Implementing High Energy Liquid Nutrition, Omega-3 Fatty Acids and Nutritional Supplements for the Treatment of Anorexia Nervosa
Abstract
Objective:
To assess the effect of different treatment approaches on the course of anorexia nervosa (AN) over time.
Methods:
The subjects were 27 hospitalized AN patients. In our retrospective analysis we compared weight gain in two groups. While one group was treated with a standard oral refeeding protocol (historical control) through January 2013 (N=16), the second group (highly standardized refeeding protocol) received a high energy liquid nutrition and nutritional supplements including omega-3 fatty acids (N=11).
Results:
On admission, the two groups were comparable in terms of height, weight, age and heart rate. At the end of our monitoring time frame of 25 days, weight gain was 121.4% higher in the highly standardized refeeding protocol group than in the historical control group (66.5 ±52.4 vs 147.3 ±55.7 grams/day; t-Test p=0.004; CI95%: 29.3-132.2). A carbohydrate rich diet clearly improved weight gain if high energy liquid nutrition was replaced by the diet according the patient’s own wishes. About 45% of our patients stated they were vegetarians at admission. However, we could not identify a vegetarian diet as a statistically significant negative prognostic factor for weight gain.
Conclusion:
The highly standardized refeeding protocol seems to be helpful in malnourished AN patients to improve weight gain without enhancing the risk of a refeeding syndrome. However, further studies with greater number of patients are needed to confirm the effectiveness of our standardized treatment protocol.
Author Contributions
Academic Editor: Agnieszka Zyromska, Nicolaus Copernicus University, Collegium Medicum, Faculty of Medicine, Chair and Clinic of Oncology and Brachytherapy
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2017 Christoph Baumann, et al
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction:
AN is reported by 1-4% of women and 0.3 – 0.7% of men in Europe. 2-3% of women have subthreshold eating disorders1. AN is a serious illness leading to substantial morbidity and mortality. The standard mortality ratios for patients with AN and a psychiatric comorbidity are 5.4 (95% confidence interval CI 95%: 4.6-6.4) and 18.1 (CI 95%: 15.2-21.3) for natural and unnatural causes of death, respectively2.
The treatment of AN is often protracted with repeated hospitalizations. Nutritional refeeding is the first therapeutic step to recovery. In case and cohort studies different low and high-calorie diet protocols exist3, 4. There are well known major complications during weight-restauration - called refeeding syndrome: a shift of electrolytes and fluid can occur in malnourished patients leading to (potentially fatal) cardiovascular, respiratory and neurological symptoms. However, prospective and retrospective studies showed that higher calorie diets instituted at admission predicted faster weight gain and less hospitalization in patients with AN5, 6, 7, 8, 9. Such a rapid refeeding protocol was well tolerated with no indicators of refeeding syndrome. Particularly in prospective and randomized trails, tube refeeding proves to be the most effective method for gaining weight without major complications10, 11. Nevertheless, the underlying etiology and pathogenesis as well as the mechanism of weight restauration are still poorly understood.
Genetics and environmental factors appear to play an important role in the pathogenesis and etiology of AN. The HTR1B gene (5-hydroxytryptamine receptor 1B) possibly has an effect on the ‘severity of anxiety in AN-spectrum patients’12. Additionally, recent prospective studies in humans show a link between maternal stress and the development of eating disorders13, 14. Emack and Mathews suggested in an animal study that ‘acute phases of maternal stress in late pregnancy’ had a significant impact on hypothalamus pituitary adrenal axis (HPA) function in adult offspring15. In 2007 Rigaud et al proved an above average increase in the thermic effect of food (TEF), ACTH, cortisol, norepinephrine and dopamine levels in AN patients after an injected gastric load16. However, none of the explanatory models of the pathophysiology has resulted in a concrete treatment plan for AN.
In our own pathophysiological model we proceed on the assumption that AN patients suffer from an autonomic imbalance. We noticed that during refeeding almost all HRV-parameters changed. It seems that AN patients experience a shift from parasympathetic to sympathetic during refeeding which comes along with a higher resting energy expenditure17.
In this study, we examined the effects of two different treatment approaches over the course of AN.
We stepwise changed our treatment protocol by introducing high liquid energy nutrition, omega-3 fatty acids and nutritional supplements according to the NICE protocol. Until now no study has examined the effect of omega-3 fatty acids on the course of AN. Even so studies with heart disease and cancer patients have shown an improved weight gain or a decrease in weight loss under supplementation of omega-3 fatty acids18, 19, 20.
Methods and Patients:
We retrospectively analyzed the clinical charts of all consecutive patients admitted for hospital refeeding due to AN (all patients met DSM criteria for restricting subtype) between 2009-2013 (N=16) and in 2016 (N=11) who had at least one 24 hour Holter ECG (N=27). Based on our recently published model of the impact of nutrition on the autonomic nervous system measured by 24 hour heart rate variability, our refeeding protocol was changed in January 2013. In May 2016 we introduced a highly standardized written refeeding protocol based on a supervision process. Because the treatment of AN was not standardized between January 2013 and May 2016 we excluded all data from this period for the comparison between the two groups. However, we decided to include the data from January 2013 until May 2016 to compare the effect of different nutritional compositions on weight gain.
Standard oral refeeding protocol up to January 2013 (historical control; group 1; N=16): Three meals and two snacks were served based upon the patient’s choice according to a daily consultation with a dietician. Physicians prescribed diets starting around 5 024 kJ (1 200 kcal) per day increased by 837 kJ (200 kcal) per day up to a normal caloric intake between 10 467 kJ (2500 kcal) and 12 560 kJ (3000 kcal) according to patients weight gain. Only two patients received high-energy liquid supplements or nasogastric tube feeding due to insufficient weight gain. Room sitters – mostly nurses – were assigned to observe patients during all meals and snacks, and for 30 minutes after consumption. The weight of each patient was measured each morning using the same weight balance under standardized conditions These conditions included consistent clothing and weighing before the patient visited the bathroom. Fluid balance, blood counts, and serum electrolytes were measured at admission and later according to the doctor’s orders. Nutritional supplements were not routinely given. We performed at least one Holter ECG in this historical control group (Pathfinder TM, Reynolds, Germany).
In May 2016 we introduced a highly standardized written refeeding protocol based upon a supervision process (group 2; N=11): In 2015 we realized a better therapeutic success after introduction of the modified NICE protocol with omega-3-fatty acid supplements. Our model about the effect of nutrition on the autonomic nervous system was published21. For further implementation of the new protocol we performed supervision by an experienced psychologist in which all professional groups participated. The results were published in an internal written protocol: All patients received a high energy liquid nutrition (Fresubin high energy) at admission starting with 42 kJ/kg/day (10 kcal/kg/day) which continuously increased within one week to reach a significant weight gain. In addition to the NICE protocol, we supplemented 2g omega-3-fatty acids per day (2 x 2Tbl Espricot TM) in all patients. Most patients received nutritional supplements: Thiamin 200-400 mg/day, vitamin B complex (Freka Vit TM), multi vitamins (Multivitamin Liechtenstein TM), Zinc, Potassium, Calcium Glycero phosphate and Magnesium. We performed a Holter at admission and before discharge. For better visualization of heart rate variability we introduced a new Holter ECG system (Medilog Darwin TM, Schiller, Switzerland).
The observation program was not changed but we realized an unconscious liberalization of our surveillance routines in response to the better weight gain of our patients.
Statistics
All patients who were admitted in our unit and agreed to the current protocol contributed data to our statistical analysis. Our primary end goal was weight gain. We also measured diet composition, energy intake, body composition and HRV-parameters. Due to staff shortage, the data for these secondary outcomes were partly fragmented. Moreover we could only capture the exact amount of calories eaten by AN patients who were in our hospital between June 2014 and January 2016 (N=22).
We checked the assumption of normality by using the Kolmogorov-Smirnov-Test and viewing boxplots and q-q diagrams. We compared our two groups using unpaired student t-test. The statistical significance threshold accounted for p=0.05. All statistical analyses were performed using SPSS 23 for Windows.
Due to different courses of disease, the length of admission of our patients varied. The in-patient care time averaged out to 30 days (as seen in Table 1). As for the major component of our statistical analysis, weight gain, we decided to make a cut after 25 days. Choosing a later moment would have shrunk the number of patients for statistical analysis, whereas an earlier moment might not have shown different impacts of our treatment regimens.
Table 1. Characteristics of the 27 AN patients| Characteristics at Baseline | Historical control N=16 | Highly standardized refeeding protocol N= 11 | All |
| Group 1 | Group 2 | ||
| Age (y) | 15.47 ±2.03(12.48-20.38) | 14.08 ±1.22 (12.39-16.56) | 14.91 ±1.86 (12.39-20.38) |
| Child´s gender | |||
| Male | 2 (12.50%) | 0 (0%) | 2 (7.41%) |
| Female | 14 (87.50%) | 11 (100%) | 25 (92.59%) |
| Height (cm) | 165.36 ±7.12 (153.50-180.00) | 160.95 ±8.12 (141.40-174.00) | 163.56 ±7.71 (141.40-180.00) |
| Percentile | 53.25 ±31.98 (10.00-99.00) | 43.27 ±28.28(3.00-92.00) | 49.19 ±30.38 (3.00-99.00) |
| Weight at admission (kg) | 40.12 ±5.06 (28.00-51.00) | 37.77 ±5.59(23.70-44.60) | 39.16 ±5.31 (23.70-51.00) |
| Percentile | 7.44 ±11.01 (0.00-31.00) | 6.00 ±8.83 (0.00-24.00) | 6.85 ±10.03 (0-31) |
| BMI (kg/m2) | 14.62 ± 1.05 (11.90-16.20) | 14.52 ±1.40 (11.90-16.80) | 14.58 ±1.18 (11.90-16.80) |
| Weight before disease (kg) | 52.18 ±8.79 (36.00-65.00) N=13 | 53.25 ±4.50 (50.00-60.00) N=8 | 52.59 ±7.33 (36.00-65.00) |
| Disease length (months) | 10.13 ±5.16 (4.00-24.00) | 10.09 ±9.76 (1.00-36.00) | 10.11 ±7.21 (1.00-36.00) |
| Vegetarian/vegan | 8(50%) | 4 (36.36%) | 12 (44.44%) |
| HR bpm | 66.13 ± 12.48 (38.00-86.00) | 62.52 ±13.11 (30.00-81.00) | 64.66 ±12.62 (30.40-86.00) |
| SDNN ms | 248.63 ±64.03 (156.00-362.00) | 242.78 ±85.45 (144.60-419.10) | 246.24 ±71.99 (144.60-419.10) |
| RMSSD ms | 57.00 ±19.93 (32.00-105.00) | 74.02 ±24.46 (47.30-109.70) | 63.93 ±23.06 (32.00-109.70) |
| Length of stay (d) | 24.00 ±8.91 (11,00-40.00) | 38.91 ±17.44 (23.00-85.00) | 30.07 ±14.78 (11.00-85.00) |
Results
During in-patient care neither refeeding syndrome nor other severe complications such as hypophosphatemia or hypokalemia were observed in any patient.
All ratio variables shown in table 1 except ‘length of stay’ ‘RMSSD’ and ‘disease length’ were normally distributed. When we compared vegetarians versus carnivores all major baseline variables were normally distributed.
At the end of our chosen monitoring time frame of 25 days, weight gain was 121.4% higher in the highly standardized refeeding group than in the historical control group (66.5 ±52.4 vs 147.3 ±55.7g/day; t-Test p=0.004; CI95%: 29.3-132.2)(Table 2)
Table 2. Weight history| Characteristics | Historical control N=16 Group 1 | Highly standardized refeeding protocol N= 11 Group 2 |
| Weight at admission (kg) | 40.12 ±5.06 (28.00-51.00) | 37.77 ±5.59(23.70-44.60) |
| Percentile | 7.44 ±11.01 (0.00-31.00) | 6.00 ±8.83 (0.00-24.00) |
| BMI at admission (kg/m2) | 14.62 ± 1.05 (11.90-16.20) | 14.52 ±1.40 (11.90-16.80) |
| Weight at discharge (kg) | 42.17 ±4.84(31.70-53.10) | 42.31 ±6.07 (27.75-49.15) |
| BMI at discharge (kg/m2) | 15.38 ±.95 (13.45-17.07) | 16.25 ±1.40 (13.88-18.84) |
| Weight at discharge/Weight at admission | 1.05 ±0 .03 (0.99-1.13) | 1.12 ±0.07 (1.04-1.28) |
| Caloric intake (kcal/d) | - | 2413.86 ±445.90 kcal N=11 10106 kJ |
| Calories (g/d/kg) | - | 62.86 ±15.83 kcal N=11 263 kJ |
| Weight gain total (kg) | 2.05 ±1.13 (-0.20-3.70) | 4.54 ±2.55 (1.50-10.50) |
| Weight gain day 25 (kg) | 1.66 ±1.31 (-0.60-3.70) N=12 | 3.68 ±1.51 (1.70-5.70) N=8 |
| Weight gain day 25 (g/d) | 66.50 ±52.36 (-24.00-148.00) N=12 | 147.25 ±55.71 (68.00-228.00) N=8 |
In order to visualize and make our study groups more comparable in spite of different baseline values, we studied weight during different days in relation to weight at admission (as seen in Table 3). On day 12 we observed a significant difference between the historical control and the highly standardized refeeding group using unpaired student t-test (p=0.04 CI95%: 0.2-4.9). Over the course of time the p-value decreased.
Table 3. Changes in body weight (percentage of baseline weight)| Days | Historical control Group 1 | Highly standardized refeeding Group 2 | Sig. | CI95% |
| Day 1 | 100 ±0.00 N=16 | 100 ±0.00 N=11 | - | - |
| Day 3 | 99.18 ±2.17 N=14 | 98.65 ±2.46 N=11 | 0.857 | -3.84 |
| Day 8 | 101.38 ±2.44 N=15 | 103.08 ±3.16 N=11 | 0.134 | 0.56-0.70 |
| Day 15 | 102.85 ±2.90 N=13 | 106.07 ±3.61 N=11 | 0.024 | 0.46-5.97 |
| Day 22 | 104.81 ±2.94 N=14 | 108.08 ±4.13 N=11 | 0.03 | 0.35-6.20 |
| Day 29 | 103.28 ±3.53 N=6 | 111.79±4.13 N=3 | 0.014 | 2.31-14.72 |
Based on anamnesis the weight before disease could be determined for 21 out of the 27 AN patients. At discharge, group 2 patients had reached 84.1% (SD: ±4.4) of their original weight compared to 82.3% (SD±10.0) by group 1 patients despite group 2 patients having a lower baseline value (74.4% ±6.3 vs 78.1% ±10.0). Nevertheless, only a single Phase 2 reached a BMI at discharge higher than 18.5 kg/m2.
In accordance with Bardone-Cone et al we observed a high rate of AN patients were vegetarian. About 45% of our patients stated eating some form of vegetarian diet. One patient out of the 27 patients ate meat products irregularly and two AN patients were pesco-vegetarian. During refeeding at our hospital unit there was no attempt to convince patients of a meat diet.
With the Kolmogorov-Smirnov-Test we showed that all variables except ‘length of stay’, ‘RMSSD’ and ‘length of disease’ were normally distributed. Using student t-test we found a significant difference in the length of disease between vegetarians and carnivores (14.63 ±9.81 versus 7.70 ±3.37 months). Although we could only detect small differences in mean calorie intake/kg/day and mean calories/day, we found that vegetarians gained less weight on average than carnivores (1.35 ±2.37 versus 1.81 ±1.42 kg). However, this incongruence was not statistically significant (unpaired student t-test).
In contrast we found a strong correlation between daily mean calorie intake and weight gain until day 20 (Pearson r=0.44 p=0.04). As seen in Figure 2 many patients initially lost weight. Over time our patients gained weight more rapidly. However, at some point most patients stopped gaining weight as rapidly. This occurred especially from day 15-20 when we observed a very low rate of weight gain per kcal compared to day 5-10 (0.02 g/kcal vs 0.11 g/kcal; paired student t-test p=0.00).
Figure 2.Necessary caloric intake for weight gain rises during refeeding. Dashed line depicts caloric intake in kcal/kg/day while solid line shows weight gain per day in % of bodyweight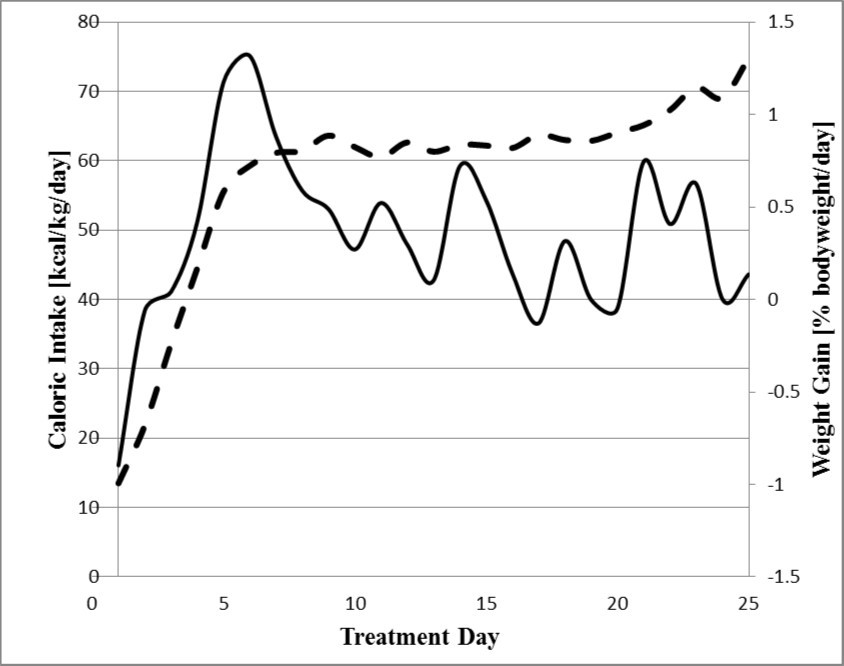
Moreover, we observed a significant better weight gain in 21 patients who received a high-carbohydrate diet with more than 50% carbohydrates and concomitantly lower fat as shown in Figure 3. The better weight gain starts after two weeks, at a time when high energy liquid nutrition was replaced by a diet according the patient’s wishes.
Figure 3.A carbohydrate rich diet improves weight gain Weight gain in 21 patients with a carbohydrate rich diet > 50% and in 16 patients with < 50% carbohydrates according the patient’s own wishes. Daily caloric intake is comparable (9 931 ±251 versus 9 416±247 kJ / 2 372 ± 60 versus 2 249 ± 59 kcal). Unpaired student t-test:p=0.03 for day 20 and p=0.01 for day 25) Carbohydrate rich diet: 14.93% (±2.83) protein, 24.13% (±8.32) fat, 58.38% (±6.85) carbohydrates Carbohydrate poor diet: 14.97% (±3.89) protein, 34.02% (±11.94) fat, 41. 19% (±3.35) carbohydrates
Discussion
This retrospective analysis is result of an internal quality management process of in hospital weight restitution in adolescents suffering from AN. As we were dissatisfied with the weight gain of our patients using a standard refeeding protocol we decided to introduce additional nutritional interventions beginning in January 2013. Our first analysis in 2015 showed a clear trend of improved weight gain but the therapy was still heterogeneous since the entire team did not stick to the changes of the new treatment plan. As a consequence, we implemented supervision by an experienced psychologist in which all professional groups participated. Our team now agreed on a standardization of the treatment protocol and a re-examination of the therapeutic results after one year.
Indeed, we were then able to show a significantly improved weight gain in 11 consecutive patients with AN who were treated with our new refeeding protocol. We did not need a nasogastric tube or anxiolytic medications in ten of the eleven patients. Together with the liberalization of our surveillance routines our refeeding protocol is extremely comfortable for the patients. For methodological reasons it must be stressed that in the past six years there have been no relevant personnel changes in the team.
The Following Considerations have Contributed to the Changes of Our Refeeding Protocol:
1) Vitamin B, Zinc, Potassium, Calcium Glycero phosphate and magnesium supplementation:
This point is based upon a standard for the prevention and management of the refeeding syndrome published from UK’s Institute for clinical Excellence (NICE) published in 2004.
In several studies prophylactic phosphate supplementation was linked to a lower risk of refeeding syndrome, even though patients started on a higher caloric intake (5, 22). Parker et al. concluded that ‘Withholding phosphate supplementation until hypophosphatemia is observed may have the potential of increasing the risk of RFS and slowing down increases in caloric intake required to promote adequate weight restoration during the admission.9
2) High energy liquid nutrition
Rigaud et al published a randomized trial which evaluates the long term effects of tube refeeding with a relatively high number of patients (n=41)(11). Compared to other refeeding protocols the study conducted by Rigaud et al showed the best weight gain by nasogastric tube feeding and seems to be the current "gold standard". We proved that oral high energy liquid nutrition can have a comparable benefit (Figure 1). At day 8 the Rigaud refeeding group achieved a 0.08 percent point higher weight gain (103.16 vs 103.08) than the highly standardized refeeding group. Over time the gap increased steadily. At day 15 the difference accounted for 1.40 percent points (107.47 vs 106.07). At day 29 the Rigaud tube refeeding group reached 116.67% of their admission weight compared to 111.79% by the highly standardized refeeding group. However, it should be taken into account that the groups compared here are very heterogeneous. Most of all, the Rigaud tube feeding group had a higher age (22.5±4.5 vs 14.74±1.93) and a lower BMI at admission (12.1±1.5 vs 14.53±1.37) than the patients admitted in our hospital unit. It should also be noted that data from another study, in which a comparable group of children were treated according to a standard refeeding protocol, was in harmony with our historical control23.
Figure 1.Similar weight gain in highly standardized refeeding group and tube refeeding patients. Comparison of bodyweight in % of day 1 between highly standardized refeeding group, historical control and the Rigaud et al refeeding group.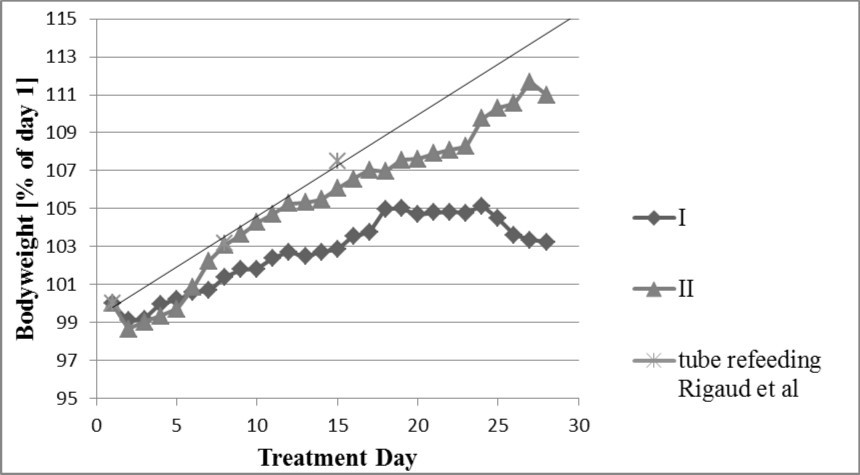
We agree with D. Rigaud et al who showed that infused gastric loads triggered a dose-dependent increase in the thermic effect of food16. Our data now confirms in a clinical setting that we need a higher caloric intake after 10 days of refeeding to reach the same weight gain as within the early refeeding. The reason behind the difficulties for AN patients in gaining weight could be an increase in plasma cortisol, ACTH and catecholamine’s after caloric intake16.
Further, our data clearly showed a better weight gain in patients with a carbohydrate rich nutrition. We are aware of the higher risk of a refeeding syndrome in these patients as recently proved by Gilles et al in an animal model24. However, we did not observe a refeeding syndrome while using supplements according the NICE guideline. Moreover omega-3 fatty acid supplementation decreases hepatic expression of lipogenic genes in a high-carbohydrate refeeding model in rats25.
Surprisingly we could not find a significant difference in weight gain between vegetarians and carnivores. Presumably we were able to compensate nutritional deficiencies in vegetarians which would have worsened the course of disease.
3) Omega-3-fatty acid supplementation
A deficit in membrane long chain poly-unsaturated fatty acids appeared in patients with AN 26. This observation may explain the beneficial effect of omega-3 fatty acid supplementation during refeeding27. While looking for a therapeutic approach to smooth the "autonomic switch" from parasympathetic to sympathicotonic28 and the increase of heart rates during refeeding we expect a beneficial effect of omega-3-fatty acids on heart rate variability as shown in adults with psychiatric disorders29, 30. Omega-3-fatty acids might help to influence the shift from parasympathetic to sympathetic because of their HRV-normalizing and neurophysiological properties31, 32, 33, 34, 35, 36.
These beneficial effects of omega-3-fatty acids on heart rate variability occur in children as well36. We implemented omega-3 fatty acid supplementation in our refeeding protocol for all patients in group 2.
For a better understanding we routinely use Holter ECG monitoring to detect the switch of the autonomic nervous system from the so called ‘hibernating circuit’ with low heart rates and high HRV to the stress reaction with higher heart rates, low HRV and probably a higher resting energy expenditure after the first ten days of refeeding. Beneath the metabolic changes in our view the ‘hibernating circuit’ seems to delete the emotional memory of the patients, which is reawakened during nutritional rehabilitation37. This ‘flash back’ of negative emotions may explain the resistance of the patients regarding nutrition therapy after reaching the caloric gain of 8 374-10 467 kJ/day (2 000-2 500 kcal/day) after the first treatment week. Routine supplementation of omega-3-fatty acids may help smooth the ‘autonomic switch’ and help the patient to tolerate the loss of the ‘vagal brake’38.
As expected weight gain in patients with AN mainly depended on caloric intake and a carbohydrate rich diet. However, caloric restriction is the leading characteristic of AN and the therapeutic goal of each therapy is to improve weight gain by a higher caloric intake without a higher risk of a refeeding syndrome. The different therapeutic approaches are characterized by different methods to improve weight gain for example by strength observation of caloric intake or nasogastric tubes. We decided to enhance caloric intake through the use of high energy liquid nutrition, nutritional supplements paired with a new therapeutic approach to weaken the autonomic switch from high vagal to low vagal activities during refeeding by using omega-3 fatty acids. We were not able to prove which therapeutic change was the most important: Omega-3 fatty acid supplementation or high energy liquid nutrition. This question must be proven by a prospective randomized trial. Moreover, our study had three major limitations: 1) The study group was too small to draw general conclusions and we had missing data 2) We did not examine the long term effects of our therapy, therefore we could not make any assertion about the relapse rate. 3) The groups were partly heterogeneous regarding ‘length of disease’, ‘RMSSD’ and ‘length of stay’. In our analysis we bypassed the problem of the differences in ‘length of stay’ by making a cut after 25 days. Considering the fact that most patients do not even approximately reach their weight before disease during refeeding, longer hospital stays should be contemplated. For the variable ‘length of disease’ it is conceivable that the statements of patients also depended on the examiner since it is often hard to determine the exact point of the start of the disease.
However, we were happy to deescalate our refeeding in patients with AN and improve weight gain with the current refeeding protocol.
Conclusions
In summary, we recommend introducing high energy liquid nutrition with a sufficient share of carbohydrates and nutritional supplements including omega-3 fatty acids for an improved weight gain during refeeding. Because of an increasing energy turnover during refeeding, caloric intake should be adjusted. In terms of a rapid weight gain nasogastric tube refeeding remains the benchmark. Nevertheless, regarding the disagreeableness accompanied by tube refeeding, the pros and cons should always be studied for each patient before inserting a nasogastric tube.
For a better understanding of the AN pathophysiology we are now analyzing the 24-hour ECGs.
Abbreviations
AN = anorexia nervosa
bpm = beats per minute
cm = centimeter
d= day
g = gram
HRV = heart rate variability
kilogram = kg
kJ = kilojoule
m = meter
ms =milliseconds
y = year
Acknowledgments
We would like to thank Kate Keller, student of University of Oregon, USA, for editorial support.
Affiliations
Christoph Baumann: University of Wuerzburg, Medical Faculty, Josef-Schneider-Straße 2, Würzburg, Germany
Christian Willaschek, Tuende Kertess-Szlaninka, Lang Johanna, Reiner Buchhorn: Caritas-Krankenhaus Bad Mergentheim, Department of Pediatrics, Uhlandstraße 7, Bad Mergentheim, Germany
References
- 1.Keski-Rahkonen A, Mustelin L. (2016) Epidemiology of eating disorders in Europe: prevalence, incidence, comorbidity, course, consequences, and risk factors. , Curr Opin Psychiatry; 29(6), 340-5.
- 2.Kask J, Ekselius L, Brandt L, Kollia N, Ekbom A et al. (2016) Mortality in Women With Anorexia Nervosa: The Role of Comorbid Psychiatric Disorders. , Psychosom 78(8), 910-9.
- 3.Garber A K, Sawyer S M, Golden N H, Guarda A S, Katzman D K et al. (2016) A systematic review of approaches to refeeding in patients with anorexia nervosa. , Int J Eat Disord; 49(3), 293-310.
- 4.Redgrave G W, Coughlin J W, Schreyer C C, Martin L M, Leonpacher A K et al. (2015) Refeeding and weight restoration outcomes in anorexia nervosa: Challenging current guidelines. , Int J Eat 48(7), 866-73.
- 5.Madden S, Miskovic-Wheatley J, Clarke S, Touyz S, Hay P et al. (2015) Outcomes of a rapid refeeding protocol in Adolescent Anorexia Nervosa. , J Eat Disord; 3, 8.
- 6.O’Connor G, Nicholls D, Hudson L, Singhal A. (2016) Refeeding Low Weight Hospitalized Adolescents With Anorexia Nervosa: A Multicenter Randomized Controlled Trial. Nutr Clin Pract.
- 7.Kohn M R, Madden S, Clarke S D. (2011) Refeeding in anorexia nervosa: increased safety and efficiency through understanding the pathophysiology of protein calorie malnutrition. , Curr Opin Pediatr; 23(4), 390-4.
- 8.Smith K, Lesser J, Brandenburg B, Lesser A, Cici J et al. (2016) Outcomes of an inpatient refeeding protocol in youth with Anorexia Nervosa and atypical Anorexia Nervosa at Children's Hospitals and Clinics of Minnesota. , J Eat Disord; 4, 35.
- 9.Parker E K, Faruquie S S, Anderson G, Gomes L, Kennedy A et al. (2016) Higher Caloric Refeeding Is Safe in Hospitalised Adolescent Patients with Restrictive Eating Disorders. , J Nutr Metab.2016: 5168978.
- 10.Born C, L de la Fontaine, Winter B, Muller N, Schaub A et al. (2015) First results of a refeeding program in a psychiatric intensive care unit for patients with extreme anorexia nervosa. , BMC Psychiatry; 15, 57.
- 11.Rigaud D, Brondel L, Poupard A T, Talonneau I, Brun J M. (2007) A randomized trial on the efficacy of a 2-month tube feeding regimen in anorexia nervosa: A 1-year follow-up study. , Clin Nutr; 26(4), 421-9.
- 12.Hernandez S, Camarena B, Gonzalez L, Caballero A, Flores G et al. (2016) A family-based association study of the HTR1B gene in eating disorders. , Rev Bras Psiquiatr; 38(3), 239-42.
- 13.O'Connor T G, Ben-Shlomo Y, Heron J, Golding J, Adams D et al. (2005) Prenatal anxiety predicts individual differences in cortisol in pre-adolescent children. , Biol Psychiatry; 58(3), 211-7.
- 14.O’Connor T G, Heron J, Glover V, Alspac Study T. (2002) Antenatal anxiety predicts child behavioral/emotional problems independently of postnatal depression. , J Am Acad Child Adolesc Psychiatry 41(12), 1470-7.
- 15.Emack J, Matthews S G. (2011) Effects of chronic maternal stress on hypothalamo-pituitary-adrenal (HPA) function and behavior: no reversal by environmental enrichment. , Horm Behav; 60(5), 589-98.
- 16.Rigaud D, Verges B, Colas-Linhart N, Petiet A, Moukkaddem M et al. (2007) Hormonal and psychological factors linked to the increased thermic effect of food in malnourished fasting anorexia nervosa. , J Clin Endocrinol Metab; 92(5), 1623-9.
- 17.Dippacher S, Willaschek C, Buchhorn R. (2014) Different nutritional states and autonomic imbalance in childhood. , Eur J Clin Nutr; 68(11), 1271-3.
- 18.Mehra M R, Lavie C J, Ventura H O, Milani R V. (2006) Fish oils produce anti-inflammatory effects and improve body weight in severe heart failure. , J Heart Lung Transplant; 25(7), 834-8.
- 19.Bayram I, Erbey F, Celik N, Nelson J L, Tanyeli A. (2009) The use of a protein and energy dense eicosapentaenoic acid containing supplement for malignancy-related weight loss in children. , Pediatr Blood Cancer; 52(5), 571-4.
- 20.Pappalardo G, Almeida A, Ravasco P. (2015) Eicosapentaenoic acid in cancer improves body composition and modulates metabolism. , Nutrition; 31(4), 549-55.
- 21.Clancy J A, Mary D A, Witte K K, Greenwood J P, Deuchars S A et al. (2014) Non-invasive vagus nerve stimulation in healthy humans reduces sympathetic nerve activity. , Brain Stimul; 7(6), 871-7.
- 22.Leitner M, Burstein B, Agostino H. (2016) Prophylactic Phosphate Supplementation for the Inpatient Treatment of Restrictive Eating Disorders. , J Adolesc Health; 58(6), 616-20.
- 23.Robb A S, Silber T J, Orrell-Valente J K, Valadez-Meltzer A, Ellis N et al. (2002) Supplemental nocturnal nasogastric refeeding for better short-term outcome in hospitalized adolescent girls with anorexia nervosa. , Am J Psychiatry; 159(8), 1347-53.
- 24.Giles E D, Hagman J, Pan Z, MacLean P S, Higgins J A. (2016) Weight restoration on a high carbohydrate refeeding diet promotes rapid weight regain and hepatic lipid accumulation in female anorexic rats. , Nutr Metab (Lond); 13, 18.
- 25.de Castro GS, Cardoso J F, Calder P C, Jordao A A, Vannucchi H. (2015) Fish oil decreases hepatic lipogenic genes in rats fasted and refed on a high fructose diet. , Nutrients; 7(3), 1644-56.
- 26.Caspar-Bauguil S, Montastier E, Galinon F, Frisch-Benarous D, Salvayre R et al. (2012) Anorexia nervosa patients display a deficit in membrane long chain poly-unsaturated fatty acids. , Clin Nutr; 31(3), 386-90.
- 27.Ayton A K, Azaz A, Horrobin D F. (2004) A pilot open case series of ethyl-EPA supplementation in the treatment of anorexia nervosa. Prostaglandins Leukot Essent Fatty Acids;. 71(4), 205-9.
- 28.Buchhorn R. (2016) The Impact of Nutrition on the Autonomic Nervous System. , Int J Food Nutr Sci 3(3), 1-16.
- 29.Carney R M, Freedland K E, Stein P K, Steinmeyer B C, Harris W S et al. (2010) Effect of omega-3 fatty acids on heart rate variability in depressed patients with coronary heart disease. , Psychosom Med; 72(8), 748-54.
- 30.Grung B, Hansen A L, Berg M, Moen-Knudseth M P, Olson G et al. (2015) Exploratory multivariate analysis of the effect of fatty fish consumption and medicinal use on heart rate and heart rate variability data. , Front Psychol; 6, 135.
- 31.Barbadoro P, Annino I, Ponzio E, Romanelli R M, D'Errico M M et al. (2013) Fish oil supplementation reduces cortisol basal levels and perceived stress: a randomized, placebo-controlled trial in abstinent alcoholics. Mol Nutr Food Res;. 57(6), 1110-4.
- 32.Goncalves C G, Ramos E J, Suzuki S, Meguid M M. (2005) Omega-3 fatty acids and anorexia. , Curr Opin Clin Nutr Metab Care; 8(4), 403-7.
- 33.Keenan K, Hipwell A E, Bortner J, Hoffmann A, McAloon R. (2014) Association between fatty acid supplementation and prenatal stress in African Americans: a randomized controlled trial. , Obstet Gynecol; 124(6), 1080-7.
- 34.Prior P L, Galduroz J C. (2012) (N-3) Fatty acids: molecular role and clinical uses in psychiatric disorders. , Adv Nutr; 3(3), 257-65.
- 35.Sauder K A, Skulas-Ray A C, Campbell T S, Johnson J A, Kris-Etherton P M et al. (2013) Effects of omega-3 fatty acid supplementation on heart rate variability at rest and during acute stress in adults with moderate hypertriglyceridemia. , Psychosom Med; 75(4), 382-9.
- 36.Christensen J H. (2011) Omega-3 polyunsaturated Fatty acids and heart rate variability. , Front Physiol; 2, 84.
Cited by (5)
This article has been cited by 5 scholarly works according to:
Citing Articles:
R. Buchhorn, Julian Buchhorn - (2020) Semantic Scholar
C. Baumann, C. Willaschek, R. Buchhorn - (2019) Semantic Scholar
R. Buchhorn, U Rakowski, C. Willaschek, C. Baumann - Journal of Heart Health (2019) Semantic Scholar
Journal of Heart Health (2019) OpenAlex
