Abstract
Cervicogenic headaches are a significant cause of head and neck pain, and occipital neuralgia is a common component of these cervicogenic headaches. Occipital injections are commonly performed at the occipital ridge, but this site does not address more proximal entrapments of the nerve in the suboccipital region. Because of the potentially dangerous structures in this region, clinicians have tended to avoid the suboccipital region, despite the pathologies seen in this region. This article discusses the pathology of the region, the alternative techniques, and the novel interventional approach developed for this region, specifically the “Stealth” approach of occipital decompression.
Author Contributions
Academic Editor: Andrea Pratico, Department of Clinical and Experimental Medicine, University of Catania, Catania, Italy
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2017 Andrea Trescot, et al
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Injections of the occipital nerve are common office procedures, perceived as safe, but there have been several devastating complications from occipital injections. In 1978, Selander and Sjöstrand1 predicted spinal cord injuries from intraneural injections, and a group of patients have developed a “locked-in” syndrome, with infarcts of the medulla, or even death, presumably due to intraneural injection of particulate steroids, with retrograde flow to the brain stem and subsequent infarct. Additional catastrophes have occurred after occipital nerve injections in patients with Arnold Chiari or posterior fossa surgeries. These injections resulted in a delayed onset respiratory arrest after apparent unrecognized subdural spread of the medication. All of these complications have been attributed to the use of sharp needles.
For these reasons, a blunt tipped needle and a suboccipital approach to the occipital nerves was developed. The suboccipital nerve decompression, also known as the Stealth decompression, is a technique using high volume injectate within the suboccipital triangle, used to treat occipital neuralgia. This treatment can be considered as part of a treatment algorithm lying between a superficial occipital nerve block and injection of the dorsal root ganglion of C2.
Epidemiology
“Headaches” is a term used to describe a wide variety of clinical entities, including intracranial headaches and “migraines”, as well as “tension headaches”, and head pain referred from the neck (“cervicogenic headaches”). A cervicogenic headache (CGH) is defined as a symptomatic unilateral headache with signs and symptoms of neck involvement, such as pain worsened with neck movements and pain with palpation of the neck and/or occipital region2. Relief of pain with diagnostic injections of the occipital or neck structures confirms the diagnosis.
The prevalence of CGH is hard to determine, because different studies have used different criteria, and thus the prevalence varies widely, from 0.4% to 80% 3. The greater occipital nerve (GON) is a significant portion of the patients with CGH, though that prevalence is also not well defined4.Because of connections with the other nerves in this region (see below), there is a great deal of overlap in pain patterns, leading to the difficulty in diagnosis. Occipital nerve pain can present as a throbbing, unilateral headache associated with photophobia, phonophobia, and nausea, which will meet the International Headache Society (IHS) criteria for migraines5. However, this is usually a bilateral condition, even if only one side is primarily symptomatic, so both are usually treated.
Anatomy
The largest of the three occipital nerves, the GON, arises from the dorsal root ganglion (DRG) of C2, running inferiorly between the arch of C1 (atlas) and the lamina of C2 (axis), lateral to the lateral atlanto-axial (AA) joint and deep to the inferior oblique capitus (IOC) muscle. The GON then curves medially and cephalad over the IOC Figure 1. As the GON travels cephalad up the neck, it travels over the posterior surface of the IOC to pierce the semispinalis capitis muscle, deep to the trapezius muscle, Figure 2 onto the posterior skull through a muscular sling formed by the aponeurosis of the sternocleidomastoid muscle (SCM) and the trapezius muscle at the skull attachment (the conjoined tendon) Figure 3. where it is joined laterally by the occipital artery. The GON can be found at this point lying in a palpable groove, immediately medial to the occipital artery and lateral to occipital prominence (the inion) Figure 1. There are several areas of potential entrapment of the GON
-Where the GON emerges from the C2 DRG, between the atlas and axis
-Between the IOC and the semispinalis capitis muscles
-Where the nerve pierces the semispinalis capitis muscle
-Where the GON exits from the aponeurosis of the trapezius
Figure 1.MRI anatomy of the upper cervical region. DI = digastric muscle; IO = inferior oblique muscle; LC = longus colli muscle; LE = levator scapulae muscle; MAS = masseter muscle; SC = semispinalis cervicis muscle; SCM = sternocleidomastoid muscle; SpC = splenius capitis muscle; TRAP = trapezius muscle; GA = great auricular nerve; GON = greater occipital nerve; GN = glossopharygeal nerve. LON = lesser occipital nerve; TON = third occipital nerve. Note the bifid spinous process of C2. (Image courtesy of Andrea Trescot, MD Clinical presentation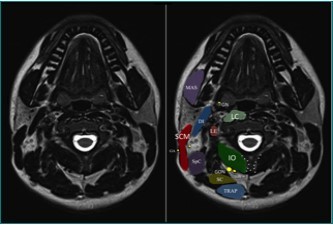
Figure 2.Path of the greater occipital nerve. (Image courtesy of Epimed International®, with permission)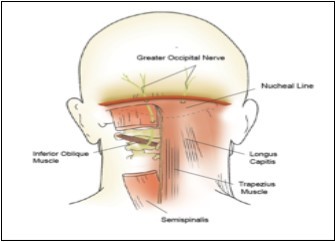
Figure 3.Anatomy of the occipital region, modified from an image from Bodies, The Exhibition, with permission. Note the connection of the greater and lesser occipital nerves. (Image courtesy of Andrea Trescot
The GON can connect with the lesser occipital nerve (LON), which arises from the cervical plexus (formed by the upper four ventral cervical rami), as well as the posterior auricular nerve Figure 3, leading to a variety of clinical presentations.
The GON is prone to trauma from flexion/tension injuries and repetitive neck contractions, which can cause entrapment and/or scarring of the GON, and a head-forward position can entrap the GON at the level of the IOC muscle Figure 4A and B.The GON canbecome inflamed and then develop adhesions in the suboccipital region, leading to further entrapment. Plastic surgeons also may “tuck the skin” around the greater and lesser occipital nerves during some cosmetic procedures, causing superficial and deep entrapment of the nerves.
Figure 4Aand B.Ergonomic issues of head position on occipital nerve entrapment (Images courtesy of Epimed International®, with permission)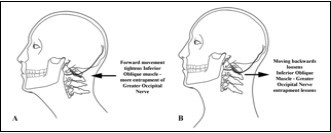
Occipital neuralgia was first described by Beruto et al.6 in 1821 as a sharp, electric-like pain radiating from the occiput to the vertex. Occipital nerve pain primarily refers to the occiput, but it may also radiate to the temporal area, forehead, and retro-orbital areas. Since the GON is made up of contributions from C1, C2, and C3 (see Anatomy), there can be a wide range of clinical presentations, including pain and paresthesias of the posterior scalp; the periorbital, temporal, and mandibular regions; and the external ear and mastoid regions, as well the neck and shoulders. Because of connections with the trigeminal cell bodies in the medulla (the cervico-trigeminal complex), the pain of occipital neuralgia can refer to structures innervated by the branches of the trigeminal nerve, such as the forehead, temples, and eyes.
Physical Exam
With the patient in a supine position, place the fingers directly under the base of the occiput as the patient actively tries to chin-tuck and retract the head. The suboccipital region is then palpated bilaterally just lateral to the spinous process of C2. Tenderness in this region should prompt the clinician to evaluate the suboccipital region as a source of pain.
Injection Technique
Because of the proximity to the vertebral artery and the potential risk of intravascular injection, few interventionalists have been interested in AA injections, and, since the proximal GON travels over the posterior arch of the atlas, even fewer have been interested in direct injections of the proximal GON. In an effort to avoid the “locked-in” syndrome complication seen with occipital injections, Dr. Gabor Racz developed a suboccipital approach to the suboccipital space in 1982, which has recently become more popular7, 8. A curved, bullet-tipped needle with side port injection, attached tubing, and wings (dubbed the “Stealth needle”™ because of its resemblance to the Stealth bomber) Figure 5 is used to approach the suboccipital triangle from above. Since the underlying pathology is usually bilateral, the procedure is usually performed bilaterally.The patient is positioned prone on the fluoroscopy table, with the neck in flexion; the hair is pulled cephalad, and the suboccipital area is sterilely prepped and draped. The inion and conjoined tendon are identified by palpation and the nuchal line is identified by fluoroscopy; the entry site should be 2cm lateral to the midline on the nuchal ridge. After making sure by palpation that the planned entry is not directly over the occipital artery, a small skin wheal of local anesthetic is injected via a 30g needle. Because the galea is so strong, an 18g needle is used to create a small stab incision in the scalp and deep fascia to facilitate the entry of the blunt-tipped needle. Standing at the head of the bed, the Stealth needle is held by the wings like a butterfly IV needle, and, with tip facing down and toward the feet Figure 6 the needle is advanced caudally. The needle position is initially identified on an anterior-posterior fluoroscopy view, with the target medial to the superior pars of C2 Figure 7 The needle is advanced through the muscle and fascial layers of the trapezius and semispinatus with confirmation of location on lateral view, aiming for the superior lamina of C2 at the level of the spinous processes Figure 8. Contrast is injected to verify needle placement Figure 9 followed by 10 cc of local anesthetics and steroids (4.5ccs 0.2% ropivicaine, 4.5ccs 1% lidocaine, and 10mg of dexamethasone on each side) to perform an adhesiolysis, freeing up the greater occipital nerves Figure 10 Because this technique is usually performed bilaterally, lower concentrations of local anesthetic are used to avoid toxicity issues.
Figure 5.The Stealth needle™ (Image courtesy of Epimed International®, with permission)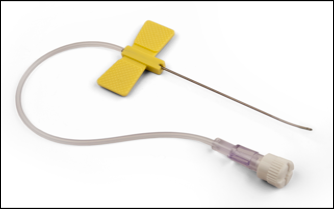
Figure 6.Placement of the Stealth needle™ from the head of the bed, under fluoroscopic control. (Image courtesy of Andrea Trescot, MD)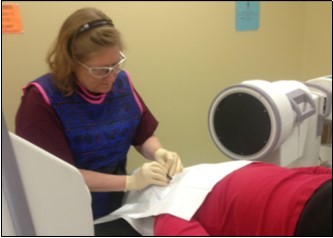
Figure 7.Anterior-posterior fluoroscopic view showing target site for Stealth needleÔ placement, medial to the superior pars of C2. (Image courtesy of Andrea Trescot, MD)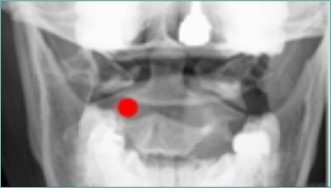
Figure 8.Lateral fluoroscopic image showing the needle in the suboccipital space; red shows the level of the spinous processes (red line). Image Courtesy of Gabor Racz, MD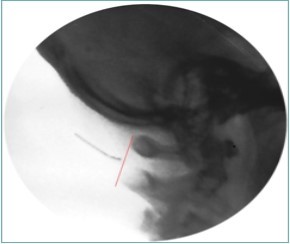
Figure 9.Lateral Fluoroscopic image showing initial contrast location deep to the suboccipital muscles; red line corresponds to the level of the inferior oblique capitus muscle. (Image courtesy of Gabor Racz, MD)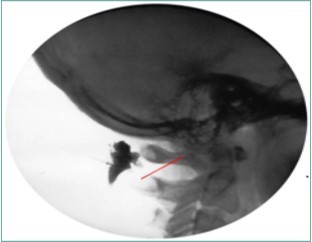
Figure 10.Lateral fluoroscopic image showing the final contrast spread; the arrow shows the outline of the greater occipital nerve at the level of the inferior oblique capitus muscle. Image Courtesy of Gabor Racz, MD
It is important to avoid positioning the needle in the area above C1 because of the rich venous plexus in that region. It is also important to monitor the flow of contrast, so that the medication does not migrate proximally into the subdural space.
Dissection of a cadaver after the injection of methylene blue showed the difference between standard occipital nerve blocks and the suboccipital decompression; the standard occipital nerve block colored only a small part of the nerve, while the Stealth needle bathed the entire nerve Figure 11
Figure 11.Methylene blue suboccipital decompression dissection . One the right is the dye on the occipital nerve form a standard occipital nerve block. On the left is the dye from the stealth decompression bathing the occipital nerve. (Image Courtesy of Gabor Racz, MD)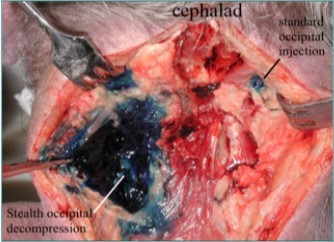
Outcomes
Justiz et al.9, performed a retrospective study of 29 patients with confirmed occipital neuralgia. By measuring pain scores, opioid consumption, and activities of daily living (ADLs), their study showed that the suboccipital decompression procedure was effective in reducing occipital headaches.
Immediately post-treatment, pain scores decreased from 7.9/10 (standard deviation =1.7) to 0.8/10 (standard deviation =1.2). Fifty-eight percent of the patients demonstrated greater than 50% pain relief at the six-month follow-up, and at the one-year follow-up, 34.5% still had ongoing relief. Additionally, the study showed that these patients had an increase in activities, with ADLs improving in 60% of the patients, as well as a significant decrease in opioid use in 56% of the patients.
More recently, Lauretti et al., directly compared the GON injections with the suboccipital decompression in 30 patients who acted as their own control10. Each patient underwent a standard occipital nerve block (which lasted only 2 weeks), and then was randomly assigned to one of 3 suboccipital decompression groups, using 5, 10, or 15cc of fluid for the adhesiolysis. They found that the standard injection of the GON gave only 2 weeks of relief, while the suboccipital injections gave 24 weeks of relief, regardless of the volume used. It is important to note that they did not use a blunt-tipped needle but rather used a standard sharp Quinke needle for both injections, which we would not recommend.
Complications
To date, no complications have been reported, though there is a theoretic risk of local anesthetic toxicity, bleeding, infection, or injection into a vessel (if using a sharp needle). Special care must be taken of patients who have undergone suboccipital surgery, such as a resection for Arnold Chiari malformation; because of the surgery, there is tenting and adhesions of the dura, with a potential spread to the subdural region, and so the procedure should be performed in a safe environment capable of managing hypotension and loss of airway reflexes, and observed for a longer period of time.
Discussion
Options for treatment of occipital neuralgia are limited. Occipital nerve blocks are one option, but they are short lived, on the order of 2 weeks.10, 11 In addition, the risks of injections with sharp needles include nerve trauma, intraneural injection, intravascular injection, and bleeding. Selander12 showed that intraneural injections of local anesthetics, especially under high pressure, could travel long distances within the nerve, all the way to the spinal cord. He proposed that this centripetal spread could explain the unexpected spinal anesthesia seen with injections near the spine (such as occipital nerve blocks). Blunt-tipped needles have been shown in animals to decrease the risk of injury to the nerves and arteries. Candido et al.13 showed that Whitacre spinal needles did not prevent intravascular injections. Heavner et al.14, 15 compared the trauma created by sharp vs. Stealth blunt-tipped needles when attempting to deliberately penetrate kidneys, liver, renal artery, intestine or spinal nerve root, and found that these blunt-tipped needles were less likely than sharp needles to pierce vital structures and/or cause bleeding.
Another common option for occipital neuralgia is the use of botulinum toxin. However, the effect is also short lived, and needs to be repeated every 12 weeks.16
Radiofrequency lesioning has been used to destroy the occipital nerve, but the pain recurs, there is a risk of neuroma formation17, and there have been reports of death and paraplegia.18 Cryoneuroablation has been used at the C2 DRG as well as the more distal occipital nerve.11 There has also recently been a published description of cryoneuroablation at the inferior oblique under ultrasound.19 However, none of these has been studied for efficacy, and destruction of the nerve may not be the most appropriate technique when the pathology is an entrapment.
Occipital stimulation has also been used effectively to treat intractable headaches, but the systems are expensive (as much as €760,00 or $97,000), with the need for multiple revisions and technical failures.20
In a 1994 presentation, Umberto Rossi described treating occipital neuralgia by dissecting down to the C1-C2 lamina, cutting the IOC to release the GON, which was flattened and pale. After this release, the nerve became round and pink, suggesting that there is a mechanical compression of the GON by the IOC 21. Ten years later, Gille et al.22 described 10 patients that were treated with IOC resection; 7 of the 10 patients were “satisfied” or “very satisfied” with the results of the surgery. However, the surgery has not had long-term studies to support it.
Suboccipital decompression addresses the underlying pathology of occipital neuralgia, and thus treats the actual entrapment of the greater occipital nerve.
Summary
The clinician has relatively poor options for the long-term treatment of occipital neuralgia. As noted above, the effect of standard occipital nerve blocks is on the order of 2 weeks, botulinum toxin injections must be repeated every 12 weeks, and, while occipital stimulation is effective, it is expensive and is associated with multiple complications and revisions. In contrast, the Stealth suboccipital decompression is effective for 24 weeks, with very low risks and little specialized equipment or expense.
Stealth suboccipital decompression is a useful technique to treat suboccipital nerve entrapment, which has been shown to be more effective than standard occipital nerve injections. Because of the approach and use of the blunt-tipped needle, there should be a significant decreased risk of nerve or artery injection, avoiding devastating complications.
References
- 1.Selander D, Sjostrand J. (1978) Longitudinal spread of intraneurally injected local anesthetics. An experimental study of the initial neural distribution following intraneural injections. , Acta Anaesth Scand 22(6), 622-634.
- 2.Antonaci F, Fredriksen T A, Sjaastad O. (2001) Cervicogenic headache: clinical presentation, diagnostic criteria, and differential diagnosis. Curr Pain Headache Rep. 5(4), 387-392.
- 3.Knackstedt H, Bansevicius D, Aaseth K, Grande R B, Lundqvist C et al. (2010) Cervicogenic headache in the general population: the Akershus study of chronic headache. , Cephalalgia 30(12), 1468-1476.
- 4.Vanelderen P, Lataster A, Levy R, Mekhail N, M van Kleef et al. (2010) 8.Occipital neuralgia. , Pain Pract 10(2), 137-144.
- 5.The International Classification of Headache Disorders: 2nd edition. , Cephalalgia.2004;24 Suppl 1, 9-160.
- 7.Racz G B, Noe C E, Justiz R. (2009) A new technique for suboccipital compartment decompression. 5th World Congress of the World Institute of Pain abstracts;March2009,New York,NY .
- 8.Racz G B, Noe C E, Justiz R. (2009) A new technique for suboccipital decompression. Pain Pract. 9(51), 29.
- 9.Justiz R, Taylor V, Racz G B, Meta G. (2009) Suboccipital decompression: A retrospective anaylysis of a novel technique for the treatment of occipital neuralgia. 5th World Congress of the World Institute of Pain abstracts;March2009,New York,NY .
- 10.Lauretti G R, Correa S W, Mattos A L. (2015) Efficacy of the greater occipital nerve block for cervicogenic headache: Comparing classical and subcompartmental techniques. , Pain Pract 15(7), 654-661.
- 11.Kim C H, Hu W, Gao J, Dragan K, Whealton T et al. (2015) Cryoablation for the treatment of occipital neuralgia. , Pain Physic 18(3), 363-368.
- 12.Selander D E. (2005) Re: combination of intraneural injection and high-injection pressure leads to fasicular injury and neurologic deficits in dogs. Reg Anesth Pain Med.30(3): 308-309;author reply. 309-310.
- 13.Candido K D, Ghaly R F, Mackerley S, Knezevic N N. (2010) A Whitacre-type spinal needle does not prevent intravascular injection during cervical nerve root injections. , South Med J 103(7), 679-682.
- 14.Heavner J E, Racz G B, Jenigiri B, Lehman T, Day M R. (2003) Sharp versus blunt needle: a comparative study of penetration of internal structures and bleeding in dogs. Pain Pract. 3(3), 226-231.
- 15.Heavner J E, Racz G B. (2010) Using procedure-specific needles: what is blunt?. , South Med J 103(7), 604.
- 17.Gekht G, Nottmeier E W, Lamer T J. (2010) Painful medial branch neuroma treated with minimally invasive medial branch neurectomy. , Pain Med 11(8), 1179-1182.
- 18.Finiels P J, Batifol D. (2016) The treatment of occipital neuralgia: Review of 111 cases. , Neuro-Chirurgie 62(5), 233-240.
- 19.Stiogicza A, Trescot A M. (2016) New technique for cryoneuroablation of the proximal occipital nerve Paper presented at: World Institute of Pain World Congress abstracts;May2016,New York,NY.
- 20.Mueller O, Diener H C, Dammann P. (2013) Occipital nerve stimulation for intractable chronic cluster headache or migraine: a critical analysis of direct treatment costs and complications. , Cephalalgia 33(16), 1283-1291.
Cited by (2)
- 1.Rupert Matthew Paul, Budwany Ryan, Abd-Elsayed Alaa, 2023, , , (), 215, 10.1016/B978-0-323-83007-2.00032-1
- 2.Racz Gabor Bela, Racz Gabor J., Racz Tibor A., 2020, , , (), 523, 10.1007/978-3-030-39982-5_20