Abstract
Background
Klippel-Feil Syndrome (KFS) is described as the clinical triad of short neck, low posterior hairline, and limitation of neck mobility.
Objective
Topresent our clinical experience with KFS patients who also had adjacent segment disease (ASD) and to propose a novel classification system for these patients.
Methods
This retrospective study was performed in the neurosurgery department of our tertiary care center. Data were gathered using the medical records of 22 KFS patients (10 males, 12 females) with ASD. Diagnosis was confirmed with imaging modalities including X-ray, computerized tomography, and magnetic resonance imaging. Clinical and radiological findings as well as therapeutic outcomes were noted.
Results
The average age of our series was 56.09 (range: 41 to 67) years. The operative technique was selected as for our novel “Yilmaz-Yucesoy Classification System”. Accordingly, one patient (4.54 %) received non-surgical treatment (Yilmaz-Yucesoy Grade 1), six cases (27.27 %) underwent anterior cervical arthroplasty, seven patients (31.81 %) underwent anterior cervical discectomy or corpectomy and fusion with cervical cage and plate (Yilmaz-Yucesoy Grade 3). Eight patients (36.36 %) with cervical spinal instability had anterior cervical discectomy or corpectomy and fusion with cervical cage and plate (Yilmaz-Yucesoy Grade 4). No mortality or remarkable complications were detected.
Conclusion
Appropriate and timely recognition and classification of patients with KFS and ASD based on our newly proposed “Yilmaz-Yucesoy Classification System” yielded promising treatment outcomes. However, further prospective, randomized, controlled trials are warranted on larger series to validate our preliminary results.
Author Contributions
Academic Editor: Chittaranjan Routray, University of North Dakota School of Medicine and Health Sciences Idaho College of Osteopathic Medicine, United States.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2021 Murat Yilmaz, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Klippel-Feil Syndrome (KFS) is a relatively rare disorder that was initially described by Maurice Klippel and Andre Feil in 1912.1 Its prevalence has not been studied in a large series; however, it may occur in every 40,000 newborns with a slight predominance of females.1, 2, 3, 4 It occurs due to the failure or incomplete and inappropriate segmentation of somites that occurs during embryogenesis between 3 to 8 gestational weeks. In other words, there is a failure of normal segmentation or formation of the cervical somites.1, 5, 6
The vast majority of KFS cases are sporadic, but there are several genetic types of KFS.7, 8, 9 KFS and ocular anomalies associated with Wildervanck syndrome may have X-linked inheritance.10 Congenital fusion of the cervical vertebrae may be a possible risk factor for the occurrence of degenerative cervical myelopathy (DCM).6
Conventionally, KFS is described as having the clinical triad of a short neck, low posterior hairline, and restriction of neck motion, which may be attributed to the fusion of cervical vertebrae.1,3,5 However, many patients do not typically display the conventional triad of KFS.
The vertebral column is comprised of fibrocartilagenous intervertebral discs and ossified vertebral units. Congenital malformations of the vertebral column involve kyphosis, scoliosis, spina bifida, and KFS.11 Klippel-Feil Syndrome can occur in a wide variety of phenotypes, its impact on the quality of life depends on the number of segmentary involvement which accompanies the deformity. 1, 3, 4
In general, cervical spinal deformity increases with the increasing number of segments involved and these patients are more likely to present with concomitant congenital problems.6 Patients with KFS are under risk for acceleration of degenerative changes and hypermobility in segments adjacent to the spinal fusion.6
The relationship between KFS and degenerative cervical myelopathy (DCM) has not been well defined, despite reports of segmental hypermobility and predisposition to KFS patients' degenerative changes in the cervical region. Since the patients with spinal fusion may be at risk for adjacent segment disease, there is a need for investigation of KFS patients for the tendency to develop neurological sequelae due to the degeneration of the neighboring segment.4
In this study, we aimed to share our experience with 22 patients who were treated for adjacent segment disease (ASD) associated with KFS. We describe the clinical and radiological findings together with our treatment outcomes and we propose “Yilmaz-Yucesoy Classification System” as a novel grading system for KFS with ASD based on our data along with a brief review of current literature.
Patients and Methods
Study Design
This retrospective study was performed after the approval of the local institutional review board (2020/15-52). Data were extracted from the medical files of a total of 22 KFS patients with concomitant ASD who were treated in the neurosurgery department of our tertiary care center between October 2004 and August 2019. The diagnosis of KFS was established based on the clinical data and radiological findings derived from plain radiography, computed tomography, (CT) and magnetic resonance imaging (MRI). Even though some patients already had direct cervical X-rays and CT images on admission, all patients underwent MRI to obtain a detailed view of the spinal cord. All patients revealed typical symptoms including neck pain, gait disturbance, numbness and weakness in the extremities, and pathological reflexes. All of these patients underwent follow-up for at least one year, and we have focused on the clinical and radiological outcomes. Patients with idiopathic fusion and acquired fusion were excluded. Patients with fused cervical vertebrae who did not demonstrate the classical wasp-waist sign were considered as either idiopathic or acquired fusion. The wasp waist sign (anterior-posterior narrowing) is an indicator of congenital spinal fusion, which is also known as block vertebra. In other words, the wasp waist sign is the narrowing of the dimensions of the bone at the level of the intervertebral disc. Moreover, the heights of the disc and vertebral body complex are the same as a normal vertebra in case of congenital fusion. In contrast, the height of the disc is lost in patients with acquired spinal fusion.11
Statistical Analysis
All of our data were analyzed using IBM Statistical Package for Social Sciences v.15 program (SPPS Inc., Chicago, IL, USA). Descriptive data were presented as mean and standard deviation or median and minimum-maximum values for quantitative variables, while categorical variables were expressed as numbers and percentages.
Results
A total of 22 KFS patients (10 males, 12 females) with ASD met the eligibility criteria for the current study. The average age of our series was 56.09 (range 41 to 67) years. The anatomical locations of the lesions, clinical data of the patients, and treatment modalities are presented in Table 1.
Table 1. An overview of clinical, radiological, and operative data in our series (n=22).| Case no. | Age | Sex | Pre-operative mJOA | Post-operative mJOA | Samartzis Type | KFS segment(s) | Adjacent segment disease(s) and level(s) | Izmir Classification System Grade | Operative technique |
| 1 | 60 | M | 15 | 17 | Type 2 | C3-4, C4-5 | C4-5 spinal stenosis | Grade 3 | Anterior, C5-6 discectomy and C5-6 instrumented fusion (PEEK cage and plate) |
| 2 | 51 | F | 11 | 15 | Type 1 | C5-6 | C4-5 spinal stenosis | Grade 2 | Anterior, C4-5 discectomy and arthroplasty |
| 3 | 66 | M | 12 | 15 | Type 1 | C5-6 | C6-7 spondylolisthesis | Grade 4 | Anterior, C6-7 discectomy and C6-7 instrumented fusion (PEEK cage and plate) |
| 4 | 41 | F | 14 | 16 | Type 1 | C3-4 | C5-6 spondylolisthesis | Grade 4 | Anterior, C6-7 discectomy and C6-7 instrumented fusion (PEEK cage and plate) |
| 5 | 55 | M | 13 | 15 | Type 1 | C6-7 | C4-5, C5-6 spinal tenosis | Grade 3 | Anterior; C4 and C5 corpectomy and C3-6 instrumented fusion (mesh cage and plate) |
| 6 | 46 | M | 15 | 17 | Type 1 | C6-7 | C4-5, C5-6 spinal stenosis | Grade 3 | Anterior, C4-5 and C5-6 discectomy and C4-6 instrumented fusion (PEEK cage and plate) |
| 7 | 66 | F | 14 | 17 | Type 1 | C6-7 | C4-5, C5-6 spinal stenosis | Grade 3 | Anterior, C4-5 and C5-6 discectomy and C4-6 instrumented fusion (PEEK 17cage and plate) |
| 8 | 61 | M | 15 | 17 | Type 1 | C5-6, C6-7 | C4-5 spinal stenosis | Grade 3 | Anterior, C4-5 discectomy and C4-5 instrumented fusion (PEEK cage and plate) |
| 9 | 67 | F | 12 | 15 | Type 1 | C5-6 | C4-5 spinal stenosis | Grade 2 | Anterior, C4-5 discectomy and arthroplasty |
| 10 | 44 | F | 17 | 17 | Type 1 | C3-4 | C4-5, C5-C6 spinal stenosis | Grade 3 | Anterior, C5 corpectomy and C4-C6 instrumented fusion (mesh cage and plate) |
| 11 | 59 | M | 15 | 16 | Type 1 | C5-6 | C4-5 spinal stenosis | Grade 2 | Anterior, C4-5 discectomy and arthroplasty |
| 12 | 54 | F | 15 | 15 | Type 3 | C4-C5,C5-6, C7-T1 | C3-4 spinal stenosis and spondylolisthesis | Grade 4 | Anterior, C4-5 discectomy and instrumented fusion (PEEK cage and plate) |
| 13 | 58 | M | 15 | 14 | Type 1 | C5-6 | C6-7 spinal stenosis and spondylolisthesis | Grade 4 | Anterior, C6-7 discectomy and instrumented fusion (PEEK cage and plate) |
| 14 | 54 | M | 15 | 15 | Type 2 | C5-6, C6-7 | C4-5 spinal stenosis and spondylolisthesis | Grade 4 | Anterior, C4-5 discectomy and instrumented fusion (PEEK cage and plate) |
| 15 | 61 | M | 16 | 16 | Type 1 | C6-7 | C4-5 spinal stenosis | Grade 2 | Anterior, C4-5 discectomy and arthroplasty |
| 16 | 48 | F | 16 | 18 | Type 1 | C6-7 | C5-6 spinal stenosis | Grade 2 | Anterior, C4-5 discectomy and arthroplasty |
| 17 | 65 | M | 15 | 17 | Type 1 | C6-7 | C3-4 and C5-6 spinal stenosis | Grade 3 | Anterior, C3-4 discectomy and instrumented fusion (PEEK cage) and C5-6 discectomy and arthroplasty |
| 18 | 57 | F | 14 | 16 | Type 1 | C5-6 | C3-4, C4-5 spinal stenosis and OPLL (needs corpectomy) | Grade 4 | Anterior, C5 corpectomy and C4-C6 instrumented fusion (otogen graft and plate) |
| 19 | 62 | F | 14 | 16 | Type 2 | C4-5, C5-6, C6-7 | C3-4 spinal stenosis and spondylolisthesis | Grade 4 | Anterior, C3-4 discectomy and instrumented fusion (PEEK cage and plate) |
| 20 | 53 | F | 16 | 18 | Type 1 | C4-5 | C5-6 spinal stenosis | Grade 2 | Anterior, C4-5 discectomy and arthroplasty |
| 21 | 56 | F | 18 | 18 | Type 1 | C6-7 | C5-6 spinal stenosis | Grade 1 | No surgical intervention |
| 22 | 50 | F | 17 | 17 | Type 1 | C3-4 | C2-3 spinal stenosis | Grade 4 | Posterior, C2 and C3 laminoplasty |
The combined imaging modalities were employed to confirm the diagnosis of KFS and patients were initially graded according to the Samartzis Classification System (Table 2).12 The distribution of the patients according to the Samartzis Classification was as follows: 18 patients (81.80 %) were Type 1, 3 cases (13.63%) were Type 2, and 1 patient (4.54 %) was Type 3.
Table 2. Samartzis classification system 12.| Type | Definition |
| I | Single-level fusion |
| II | Multiple, non-contagious fusion |
| III | Multiple, contagious fused segments |
The operative technique was selected concerning our novel “Yilmaz-Yucesoy Classification System” (Table 3). Accordingly, 1 patient (4.54 %) had non-surgical treatment (Yilmaz-Yucesoy Grade 1), 6 patients (27.27 %) received anterior cervical arthroplasty (Yilmaz-Yucesoy Grade 2) (Figure 1), 7 cases (31.81 %) received anterior cervical discectomy or corpectomy and fusion with cervical cage and plate (Yilmaz-Yucesoy Grade 3) (Figure 2). These latter patients did not have cervical spinal instability but had spinal stenosis at more than 1 level. They needed at least 2 level discectomy or corpectomy due to the ossification of posterior longitudinal ligament or osteophyte formation. Eight patients (36.36 %) with cervical spinal instability were performed anterior cervical discectomy or corpectomy and fusion with cervical cage and plate (Yilmaz-Yucesoy Grade 4) (Figure 3).
Table 3. The novel “Yilmaz-Yucesoy Classification System” for Klippel Feil Syndrome (KFS).| Grade | Radiological diagnosis of KFS | Radiological instability | Clinical finding(s) | Additional information | Treatment |
| 1 | + | - | - | - | Conservative, symptom driven, muscle strengthening |
| 2 | + | - | + | - | Discectomy, arthoplasty |
| 3 | + | - | + | Spinal stenosis involving more than 2 levels. At least 2 level discectomy or corpectomy needed due to OPLL or osteophyte formation. | Multilevel discectomy with cage or corpectomy and instrumented fusion with cage and plate) |
| 4 | + | + | + | If necessary, posterior laminectomy and stabilization or laminoplasty can be employed. | Discectomy and fusion with cage and cervical plate or corpectomy and instrumented fusion with cage and plate, and/ or laminectomy and screw- rod fixation. |
Figure 1A, B, C. X-ray, computerized tomography, and magnetic resonance imaging views of a Yilmaz-Yucesoy Classification System Grade 2 patient.
Figure 2. Computerized tomography, and magnetic resonance imaging views of a Yilmaz-Yucesoy Classification System Grade 3 patient who underwent anterior cervical discectomy and fusion with cervical cage and plate.
Figure 3. Computerized tomography scans of a Yilmaz-Yucesoy Classification System Grade 4 patient who underwent anterior cervical discectomy and fusion with cervical cage and plate.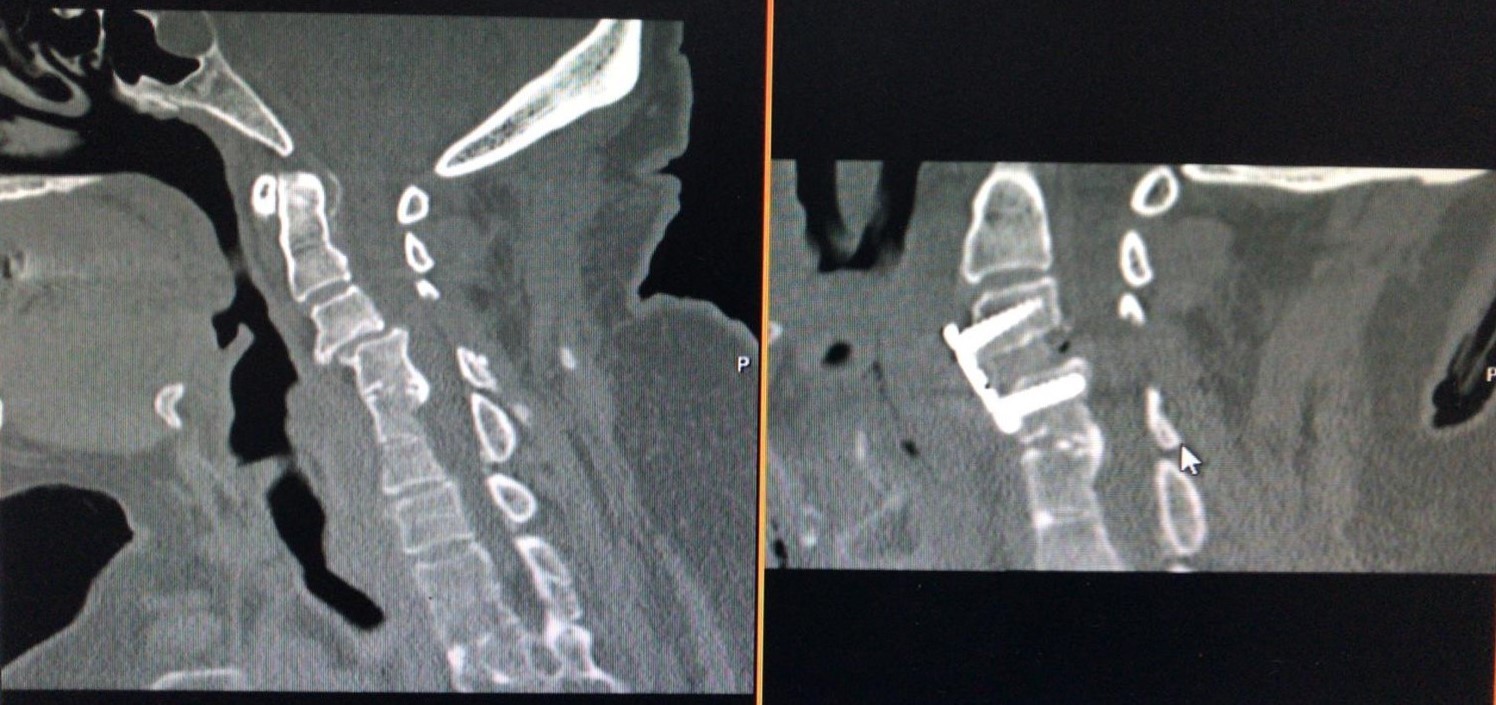
Spinal cord intensity changes were found in seventeen (77.27 %) patients in the MRI. The preoperative and postoperative condition of the patients was assessed with the modified Japanese Orthopaedic Association Scale (mJOA).13 Preoperative average mJOA scores were 14.5±1.4 and postoperative third month mJOA scores 16.2±1.6. In our series, no perioperative mortality was reported and no complications occurred during operations and early postoperative period. The mean follow-up period was 40 months (range: 12 months to 12 years). A satisfactory degree of recovery was observed in all patients.
Discussion
This study was performed to present our clinical experience with a series of KFS patients with concomitant ASD. Based on our clinical, diagnostic, and therapeutic data, we propose a new classification system that can be used to recognize the patients accurately and to tailor the appropriate treatment strategy in order to optimize the treatment outcomes.
Evaluation of KFS patients necessitates a complete and detailed physical examination. This disorder can be accompanied by various pathologies such as Sprengel’s Deformity, Duane Syndrome, renal agenesis, Wildervanck Syndrome, renal, vascular, and cardiac malformations. Notably, nearly half of KFS patients may suffer from concurrent scoliosis and atlantoaxial instability.1 The radiological assessment of the cervical spine in KFS patients consists of plain radiographs, CT, and MRI. Plain radiographs on flexion and extension may exhibit details of spinal stability and mobility.1 A wasp-waist sign may be present on radiographs and flexion/extension X-ray may illustrate the status of spinal stability and movement in a clinically stable patient.1 No specific etiologic factors have yet been identified for KFS.4
Possible multisystemic involvement and syndromic presentation must be remembered in KFS patients with ASD. Therefore, a multidisciplinary approach and close collaboration between various branches are crucial in the evaluation of these patients and particularly candidates for surgical treatment.1 Patients with persistent neurological pain, myelopathy, abrupt and recent onset of muscular weakness, and documented spinal instability are possible candidates for surgical treatment. In addition to spinal instability, deformities are important during the decision making for surgical treatment. Cervical fusion may occur from either anterior or posterior approaches subsequent to evaluation. Anterior approach can involve either cervical fusion or corpectomy with the placement of synthetic or bone grafts. Cervical total disc arthroplasty can be considered as a surgical alternative that may be beneficial for the quality of life and prevention of ASD especially in the adult population with degenerative changes. On the other hand, posterior approaches can be employed including decompression and fusion by means of different instrumentation procedures. Selected cases with severe deformities may be treated with a combined anterior-posterior approach. Healing osteomyelitis or discitis, previous fusion without instrumentation, ankylosing spondylitis, and juvenile idiopathic arthritis must be considered in the differential diagnosis.6, 14
Congenital fusion of cervical vertebrae, such as KFS, is a possible risk factor for the development of degenerative cervical myelopathy (DCM).6 Klippel-Feil Syndrome occurs due to the failure or incomplete segmentation of somites early during the gestational development of the spinal column.6, 15
KFS can present in a wide phenotype spectrum and its impact on the quality of life is associated with the severity of the condition, accompanying deformity, and the possibility for compression of spinal cord and myelopathy. Typically, the cervical spinal deformity increases in parallel with the number of levels involved.6 Congenital defects that may influence visceral, musculoskeletal, otolaryngological, and neurological systems may be observed in these patients.3 It has been postulated that KFS patients are vulnerable to accelerated degenerative changes and hypermobility at segments adjacent to the spinal fusion.6
Patients with KFS are prone to the development of cervical joint degeneration. Even though the mechanism for this association is still obscure, the increased biomechanical stress on nonfused segments may be contributory.8
The exclusion of patients with idiopathic cervical fusion (ICF) of vertebrae who did not demonstrate the typical “wasp-waist sign” associated with KFS was based on the assumption that none of these subjects displayed any signs of degenerative changes at the fused level. It must be remembered that ICF may constitute a distinct clinical entity rather than simply representing a broader spectrum of phenotypes associated with KFS.6 In addition, ICF, which refers to congenital fusions that do not exhibit any shreds of evidence for degenerative fusion, have not been previously reported.
The relationship between KFS and DCM has not been completely elucidated but there are reports indicating segmental hypermobility and a tendency for degenerative changes in the cervical spine of patients with KFS.1, 3 The recognition that patients with the surgical fusion of vertebrae may be at risk for ASD further supports that patients with KFS should be investigated for an increased risk to develop neurologic sequelae linked with degenerative changes.4
Limited mobility, usually due to bone and soft tissue restrictions associated with KFS, may lead to excessive movement and shear stress in unfused segments that may appear clinically as degenerative disc disease later in life.5
Operative management strategies have included arthrodesis and stabilization of the cervical spine. Other surgical treatment options have been attempted, including cervical disc replacement, in order to restore physiological movement and take action on further loss of motion and development of adjacent segment disease.5 The relatively high prevalence of KFS in surgical series and their more obvious MRI findings remind that these patients are more vulnerable for the development of cervical spondylotic myelopathy.4
The literature has noted that up to half of KFS cases may not present with typical findings such as “wasp-waist sign” and the reason behind such assertions remains speculative.16
The strengths of the current study involve data integrity and adequate duration of follow-up. However, limitations such as retrospective design, data restricted to the experience of a single-center, and possible confounding effects of ethnic, genetic, and socioenvironmental factors must be remembered during extrapolation of our data to larger populations.
Conclusion
In conclusion, recognition and classification of KFS patients with ASD is critical for the establishment of diagnosis accurately and tailoring the individualized treatment strategy without delay. Our preliminary data yielded that the novel “Yilmaz-Yucesoy Classification System” can be useful to categorize these patients and to optimize therapeutic outcomes. However, further multi-centric, prospective, randomized trials on larger series are warranted to confirm and validate our newly proposed classification system.