 7d) is rich in polyphenols that exhibited strong anticancer effect with enzyme modulating properties 379. Jazzara et.al, 2016 concluded that λ-carrageenan (Figure 6f) (sulfated galactans found in certain red seaweeds) could be a promising bioactive polymer 380, showed a remarkable inhibitory effect on MDA-MB-231(triple negative breast cancer cell line) cell migration 381. Several studies support polyphenols 382, 383, 384, 385, 386, flavonoids 387, 388, 389, 390, 391, 392, 393, 394, 395, fucoidan 396, 397, 398, 399, 400, 401, 402, 403, 404, 405, 406, 407, lutein/zeaxanthin 408, 409, 410, 411, 412, other seaweed alkaloids, peptides, tannins and polysaccharides 413, 414, 415, 416, 417, 418, 419, 420, 421, 422, 423, 424, 425 in breast cancer management.
Figure 7. Plants studied in breast cancer (a). Satureja khuzistanica (Source: Med P Group)
Figure 7(b). Casearia Sylvestris (Source: Árvores do Bioma Cerrado)
Figure 7(c). Podophyllum hexandrum (Source: Wikimedia Commons)
Figure 7(d). Eucheuma cottonii (Source: tradekey.com)
Figure 7(e). Cymopolia barbata (Source: Melev's Reef)
Colorectal Cancer
Colorectal cancer (CRC) is the third most common cancer worldwide and the fourth most common cause of cancer death 426. It is the second leading cause of death in US, affecting some 135,000 estimated new patients with more than 50000 deaths every year 427, 428, 429. In 2015, there were 376,000 new cases and 191,000 deaths in China 430. The overall incidence of CRC is decreasing in many high-income countries, although reported significant increase in Denmark, New Zealand, Australia, UK and Canada, mainly driven by increases in distal (left) tumors of the colon and predominant in 431, 432, 433, 434, 435, 436, 437, 438. Lifestyle determines around 50% to 60% incident of CRC irrespective of age 439, 440, 441, 442. Physical activity may prevent approximately 15% of the colon cancers 443. Fish, poultry, cheese, fruit, vegetables, tea and coffee were not associated with colorectal-cancer risk 444. Alcohol consumption, red meat/processed meat, junk food, smoking, diabetes and obesity potentiate the same risk 445, 446, 447, 448. In 2018, the estimated national expenditure was $16.6 billion in US, which was $4.5 billion to $9.6 billion in 2009 and projected to be more than $20 in 2020 449, 450, 451. There were over 1.8 million new cases in 2018. Hungary, North Korea, Slovakia, Norway, Denmark, Portugal, Japan are in the top-ranking positions 452. 5-year survival for patients with stage IV CRC is less than 10% 453. The overall risk of CRC among patients with ulcerative colitis is about ten times higher than that of the general population 454. A recent study reveals that chili peppers does not increase or decrease the risk of CRC 455. Previous studies say capsaicin has both carcinogenic and anticancer effects. Table 4.
Table 4. Traditional Plants Used in Colorectal Cancer| Plant | Plant Parts Used | Important Constituents | Mode of Action/ Pathway Modulation/ Study Results | |
| Inula Viscosa | Leaves | Polyphenols (caffeoylquinic acid (Figure 8a), dicaffeoylquinic acid, flavonoids, terpens, lactones) and sesquiterpens (Tomentosin and Inuvisocolide) | Affects the cell cycle progression and induces apoptosis by activation of caspases in colon cancer cells. Moreover, IV extract exhibits anti-tumor activities in an animal model, and it is safe for use 456. | |
| Matricaria chamomilla | Flowers | Flavonoids | Apigenin (4′,5,7-trihydroxyflavone) suppressed the EMT, migration, and invasion of human colon cancer by inhibiting the NF-κB/Snail pathway 457. | |
| Curcuma longa | Rhizome | Curcumin | Inhibit activation of NF-κB, downregulates anti-apoptotic, cell-proliferative, invasive, and angiogenic gene products, suppress activation of STAT3, HIF-1, PPAR and expression of TNF, IL-1, IL-6 458. | |
| Zingiber officinale | Rhizome | 6-gingerol and 6-paradol, shogaols and zingerone | 6-gingerol, regulate the molecules in cellular signal transduction pathways, including NF-κB, AP-1, growth factors, chemokines, MAPK, p53, cyclin D1, VEGF, COX-2 and iNOS pathways. 6-gingerol and 6-paradol have been found to induce cancer cell apoptosis 458. | |
| Alpinia galanga | Rhizome | 1′-Acetoxychavicol acetate (ACA) (Figure 8b) | ACA has been shown to induce apoptosis in CRC cell lines, inhibits DNA synthesis, thereby inhibiting cell proliferation. In rat intestine epithelial cells (IEC6), ACA induced glutathione S-transferase and NAD(P)H: quinone oxidoreductase 1 (NQO1) activities, increased intracellular glutathione levels, and upregulated intranuclear Nrf2 and cytosolic p21. It also has the ability to inhibit azoxymethane-induced colon tumorigenesis in rats 458. | |
| Piper longum(Long pepper or pipli) | Dried fruit | Piperlongumine (Figure 8c) | Piperlongumine targets Ras/PI3K/Akt/mTOR signaling axis to inhibit tumor cell growth and proliferation in DMH/DSS induced experimental colon cancer 459. | |
| Rhizomacoptidis(Huang Lian) | Rhizome | Berberine (Figure 8d) | Downregulates β-catenin-induced proliferation by binding RXR, cell proliferation by inducing the G2/M phase arrest and down-regulated the expression of the related cyclins 460. Promotes apoptosis of CRC via regulation of the long non-coding RNA (lncRNA) cancer susceptibility candidate 2 (CASC2)/AU-binding factor 1 (AUF1)/B-Cell CLL/Lymphoma 2 (Bcl-2) axis 461. | |
| Punicagranatum(Pomegranate) | Fruit | Ellagitannins, ellagic acid | CDKN1A (p21, Cip1) induction followed by cell-unique downregulation of miR-224 or upregulation of miR-215 462. Pomegranate ellagic acid and their microbiota metabolites urolithins exert anticancer effects in preclinical CRC models, and target normal and malignant colon tissues in CRC patients 463. Consumption of the pomegranate extract was significantly associated with a counterbalance effect in the expression of CD44, CTNNB1, CDKN1A, EGFR and TYMs 464. | |
| Phaseolus vulgaris(Navy bean) | Beans | Anacardic acid (Figure 8e) and nobiletin | Imparts glutathione regulation, and involved cancer control mechanisms such as detoxification of xenobiotics, antioxidant defense, proliferation, and apoptosis. Metabolic pathways involving lysine, and phytochemicals were also modulated by navy bean intake in CRC survivors 465. Anacardic acid efficiently repressed expression of CD44 and MMP14 in HCT116 colon cancer cells, with repression of the SUMO-conjugated TFAP2A isoform and elimination of CD44+/hi/ALDH+/hi cells 466. | |
| Macleaya cordata | Leaves | Sanguinarine (8f) | Sanguinarine decreased the tumor size in a dose-dependent manner in orthotopical colorectal carcinomas through intrinsic apoptosis pathway in BALB/c-nu mice. It dephosphorelates STRAP and MELK and disassociates the interaction between them to trigger intrinsic apoptosis. Overexpression of STRAP and MELK may be markers of CRC and their disassociation may be a determinant of therapeutic efficacy 467. | |
| Betula alba (white birch) | Bark | Betulinic acid (Pentacyclic triterpenoid) | Significantly reduced the expression of matrix metalloproteinase (MMPs) and increased the expression of MMPs inhibitor (TIMP-2), MMP-2+ cells and Ki-67+ cells were reduced and cleaved caspase-3+ cells were increased in tumor tissues of mice. Betulinic acid promoted the apoptosis of CRC cells and also inhibited the metastasis of cancer cells 468. Nano-capsulated analogue (2c) with better therapeutic efficacy than parent molecule to colon carcinoma cells has been reported 469. | |
| Boswellia serrata | Oleo-gum-resin | 3 acetyl-11-keto-β-boswellic acid (AKBA) | AKBA induced upregulation of tumor-suppressive miR-34a and downregulation of miR-27a in CRC cells, inhibited cellular proliferation, induced apoptosis and cell-cycle arrest in CRC cell lines, and these effects were significantly enhanced with combined treatment of AKBA and curcumin 470. | |
| Toxicodendronvernicifluum(formerly known asRhusverniciflua) | Stokes | Butein* (3,4,2',4'-tetrahydroxychalcone, aromatic ketone) | The depletion of securing (onchoprotein) enhances butein-induced apoptosis and tumor inhibition in human CRC. 471, 472, 473. Synergistic effects of butein and cisplatin, induce combined inhibition of p38α and MEK specifically induced apoptosis through caspase-3 in CRC cells 474. Butein ameliorated colitis (most important risk factor of CRC) in IL-10(-/-) mice by regulating IL-6/STAT3 and MMP-9 activation 454. | |
| Capsicum annuum(Chili pepper) | Fruit | E-capsaicin (8-methyl-N-vanillyl-trans-6-nonenamide) | Chili peppers can promote digestive juice to secrete and accelerate bowel movements, which may reduce the risk of CRC. Capsaicin can downregulate the expression of COX-2 and B-catenin mRNA, promoting apoptosis through caspase 3 activation and inhibiting the proliferation of cells 455. Capsaicin Mediates Cell Cycle Arrest and Apoptosis in Human Colon Cancer Cells via Stabilizing and Activating p53 475, 476. Low concentration capsaicin promotes CRC cell migration and invasion by triggering production of ROS 477. | |
| Colchicum autumnale (meadow saffron) or Gloriosa superba (glory lily) | Seeds and bulbs | Colchicine | The cellular uptake and apoptotic efficiency of colchicine is correlated with downregulation of MMP-9 mRNA Expression in SW480 CRC cells 478. Colchicine induces apoptosis via MMP loss in HT-29 cells, ROS production, caspase-3 activation, upregulation of pro-apoptotic Bax, downregulation of anti-apoptotic Bcl-2 and phosphorylation of p38, which indicates an involvement of p38-regulated intrinsic apoptosis pathway 479, 480. | |
| Vitis vinifera(Grape) | Seed | Resveratrol | Resveratrol was found to upregulate miR-96 in a genetically engineered mouse model for sporadic CRC, which caused the downregulation of KRAS, an oncogene associated with tumor aggressiveness and chemoresistance 481. Upregulation of miR-101b and miR-455, which in turn led to decreased levels of IL-6 and TNF-α; these are pro-inflammatory proteins known to be promoters of colon cancer 482, 483. Additionally, a combination of resveratrol and grape seed extract has been reported to suppress Wnt/β-catenin signaling and increase mitochondria-dependent apoptosis in in vitro and in vivo models 484. Finally, resveratrol has been shown to increase miR-34a levels in DLD-1 and SW480 cells 485. | |
| Allium cepa(Red onion) | Bulb | Quercetin** | Cytotoxic activity of Quercetin on two human colonic cancer cell lines, HT29 and HCT15, depends on COX-2 dependent ROS generation that induces apoptosis and inhibits cell survival 486. In HT-29 colon cancer cells quercetin treatment decreases cell viability, arrests the cell cycle at the G1 phase and induces apoptosis, also Decreases the expression of CSN6, a subunit of the constitutive photomorphogenesis 9 multiprotein complex 487. Additionally, treatment of HT-29 cells with quercetin upregulates AMP-activated protein kinase, a physiological cellular energy sensor, which markedly suppresses cell proliferation 488. Quercetin has also been demonstrated to suppress the Wnt/β-catenin and NF-κB pathways in CRC cells 489, 490. | |
| Elettaria cardamomum | Bark | Cardamonin (Figure 8g) | Induction of apoptosis, cell cycle arrest, ROS generation, downregulation of MAPK signaling, induction of Bax translocation and loss of mitochondrial integrity were the mechanisms behind the anti-proliferative effect of cardamonin in human CRC cell lines. Also, activation of both p38 and JNK and in presence of ROS scavenger the activation was attenuated 491. Suppression of β-catenin dependent gene expression in human CRC cell lines 492. Cardamonin reduces chemotherapy resistance of colon cancer cells via the TSP50/NFκB pathway in vitro 493. | |
| Tripterygium wilfordii Hook F (Mandarin) | Root | Celastrol (Figure 8h) | Inhibit the growth, adhesion, and metastasis of human CRC cells through the inhibition of TGF-β1/Smad signaling 493. Suppression of the expression of key genes (TYMP, CDH5, THBS2, LEP, MMP9, and TNF) and proteins (IL-1b, MMP-9, PDGF, Serpin E1, and TIMP-4) involved in the angiogenesis pathway 495. Reduces the cell size of the SP (side population) increases frequency of apoptosis and binds to Pgp protein in cell membranes inhibiting its transport function 496. Inhibits CRC cell proliferation and migration through suppression of MMP3 and MMP7 by the PI3K/AKT signaling pathway 497. Effectively inhibited SW480 CRC cell proliferation, down-regulation of Shoc2 expression also significantly inhibited proliferation, colony formation, and migration functions of tumor cells 498. | |
| Mundulea sericea | Bark, roots, and leaves | Deguelin (Figure 8i) (Rotenoid, flavonoid) | Deguelin inhibited CRC cell growth by inducing apoptosis via activation of p38 MAPK pathway 499. Promoted cell cycle arrest at G0/G1 phase in colon cancer cells 500. Deguelin has been found to regulate cell cycle in colon cancer cells by stimulating p27 expression. Cyclin D1 and cyclin E is dramatically downregulated with treatment of deguelin 501. Deguelin exerted anticancer activity of human gastric cancer MGC-803 and MKN-45 cells in vitro 502. | |
| Trigonellafoenum graecum (Fenugreek) | Seed | Diosgenin (Figure 8j)(phytosteroid sapogenin) | Diosgenin induces apoptosis due to HMG (3-hydroxy-3-methylglutaryl) CoA suppression in human colon carcinoma cells 503. Diosgenin induced apoptosis in colorectal cancer cell lines HCT-116 and HT-29 504. HT-29 is sensitized by diosgenin to TRAIL induced apoptosis 505. Acts on colon carcinoma (HCT-15) cells, induces apoptosis via mitochondrial dependent pathway 506. | |
| Darmera peltate (Indian Rhubarb) andRheum palmatum (Chinese Rhubarb) | Bark and roots | Emodin (Figure 8k) (Trihydroxy-anthraquinone) | Inhibited the invasion and migration abilities of RKO cells and decreased the expression of MMP-7, MMP-9, and suppressed the growth of colorectal cancer cells by inhibiting VEGFR2 507, 508, decreased viability of CoCa cells and induced apoptosis in a time and dose-dependent manner, down regulated Bcl-2 family expression 509. Mitochondrial dysfunction and ROS accumulation in colon cancer cells also reported 510. | |
| Embeliaribes (White-flowered Embelia) | Fruit | Embelin (Figure 8l) | In tumor milieu, embelin increased the infiltration of CD8+ T cells, NK cells and mature dendritic cells whilst depleted the regulatory T cells. Moreover, embelin could directly interfere with the generation and function of MDSCs in vitro 511. In colon cancer cells, embelin diminished both the constitutive and IL-6-induced STAT3 activation by stimulating Src homology domain 2-containing protein tyrosine phosphatase (SHP2) activity 512. Embelin potently inhibited NF-κB signaling in macrophages and decreased the production of key pro-inflammatory cytokines and tumorigenic factors involved in CAC, such as TNFα, IL-6 and COX-2 513. | |
Aloe-emodin, a natural compound extract from Aloe Vera, has been discovered to suppress cell proliferation and accelerate apoptosis in a variety of tumor cells 514. Camptothecin induces the upregulation of Programmed Death-Ligand 1(PD-L1) and other cytokines that modulate the attraction, migration, and functions of immune cells, primarily T-cells 515. Scutellarin (Figure 8m) is a flavonoid isolated from a medicinal herb Scutellariabarbata(Figure 9a), downregulates the anti-apoptotic protein Bcl-2 and induces apoptosis by activating p53, which upregulates Bax to activate caspase 3 via the mitochondrial pathway 516. Treatment of HT-29 cells with luteolin (flavonoid, exist in fruits, vegetables and medicinal herbs) results in a loss of the mitochondrial membrane potential, an increase in mitochondrial Ca2+ level, upregulation of Bax, downregulation of Bcl-2, release of cytochrome c from the mitochondria to the cytosol and an increase in the levels of the active forms of caspase-9 and caspase-3 517. Treatment of Caco-2 CRC cells with extra virgin olive oil (rich in hydroxytyrosol and oleuropein) miR-23a and miR-301a, which were predicted to target type 1 cannabinoid receptor (CB1) in colon cancer 518, important implications in chemoprevention. Gambogic acid (Figure 8n), a xanthonoid extracted from the resin of Garcinia hanburyi (Figure 9b) inhibits HT-29 proliferation via induction of apoptosis 519. Walnuts (genus Juglans) have been shown to suppress colon cancer in mice models through the decreased expression of miR-1903, miR-467c, and miR-3068, as well as the increased expression of miR-297a in athymic nude mice injected subcutaneously with HT-29 CRC cells 520. Also, Aggarwal et.al, 2013 listed phytochemicals like garcinol, gossypol, gossypin, guggulsterone, indole-3-carbinol, morin, naphthoquinone, nimbolide, noscapine, oleandrin, piperine, piceatannol, pinitol, plumbagin, pomegranate, retnoids, honokiol, sesamin, silymarin, simvastatin, terpenoid, thymoquinone, tocotrienol, triptolide, ursolic acid, withanolides, xanthohumol, and zerumbone 458 having potentials in CRC. Seaweeds like U. pinnatifida175, 401, 521, 522, 523, 524, 525, 526, 527, Saccharina latissimi (9c) 528, Fucusvesiculosus176, 295, 529, 530, Sargassum hemiphyllum (Figure 9d) 531, 532, 533 have proven efficacy in this situation. Also, Algae derived astaxanthin 534, 535, 536, 537, 538, 539, 540, fucoxanthin 541, 542, 543, 544, 545, lutein and zeaxanthin 546, 547, 548, 549, polyphenols 550, 551, 552, 553, 554 shown individual excellence.
Figure 8. Plant derived biomolecules studied in colorectal cancer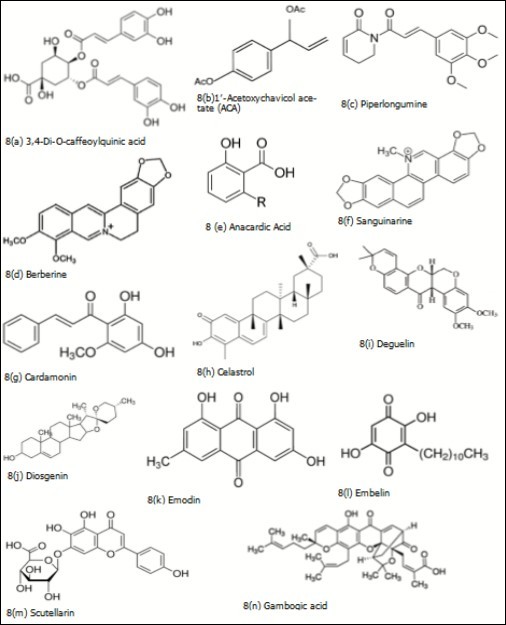
Figure 9(a). Scutellaria barbata (Source: Strictly Medicinal Seeds)
Figure9(b). Garcinia hanburyi (Source: Vitamin Supplement Ingredients Information)
Figure 9(c). Saccharina latissimi (Source: Nature Picture Library Print Store)
Figure 9(d). Sargassum hemiphyllum (Source: natural-history.main.jp)
Other Bioactive Non-Plant Compounds
Caffeic acid phenethyl ester (Figure 10a) is a central active component of propolis from honeybee hives. Propolis is a well-known health supplement that is extremely popular in Australia and New Zealand. It is constantly marketed in Japan with sales exceeding US$300 million/year 555. It can impart strong antimitogenic activity in lung cancer, breast cancer 556 and apoptosis in colon cancer 557. Peripheral neuropathy is a common side effect of many chemotherapeutic agents including paclitaxel. Poor nutritional status and obesity increase the risk of paclitaxel induced neuropathy 558. PEGylated liposomes of paclitaxel were successfully developed and demonstrated reduced neurotoxicity in-vitro in neuronal cells and prevented development of peripheral neuropathy in-vivo 559. Glutathione 560 and gallic acid 561 may ameliorate paclitaxel-induced neuropathic pain. Di(2-ethylhexyl) phthalate (DEHP) (Figure 10b), estrogen receptor alpha (ERα) agonist due to its ability to interact with ERα and promote the cell proliferation of ERα-positive breast cancer cells, significantly protected MCF-7 cells against the genotoxicity of camptothecin 562. Actinomycin D obtained from various Streptomyces strains decreases Mcl-1 expression in lung cancer cells 563, induces p53-independent cell death in leukemia 564, synergistically suppressed multiple metastasis of TRAIL-resistant colon cancer in the liver with soluble TRAIL gene 565. Hyperthermic intraperitoneal chemotherapy (HIPEC) with cisplatin and mitomycin C (Figure 10c) (obtained from Streptomyces caespitosus) is the only protocol to demonstrate an adjuvant HIPEC benefit in colorectal cancer patients at high risk for peritoneal failure and an alternative to high-dose and short-term oxaliplatin 566. 5-FU plus mitomycin remains the preferred chemotherapy in most patients with anal cancer 567. Bleomycin (Figure 10e) is an antibiotic complex of several glycopeptides derived from Streptomyces verticillus, gained FDA approval in July 1973. The extract from Streptomyces sp. MUM265- represents a valuable bioresource of bioactive compounds for the future development of chemo-preventive agents, with particular promise suggested for treatment of colon cancer 568. Bleomycin is an indispensable antineoplastic agent for the treatment of germ cell tumors and lymphomas. Pirfenidone (Figure 10d) (novel orally available antifibrotic drug approved by the FDA in 2014) is currently the only approved therapy for idiopathic pulmonary fibrosis (IPF), considered as a salvage drug for refractory cases of bleomycin-induced lung injury 569. In a similar study, Yu et.al, 2019 reported EZY-1 (16-amino-acid peptide was isolated from Eucheuma) can inhibit the IPF induced by bleomycin 570.
Figure 10. Biomolecules from non-plant origin studied or used in different types cancers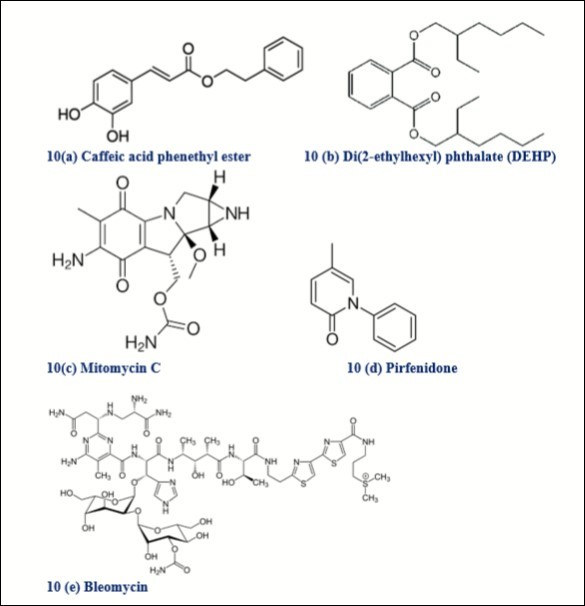
Alternative Therapies and Mind-Body Interventions
According to the Global Health Observatory Report from the WHO, insufficient physical activity is the 4th leading risk factor for mortality. Participation in 150 minutes of moderate physical activity a week or its equivalent is estimated to reduce risk of breast and colon cancer by 21%–25% 571. Approximately 50% of all leukemia, lymphoma, colorectal- and breast cancer patients are affected by CIPN. Sensorimotor training (SMT) or whole-body vibration (WBV) can reduce the symptoms of CIPN and attenuate motor and sensory deficits 572. Hypnosis, music (Figure 11) and relaxing video reduced anxiety and pain associated with colonoscopy and need for sedation during colon cancer screening 573, 574, 575, 576, 577, 578, 579, 580, 581. Impaired cognitive function, change in brain metabolism and change in brain structure are associated with cancer treatment. CBT moderately improved anxiety and depression in patients with early-stage breast cancer 582, significantly improved tumor associated fatigue levels after 8 weeks 583, improved QoL 584, improved cognitive function 585, improved insomnia 586, 587, reduced fear of cancer recurrence 588 and most importantly, reduced pain and distress 589. Mindfulness-based approaches and hypnosis reduced demonstrated efficacy in reducing anxiety and depressive symptoms. 40% to 50% CRC patients reported fear of cancer recurrence, tends to increase around the time of scans or other testing for recurrence 590. Also, CRC patients have unique psychosocial needs (e.g., isolation, embarrassment) related to altered eating and bowel habits and sexual dysfunction that warrant clinical attention 591. Acceptance and commitment therapy, meta-cognitive therapy, and mindfulness-based therapies emphasize mindfulness, acceptance, cognitive flexibility, and patient values changes in self-efficacy or confidence in using coping skills targeted by the intervention, acceptance of unwanted thoughts and feelings, or enhanced social support as well as physiological mechanisms (e.g., decreased arousal to negative thoughts and feelings about cancer) 592. Combined CBT/GET improves fatigue and functional outcomes for a subset of patients with post-cancer fatigue in breast or colon cancer 593. CBT intervention has the potential to ease acute anxiety during the often-challenging re-entry phase and to prevent the development of chronic, debilitating, and costly anxiety 594. Physical activity interventions also reduce depressive and anxiety symptoms in breast cancer survivors 595. Telehealth approaches may improve access to mental health resources especially for those with limited online access or lack of online skill 596. Yoga has a solid effect on cancer-related fatigue in patients with breast cancer 597. It is one of the most prevalent complementary therapies used in breast cancer care, seems to be as effective as other exercise modalities for improving the QoL of women with breast cancer 598. Wei et.al, 2019 reported significant improvement in lymphedema status, range of shoulder motion and spinal mobility after an 8-week yoga intervention 599. Although, yoga could not improve HRQoL in patients with colorectal cancer 600 but research supports that yoga is a promising intervention for reducing fatigue and sleep disturbances in this patient group 601.
Figure 11. Music Therapy: Pain or Distress Management 619. In the UK, music therapists are trained to master’s level and are registered with Health and Care Professions Council as allied health professionals. Aristotle recognized the innate ability of melodies to surpass “feelings such as pity and fear, or enthusiasm,” and thus “heal and purify the soul.” The Greeks identified Apollo as the father of both healing and music, alongside his many other accolades (as God of light, sun, truth, prophecy, plague and poetry).
Dyadic yoga may offer effective relaxation techniques for lung cancer patients and their caregivers who were undergoing an extreme stressor in addition to the cancer experience 602, feasible and beneficial for patients having toxic thoracic radiography 603. Approximately 20% of breast cancer survivors develop breast cancer-related lymphedema (BCRL) 604. Acupuncture is safe and effective at reducing breast cancer-related lymphoedema in patients after breast cancer treatment 605 and managing joint stiffness. Acupuncture use among breast cancer patients in the US is currently as high as 16% to 63% 606. At the current time breast cancer related lymphedema is incurable but well manageable by a number of physical therapy modalities, especially complete decongestive therapy (CDT) 607. Obesity is a factor that deteriorates the CDT efficacy. Early treatment, before developing fat accumulation and fibrosis, must be primary goal in the treatment of BCRL 608. The first “intensive treatment” phase aims to decongest the swollen arm through two or more weeks of daily therapist-delivered treatment including multi-layer compression bandaging and manual lymph drainage (MLD). This is followed by a “maintenance” phase of patient self-treatment, with compression usually in the form of hosiery 609. The mindfulness component may enhance the positive impact of exercise on cognitive function in breast cancer 610, 611. Tai Chi is accessible to most people and does not require special facilities or expensive equipment 612. Healthier dietary choices were the most frequently reported change already made by people affected by CRC, followed by increased physical activity, stress management, quitting smoking and alcohol and therapies including meditation, tai chi, and naturopathy 613. Greater shoulder muscular strength was significantly associated with better functional well-being in breast cancer survivors with TC Qigong training 614. Massage with or without aromatherapy have been suggested by a few studies in breast cancer to ameliorate anxiety and other symptom relief 615 and immunologic state 616 that needs further investigation. TENS was found valuable in lung cancer patient underwent standard posterolateral thoracotomy 617. Animal-assisted activities (Figure 12) has potential benefit children with cancer because pediatric oncology patients often suffer from distress due to physical examinations, venipuncture, chemotherapy infusions, spinal taps, surgery, hospitalization, pain, fear of medical procedures, unpleasant physical symptoms, uncertainty, and worry about death 618.
Figure 12. Hospitalized kid on animal visit 620. Understanding whether AAA is safe and effective for pediatric cancer patients is critical, especially because of concern about infection in immunosuppressed persons. Conducting AAA research in pediatric oncology requires understanding current regulations and variations in practice. Knowledge of regulations helps us understand elements required for intervention protocols (e.g., hand-cleaning), whereas knowledge of practice variation can help us identify research opportunities.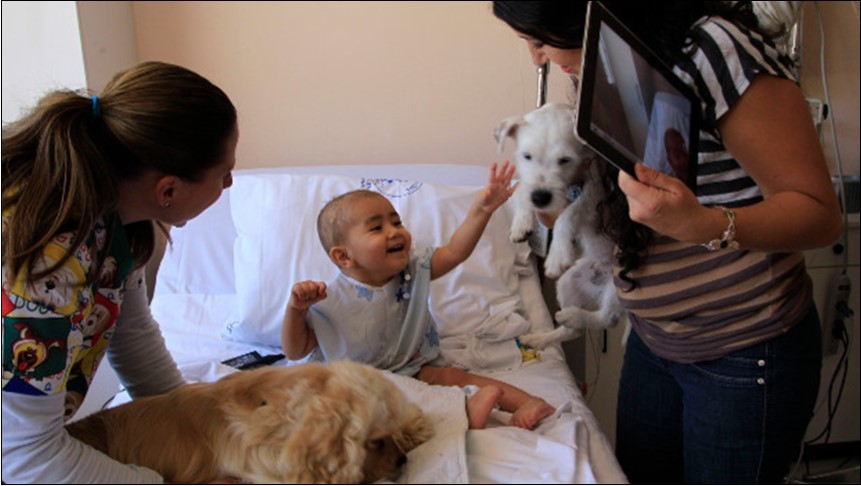
Conclusion
The journey from diagnosis to treatment of cancer affects the patients' lives in a variety of ways. Debilitating symptoms arising both due to disease and its treatments consistently hamper their QoLs. CAM treatment aims to restore body’s ability to protect, regulate and heal itself. Since almost 50% of existing medicine is derived from plants, it is clear that natural sources, especially plants can be investigated for effective medicines in cancer treatment. These data can equip providers and patients with the information they need to have informed conversations regarding non-drug approaches for treatment of specific cancer conditions. The use of CAM by cancer patients is becoming widespread. This is a reflection of the many needs and concerns that are currently not being met by conventional medical practice. Significant proportions of cancer patients in developed countries use complementary therapies as adjuncts to conventional symptom management to improve their QoL. India’s indigenous systems of medicine, such as Ayurveda, Siddha, and Unani, are more than 5,000 years old, and in rural areas, the Indian population has relied heavily on these practices, particularly Ayurveda. In addition, CAM has the potential for the primary and secondary prevention of cancer through counselling on healthy lifestyle, nutrition and supporting the human power of ‘salutogenesis’ throughout life. The lack of communication about CAM use may be due to fear of a negative response, physicians being perceived as not supportive nor helpful, or physicians and patients having differing views about CAM. Discussions of CAM therapies may have additional benefits for the patient-provider relationship, as studies have shown it indicates use of participatory decision-making, patient-centered communication, and thus greater patient satisfaction, not only in cancer care but also in other arenas of healthcare.
Financial Disclosure or Funding
N/A
Informed Consent
N/A
Author contributions
N/A
Acknowledgements
I’m thankful to Dr. Alexandra McCarthy, The University of Auckland, New Zealand for her valuable time to audit my paper and for her thoughtful suggestions. I’m also grateful to seminar library of Faculty of Pharmacy, University of Dhaka and BANSDOC Library, Bangladesh for providing me books, journal and newsletters.
Abbreviations
Mortality and Prevalence (GLOBOCAN)
Chronic Myeloid Leukemia (CML) Leukemia & Lymphoma Society (LLS) cleaved Poly (ADP-ribose) Polymerase (c-PARP) Immunogenic Cell Death (ICD) Tyrosine Kinase Inhibitors (TKI) Bruton Tyrosine Kinase (BTK) Glycogen Synthase Kinase-3β (GSK3β) Myeloid cell leukemia-1 (Mcl-1) 'Tamm-Horsfall Protein 1' (THP-1) mammalian target of rapamycin (mTOR) Src-Homology 2-Containing Inositol-5-Phosphatase 2 (SHIP2) Phosphatase and Tensin homolog (PTEN) Plant Homeodomain Finger 6 (PHF6) T-cell acute lymphoblastic leukemia (T-ALL) NADPH Quinone Dehydrogenase 1 (NQO1) Nuclear factor erythroid 2-related factor 2 (Nrf2) Rb tumor suppressor protein (pRb) c-Jun NH2-terminal kinase (JNK) Human Leukocyte Antigen (HLA) Epigallocatechin Gallate (EGCG) BReast CAncer gene-1 (BRCA-1) Chemotherapy-induced peripheral neuropathy (CIPN) Sensorimotor Training (SMT) Whole Body Vibration (WBV) Neoadjuvant Chemotherapy (NCT) Di(2-ethylhexyl)phthalate (DEHP) Programmed Death-Ligand 1(PD-L1) cyclin-dependent kinase inhibitor 1A gene (CDKN1A) Transcription Factor AP-2α (TFAP2A) Serine-threonine kinase receptor-associated protein (STRAP) Maternal embryonic leucine zipper kinase (MELK) COP9 signalosome subunit 6 (CSN6) TNF-related apoptosis-inducing ligand (TRAIL) Idiopathic Pulmonary Fibrosis (IPF) Graded Exercise Therapy (GET) Manual Lymph Drainage (MLD) Transcutaneous electrical nerve stimulation (TENS) Animal-Assisted Activities (AAA)