Abstract
Background:
Chest wall deformities may be managed with skeletal manipulation, which risks life-threatening complications. Custom-made prostheses are a less invasive surgical option, manufactured from silicone elastomer using 3D computed tomographic reconstruction and 3D-printed thoracic models.
Methods:
All patients undergoing custom-made implants between January 2010 and March 2017 were identified from the prosthetic department records. A retrospective review of the clinical records was performed. Mean follow up period was 1.8 years. A comparison was made with our earlier results from 1995 to 2009.
Results:
Twenty-six patients underwent insertion of custom-made implants for chest wall deformity. Pectus excavatum was present in 50% (n=13), and Poland syndrome 42% (n=11). All 11 female patients underwent 3D reconstruction and 3D printed models, and 3 of 15 males. Four underwent simultaneous bilateral breast augmentation, and three had staged breast augmentation. Seroma occurred in 27% (n=7), and hypertrophic scar in 12% (n=3). The reoperation rate was 23% (n=6), including autologous fat graft in two patients. Surgical suction drains were used in 42% (n=11) patients, of whom 36% (n=4) developed seroma, compared with 17% (n=2) of those without drains (p=0.08).
Conclusions:
Custom-made prostheses are an effective and safe option for patients with chest wall deformities. The majority have a short postoperative inpatient stay (81%) and are satisfied with the outcome (77%). Seroma was the commonest complication (27%), and drains did not reduce seroma risk. Single dose intravenous antibiotic prophylaxis is adequate. A minority of patients opt for further aesthetic procedures.
Author Contributions
Academic Editor: Marcos Gomez Ruiz, Hospital Universitario Marques de Valdecilla, Santander, Spain.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2019 Dallan P Dargan, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Adult patients with chest wall deformities often present with aesthetic concerns. Often the concern is asymmetry of the chest wall or sternal concavity, with implications for self-confidence and social situations. In females, there is often associated breast hypoplasia or asymmetry, and breast augmentation may also be requested.
The anatomical aspects of chest wall deformities, classifications, genetic and histological features have been described1. Poland syndrome and pectus excavatum are the most common chest wall deformities. Poland syndrome is treated with a range of soft-tissue reconstructive techniques2. Mild cases of pectus excavatum may benefit from fat transfer, or in female patients with breast implants placed more medially. Severe symptomatic pectus excavatum cases are treated with skeletal reconstruction as reported mainly in the paediatric surgical literature3. However, skeletal reconstruction has significant post-operative complications including pneumothorax and haemothorax4, cardiac perforation or massive haemorrhage5,6,7,8, and the deformity may recur. Severe non-symptomatic cases of pectus excavatum present a reconstructive challenge especially in female patients with breast hypoplasia. A customised pre-sternal prosthesis may ameliorate the perceived anomaly sufficiently to provide a satisfactory result without the potential risks of skeletal remodelling.
This study outlines the results of 26 patients who had customised prostheses for chest wall deformities in our unit performed during 2010-2017. This article presents an updated technique for manufacture of custom prostheses of the chest wall and the variations in aspects of surgical technique which are utilised. In particular, the use of 3D CT scanning and 3D printing has broadened the patient cohort for which this technique may be suitable. A comparison with our previous 28 patients in 1995-2009, which includes the 13 cases published in 20089, is also provided.
Methods
The clinical records of all patients undergoing chest wall deformity surgery with a custom-made chest prosthesis between January 2010 and March 2017 were retrospectively reviewed from the prosthetic department records. The type of chest wall deformity, use of computer aided design, incision site, prophylactic antibiotics, surgical drains, length of stay, complications, and subsequent procedures were evaluated. The outcomes were compared with those collected at our institution between 1995 and 2009. Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines10 were adhered to in the reporting of this study.
Manufacturing Technique
Patients referred for consideration of custom-made chest wall implant underwent an initial consultation with the prosthetists. Three-dimensional computed tomography (3D CT) was performed to assess the skeletal features of the chest wall, using Materialise Mimics software (Materialise, Leuven, Belgium) (Figure 1 a, b, c). A model of the bone structure was then created using a 3D printer, Objet Eden 260vs (Stratasys, Eden Prairie, MN, USA). Alginate was poured onto the 3D model to create an alginate impression (Alginate Colour, DE Healthcare Ltd, Kingthorpe, UK), and a plaster model of the chest wall created from this alginate impression. In lean patients with pectus excavatum, or in Poland syndrome with soft tissue deformity alone, an alginate impression of the whole chest was taken without CT, and the plaster model of the chest created from this. Once a plaster or 3D-printed model was available, a wax prototype of the prosthesis was manually crafted on the plaster model, using carving tools and a blowtorch.
Figure 1. a) Anterior portion of axial computed tomography image of thorax, demonstrating pectus excavatum, with portion used for reconstruction highlighted in yellow. b) and c) three dimensional computer-aided reconstruction of the chest wall model.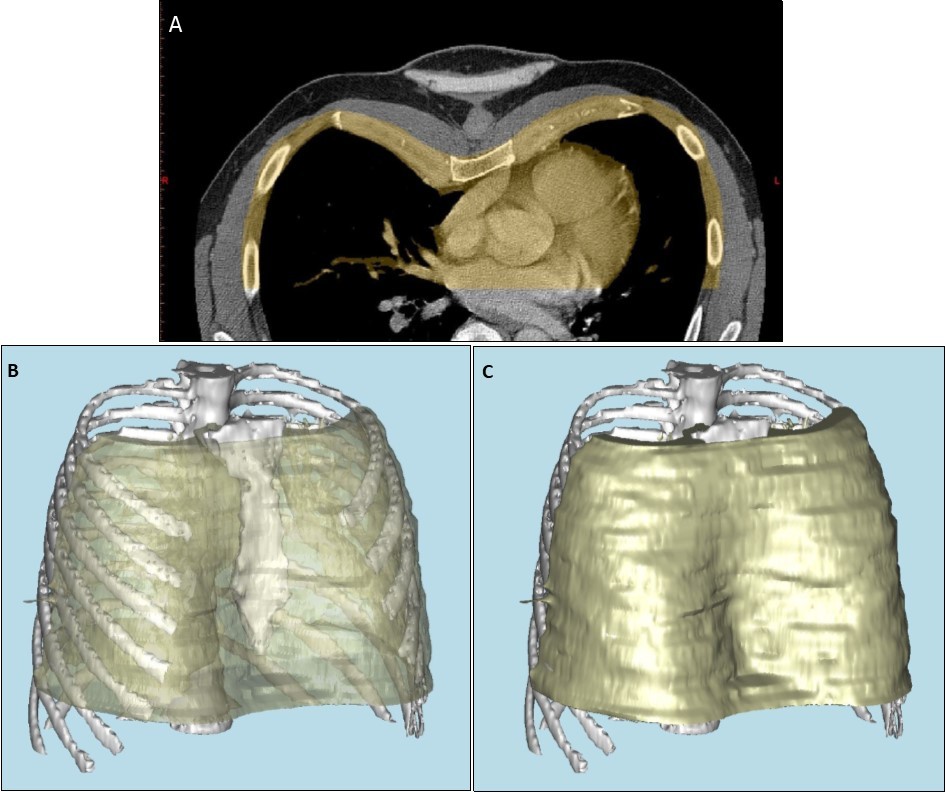
The patient was then invited for a further consultation, together with the consultant surgeon. Patients were advised to bring a tight-fitting t-shirt to help assess the size and shape of the implant. The wax prototype was placed on the on 3D-printed model to aid discussion.
Once the wax model shape was finalised, a two-part mould was made in Crystacal® gypsum (Saint-Gobain Formula, Kutzhütte, Germany). The mould was made in a two-part metal flask to allow the silicone to be pressed under pressure without cracking or fracture of the plaster. The wax model was placed in wet plaster, smoothed and allowed to set, then separated using a separating medium such as Vaseline® (Unilever, London, United Kingdom), or Medi Mould (Polymed Limited, Cardiff, United Kingdom). The top half of the flask was filled with plaster. The flask was boiled to remove the wax (lost wax technique), leaving a plaster relief of the implant. The plaster surface was roughened with a 2mm drill burr in multiple places to discourage capsulation of the prosthesis within the body (Figure 2a and b). The model was separated using Medi Mould to prevent sticking of the plaster to the silicone.
Figure 2. a) and b) Mould created in the plaster flask, using the lost wax technique. Holes are present in the surface to roughen the implant and help prevent capsulation.
Once the two halves of flask were prepared, the silicone was mixed and inserted under pneumatic pressure using a silicone gun. MED-4805, an implantable medical grade silicone elastomer (Polymer Systems Technology Limited, High Wycombe, United Kingdom) was used to form the implant. The mould was clamped under pressure to remove excess silicone and air and placed in a conventional oven at 100°C for four hours, then removed and left to cool. Once cooled, the clamps were removed, and the mould opened. Any excess silicone is trimmed, and the implant washed with Fairy liquid (Proctor & Gamble, Cincinnati, OH, USA) and water to remove any debris. Holes were pierced in the implant again to help prevent capsulation and migration (Figure 3).
Figure 3. A custom prosthesis with holes pierced in the surface to prevent capsulation and migration.
Completed prostheses were reviewed, with the 3D chest wall model, by the consultant plastic surgeon (Figure 4). Once accepted, the prosthesis was sterilised at Synergy Healthcare (Preston, United Kingdom), at 134°C for three minutes. The packaged, sterilised prosthesis was returned to our institution, and opened during the operation. Implants were re-sterilised if past expiration date. The prosthetists and the 3D models were both available in the hospital on day of surgery to confirm orientation of implants.
Figure 4. Demonstration of the implant position on a reconstructed model of the thorax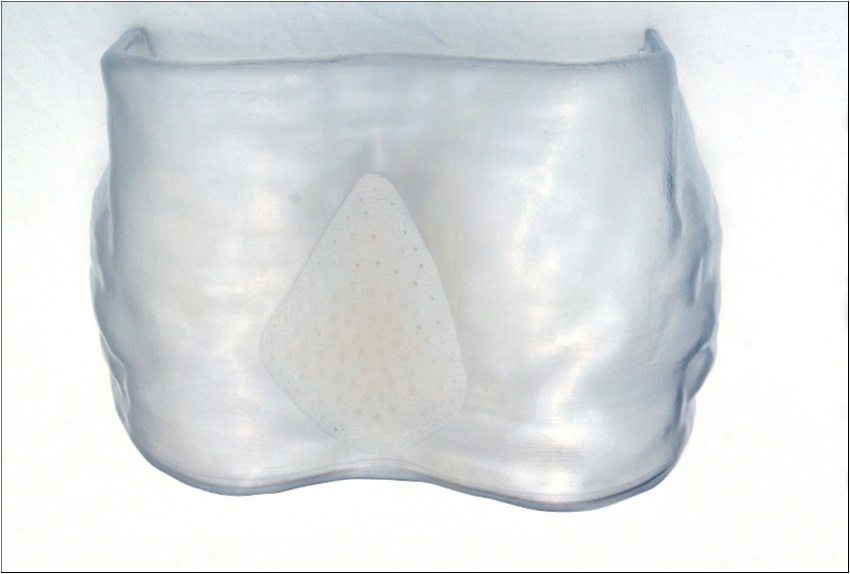
Statistical Analysis
Normally distributed categorical data was assessed using Fisher’s exact test. Microsoft® Excel® (Microsoft Corporation, Redmond, WA, USA) was used for statistical analysis. Statistical significance was set at p<0.05.
Results
A total of 39 individuals with chest wall deformities were identified who were referred to the prosthetic service for consideration of a custom-made implant during the study period (24 pectus excavatum, 13 Poland syndrome, 1 pectus carinatum, and 1 other chest wall deformity).
Thirteen patients had not undergone insertion of a custom implant by March 2017 and were excluded: five were awaiting implant surgery for pectus excavatum; one declined the custom prosthesis as breast augmentation funding had not been secured; two had opted for fat transfers without an implant; two had not attended appointments; and one was deferred for pre-operative cardiology investigation.
The remaining 26 patients underwent insertion of a custom-made implant during the period studied (Table 1). Mean follow-up was 1.8 years (13 days to 8.1 years); six patients had less than 60 days follow-up due to failure to attend clinic appointments. 50% (n=13) had pectus excavatum and 42% (n=11) had Poland syndrome, 4% (n=1) had pectus carinatum with multiple past episodes of rib fractures, and 4% (n=1) had a chest wall deformity with thoracic scoliosis and unilateral breast hypoplasia. 54% (n=14) underwent 3D CT, and 3D printed reconstruction, including 11 females and 3 males. 46% (n=12) had plaster models created directly from the patient’s chest, all twelve of whom were male. All 26 patients had wax models made of the prosthesis. 23% (n=6) had previous chest wall or breast procedures, including two with previous custom implants, 8% (n=2) had previous surgery for gynaecomastia, and 8% (n=2) were pectus excavatum patients with previous chest wall reconstruction under the cardiothoracic team (one Ravitch procedure11, one Nuss pectus bar12).
The distribution of implant locations were: 31% (n=12) pre-sternal; 31% (n=8) right chest wall; 23% (n=6) left chest wall. Four female patients with pectus excavatum underwent simultaneous bilateral breast augmentation with mammary prostheses in addition to the custom-made chest prosthesis. Three patients required a subsequent staged breast augmentation (two unilateral and one bilateral).
Incision Site and Plane of Prosthesis Insertion
The majority of incisions (50%, n=13) were inframammary, although a variety of approaches were used (Table 1). Regarding the anatomical plane for the implant pocket, details were available for 22 of 26 patients. Custom prostheses in were placed in the submuscular plane, or supra- or sub-fascial. In all four cases with simultaneous bilateral breast augmentation, the mammary prostheses were placed in a subglandular plane. Most patients had a short hospital stay, and 81% (n=21) were discharged by the second post-operative day.
Follow-up and Complications
Mean follow-up time was 641 days (range 13 days to 8.1 years). 15% (n=4) had less than 30 days follow-up, due to failure to attend clinic appointments. 62% (n=16) developed one or more complications; excluding seromas and hypertrophic scars the complication rate was 31% (n=8). Post-operative complications are outlined in Table 1. The incidence of seroma was 27% (n=7) ((n=5) in pectus excavatum (n=2) in Poland syndrome). 12% (n=3) had seromas which required serial aspiration, and 15% (n=4) had seromas which resolved spontaneously.
Table 1. Comparison of cohorts and complication rates in patients undergoing custom made chest wall implants between 1995-2009 and 2010-2017.| Factors evaluated | 1995-2009*(n=28) | 2010-2017(n=26) | 1995-2017 (n=54) |
| Male : female ratio | 21:7 | 15:11 | 36:18 |
| Age range | 17-58 | 17-51 | 17-58 |
| Pectus excavatum | 18 | 13 | 31 (57%) |
| Poland syndrome | 7 | 11 | 18 (33%) |
| Poland and pectus excavatum combined | 1 | 0 | 1 (2%) |
| Other | 7 | 2 | 9 (17%) |
| Surgical incision** | |||
| Unilateral inframammary | - | 8 | - |
| Bilateral inframammary | - | 5 | - |
| Inferolateral | - | 2 | - |
| Axillary | - | 3 | - |
| Transverse upper epigastrium*** | - | 1 | - |
| Combined medial and lateral for severe Poland deformity | - | 1 | - |
| Existing lateral chest scar | - | 1 | - |
| Lateral | - | 1 | - |
| Unknown | - | 4 | - |
| Minor complications | |||
| Seroma | 7 | 7 | 14 (26%) |
| Hypertrophic scar | - | 3 | 3 (6%) |
| Complications requiring further surgery | |||
| Replacement of implant due to movement | 2 | 0 | 2 (4%) |
| Replacement of implant due to insufficient size/asymmetry | 1 | 0 | 1 (2%) |
| Additional implant insertion due to asymmetry | 0 | 1 | 1 (2%) |
| Removal due to recurrent seroma | 1 | 1 | 2 (4%) |
| Removal due to generalised lymphadenopathy | 0 | 1 | 1 (2%) |
| Asymmetry or contour defect requiring autologous fat graft | 0 | 2 | 2 (4%) |
| Trimming of palpable inferior edge of implant after migration | 0 | 1 | 1 (2%) |
| Reposition of breast implants and modifications of chest wall implant for symmastia and right breast implant migration | 0 | 1 | 1 (2%) |
Further Surgery
A second admission for surgery was required in 23% (n=6) patients. 4% (n=1) developed generalised reactive lymphadenopathy within days of implant insertion which required urgent removal of the implant. Symmastia of breast prostheses and communication with the custom prosthesis pocket occurred in 4% (n=1), requiring implant repositioning and refashioning of the suprasternal pocket in pectus excavatum. 4% (n=1) underwent insertion of an additional prosthesis to correct asymmetry. 4% (n=1) underwent staged exchange of implants for asymmetry and serial seroma, and 8% (n=2) required autologous fat grafting following the customised prosthesis to improve contour or symmetry, one of whom required serial fat graft procedures. 8% (n=2) developed implant migration, of whom one required elective trimming of the implant, and the other settled spontaneously. In total, asymmetry was noted in 15% (n=4). One patient with asymmetry did not require intervention. No direct cardiorespiratory or rib complications were observed in this series, nor was silicone leakage noted.
Prophylactic Antibiotics
Intravenous antibiotic prophylaxis was used in most cases (Table 2), although some received additional post-operative oral prophylaxis. A suspected superficial surgical site infection was noted in 4% (n=1), following perioperative prophylactic intravenous and oral prophylactic antibiotics, and a course of post-operative oral flucloxacillin and phenoxymethylpenicillin was prescribed.
Table 2. Use of antibiotic prophylaxis in patients undergoing customised implants in 2010-2017| Intravenous antibiotics at induction | Number of patients (n=26) | Prophylactic post-op. oral antibiotics | Number of patients (n=26) |
| Cefuroxime | 11 (42%) | Cefalexin | 3 (12%) |
| Co-amoxiclav | 6 (23%) | Co-amoxiclav | 5 (19%) |
| None | 4 (15%) | None | 14 (54%) |
| Not recorded | 5 (19%) | Not recorded | 4 (15%) |
Surgical Drains
Surgical drains (closed-suction) were used in 42% (n=11), not used in 46% (n=12), and drain status was unrecorded in 12% (n=3). Of those with drains, a higher proportion developed seroma, 36% (n=4), compared with 17% (n=2) of the 12 patients without drains, although this was not statistically significantly different on Fisher’s exact test (p=0.08).
Patient Satisfaction
A subjective assessment of patient satisfaction was obtained at completion of follow-up from the clinical records of outpatient reviews for 22 of the 26 patients. 77% (n=17) were satisfied with the outcome, 19% (n=5) had some ongoing concerns recorded including 8% (n=2) regarding residual asymmetry, 8% (n=2) reported some discomfort or pain, and 4% (n=1) with implant migration.
Discussion
The number of patients undergoing custom-made implants annually has doubled from approximately two per year (28 implants in 15 years) to four (26 in under 8 years), although complication rates appear relatively unchanged in our institution. Seroma incidence (27% (n=7)) following insertion of chest wall implant, is considerably higher than in bilateral breast augmentation, estimated at 2.8% (15 of 539 patients), with smoking, body mass index and pocket position potentially related to breast augmentation seroma formation in the latter13. A similar series of 26 patients undergoing custom implants, by Soccorso et al. in Birmingham UK, included 18 individuals with pectus excavatum and 6 with Poland syndrome, of which six were adolescents, and reported only two seromas14. The largest series of customised implants for pectus excavatum from the team by Chavoin et al. in Toulouse15 demonstrated a significant improvement in malformation correction following the implementation of 3D CT and computer-aided design. Social and emotional improvements were recorded following prosthesis insertion (using the Short Form 36 assessment16) and seroma was detected clinically in every case15.
The variation in incision types and the plane of pocket formation for this group reflects the variety of previous surgeries, deformity types and implant shapes which are customised for the chest wall in these individuals. In a cohort of 63 patients with Poland syndrome2 classified cases into simple (55 cases) or complex (8 cases), of whom 42 opted for reconstruction. Complex Poland syndrome was defined including features such as a smaller hemithorax, ipsilateral upper limb shortening and brachysyndactyly. Males were treated mainly with latissimus dorsi pedicled transfer, and females with a variety of combinations of procedures. Only five patients in this series were treated with custom prostheses, and all five had removal of implants within six months due to discomfort and contour difficulties2.
Antibiotic Prophylaxis
A retrospective review of antibiotic prophylaxis in prosthetic augmentation of 3256 breasts17 found a lower incidence of superficial and periprosthetic infections in patients who had received a single dose of intravenous antibiotics, compared with those who had received a combination of intravenous and oral antibiotics. Antibiotic prophylaxis according to the published antimicrobial guidelines for our institution18,19 for implants in plastic surgery patients are a single dose of 1.5 grams of intravenous cefuroxime at induction of anaesthesia, without prophylactic oral antibiotics. The absence of infective complications in this series, and the low rate of infection in published series2,9,15,20, suggests that post-operative oral antibiotic prophylaxis is not necessary for this cohort. Margulis et al. (2006) from Israel reported good outcomes from seven patients with custom implants for pectus excavatum, and used cefazolin 1 gram at induction, and two suction drains for each patient20. Drain use in prosthetic breast augmentation to reduce the incidence of haematoma formation or early capsule formation, has shown no association with infection, at least with cohesive gel implants21.
Limitations
It is possible that other superficial infections and minor seromas may have occurred and been under-reported. Regarding drains, selection bias may account for the increased seroma rate in those with drains: the more extensive procedures may have necessitated surgical drains. The duration of follow-up for some participants may not be sufficient to detect incidences of capsular contracture or leakage, and future studies should aim to report long term outcomes.
Conclusions
Most patients with pectus excavatum and Poland syndrome who are referred with chest wall deformity are suitable for customised chest wall prosthesis. The majority have a short postoperative inpatient stay (81%) and are satisfied with the outcome (77%). 3D CT and 3D printed reconstruction was beneficial for all 11 female patients in this cohort, and 3 of 15 males. Surgical drain use was associated with a higher incidence of seroma formation, although this did not reach statistical significance, it suggests that drains may not be necessary in many cases. A single dose of intravenous antibiotic as prophylaxis at induction appears sufficient for custom-made implants. Seroma was the commonest complication (27%), and some patients requested further aesthetic procedures to improve the chest wall or breast appearance.
Acknowledgements
Gina Woolley FdSc, Jane McPhail BSc (Hons), Elaine Goldsworthy BSc (Hons), for presenting the 1995-2009 data at the International Anaplastology Association congress in Paris in 2009, and Nina Sykes and Anthony Simpson of the Prosthetics department in Whiston Hospital for their assistance with the changes to the prosthesis manufacturing technique.
Mr P McArthur, Mr A Iqbal, Mr K Hancock,Mr R Pritchard-Jones, Mr A Benson, and Mr I James, consultant surgeons in Mersey Regional Burns and Plastic Surgery Unit during the periods reviewed, for contributing patients to the study.
Funding
None