Secular Trend in the Incidence of Japanese Employees with Thyroid Cancer Undergoing Thyroidectomy from 2005 to 2014: a Retrospective Descriptive Study Using an Employment-Based Insurance Claims Database
Abstract
This study aimed to investigate the trend of patients with thyroid cancer undergoing thyroidectomy and those undergoing fine needle aspiration (FNA) and its relationship with thyroid cancer, using a large-scale, real-world database established based on the employment-based health insurance claims data in Japan. In this retrospective descriptive study, annual incidence rates of patients with thyroid cancer undergoing thyroidectomy and FNA from 2005 to 2014 were calculated. Among the 3,130,757 enrollees, 926 patients were diagnosed with thyroid cancer and underwent thyroidectomies. The annual incidence of patients with thyroid cancer undergoing thyroidectomy increased from 5.4 (95% confidence interval 95% CI, 3.4–8.5) in 2005 to 11.7 (10.1–13.5) per 100,000 patient-years in 2013, with a gradual increase among patients in their 20s and 30s and with the most notable increase among those in their 40s and 50s. The annual FNA rate also increased during those years, whereas no noticeable change was observed in the thyroid cancer detection rate. The incidence rates of patients with thyroid cancer undergoing thyroidectomy and those undergoing FNA were strongly correlated, with Pearson’s correlation coefficients of 0.935 for men and 0.886 for women. In conclusion, an increasing trend in the incidence of patients with thyroid cancer undergoing thyroidectomy was observed from 2005 to 2013 among a large group of Japanese employees of working age and their dependents. These results provide useful information on the impact of patients with thyroid cancer undergoing thyroidectomy on the working population.
Author Contributions
Academic Editor: Giovanni Mauri, European Institute of Oncolgy, Italy.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2019 Yasushi Noguchi, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Thyroid cancer is the most common form of endocrine cancer. Its incidence in the last few decades has increased worldwide 1, 2, with an average increase of 48.0% and 66.7% among men and women, respectively, over two periods, 1973–1977 and 1998–2002 in the Americas, Asia, Europe, and Oceania 1. The estimated incidence rate in Japan also continuously increased from 5.7 to 10.9 per 100,000 individuals from 2002 to 2012. However, the estimate was based on patient statistics derived from prefectural registries and did not include all prefectures 3.
One of the possible explanations behind the increasing incidence is the detection of a greater number of thyroid cancer cases due to an increased number of individuals who underwent screening and diagnostic tests 4. A growing number of screening tests for thyroid cancer, including ultrasound, are performed globally 4, 5, 6. In Japan, thyroid cancer screening has been increasingly conducted in mass screenings and comprehensive medical examinations 7, 8, leading to increased detection of thyroid nodules, detected by ultrasound in 6.9–31.6% of individuals during screening tests or incidentally during medical examinations 9. Thyroid nodules suspected to be malignant are subjected to fine needle aspiration (FNA), which is indicated to obtain a cytological diagnosis and recommended for thyroid cancer diagnosis in Japan 10. A US population-based estimate reported a rapid increase in FNA and operative procedures, such as thyroidectomy, associated with an increased incidence of thyroid cancer 11.
To our knowledge, nationwide studies on the recent trend of thyroid cancer incidence are limited in Japan. Aside from cancer surveillance data, real-world data such as healthcare claims databases are currently available in Japan. The JMDC Inc. (Tokyo, Japan; previously known as Japan Medical Data Center Co., Ltd.) has established a database based on employment-based health insurance claims data since 2005 12. The database covers 3 million employees of middle- to large-sized companies and their dependents aged <75 years. Using this large-scale real-world data, the incidence trend of patients with thyroid cancer undergoing thyroidectomy and that of FNA and its relationship with thyroid cancer are investigated.
Materials and Methods
Design and Data Source
This is a retrospective descriptive study using the JMDC database, which consists of enrollment and claims records for in- and out-patient care at hospitals and clinics as well as for pharmacy prescriptions of corporate employees’ health plans affiliated with the Health Insurance Associations. Data of approximately 3 million employees and their dependents aged <75 years accounted for 1.3% of the national population as of 2014. All of the individuals enrolled in the JMDC database from January 2005 to October 2014 (study period) were included in our analysis. Elderly people aged ≥75 years in Japan are covered by a separate public insurance system and, therefore, are not included in the database. The database has been widely used in epidemiological studies and by local regulatory authorities 13, 14. All of the claims data were anonymized during retrieval from the insurers. Diagnoses, procedures, and drugs are coded according to the International Classification of Disease, tenth revision (ICD-10), the Japanese national reimbursement scheme, and the Anatomical Therapeutic Chemical classification of the European Pharmaceutical Marketing Research Association, respectively.
As this study used pre-existing and de-identified data, the requirement to obtain informed consent from individual enrollees was exempted according to the local ethical guidelines for medical and health research involving human subjects 15. This study and the waiver of informed consent were approved by the Keio University Faculty of Pharmacy ethics committee for research involving humans, Tokyo, Japan (No. 150324-1).
Variables
Thyroid cancer was defined using the ICD-10 diagnosis code for malignant neoplasm of the thyroid gland (C73), combined with the Japanese national insurance reimbursement scheme code for thyroidectomy (K463) 16. The occurrence of thyroid cancer was identified as the first record of malignant neoplasm in the thyroid gland, followed by thyroidectomy each year in the database. The date of the first thyroidectomy was set as the index date. FNA of the thyroid gland was identified using the Japanese national insurance reimbursement scheme code for thyroid gland puncture and needle biopsy (D411). Although both ultrasound and FNA are used for standard thyroid cancer diagnosis 17, 18, only FNA was identified because of the lack of imaging data in the database to identify the purpose of ultrasound tests. Only the first FNA record for that patient within the database each year was identified as the first FNA.
Statistical Analysis
Patient demographics and clinical characteristics were summarized for the whole study period, using descriptive statistics.
The annual incidences of patients with thyroid cancer undergoing thyroidectomy and the annual FNA rates were both expressed as incidence rate per 100,000 patient-years from April to the following March each year. The risk period for annual incidence and annual FNA rate started on April 1 and continued until the index date (the date of the first thyroidectomy). If no occurrence of thyroid cancer was recorded, the risk period ended on March 31 of the following year. Both the incidence rates of patients with thyroid cancer undergoing thyroidectomy and FNA rates were stratified by sex and age (<20, 20–39, 40–59, and 60–74 years).
Detection rate was determined by dividing the number of patients with thyroid cancer who underwent both FNA and thyroidectomy by the number of patients who underwent FNA for each year. The annual incidence rates of patients with thyroid cancer undergoing thyroidectomy were scatter-plotted against the annual FNA rates according to sex, and Pearson’s correlation coefficients were then calculated. Data analyses were performed using SAS (SAS Institute Inc.; Cary, NC, USA) version 9.3.
Results
Among the 3,130,757 enrollees, 12,148 had the diagnosis code for malignant neoplasm in the thyroid gland during an approximate period of 10 years (Figure 1). Among those recorded to have malignant neoplasms in the thyroid gland, 926 (7.6%) with thyroid cancer underwent thyroidectomy. Among the patients with thyroid cancer undergoing thyroidectomy, 69.8% were women, with the mean (± standard deviation SD) age of 48.7 ± 11.2 [Table 1]. Papillary thyroid cancer was the most commonly observed pathology, accounting for 52.7% of patients. A mean of 30.1 ± 23.2 months elapsed from the first diagnosis to thyroidectomy.
Table 1. Demographics and clinical characteristics of patients with thyroid cancer undergoing thyroidectomy| Patients with thyroid cancer who underwent thyroidectomy (n = 926) | |
| Sex (female) | 646 (69.8) |
| Age (year), mean ± SD | 48.7 ± 11.2 |
| <20 | 6 (0.6) |
| 20–39 | 173 (18.7) |
| 40–59 | 575 (62.1) |
| 60–74 | 172 (18.6) |
| Database enrollment period (year), mean ± SD | 4.7 ± 2.2 |
| Interval from first diagnosis to thyroidectomy (month), mean ± SD | 30.1 ± 23.2 |
| Duration from thyroidectomy to last visit (month), mean ± SD | 25.5 ± 21.5 |
| Disease namea | |
| Thyroid cancer | 372 (40.2) |
| Papillary thyroid cancer | 488 (52.7) |
| Medullary thyroid cancer | 28 (3.0) |
| Follicular thyroid cancer | 21(2.3) |
| Thyroid malignancy | 7 (0.8) |
| Malignant thyroid tumor | 8 (0.9) |
| Anaplastic thyroid cancer | 2 (0.2) |
| Thyroid hormone replacement | 549 (59.3) |
The annual incidence rates of patients with thyroid cancer undergoing thyroidectomy increased between 2005 and 2013 from 5.4 (95% confidence interval 95% CI, 3.4–8.5) to 11.7 (10.1–13.5) per 100,000 patient-years (Table 2). The incidence rates increased for both sexes (Figure 2 (a)), whereas the incidence rates in women were approximately two to three times higher than those in men. The incidence rates of patients aged 20–39 years showed a gradual increase and that of 40–59 years showed the most notable increase between 2005 and 2013 (Figure 2 (b)). Unlike the younger age groups, the incidence rates of patients aged 60–74 years were consistently higher but fluctuated throughout the study period. Lower incidence rates were observed in this age group between 2007 and 2009.
Table 2. Annual incidence rates of patients with thyroid cancer undergoing thyroidectomy and annual FNA rates| Year1) | Patients who underwent FNA2), n | Total observed patient-years | Annual FNA rate3), per 100,000 patient-years (95% CI) | Patients with thyroid cancer who underwent thyroidectomy 4), n | Total observed patient-years | Annual incidence rate5), per 100,000 patient-years (95% CI) | Detection rate6)(%) |
| 2005 | 82 | 334,817 | 24.5 (19.7–30.4) | 18 | 334,863 | 5.4 (3.4–8.5) | 19.5 |
| 2006 | 92 | 348,661 | 26.4 (21.5–32.4) | 19 | 348,774 | 5.5 (3.5–8.5) | 18.5 |
| 2007 | 118 | 438,127 | 26.9 (22.5–32.3) | 19 | 438,310 | 4.3 (2.8–6.8) | 12.7 |
| 2008 | 217 | 730,154 | 29.7 (26.0–34.0) | 36 | 730,450 | 4.9 (3.6–6.8) | 11.1 |
| 2009 | 282 | 930,327 | 30.3 (27.0–34.1) | 67 | 930,764 | 7.2 (5.7–9.2) | 18.8 |
| 2010 | 570 | 1,512,924 | 37.7 (34.7–40.9) | 120 | 1,513,610 | 7.9 (6.6–9.5) | 16.1 |
| 2011 | 807 | 1,809,334 | 44.6 (41.6–47.8) | 159 | 1,810,475 | 8.8 (7.5–10.3) | 16.5 |
| 2012 | 815 | 1,831,248 | 44.5 (41.6–47.7) | 203 | 1,832,837 | 11.1 (9.7–12.7) | 23.6 |
| 2013 | 802 | 1,588,910 | 50.5 (47.1–54.1) | 186 | 1,590,713 | 11.7 (10.1–13.5) | 21.2 |
| Total | 4,214 | 10,457,493 | 40.3 (39.1–41.5) | 926 | 10,465,015 | 8.9 (8.3–9.4) | 19.0 |
Figure 2.Annual incidence of patients with thyroid cancer undergoing thyroidectomy according to sex (a) and age (b)
The annual FNA rates also increased between 2005 and 2013 from 24.5 (95% CI, 19.7–30.4) to 50.5 (47.1–54.1) per 100,000 patient-years (Table 2). The FNA rates increased in both sexes, which were approximately two to four times higher in women than in men (Figure 3(a)). FNA rates were also higher among the older age groups throughout the study period. The FNA rate of patients aged 40–59 years notably increased between 2005 and 2013 (Figure 3(b)). Throughout the study period, the highest FNA rates with fluctuations were observed in patients aged 60–74 years. Their FNA rates were particularly lower in 2008 and 2009.
Figure 3.Annual FNA rates by sex (a) and age (b)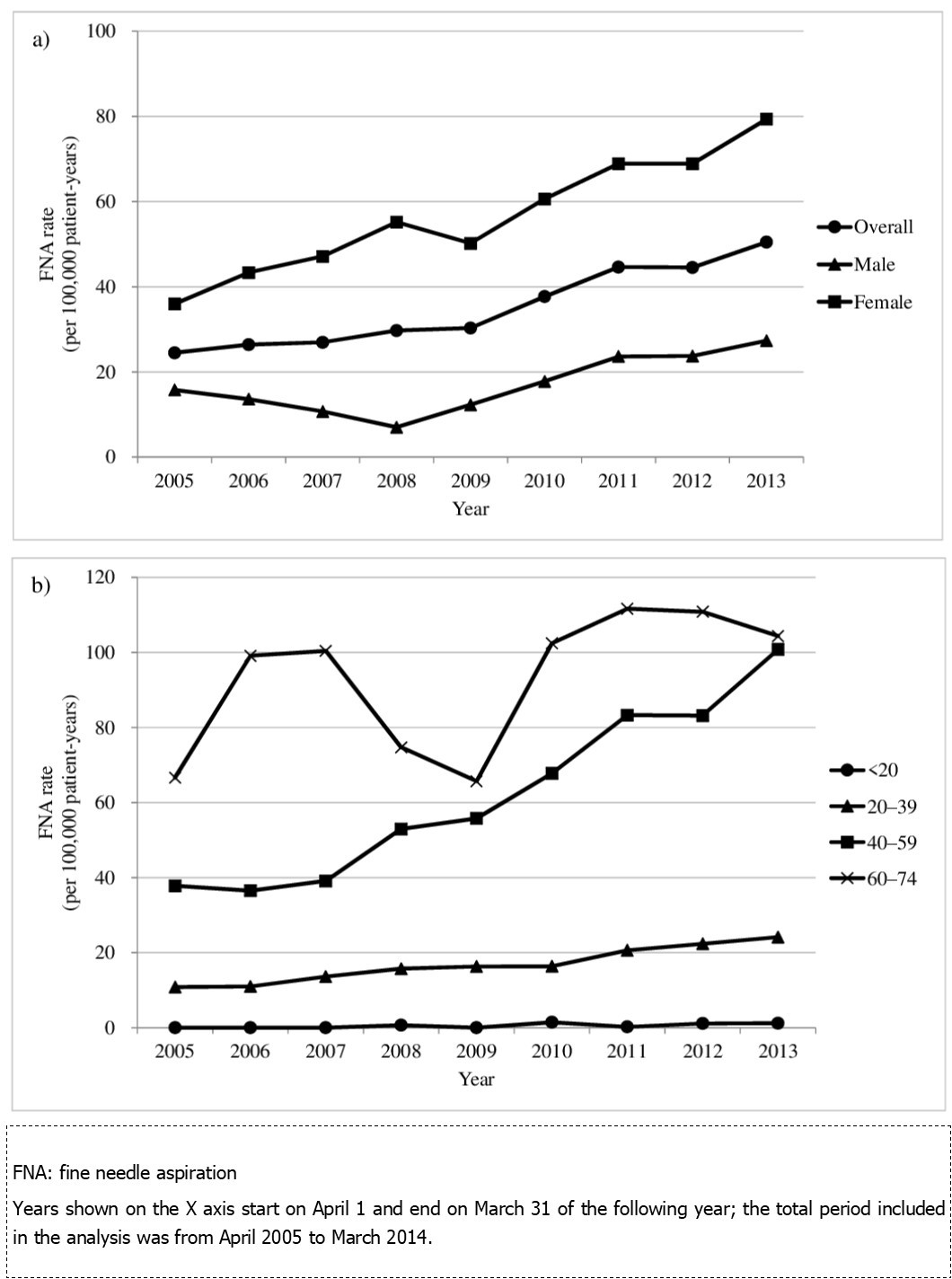
Detection rates of thyroid cancer in patients who underwent thyroidectomy ranged from 11.1% to 23.6% between 2005 and 2013, without any noticeable trend (Table 2).
Correlation analysis between the annual incidence rate of patients with thyroid cancer undergoing thyroidectomy and the corresponding annual FNA rate showed a high Pearson’s correlation coefficient in both sexes: 0.935 in men and 0.886 in women (Figure 4).
Figure 4.Correlation between the incidence rate of patients with thyroid cancer undergoing thyroidectomy and FNA rate from 2005 to 2013.
Discussion
The incidence rates in patients with thyroid cancer undergoing thyroidectomy, FNA rates, and the between-rate association were investigated using a large employment-based database of health insurance claims in Japan. The annual incidence rates in patients with thyroid cancer undergoing thyroidectomy increased from 2005 to 2013 and paralleled the increased FNA rate.
This is the first study estimating the incidence trend of thyroid cancer patients undergoing thyroidectomy using a large-scale real-world data in Japan. Since the individuals included in the JMDC database are employees, who worked in middle- to large-sized enterprises with relatively higher income, and their dependents, they perhaps do not represent the general Japanese population. However, they may partially represent Japanese workers of working age and their dependents because most employees commonly retire after the age of 65 years in Japan. The prevalence of thyroid cancer among Japanese people, especially women, gradually increases above the age of 30 years and peaks at approximately 65 years as reported by the National Cancer Center Japan 19. A consistent trend that the incidence of thyroid cancer patients undergoing thyroidectomy increased among patients in their 20s to 50s was observed in our results. Considering that individuals in the study database partially include Japanese workers of working age, particularly those in their 20s to 50s (Figure s), our study results provide useful information on the impact of thyroid cancer patients undergoing thyroidectomy on the workplace, for example, the ability to remain in or return to work after they or their family members experience thyroid cancer entailing thyroidectomy.
The trend of a unidirectional increase in the incidence of thyroid cancer was observed in this study. This increasing trend was consistent with the incidence of thyroid cancer previously reported worldwide in the last few decades 1, 2 and with the estimated incidence of thyroid cancer in Japan 3. However, the annual incidence rates of thyroid cancer patients undergoing thyroidectomy in this study were lower than the annual incidence of thyroid cancer previously reported in the United States between 2005 and 2012 (12.8–21.5 per 100,000 individuals) 4 and in Japan during the same period (7.2–10.9 per 100,000 individuals) 3. This may simply be because this study focused on patients with thyroid cancer who underwent thyroidectomy to exclude cases that used the diagnosis code for thyroid cancer for reimbursement purposes only. For this reason, our incidence rates of thyroid cancer should not be considered for “wait-and-watch” patients diagnosed with thyroid cancer.
Paralleled with the incidence of thyroid cancer patients undergoing thyroidectomy, this study also demonstrated increased FNA rates from 2005 to 2013, which was highly correlated with the incidence of thyroid cancer patients undergoing thyroidectomy without any remarkable changes in the detection rate. Although whether FNA was appropriately implemented on nodules measuring ≥1 cm as recommended in the guidelines 17 could not be assessed owing to the unavailability of imagining data and nodule size in the database, increased screening may have led to a higher number of suspicious cases referred for FNA, which consequently increased the identification of malignant thyroid cancer cases resulting in thyroidectomy. A previous study, in fact, reported that the number of individuals who underwent screening tests for thyroid cancer, namely, through palpation and/or ultrasound, increased in Tokyo 7. The increased screening procedures might be partially due to the increasing awareness of cancer through governmental campaigns for cancer prevention and screening to reduce cancer mortality, which was initiated under the Cancer Control Act of 2007 20. Although thyroid cancer itself is not the target of governmental campaign for cancer prevention and screening, thyroid nodules can be detected in the course of screening for breast cancer. Another possibility is the heightened concern among the public on the release of radiation from the nuclear power plant after the March 2011 earthquake. However, the unavailability of geographical information in the database and our study period lacking sufficient follow-up time after the nuclear fallout prevented the evaluation of the fallout effects on thyroid cancer. Further studies are needed to assess the screening trend.
The incidence rates of thyroid cancer patients undergoing thyroidectomy and FNA rates at aged 60–74 years were high compared with those of the other age groups and fluctuated throughout the study period. These fluctuations may be ascribable to an artifact caused by a disproportionally small representation of the elderly population and the increasing size of the JMDC database during the study period. Individuals aged ≥60 years accounted for only 9.2% (288,741/3,130,757) of all individuals included in the database during the study period. In addition, the number of individuals in this age group contained in the database is substantially smaller in 2007 or earlier than in 2008 or later. Therefore, the estimates on incidence rates of thyroid cancer patients undergoing thyroidectomy and FNA rates may be unstable in the elderly.
This study has some limitations that warrant consideration when interpreting the results. First, the generalizability of our findings may be limited. Our results may not be generalizable to populations outside the JMDC but may be partially generalizable to Japanese working populations of working age, particularly for those aged <60 years. Second, the possible misclassification and inaccuracy of the diagnoses recorded in the study database cannot be avoided. As in any claim-based database studies, codes are assigned for reimbursement purposes. In this study, the ICD-10 code for malignant neoplasm of the thyroid gland was used to identify thyroid cancer combined with a medical record of thyroidectomy to enhance diagnosis validity. Further, summary statistics of lymph node or distant metastases were not presented because of this limitation; a diagnostic code for metastases associated with thyroid cancer is not necessarily recorded in the study database unless claims for the treatment of these metastases have been issued. The association between thyroid and metastatic cancers is also unclear in the study database. In addition, due to a lack of nodule size and cytology results (i.e., Bethesda class) and the necessity to maintain anonymity due to the privacy protection rule in the database, the accuracy of these diagnostic codes with respect to severity cannot be validated. Third, specific histological types of thyroid cancer were not available in patients simply coded as “thyroid cancer.” However, we believe that those coded as “thyroid cancer” may mostly fall into the papillary thyroid cancer subtype. This estimation is based on the reports that papillary thyroid cancer was the most predominant subtype of thyroid cancer accounting for >90% in Japan 21. Therefore, we believe that our results would indicate the annual incidences of differentiated thyroid cancer. However, due to the histological data unavailability, whether the increased annual incidence is attributed to an increase in a specific subtype cannot be identified.
Conclusion
A trend of the increasing incidence of patients with thyroid cancer undergoing thyroidectomy was observed from 2005 to 2013 among a large group of Japanese employees of working age and their dependents. Our results may help understand the impact of thyroid cancer patients undergoing thyroidectomy on the working population. This is the first study assessing the incidence of thyroid cancer focusing on thyroidectomy and FNA rates, using a large-sized real-world database. Due to these limitations, further studies to track thyroid cancer incidence may be warranted to determine the trend of thyroid cancer incidence in the Japanese population.
Acknowledgments
This study was funded by Eisai Co., Ltd. (Tokyo, Japan). The authors would like to thank Gen Terashima from JMDC Inc. (Tokyo, Japan; previously Japan Medical Data Center Co., Ltd.) for assistance with JMDC database interpretation. The current affiliation of Hitomi Nagamune is Bayer Yakuhin, Ltd.
Supplementary Information
References
- 1.Kilfoy B A, Zheng T, Holford T R, Han X, Ward M H. (2009) International patterns and trends in thyroid cancer incidence. , Cancer Causes Control 20, 525-531.
- 2.Ferlay J, Bray F, Steliarova-Foucher E, Forman D. (2015) . Cancer Incidence in Five Continents, CI5plus: IARC CancerBase No. 9 [Internet]. Lyon, France:International Agency for Research on Cancer; Accessed onSeptember1,2018.Availablefrom:http://ci5.iarc.fr .
- 3.Cancer Information Service.National Cancer Center, Japan. (n.d.) Cancer registry and statistics: Cancer incidence (1975-2012). Accessed on September1,2018. Available. from: http://ganjoho.jp/en/professional/statistics/table_download.html
- 4.Zevallos J P, Hartman C M, Kramer J R, Sturgis E M, Chiao E Y. (2015) Increased thyroid cancer incidence corresponds to increased use of thyroid ultrasound and fine-needle aspiration: a study of the Veterans Affairs health care system. , Cancer 121, 741-746.
- 5.Ahn H S, Kim H J, Welch H G. (2014) Korea's thyroid-cancer "epidemic"–screening and overdiagnosis. , N. Engl. J. Med 371, 1765-1767.
- 6.Leenhardt L, Bernier M O, Boin-Pineau M H, Conte Devolx B, Maréchaud R. (2004) Advances in diagnostic practices affect thyroid cancer incidence in France. , Eur. J. Endocrinol 150, 133-139.
- 7. (2015) Tokyo Health Service Association. Tokyo Health Service Association annual report. [in Japanese]. Accessed onSeptember1,2018. Available from: http://www.yobouigaku-tokyo.or.jp/nenpo/pdf/2015/05_05.pdf 44, 111-116.
- 8.Miki H, Inoue H, Komaki K, Uyama T, Morimoto T. (1998) Value of mass screening for thyroid cancer. , World J. Surg 22, 99-102.
- 9.Shimura H. (2010) Nihon ni okeru koujyousensyuyou no hindo to keika ― ningen dokku karano deta. [in Japanese] , J. of the Japan Thyroid Association 1, 109-113.
- 10. (2010) Japan Association of Endocrine Surgeons, Japanese Society of Thyroid Surgery.Koujyousen syuyou sinryou gaidorain. [in Japanese] , Tokyo, KANEHARA & CO., LTD
- 11.Sosa J A, Hanna J W, Robinson K A, Lanman R B. (2013) Increases in thyroid nodule fine-needle aspirations, operations, and diagnoses of thyroid cancer in the United States. , Surgery 154, 1420-1426.
- 12.Kimura S, Sato T, Ikeda S, Noda M, Nakayama T. (2010) Development of a database of health insurance claims: standardization of disease classifications and anonymous record linkage. , J. Epidemiol 20, 413-419.
- 13.Shibata N, Kimura S, Hoshino T, Takeuchi M, Urushihara H. (2018) Effectiveness of influenza vaccination for children in Japan: Four-year observational study using a large-scale claims database. , Vaccine 36, 2809-2815.
- 14.Pharmaceutical, Medical Devices Agency. (2014) Report of trial survey for risk assessment methods of adverse event onset using receipt data. Accessed onNovember2,2017.Available from:https://www.pmda.go.jp/files/000205629.pdf
- 15.Ministry ofHealth, Labour and Welfare. (n.d.) Ethical Guidelines for Medical and Health Research involving Human Subjects. Accessed onNovember2,2018.Availablefrom:http://www.mhlw.go.jp/file/06-Seisakujouhou-10600000-Daijinkanboukouseikagakuka/0000080278.pdf.
- 16.Health Insurance Bureau.Ministry of Health, Labour, and Welfare. (n.d.) Various Information of Medical Fee. Accessed onJune1,2018. Available from: http://www.iryohoken.go.jp/shinryohoshu/.
- 17.Haugen B R, Alexander E K, Bible K C, Doherty G M, Mandel S J. (2016) American Thyroid Association management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: The American Thyroid Association Guidelines Task Force on thyroid nodules and differentiated thyroid cancer. , Thyroid 26, 1-133.
- 19.Cancer Information Service, National Cancer Center, Japan.Cancer registry and statistics: age-specific incidence of cancer by year. Accessed onSeptember19,2018. Available from http://gdb.ganjoho.jp/graph_db/gdb1?dataType=30.

