Kynurenines and Vitamin B6: Link Between Diabetes and Depression.
Abstract
The increased association between depression and diabetes mellitus is generally acknowledged. Recent studies suggest that depression leads to diabetes.However, the underlying molecular mechanisms for this association remain unclear.Literature and our data indicate that inflammatory and/or stress factors in depression up-regulate tryptophan (TRP) conversion into kynurenine (KYN), a substrate for nicotinamide adenine dinucleotide (NAD) biosynthesis. Deficiency of vitamin B6, a co-factor of the key enzymes of KYN – NAD pathway, shunts KYN metabolism from formation of NAD towards production of xanthurenic (XA) and kynurenic (KYNA) acids. Human and experimental studies reveal that XA, KYNA and their metabolites interfere with production, release and biological activity of insulin. We propose that inflammation- and/or stress-induced up-regulation of TRP – KYN metabolism in combination with vitamin B6 deficiency is one of the mechanisms mediating increased risk of diabetes in depression. Consequently, monitoring formation of diabetogenic KYN derivatives might help to identify subjects-at-risk for the development of diabetes. Pharmacological down-regulation of the TRP – KYN – NAD pathway and maintenance of adequate vitamin B6 status might help to prevent the development of diabetes in depression and other conditions associated with inflammation/stress–induced excessive production of KYN and vitamin B6 deficiency, e.g., obesity, cardiovascular diseases, aging, menopause, pregnancy, and hepatitis C virus infection.
Author Contributions
Academic Editor: Lu Qi, Assistant Professor of Medicine; Harvard Medical School Assistant Professor of Nutrition; Harvard School of Public Healthe
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2013 Gregory Oxenkrug, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
Paul Summergrad is a non-promotional speaker for CME outfitters, Inc., and consultant and non-promotional speaker for Pri-med, Inc.
Citation:
Introduction
The increased association between depression and diabetes mellitus is generally acknowledged 1, 2. Observation of 65% increase risk for development of (mostly type 2) diabetes in a prospective study of clinically depressed patients 3 supports the hypothesis that depression leads to diabetes 4. Molecular mechanisms that mediate the increased risk of diabetes in depression remain undetermined although some hypotheses discussed elsewhere 2. Current review focuses on tryptophan (TRP) – kynurenine (KYN) – nicotinamide adenine dinucleotide (NAD) metabolism in depression; the effects of vitamin B6 on KYN – NAD metabolism, and on the effects of KYN and some of its derivatives on production, release and biological activity of insulin. Literature and our data suggest that depression is associated with the increased production of KYN from TRP in response to activation of rate-limiting enzymes of the TRP – KYN pathway induced by pro-inflammatory cytokines and/or stress hormones, and with deficiency of vitamin B6, a co-factor to the key enzymes of KYN - NAD metabolism. These two conditions are necessary for the formation of diabetogenic KYN derivatives.
Tryptophan – Kynurenine Metabolic Pathway.
Tryptophan (TRP) is an essential (for humans) amino acid. About 5 % of non-protein routes of TRP metabolism is utilized for formation of methoxyindoles: serotonin, N-acetylserotonin and melatonin 5, 6, Figure 1.
Figure 1.Vitamin B6 deficiency-induced shift of post-KYN metabolism from biosynthesis of NAD towards formation of diabetogenic KYN derivatives.
The major non-protein route of TRP metabolism is formation of KYN, catalyzed by rate-limiting enzymes: indoleamine 2,3-dioxygenase (IDO) or TRP 2,3-dioxygenase (TDO) 6. IDO is activated by pro-inflammatory factors, e.g., interferon-gamma (IFNG), tumor necrosis factor-alpha, IL-1 beta, and lipopolysaccharide, while TDO is inducible by stress hormones, e.g., cortisol, prolactin, and by substrate, TRP 5 .
Kynurenine – Nicotinamide Adenine Dinucleotide Metabolic Pathway.
KYN is substrate for two post-KYN metabolic pathways:
1). Formation of kynurenic acid (KYNA), catalyzed by KYN aminotransferases (KAT) 7. KYNA -is a NMDA 6 and α-7 Nicotinic Acetylcholine Receptors antagonist 7 and a precursor of quinaldic acid (QA) 8; and
2). Formation of 3-hydroxyKYN (3-HK) catalyzed by KYN 3-monooxygenase5, 6.
3-HKis a substrate for two metabolic pathways:
1). Formation of nicotinamide adenine dinucleotide (NAD). The first step of the 3-HK – NAD pathway catalyzed by kynureninase; and
2). Formation of xanthurenic acid (XA), catalyzed by 3-HK-transaminase 9. XA is a precursor of 8-hydroxyquinaldic acid (8-HQ) 10.
Pyridoxal 5'-Phosphate and KYN – NAD Metabolic Pathway.
Pyridoxal 5'-phosphate (P5P), an active form of vitamin B6, is a cofactor for >100 metabolic reactions, including key enzymes of post-KYN metabolism: KYN 3-monooxygenase,KAT and kynureninase, - the latter enzyme is particularly sensitive to dietary vitamin B-6 restriction 11. Down-regulation of kynureninase, caused by P5P deficiency, shifts 3-HK metabolism from formation of NAD to production of XA and KYNA 12. P5P deficiency combined with up-regulated TRP conversion to KYN leads to increased availability of 3-HK as substrate for formation of XA and 8-HQ, and increased availability of KYN as substrate for KYNA and QA in the cerebellum, corpus striatum, frontal cortex, and pons/medulla 13, blood 14 and peripheral organs 15, 17 including pancreatic islets 15.
Vitamin B6 depletion results in drastic increase while vitamin B6 supplementation normalizes urinary 3-HK and XA after TRP load in cardiac 19 and obese 20 patients and rats 16.The other consequence of P5P deficiency-induced down-regulation of kynureninase is the decreased formation of NAD that subsequently inhibits synthesis and secretion of insulin and triggers death of pancreatic beta-cells 21, 22. Considering that NAD inhibits TDO, decreased formation of NAD caused by P5P deficiency might result in further activation of TDO and increased production of KYN 23.
Besides P5P deficiency, kynureninasemight be inhibited by XA, thus sustaining the accumulation of 3-HK, KYNA, KYN and XA at the expense of NAD production 24. Additionally, XA might perpetuate P5P deficiency by inhibiting pyridoxal kinase, the enzyme which catalyzes the formation of P5P from vitamin B6 25.
Diabetogenic Effects of KYN Derivatives.
Xanthurenic Acid
XA was the first KYN metabolite to be observed in the increased amounts in the urine samples of type 2 diabetes patients in comparison with in healthy subjects 26. Recent study found the increased levels of XA precursors, KYN and 3-HK in serum samples of diabetic rethinopathy patients 27. XA induced experimental diabetes in rats 28.
The possible mechanisms mediating XA contribution to the development of diabetes are 1). Formation of chelate complexes with insulin (XA-In). As antigens, XA-In complexes are indistinguishable from insulin but have 49% lower activity than pure insulin 28; 2). Formation of Zn++-ions – insulin complexes in β-cells that exert toxic effect in isolated pancreatic islets 29, 30; 3). Inhibition of insulin release from rat pancreas 15; and 4). Induction of pathological apoptosis of pancreatic beta cells through caspase-3 dependent mechanism 31, 32.
Kynurenic Acid
KYNA was found to be increased in urine of nonhuman primate and mouse models of type 2 diabetes mellitus in a recent metabolomic study 33, and in patients with diabetic retinopathy 27. The possible mechanisms of diabetogenic effect of KYNA might be related to KYNA ability to block NMDA receptors. Thus, NMDA antagonist and pharmacological precursor of KYNA, 7-chlorokynurenic acid, 6 and NMDA antagonist, MK-801, negated the inhibition of glucose production by NMDA agonists injected into dorsal vagal complex in rodents 34.
In addition, XA, KYNA, and their derivatives, QA and 8-HQ, inhibit pro-insulin synthesis in isolated rat pancreatic islets 35
Recent study revealed elevated expression of IDO in serum samples of diabetic retinopathy patients 27. In the same vein, surplus dietary TRP, the substrate for formation of KYNA and XA, induced insulin resistance in pigs 36.
KYN/TRP Ratio and Neopterin As Clinical Markers of Up-Regulation of TRP – KYN Pathway.
Plasma (serum) ratio of KYN to TRP (KTR) is a generally accepted clinical marker of IDO activity 14. Considering that both IDO and TDO regulate the rate of TRP conversion into KYN 37, plasma concentrations of TRP and KYN might be affected by the activity of stress hormone inducible TDO as well. However, KTR does reflect IDO activity in conditions associated with inflammation 38.
Concurrently with induction of IDO, pro-inflammatory factors (e.g., IFNG, TNF-alpha, IL-1beta) induce guanosine triphosphate cyclohydrolase I (GTPCH), a rate-limiting enzyme in the biosynthesis of tetrahydrobiopterine (BH4), the obligatory cofactor of nitric oxide (NO) synthase (NOS) 39. GTPCH catalyzes GTP conversion into 7,8-dihydroneopterin (BH2) 39. Pro-inflammatory factors-induced activation of GTPCH results in increased formation of neopterin, a stable, water-soluble derivative of BH2 39. Therefore, inflammation increased formation of neopterin might be considered not simply a clinical marker of inflammation, but an indirect marker of IDO activation as well 35.
Blood neopterin levels correlate with KTR in healthy humans 40, and cardiovascular patients 14. We found similar strong (r=0.68) and highly significant (p<0.0001) correlation between serum KTR ratio and neopterin in 80 hepatitis C virus (HCV) patients treated with interferon-alpha (unpublished data).
Kynurenine/Tryptophan Ratio, Neopterin and P5P Levels in Depression and Diabetes.
Diabetes
Clinical and experimental data suggest there is increased metabolism of TRP in diabetes, most likely, resulting from up-regulation of TRP – KYN pathway. Thus, impaired accumulation of TRP in the brain concomitantly with a much faster disappearance of the administered TRP from the bloodstream was observed in streptozotocin-diabetic rats after TRP load 41. Similarly, post-loading levels of plasma TRP (but not of other large neutral amino acids) increased less in diabetic patients than in healthy controls. 42
Recent studies reveal decreased plasma TRP concentrations and increased KYN and KTR in 21 hemodialysis patients with diabetes in comparison with 40 healthy controls patients. An increase in neopterin was correlated with KYN concentrations (r = 0.393, p < 0.01), suggesting that increased TRP degradation is a result of IDO activation 43. In the same vein, neopterin but not C-reactive protein, an inflammation marker not related to IDO/GTPCH activation, increased in diabetic in comparison with non-diabetic patients with critical limb ischemia 44.
Decreased kynureninase activity was observed in liver and kidney of alloxan diabetic rabbits 45. Additionally, XA was identified in pre-diabetes subjects 46.
Neopterin correlated with insulin resistance, an early event in the pathogenesis of type 2-diabetes,in Caucasian population 14, 47, 48. We observed correlation between plasma neopterin concentrations and IR (HOMA-IR, r=0.08, P <0.03), and P5P (r =−0.13, P = 0.002) in 592 adult (45–75 years of age) participants of community dwellers in the Boston Puerto Rican Health Study. The strongest (r=0.15) and most significant (P<0.0002) correlation was recognized between HOMA-IR and neopterin/P5P ratio (a combined index of increased inflammation and P5P deficiency) 49. Low plasma concentrations of P5P have been reported in conditions associated with increased fasting glucose and glycated hemoglobin 50.
These results are in line with our hypothesis that combined up-regulation of the TRP – KYN pathway (as indirectly assessed by neopterin concentrations) and P5P deficiency serves as one of the mechanisms promoting the development of diabetes in depression.
Depression
It was initially suggested in 1969 that stress-induced TDO activation shunts TRP metabolism from formation of serotonin towards production of KYN in depression 34, 51. The discovery of inflammation-inducible IDO added another mechanism of up-regulation of KYN formation from TRP in depression 52. Association of depression with an increased production of cortisol 53 and inflammatory factors 54 is described elsewhere. Both IDO and TDO activation leads to the same major consequences: 1). Deficiency of formation of serotonin (and its metabolites, melatonin and N-acetylserotonin) contributing to insomnia, dysregulation of biological rhythms and impaired neurogenesis observed in depression 55, 56, 57; 2). Up-regulated formation of KYN and its neuroactive derivatives which exert anxiogenic, pro-oxidative and cognitive impairment effects typical for depression 58, 59, Figure 2.
Figure 2.Shift of tryptophan metabolism in depression.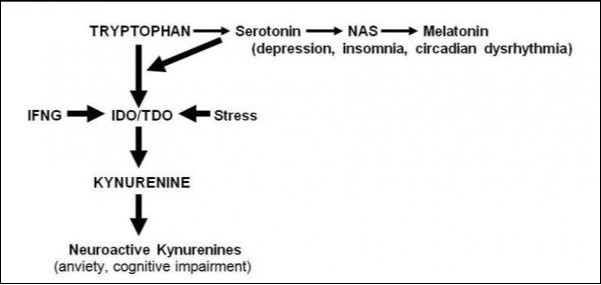
Increased plasma neopterin levels were reported in depressed patients, further supporting the notion of IDO activation in depression 60.
Low plasma concentrations of P5P have been reported in depression 61. An increase in KTR and a deficiency in vitamin B6 might explain the increased production of XA in depressed patients 62.
Hepatitis C virus, Depression and Diabetes
Depression is the often side-effect of interferon (IFN)-alpha administration to patients with hepatitis C virus (HCV) infection. There is a strong correlation between increased production of KYN (and its derivatives) and severity of IFN-alpha-induced depression 63. We found an increased frequency of the carriers of high producer (T) allele, of IFNG(+874) T/A gene, that encodes production of IFNG protein, among HCV patients suffering from depression as a side-effect of IFN-alpha treatment 64. Serum neopterin concentrations were higher in HCV versus the non-HCV patient population 65 and predicted resistance to IFN-alpha therapy 66. We found strong (r=0.68) and significant (p<0.0001) correlation between the serum KTR ratio and neopterin in 80 hepatitis C virus (HCV) patients treated with interferon-alpha (unpublished data).
Incidence of diabetes is higher among HCV patients than in non-HCV population 67. HCV infection significantly lowers vitamin B6 levels 68. IFN-alpha treatment was associated with increased risk of developing insulin resistance and higher incidence of type 2 diabetes in comparison with the groups of both non-viral chronic liver disease 69 and patients with chronic hepatitis B virus 70. Moreover, antecedent HCV infection markedly increases the risk of developing diabetes in susceptible subjects, while even non-diabetic HCV patients have insulin resistance and specific defects in the insulin-signaling pathway 71. These data are in line with the current hypothesis that increased risk of IR in HCV patients depends on a combination of inflammation (e.g.,IFNG)-triggered up-regulation of TRP – KYN metabolism with P5P deficiency-induced dysregulation of KYN – NAD metabolic pathway.
Therapeutic Interventions
Current hypothesis suggests that prevention and treatment of diabetes in depression and other conditions associated with chronic stress or chronic Th-1 type inflammation should include the supporting of adequate vitamin B6 status 72 and pharmacological modifications of TRP – KYN metabolism, aimed at down-regulating the formation of diabetogenic KYN derivatives. The latter may be achieved by administration of IDO and TDO inhibitors. The known IDO inhibitor, 1-methyl-L-TRP 73, is not available for human use. There are two IDO inhibitors available for human use: minocycline, an antibiotic with anti-inflammatory action 74, 75, and antidepressant, wellbutrin 76. It is noteworthy that wellbutrin, contrary to tricyclic antidepressants, has a favorable metabolic profile 77. The strongest IDO inhibitor is berberine, an isoquinoline alkaloid isolated from Berberisaristata, an herb widely used in Indian and Chinese systems of medicine 78. Berberine exerts therapeutic potential in diabetic hamsters 79 and diabetic patients 80, 81. It is noteworthy that both berberine and minocycline prolonged life span and improved health span in the Drosophila model 82, 83. Consistent with our hypothesis, is observation that vitamin B6 supplementation dose-dependently decreased insulin levels and improved insulin resistance in KK-A(y) mice, an animal model of obese, type 2 diabetes 84, 85. Current hypothesis is in line with the previously published reports of the neuroprotective effect of vitamin B6 86 and its contribution to regulation of choline and docosahexaenoic acid concentrations 87, 88.
Conlusions
Review of literature and our data suggest that one of the mechanisms for the increased incidence of diabetes in depressed subjects might be up-regulation of TRP - KYN metabolism in combination with P5P deficiency, resulting in excessive formation of diabetogenic KYN derivatives.
Monitoring of KYN/P5P status and formation of XA and KYNA in depressed patients might help to identify subjects-at-risk for the developing of diabetes.
Support for adequate B6 status 89 and inhibition of TRP- KYN metabolism might prevent development of diabetes associated with depression.
This proposed biochemical mechanism may explain the increased risk of diabetes not only in depression, but in other conditions associated with the combined up-regulation of KYN production and P5P deficiency, e.g., obesity, cardiovascular disorders 90, menopause, aging , HCV and treatment with IFN-alpha 91. It is noteworthy that pro-inflammatory activity of adipokines 92 and KYN production in white adipose tissue 93 has been reported along with higher plasma KTR in obese, but not lean, subjects 94.
Abbreviations
TRP – tryptophan; IFNG – interferon-gamma; IDO – indoleamine 2,3-dioxygenase; KYN – kynurenine; KMO – KYN-3-monooxygenase;3-HK – 3-hydroxyKYN; P5P – pyridoxal 5'-phosphate; QUIN – quinolinic acid; NAD – nicotinamide adenine dinucleotide; KYNA – kynurenic acid; XA – xanthurenic acid; QA - quinaldic acid; 8-HQ – 8-hydroxyquinaldic acid; GTP – guanosine triphosphate; GTPCH – GTP cyclohydrolase I; BH2 - 7,8-dihydroneopterin;. BH4 – tetrahydrobiopterin; NOS – nitric oxide synthase
Acknowledgements
GF Oxenkrug is a recipient of NIMH099517 grant.
References
- 3.Campayo A, P de Jonge, J F Roy, Saz P, de la Camara et al. (2010) . , Am. J. Psychiatry 167, 580-588.
- 7.Albuquerque E X, Schwarcz R. (2012) Biochem,Pharmacol.2012Dec24. [Epub ahead of print]. 0006-2952.
- 21.J P Gray, K N Alavian, E A Jonas, E A Heart. (2012) . , Am. J. Physiol. Endocrinol. Metab 303, 191-9.
- 24.Shibata Y, Ohta T, Nakatsuka M, Ishizu H, Matsuda Y. (1996) . Advances in Experimental Medicine and Biology 40, 55-58.
- 27.P K Munipally, S G Agraharm, Valavala V K, Gundae S, N R Turlapati. (2011) . , Archives of Physiology and Biochemistry 117, 254-258.
- 32.Wang Q, Chen J, Wang Y, Han X, Chen X. (2012) . PLoS One 7:e38522. doi: 10.1371/journal.pone.0038522
- 39.Sucher R, Schroecksnadel K, Weiss G, Margreitera R, Fuchs D et al. (2010) . , Cancer Letters 287, 13-22.
- 41.Masiello P, Balestreri E, Bacciola D, Bergamini E. (1987) Influence of experimental diabetes on brain levels of monoamine neurotransmitters and their precursor amino acids during tryptophan loading. , Acta Diabetol Lat 24, 43-50.
- 42.Fierabracci V, Novelli M, A M Ciccarone, Masiello P, Benzi L. (1996) . , Diabetes Metab 22, 51-6.
- 43.Koenig P, Nagl C, Neurauter G, Schennach H, Brandacher G et al. (2010) . , Clin. Nephrol 74, 465-70.
- 44.Bertz L, Barani J, Gottsäter A, P M Nilsson, Mattiasson I et al. (2006) . , Int. Angiol.25 370-7.
- 45.Allegri G, Zaccarin D, Ragazzi E, Froldi G, Bertazzo A et al. (2003) . , Adv. Exp. Med. Biol 527, 387-93.
- 48.Schennach H, Murr C, Gächter E, Mayersbach P, Schönitzer D et al. (2002) . , Clinical Chemistry 48, 643-645.
- 49.Oxenkrug G, K L Tucker, Requintina P, Summergrad P. (2011) . , American Journal of Neuroprotection and Neuroregeneration 3, 48-52.
- 64.Oxenkrug G, Perianayagam M, Mikolich D, Requintina P, Shick L. (2011) . , J. Neural. Transm 118, 271-274.
- 66.G F Oxenkrug, Requintina R J, D L Mikolich, Ruthazer R, Viveiros K. (2012) . Hepatitis Research and Treatment. 2012, 4 pages, Article ID 619609.
- 76.Brustolim D, Ribeiro-dos-Santos R, R E Kast, E L Altshuler. (2006) . , Int. Immunopharmacol 6, 903-90.
- 81.F Di Pierro, Villanova N, Agostini F, Marzocchi R, Soverini V. (2012) Pilot study on the additive effects of berberine and oral type 2 diabetes agents for patients with suboptimal glycemic control. , Diabetes Metab Syndr Obes 5, 213-7.
- 82.NavrotskayaV V, Oxenkrug G, Vorobyova L I, Summergrad P. (2012) Berberine prolongs life span and stimulates locomotor activity of Drosophila melanogaster. , Amer J Plant Sci 3, 1037-1040.
- 83.Oxenkrug G, Vorobyova V Navrotskaya L, Summergrad P. (2012) . Minocycline Effect on Life and Health Span of Drosophila Melanogaster. Aging and Disease 3: 352 – 359 .
- 84.Unoki-Kubota H, Yamagishi S, Takeuchi M, Bujo H, Saito Y. (2010) . , Protein Pept. Lett 17, 1177-81.
- 85.Murakoshi M, Tanimoto M, Gohda T, Hagiwara S, Ohara I. (2009) Diabetes Res. Clin. Pract. 83,183-9
- 88.N van Wijk, C J Watkins, R J Hageman, J C Sijben, R J Wurtman. (2013) . , Nutr Metab (Lond) 9, 10-1186.
- 90.Niinisalo P, Raitala A, Pertovaara M, Oja S S, Lehtimäki T. (2008) . , Scand. J. Clin. Lab. Invest 68, 767-770.
Cited by (30)
This article has been cited by 30 scholarly works according to:
Citing Articles:
Pharmacological Research (2025) Crossref
Luqi Qin, Bei Fan, Yixia Zhou, Jiahuan Zheng, Rao Diao et al. - Pharmacological Research (2025) Semantic Scholar
Behavioural Brain Research (2024) Crossref
Joanna Mikulska, Diana Pietrzak, Paweł Rękawek, Krystian Siudaj, Łucja Justyna Walczak-Nowicka et al. - Behavioural Brain Research (2024) Semantic Scholar
Biomedicines (2023) Crossref
Kathleen Mikkelsen, Narges Dargahi, S. Fraser, V. Apostolopoulos - Biomedicines (2023) Semantic Scholar
K. Kozieł, E. Urbanska - Cells (2023) Semantic Scholar
Frontiers in Microbiology (2023) Crossref
S. Miri, JuDong Yeo, Sarah Abubaker, R. Hammami - Frontiers in Microbiology (2023) Semantic Scholar
Nature Communications (2022) Crossref
Teng Huang, Jia‐xin Song, Jia Gao, Jia Hua Cheng, Hao Xie et al. - Nature Communications (2022) Semantic Scholar
Frontiers in Oncology (2022) Crossref
Yun Chen, Jianliang Chen, Dainian Guo, Peixuan Yang, Shuang Chen et al. - Frontiers in Oncology (2022) Semantic Scholar
Progress in Neuro-Psychopharmacology and Biological Psychiatry (2021) Crossref
Leszek Rudzki, T. Stone, Michael Maes, B. Misiak, J. Samochowiec et al. - Progress in Neuro-psychopharmacology and Biological Psychiatry (2021) Semantic Scholar
Progress in Neuro-Psychopharmacology and Biological Psychiatry (2021) Crossref
Brain, Behavior, and Immunity (2020) Crossref
M. Morgese, S. Schiavone, A. B. Maffione, P. Tucci, L. Trabace - Brain, behavior, and immunity (2020) Semantic Scholar
M. Więdłocha, P. Marcinowicz, M. Janoska-Jaździk, A. Szulc - Progress in Neuro-psychopharmacology and Biological Psychiatry (2020) Semantic Scholar
Molecular Neurobiology (2020) Crossref
Leszek Rudzki, M. Maes - Molecular Neurobiology (2020) Semantic Scholar
Journal of Nutrition and Health (2020) Crossref
신 필경, 천 수경, 김 명선, 박 선주, 김 민정 et al. - Journal of Nutrition and Health (2020) Semantic Scholar
Psychoneuroendocrinology (2019) Crossref
Leszek Rudzki, L. Ostrowska, D. Pawlak, A. Małus, K. Pawlak et al. - Psychoneuroendocrinology (2019) Semantic Scholar
Endocrine Journal (2019) Crossref
Progress in Neuro-Psychopharmacology and Biological Psychiatry (2019) Crossref
A. Lakstygal, M. D. Abreu, Dmitry A. Lifanov, Edina A. Wappler-Guzzetta, Nazar Serikuly et al. - Progress in Neuro-psychopharmacology and Biological Psychiatry (2019) Semantic Scholar
Edward Yu, C. Papandreou, M. Ruíz-Canela, M. Guasch-Ferré, C. Clish et al. - Clinical Chemistry (2018) Semantic Scholar
Clinical Chemistry (2018) Crossref
Biological Research For Nursing (2018) Crossref
M. Groer, D. Fuchs, A. Duffy, Adetola F. Louis-Jacques, Amy L. D’Agata et al. - Biological Research for Nursing (2018) Semantic Scholar
Cytometry Part A (2018) Crossref
Qian Li, Chun Huang, Liwei Liu, Rui Hu, J. Qu - Cytometry Part A (2018) Semantic Scholar
Metabolic Brain Disease (2018) Crossref
Yanjie Qin, Nanxi Wang, Xinlin Zhang, Xuemei Han, Xuejia Zhai et al. - Metabolic brain disease (2018) Semantic Scholar
Pharmacological Research (2018) Crossref
Journal of Affective Disorders (2018) Crossref
L. van Lee, S. Cai, S. Loy, E. H. Tham, F. Yap et al. - Journal of Affective Disorders (2018) Semantic Scholar
Neuropharmacology (2017) Crossref
F. Fazio, L. Lionetto, M. Curto, L. Iacovelli, C. Copeland et al. - Neuropharmacology (2017) Semantic Scholar
European Journal of Pharmacology (2017) Crossref
M. Morgese, S. Schiavone, L. Trabace - European Journal of Pharmacology (2017) Semantic Scholar
M. Morgese, S. Schiavone, E. Mhillaj, M. Bove, P. Tucci et al. - Pharmacological Research (2017) Semantic Scholar
Nutrients (2017) Crossref
J. Plows, F. Budin, Rebecka A. M. Andersson, Valerie J. Mills, K. Macé et al. - Nutrients (2017) Semantic Scholar
Molecular Neurobiology (2017) Crossref
Molecular Neurobiology (2016) Crossref
Isabella Caroline Silva Dias, Bruno Carabelli, D. Ishii, Helen Morais, M. C. Carvalho et al. - Molecular Neurobiology (2016) Semantic Scholar
G. Morris, M. Berk, A. Carvalho, J. Caso, Y. Sanz et al. - Molecular Neurobiology (2016) Semantic Scholar
Molecular Neurobiology (2015) Crossref
G. Oxenkrug - Molecular Neurobiology (2015) Semantic Scholar
(2015) Crossref
G. Oxenkrug - (2015) Semantic Scholar
S. Mittal - Cambridge International Law Journal (2015) Semantic Scholar
